Abstract
The level of the specific activity of digestive enzymes and expression of their corresponding glucanase genes (Cg-glu1 ) were studied in Grays mussels, Crenomytilus grayanus , collected from the mussels' assemblages in different parts of Peter the Great Bay (northwestern part of the Sea of Japan). Both methods demonstrated similar independent results: there is variability in the contribution of phytoplankton to the nutrition of mussels in different parts of Peter the Great Bay. The highest level of the specific activity of 1,3-β-D-glucanases and expression of the glucanase genes were detected in the mussels from Amursky Bay: the differences were significantly increased (p < 0.05) compared with the level of the specific activity of the digestive enzyme 1,3-β-D-glucanases in mussels from Vostok Bay and Ussuriisky Bay. In all three bays, young mussels consumed phytoplankton more actively than 20-year-old mussels.
Keywords
Grays mussel ; Crenomytilus grayanus ; Phytoplankton ; 1,3-β-D-glucanases ; Glucanase genes Cg-glu1
Introduction
Bivalve molluscs are among the most dominant filter-feeders in coastal marine environments (Seed, 1976 ). There have been several investigations that have focused on the diet of this important group of marine invertebrates (Seed and Suchanek, 1992 ). Mussels obtain nutrients from dissolved organic substances, bacteria, phytoplankton, and a number of zooplankton species of various sizes (Davenport et al ., 2000 ; Roditi et al ., 2000 ; Wong et al ., 2003 ). Phytoplankton is the main component of the diet of many suspension-feeders, including bivalves that inhabit shallow marine environments (Bricelj and Shumway, 1991 ; MacDonald and Ward, 1994 ). Modern methods that have been applied to the study of trophic structures of an ecosystem are mostly aimed at identifying food sources (Kharlamenko et al ., 1995 ; Kharlamenko et al ., 2001 ); however, the quantity of phytoplankton has rarely been identified from these studies. Phytoplankton provides a key link between primary production and higher trophic levels. Both the quality and quantity of seston are important determinants of food resources of bivalves; furthermore, both of these factors are highly variable in marine environments (Bayne, 1993 ). Therefore, modern methods are required to estimate the nutrition components of marine invertebrates.
Grays mussel, Crenomytilus grayanus (Dunker 1853), is the dominant mussel species of the coastal benthic communities of the Sea of Japan; it plays an important role in the function of the marine ecosystems ( Skarlato, 1981 ). Additionally, this common mussel has commercial value in the Far East federal disrict of Russia. Previous studies regarding the biology and ecology of this species have been performed. Little information is available regarding the diet and feeding behaviour of this mussel and its trophic relationships with phytoplankton communities. At the present time, the study of the mytilids' role as consumers of seston is required for understanding the trophic structure of the marine coastal communities, particularly in those areas where the bivalve molluscs of the family Mytilidae are abundant. However, little is known about seston composition of the mussels diet; moreover, their feeding behaviours in its trophic relationships with phytoplankton communities remain unclear. 1,3-β-D-glucanases of marine invertebrates take part in the digestion of dietary polysaccharides; these play an important role in embryogenesis. On the other hand, it is known that phytoplankton, including diatoms and phytoflagellates, contain 1,3-β-D-glucans, which are the main nutritional components of mussels ( Elyakova et al ., 1981 ; Myklestad et al ., 1982 ; Whyte, 1987 ). Therefore, the study of the activity of enzymes that catalyse the transformation of this polysaccharide will potentially benefit the field of marine biology and can greatly expand the fundamental knowledge regarding the digestion process of hydrobionts.
The aim of this study is to investigate the 1,3-β-D-glucanases activity of extracts and to analyse the expression of the 1,3-β-D-glucanase genes in the common representative benthic communities of the Sea of Japan Grays mussel Crenomytilus grayanus in different habitats. It is known that the digestive enzyme 1,3-β-D-glucanases catalyses the cleavage of the major polysaccharides 1,3;1,6-β-D-glucans (laminarans), which have been found in the composition of phytoplankton. This paper is the first of its type to attempt to study the nutrition requirements of the common mussel C. grayanus using two independent methods of analysis; the goal is to determine the amount of phytoplankton that acts as the nutritional source of mussels and to compare its value between mussels of different ages from three bays. The analysis of 1,3-β-D-glucanase gene expression and its activity allows one to evaluate marine organisms as consumers of phytoplankton, which are the primary producers of energy in the marine ecosystems.
Material and Methods
Study Area and Sampling Sites
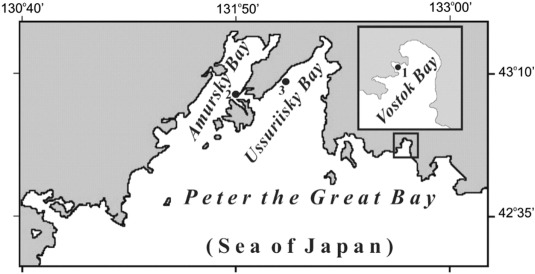
Mussels were collected in August of 2006, 2007, and 2009 at a water temperature of 19 ± 2 °C from the mussels' beds in different parts of Peter the Great Bay (northwestern part of the Sea of Japan). Mussels were collected by scuba diving at three open sites near their typical habitats (Fig. 1 ). Sampling site 1 (Vostok Bay) consisted of large boulders with a clumpy pile at the open coast that was subjected to frequent surf; molluscs were collected at a depth of 7 m. Sampling site 2 (Amursky Bay) also consisted of a hard substrate of the cape that was subjected to surf; mussels were collected at a depth of 6–9 m. Sampling site 3 (Ussuriisky Bay) represented a mix of the boulders and pebbles; mussels were collected at a depth of 6–7 m. These bottom sites corresponded to variations of bionomic type III subtidal zones.
|
|
|
Fig. 1. Map of the study area and the positions of the sampling sites. |
Determination of Enzymatic Activity
The activity of 1,3-β-D-glucanases, as it relates to catalysing the hydrolysis of laminaran, was estimated by an increase of the reducing saccharides amounts by the Nelson–Somogyi method (Nelson, 1944 ; Somogyi, 1952 ). All molluscs were collected and immediately delivered to the laboratory. Each mussel was opened and the digestive glands were excised. The digestive glands were homogenized and extracted with 0.025 M Na-succinate buffer (pH 5.2) at a 1:3 ratio. The extract was centrifuged for 15 min at 9,000 rpm at 4 °C; 1 ml of the supernatant was applied to the column with Sephadex G-25, which was performed to remove low-molecular-weight substances (Sova et al., 1970 ). Laminaran from the brown alga Laminaria cichorioides was obtained as previously described ( Zvyagintseva et al., 1999 ) and was used as a substrate.
The incubation mixture contained 100 μl of the enzyme and 400 μl of the laminaran solution (1 mg/ml in 0.05 M Na-succinate buffer, рН 5.2). The time of incubation did not exceed that needed to cleave 10% of the substrate glycosidic bonds.
A unit of enzyme activity was defined as the amount of the enzyme that was required to catalyse the formation of 1 μmol of glucose in 1 min under standard conditions. The activity was expressed as a unit/mg protein.
The protein concentration was measured according to the Lowry procedure using BSA as a standard (Lowry et al., 1951 ).
The digestive activity of mussels was estimated based on the level of the enzymatic activity of 1,3-β-D-glucanases (laminarinases) in homogenized hepatopancreas, which had the highest enzymatic activity compared to other О-glycoside hydrolases.
Different shell lengths were used to estimate the digestive enzyme activity based on the age of mussels. To determine the age of each mussel, the growth rings on the outer shell surface and the structural marks on the longitudinal slices of each shell were examined (Zolotarev, 1989 ). Each sample of the digestive gland, containing 3–5 living specimens, was analysed three times.
Reverse Transcriptase PCR (RT-PCR)
Total RNA isolation was performed as previously described (Dubrovina et al., 2010 ) using YellowSolve extraction (Clonogen, St. Petersburg, Russia); however, there were minor differences: each cDNA library was obtained from the tissues of the hepatopancreas of 3 mussels (0.5–0.6 g, fresh weight), which was homogenized with 0.5–1 ml of YellowSolve. The homogenized solution (0.2 ml) was used to further isolate the RNA.
The semiquantitative RT-PCR analysis was performed as has been previously described (Odintsova et al., 2006 ). The degenerate primers 5′-GGC GAG ATC GAY ATY ATG (Gluc1) and 5′-TAG AAC GGC TTR TCG AAY GG (Gluc2) were designed on the basis of amino acid sequences GEIDIM and PFDKPFY, which are conserved for 1,3-β-D-glucanases (Kovalchuk et al., 2006 ). These primers were used for the amplification of the 347-bp glucanase transcripts, Ta = 50 o С, elongation time = 30 sec.
Based on the known Mytilus edulis mRNA for the actin gene (AB257134), we designed specific primers, Actin1: 5′-CGAGCAGGAAATGTCAACCG and Actin2: 5′-TGTCGGCAATACCTGGGAAC. These primers were used for the amplification of the 270-bp actin transcripts, Ta = 56 o С, elongation time = 20 sec. The RT-PCR products of the C. grayanus actin gene (Cg-actin1 ) share 96% sequence identity with the known M. edulis mRNA (AB257134). The data pertaining to the glucanase gene expression are presented as relative fluorescent units normalized to the expression of the corresponding Cg-actin1 gene ( Kiselev et al., 2006 ). We used actin for the normalization of quantitative or semiquantitative RT-PCR for the mussels because it is a commonly used house-keeping genes (Bigot et al., 2010 ).
PCR products were collected after 25, 30, 35, 40, and 45 cycles; the linearity of PCR was determined using a quantitative analysis by GelDoc Quantity One 4.6.2 software (Boi-Rad, USA). The linearity of PCR was determined to be between 35 and 40 cycles for the glucanase genes and between 30 and 35 cycles for the actin genes.
Sequence Analysis
The RT-PCR products were sequenced by methods that were described (Kiselev et al., 2006 ) at the Instrumental Centre of Biotechnology and Gene Engineering of IBSS FEB RAS using an ABI 3130 Genetic Analyzer (Applied Biosystems, Foster City, USA). Sequencing of each gene was performed at least three times. The BLAST search program was used for sequence analysis. Multiple sequence alignments were performed using the BioEdit 7.0.8 program (http://www.mbio.ncsu.edu/BioEdit/bioedit.html ).
Statistical Analysis
Statistical analysis was performed using the Statistica 8.0 program and Microsoft Excel. The results were represented as the mean ± standard deviation (S.D.) and were tested by a paired Students t-test. The percentage ratio of the level of the 1,3-β-D-glucanases activity and the gluconase gene expression among all of the age groups of mussels were compared. The 0.05 level was selected as the point of minimal statistical significance in all analyses.
Results
Sequence Analysis and Comparisons
The degenerate primers 5′-GGC GAG ATC GAY ATY ATG (Gluc1) and 5′-TAG AAC GGC TTR TCG AAY GG (Gluc2) were designed for the semiquantitative RT-PCR analysis, which led to the amplification of the 347-bp glucanase transcripts; these transcripts were based on the amino acid sequences GEIDIM and PFDKPFY, which are conserved for 1,3-β-D-glucanases (Kovalchuk et al., 2006 ). Based on the known Mytilus edulis mRNA for the actin gene (AB257134), we designed specific primers, 5′-CGAGCAGGAAATGTCAACCG (Actin1) and 5′-TGTCGGCAATACCTGGGAAC (Actin2), which were used for the amplification of the 270-bp actin transcripts. The RT-PCR products of the C. grayanus actin gene (Cg-actin1 ) share 96% sequence identity with the known M. edulis mRNA (AB257134).
We sequenced several transcripts of glucanase gene transcripts from various individuals that differed in 1–4 nucleotides (347-bp PCR products). Only one glucanase sequence was repeated several times in the independent transformations. We deposited only one glucanase sequence, which was repeated several times in the independent transformations, to GeneBank (GenBank Accession No. FJ711171). It is the first deposited sequence for a glucanase of Crenomytilus grayanus . Comparison of the deduced amino acid sequences of these fragments with the previously deposited glucanase sequences in GenBank indicates that the transcripts encode 1,3-β-D-glucanases ( Table 1 ). The 1,3-β-D-glucanase gene, which was identified in the cells of C. grayanus for the first time and designated Cg-glu1 , shares 38–50% sequence identity at the protein level with previously characterized 1,3-β-D-glucanases ( Table 1 ). The deduced amino acid sequence of the Cg-glu1 gene shows 50% identity with 1,3-β-D-glucanase from the scallop Mizuhopecten yessoensis ( Kovalchuk et al., 2006 ).
| Mizuhopecten yessoensis (AY848857) | Chlamys rosealbus (AAZ04386) | Pseudocardium sachalinensis (AAP74223) | Strongylocentrotus purpuratus (AAC47235) | |
|---|---|---|---|---|
| Crenomytilus grayanus'Cg-glu1 (FJ711171) | 50 | 50 | 38 | 40 |
Enzymatic Activities of Glucanases in Extracts and the Expression of Glucanase Genes in the Hepatopancreas of Mussels in Ontogenesis
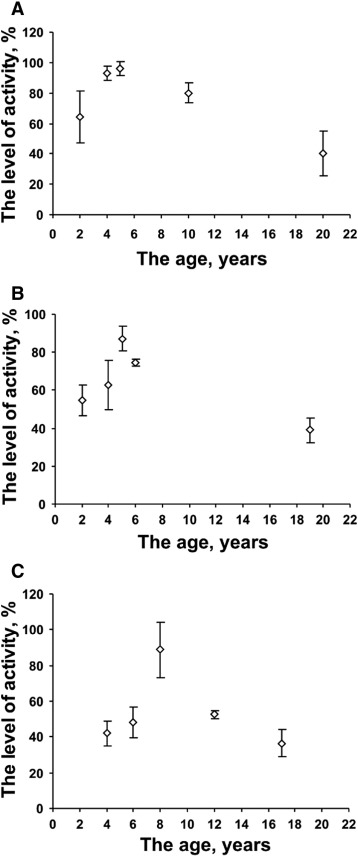
Fig. 2 (a–c) shows that this mussel species has high levels of specific activity 1,3-β-D-glucanases in all three bays independent of the age of the specimens; however, this parameter is changed in the process of ontogenesis. Relatively low 1,3-β-D-glucanase activity was noticed in extracts from all of Grays mussel specimens at the early stage of ontogenesis and from specimens of the oldest age group in all three bays. For example, in mussels from the II and the III age groups (Fig. 2 a), which were collected from Amursky Bay, the mean specific activity of 1,3-β-D-glucanases in extracts was three times higher (p < 0.05) than in 16-year-old mussels. Fig. 2 b simultaneously shows that the level of the specific activity of 1,3-β-D-glucanases in Vostok Bay in extracts was twofold higher in 5-year-old mussels (p < 0.05) compared to the activity of 19-year-old specimens. However, in Ussuriisky Bay, the changes in the specific activity of 1,3-β-D-glucanases in extracts of ontogenesis in C. grayanus were different than in two previous bays ( Fig. 2 b). The highest level of the specific activity of 1,3-β-D-glucanases in extracts was detected in age group III, 7-year-old mussels with a shell length of 70 mm (p < 0.05); the levels in mussels from age group V were significantly lower (p < 0.05) than in mussels from age group IV (Fig. 2 c).
|
|
|
Fig. 2. Activity of the 1,3-β-D-glucanases (mean ± S.D.) in the mussel Crenomytilus grayanus, grouped by different ages from three different bays: Amursky Bay (A), Vostok Bay (B), and Ussuriisky Bay (C). Note: Mussels from Amursky Bay were divided into 5 groups: I – 2-year-old mussels (shell length approximately 30 mm); II – 4-year-old mussels (40 mm); III – 5-year-old mussels (55 mm); IV – 10-year-old mussels (85 mm); and V – 16-year-old mussels (110 mm). Mussels from Vostok Bay were divided into age groups: I – 2-year-old mussels (30 mm); II – 4-year-old mussels (45 mm); III – 5-year-old mussels (58 mm); IV – 6-year-old-mussels (65 mm); and V – 19-year-old mussels (approximately 110 mm). Mussels from Ussuriisky Bay were arranged by age groups: I – 4-year-old mussels (40 mm); II – 6-year-old mussels (50 mm); III – 7-year-old mussels (68 mm); IV – 12-year-old mussels (85 mm); and V – 17-year-old mussels (100 mm). |
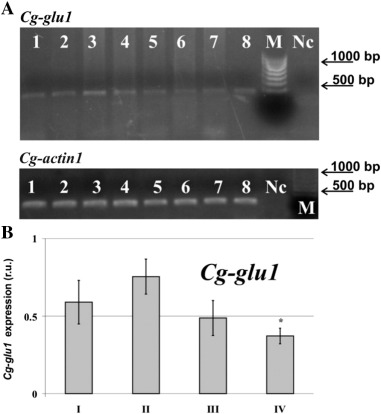
These results are consistent with the data results of analysis of the Cg-glu1 gene expression ( Fig. 3 lines 1,3), which showed that the highest level of expression of the Cg-glu1 gene corresponded to the youngest specimens, 3-year-old and 4–5-year-old in age groups I and II, respectively. The gene expression in mussels from group II was twice as high (p < 0.05) as that of the mussels from group IV (Fig. 3 ).
|
|
|
Fig. 3. Expression of the glucanase gene in the hepatopancreas of the mussel Crenomytilus grayanus of different age groups from Amursky Bay. (A) Electrophoretic separation of the Crenomytilus grayanus RT-PCR products of Cg-glu1 and Cg-actin1 . Lanes 1, 3, 5, and 7: I, II, III, and IV mussel groups, 2 μl of the templates diluted with H2 O 1:4. Lanes 2, 4, 6, and 8: the groups are presented in the same order, 2 μl of undiluted templates. Nc, negative control (PCR mixture without mussel cDNA); M, synthetic marker. (B) Quantification of the Cg-glu1 transcripts by GelDoc Quantity One. The designation r.u. indicates relative fluorescence units. The data obtained from three independent experiments were averaged and are presented as the mean ± SEM. * p < 0.05, vs. values of II mussel group. See text for comment. |
Enzymatic Activity of the Glucanases in the Extracts and the Expression of the Glucanase Genes in the Hepatopancreas of Mussels Collected from Three Bays of the Sea of Japan
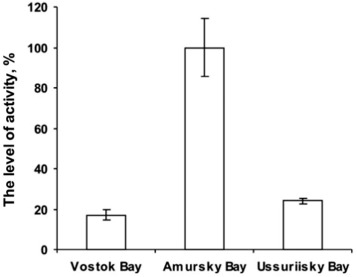
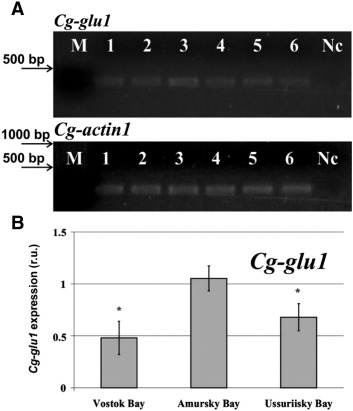
To investigate the consumption of phytoplankton by the mussel Crenomytilus grayanus from three different bays of the Sea of Japan, we analysed the activity level of 1,3-β-D-glucanases and the glucanase gene expression ( Fig. 4 ; Fig. 5 ). The highest activity level of 1,3-β-D-glucanases in extracts was detected in mussels from Amursky Bay, where the differences were significantly (p < 0.05) higher compared with those in mussels from Vostok Bay and Ussuriisky Bay (Fig. 4 ). The lowest level of 1,3-β-D-glucanases activity in the extracts was detected in mussels collected from Vostok Bay. Between Vostok Bay and Ussuriisky Bay, statistical significance was assumed as p < 0.01.
|
|
|
Fig. 4. Comparison of the levels of the 1,3-β-D-glucanases activity in the mussel Crenomytilus grayanus from three bays of the Sea of Japan. Note: vertical lines – S.D. |
|
|
|
Fig. 5. Expression of the glucanase gene in the hepatopancreas of the mussel Crenomytilus grayanus collected from three bays of the Sea of Japan. (A) Electrophoretic separation of the C. grayanus RT-PCR products of Cg-glu1 and Cg-actin1 . Lanes 1, 3, and 5: Vostok, Amursky, and Ussuriisky bays, 2 μl of the templates diluted with H2 O 1:4. Lanes 2, 4, and 6: Vostok, Amursky, and Ussuriisky bays, 2 μl of undiluted templates. Nc, negative control (PCR mixture without mussel cDNA); M, synthetic marker. (B) Quantification of the Cg-glu1 transcripts by GelDoc Quantity One. The designation r.u. indicates relative fluorescence units. *p < 0.05, vs. values of Amursky Bay. The data obtained from three independent experiments were averaged and presented as the mean ± S.D. |
Comparison analysis showed (Fig. 5 ) that among three bays of the Sea of Japan, the highest level of glucanase gene expression in the digestive gland was detected in 7-year-old specimens of C. grayanus with a shell length of approximately 70–75 mm, which was collected in Amursky Bay (Fig. 5 a). Our results show statistical significance at 95 % level between the differences in the glucanase gene expression in C. grayanus from Amursky Bay, Vostok Bay, and Ussuriisky Bay ( Fig. 5 b). In Vostok Bay, the level of glucanase gene expression was 2 times lower than in Amursky Bay (p < 0.05), whereas the differences between glucanase gene expression in mussels from Ussuriisky Bay and Vostok Bay were 1.5 times greater (Fig. 5 b).
Discussion
Currently, the study of glucanase activity in marine invertebrates, particularly in bivalve molluscs, is an important object of research, as these enzymes can be used to produce biologically active fragments of 1,3; 1,6-β-D-glucan from marine invertebrates (Menshova et al., 2014 ) and may lead to determining the exact structure of the polysaccharides (Menshova et al., 2014 ); however, our study was conducted in small quantities of similar research. In a number of studies, high activity levels of 1,3-β-D-glucanases in hepatopancreas in bivalve molluscs have been detected (Elyakova et al ., 1981 ; Sova et al ., 2013 ). This is likely related to the peculiarities of ration feeding, which include organisms with high compositions of laminaran; laminaran is a storage mechanism for polysaccharides and is an important source of glucose for algae feeders. Prior to this study, the activity of glucanases has only been investigated in two commercial species of bivalves: the scallops Mizuhopecten yessoensis and Pseudocardium sachalinensis (Spisula sachalinensis ) ( Sova et al ., 2013 ; Kovalchuk et al ., 2006 ). According to the opinion of previous authors (Sova et al., 2013 ), these species have high levels of 1,3-β-D-glucanases activity in the hepatopancreas; this corresponds to enzyme hyperactivity in the crystalline stalks as has been confirmed by a molecular study. High levels of 1,3-β-D-glucanases activity in the crystalline stalks can be explained by the storage functions of the enzymes ( Sova et al ., 2013 ; Kovalchuk et al ., 2006 ). On the other hand, the level of activity in other enzyme proteinases, as well as chitinases, was appreciably low in bivalves compared to the levels of the enzyme activity in the digestive glands of specimens Crustaceae and Asteroidea ( Elyakova, 1972 ; Kozlovskaya and Vaskovsky, 1970 ), which are predators in nature. High levels of glucanase activity correlate with the fact that the main component of food in bivalves is phytoplankton rich in the content of 1,3-β-D-glucans ( Elyakova et al ., 1981 ; Myklestad et al ., 1982 ; Whyte, 1987 ).
The sessile organism mussel C. grayanus lives in the upper part of the intertidal zone at a depth of 1–60 m; they attach themselves to the boulders using the byssal threads (Skarlato, 1981 ). As with other species of mytilids, C. grayanus appears to be a suspension filter-feeder that continuously filters tiny particles, including phytoplankton, from the ambient water ( Shumway et al ., 1985 ; Bricelj and Shumway, 1991 ; MacDonald and Ward, 1994 ; Davenport et al ., 2000 ). In this study, the RT-PCR results showed that the glucanase gene expression was twofold higher in young 5-year-old mussels than in older 16-year-old mussels (Fig. 3 , lines 3, 7); this was confirmed by the amount of 1,3-β-D-glucanase activity, which was isolated in mussels independent of their habitat (Fig. 2 A–C). As can be seen in the figures, the activity of the digestive enzyme 1,3-β-D-glucanase and the gene expression of glucanase during the ontogenesis of mussels were similar. At the beginning of ontogenesis, the activity level of the digestive enzyme 1,3-β-D-glucanase and expression of the glucanase gene were increased C. grayanus , which reached a maximum value in 5–8-year-old mussels; at the end of ontogenesis, their values gradually decreased, with levels reaching 30–50% maximum values.
The results of both analyses have showed that these mussels consume different amounts of phytoplankton during ontogenesis, independent of their location. At the same time, they consume phytoplankton more actively at the early stages of development than at the older stages in all bays. These differences, which observed in different amounts of phytoplankton, consumed by the mussels during ontogenesis probably can be explained such biological processes as growth and reproduction activity. At the early stage of ontogenesis, C. grayanus feeds on phytoplankton at a higher rate compared to adults. During reproductive period the feeding trends in C. grayanus decreased when mussels reach mature condition ( Seed, 1976 ). This may be due to the diet of bivalves, which usually includes diverse species of phytoplankton and other particles of seston, depending on the temporal changes in the environmental conditions ( Bayne, 1993 ; Davenport et al ., 2000 ; Wong et al ., 2003 ). Additionally, many factors may influence the filtration rate of mussels under environmental conditions where a wide spectrum of changing factors may influence the feeding behaviour.
It is widely known that bivalve molluscs can exert significant control over phytoplankton population (Safi and Gibbs, 2003 ). Their filter feeding activity effectively prevents the occurrence of phytoplankton blooms. In many countries, marine mussels are used as a natural control of eutrophication in coastal areas. Our results shown that the contribution of phytoplankton to the mussels' diet varied for the species collected from different parts of Peter the Great Bay.
The results of our analyses have showed that among 7-year-old mussels, C. grayanus consumes phytoplankton most actively in Amursky Bay compared with Vostok Bay and Ussuriisky Bay ( Fig. 4 ; Fig. 5 ). Presumably, the observed differences are result from various trophic levels in these bays. Additionally, our results were confirmed by the data on the density and biomass of phytoplankton from Peter the Great Bay from July to August ( Selina, 1992 ; Stonik and Orlova, 1998 ; Begun, 2004 ).
The phytoplankton data observed in Amursky Bay demonstrated the highest values of biomass (11,000–14,000 mg m3 ) and density of algal cells (9.4–14.6 mln. cells L− 1 ) (Stonik and Orlova, 1998 ) compared to those observed in Ussuriisky Bay, where phytoplankton had the smallest values of biomass (60–1250 mg m3 ) and density of algal cells (0.989–0.990 mln. cells L− 1 ) (Begun, 2004 ). As shown in a previous study (Selina, 1992 ) in Vostok Bay, these parameters were two times higher than in Ussuriisky Bay. Therefore, the high biomass phytoplankton values found in these waters appear to confirm the eutrophic and oligotrophic features of these coastal areas.
Based on the data regarding high glucanase activity and expressions of the glucanase genes in the analysed cells, we suggest that phytoplanktons are an essential nutritional component of the mussel C. grayanus . However, the amounts of phytoplankton that are consumed by mussels depended on the respective habitats and age. Two factors that determine the diet of mussels are the quality and amount of seston. These factors have considerable variability due to the ever-changing environmental conditions of the marine coastal zones. In response to the unstable nutritional environment, many filter feeding invertebrates, including the widespread groups of mytilids bivalves, have developed an elaborate and selective feeding strategy ( Shumway et al ., 1985 ; Prins et al ., 1991 ).
Conclusions
In conclusion, in this study, we have undertaken preliminary steps to advance our understanding of the feeding activity of C. grayanus in different bays of the Sea of Japan; additionally, we tried to quantitatively evaluate the consumption of phytoplankton by this mussel species. Thus, our investigation of the activity of the digestive enzyme 1,3-β-D-glucanase and the expression of the glucanase genes in marine mussels can be used as a convenient method to determine the contribution of phytoplankton as a nutritional source for marine invertebrates.
Our study has demonstrated that described techniques can successfully be used as alternative pathways to determine the contribution of phytoplankton to the nutritional requirements of mussels. The results of successfully applying quantitative PCR for a diet analysis in other invertebrates have been documented in the literature (Martin et al ., 2006 ; Nejstgaard et al ., 2008 ).
Acknowledgements
We would like to thank a number of people for their help with the field and laboratory analyses for this project. This research was supported by grants (10-III-В-06-114 , 09-III-А-06-210 ) of Far East Branch of Russian Academy of Sciences (FEB RAS), Program of Russian Academy of Sciences Molecular and Cellular Biology, and Russian Foundation for Basic Research (09-04-08349-3 , 10-04-00427 ) grants. Additionally, we thank the Instrumental Centre of Biotechnology and Gene Engineering of Institute of Biology and Soil Science FEB RAS. Finally, we are especially grateful to Dr. Alexandra Dubrovina, who improved the English of the manuscript.
References
- Bayne, 1993 B. Bayne; Feeding physiology of bivalves: time-dependence and compensation for changes in food availability; ,in: R.F. Dame (Ed.), Bivalve filter-feeders in estuarine and coastal ecosystem processes, NATO ASI Series G: Ecological Sciences, vol. 33, , Springer-Verlag, Heidelberg (1993), pp. 1–24
- Begun, 2004 A.A. Begun; Phytoplankton in the Golden Horn Bay and the Ussuri Bay (Japan Sea) under condition of anthropogenic population; Izv. TINRO, 138 (2004), pp. 320–344
- Bigot et al., 2010 A. Bigot, P. Vasseur, F. Rodius; SOD and CAT cDNA cloning, and expression pattern of detoxification genes in the freshwater bivalve Unio tumidus transplanted into the Moselle river ; Ecotoxicology, 19 (2010), pp. 369–376
- Bricelj and Shumway, 1991 V. Bricelj, S. Shumway; Physiology. Energy acquisition and utilization; S.E. Shumway (Ed.), Biology, Ecology and Aquaculture of Scallops, Elsevier, Amsterdam (1991), pp. 305–345
- Davenport et al., 2000 J. Davenport, R.J.J.W. Smith, M. Packer; Mussels Mytilus edulis : significant consumers and destroyers of mesozooplankton ; Mar. Ecol. Prog. Ser., 198 (2000), pp. 131–137
- Dubrovina et al., 2010 A.S. Dubrovina, A.Y. Manyakhin, Y.N. Zhuravlev, K.V. Kiselev; Resveratrol content and expression of phenylalanine ammonia-lyase and stilbene synthase genes in rol C transgenic cell cultures of Vitis amurensis; Appl. Microbiol. Biotechnol., 88 (2010), pp. 727–736
- Elyakova, 1972 L.A. Elyakova; Distribution of cellulases and chitinases in marine invertebrates; Comp. Biochem. Physiol. B, 43 (1) (1972), pp. 67–70
- Elyakova et al., 1981 L.A. Elyakova, N.M. Shevchenko, S.M. Avaeva; A comparative study of carbohydrase activities in marine invertebrates; Comp. Biochem. Physiol. B, 69 (4) (1981), pp. 905–908
- Kharlamenko et al., 1995 V.I. Kharlamenko, N.V. Zhukova, S.V. Khotimchenko, V.I. Svetashev, G.M. Kamenev; Fatty acids as markers of food sources in shallow-water hydrothermal ecosystems (kraternaya Bight, Yankich Island, Kurile islands); Mar. Ecol. Prog. Ser., 120 (1995), pp. 231–241
- Kharlamenko et al., 2001 V.I. Kharlamenko, S.I. Kiyashko, A.B. Imbs, D.I. Vyshkvartzev; Identification of food source of invertebrates from the sea grass Zostera marina community using carbon and sulphur stable isotope ratio and fatty acid analyses ; Mar. Ecol. Prog. Ser., 220 (2001), pp. 103–117
- Kiselev et al., 2006 K.V. Kiselev, M.I. Kusaykin, A.S. Dubrovina, D.A. Bezverbny, T.N. Zvyagintseva, V.P. Bulgakov; The rol C gene induces expression of a pathogenesis-related B-1,3-glucanase in transformed ginseng cells; Phytochemistry, 67 (2006), pp. 2225–2231
- Kovalchuk et al., 2006 S.N. Kovalchuk, E.V. Sundukova, M.I. Kusaykin, K.V. Guzev, S.D. Anastiuk, G.N. Likhatskaya, E.V. Trifonov, E.A. Nurminski, V.B. Kozhemyako, T.N. Zvyagintseva, V.A. Rasskazov; Purification, cDNA cloning and homology modeling of endo-1,3-beta-D-glucanase from scallop Mizuhopecten yessoensis; Comp. Biochem. Physiol. B, 143 (4) (2006), pp. 473–485
- Kozlovskaya and Vaskovsky, 1970 E.P. Kozlovskaya, V.E. Vaskovsky; A comparative study of proteinases of marine invertebrates; Comp. Biochem. Physiol. B, 34 (1) (1970), pp. 137–142
- Lowry et al., 1951 O.H. Lowry, N.J. Rosebrough, A.L. Farr, R.J. Randall; Protein measurement with Folin phenol reagent; J. Biol. Chem., 193 (1951), pp. 265–275
- MacDonald and Ward, 1994 B. MacDonald, J. Ward; Variation in food quality and particle selectivity in the sea scallop Placopecten magellanicus (Mollusca: Bivalvia) ; Mar. Ecol. Prog. Ser., 108 (1994), pp. 251–264
- Martin et al., 2006 D.L. Martin, R.M. Ross, L.B. Quetin, A.E. Murray; Molecular approach (PCR-DGGE) to diet analysis in young Antarctic krill Euphausia superba; Mar. Ecol. Prog. Ser., 319 (2006), pp. 155–165
- Menshova et al., 2014 R.V. Menshova, S.P. Ermakova, S.D. Anastyuk, V.V. Isakov, Y.V. Dubrovskaya, M.I. Kusaykin, B.H. Um, T.N. Zvyagintseva; Structure, enzymatic transformation and anticancer activity of branched high molecular weight laminaran from brown alga Eisenia bicyclis; Carbohydr. Polym., 99 (2014), pp. 101–109
- Myklestad et al., 1982 S. Myklestad, R. Djurhuus, A. Mohus; Demonstration of exo-(β-1,3)-D-glucanase activity in some planctonic diatoms; J. Exp. Mar. Biol. Ecol., 56 (1982), pp. 205–211
- Nejstgaard et al., 2008 J.C. Nejstgaard, M.E. Frischer, P. Simonelli, C. Troedsson, M. Brakel, F. Adiyaman, A.F. Sazhin, L.F. Artigas; Quantitative PCR to estimate copepod feeding; Mar. Biol., 153 (2008), pp. 565–577
- Nelson, 1944 N. Nelson; A photometric adaptation of the Somogyi method of determination of glucose; J. Biol. Chem., 153 (1944), pp. 375–381
- Odintsova et al., 2006 N. Odintsova, V. Dyachuk, K. Kiselev, N. Shelud'ko; Expression of thick filament proteins during ontogenesis of the mussel Mytilus trossulus (Mollusca: Bivalvia) ; Comp. Biochem. Physiol. B, 144 (2006), pp. 238–244
- Prins et al., 1991 T.C. Prins, A.C. Small, A.J. Pouwer; Selective ingestion of phytoplankton by the bivalves Mytilus edulis L., and Cerastoderma edule (L.) ; Hydrobiol. Bull., 25 (1991), pp. 93–100
- Roditi et al., 2000 H.A. Roditi, N.S. Fisher, S.A. Sando-Wilhelmy; Uptake of dissolved organic carbon and trace elements by zebra mussels; Nature, 407 (2000), pp. 78–80
- Safi and Gibbs, 2003 K.A. Safi, M.M. Gibbs; The importance of different size classes of phytoplankton in Beatrix Bay, Pelorus Sound and the potential implications for the aquaculture of the mussel Perna canaliculus; N. Z. J. Mar. Freshw. Res., 37 (2003), pp. 267–272
- Seed, 1976 R. Seed; Ecology; Marine mussels: their ecology and physiology (1976), pp. 13–65 (Lond. etc.)
- Seed and Suchanek, 1992 R. Seed, T.H. Suchanek; Population and community ecology of Mytilus; E.M. Gosling (Ed.), The mussel Mytilus : ecology, physiology, genetics and culture, Elsevier, Amsterdam (1992), pp. 87–157
- Selina, 1992 M.S. Selina; Phytoplankton in the area of a mussel farm in Vostok Bay of the Sea of Japan; Russ. J. Mar. Biol., 5–6 (1992), pp. 15–24
- Shumway et al., 1985 S. Shumway, T. Cucci, R. Newell, C. Yentsch; Particle selection, ingestion, and absorption in filter-feeding bivalves; J. Exp. Mar. Biol. Ecol., 91 (1985), pp. 77–92
- Skarlato, 1981 O.A. Skarlato; Bivalve Mollusks of the Temperate Latitudes of Western Pacific; Nauka, Leningrad (1981), pp. 1–479
- Somogyi, 1952 M. Somogyi; Estimation of sugars by colorimetric method; J. Biol. Chem., 200 (1952), p. 245
- Sova et al., 1970 V.V. Sova, L.A. Elyakova, V.E. Vaskovsky; The distribution of laminarinases in marine invertebrates; Comp. Biochem. Physiol., 32B (1970), pp. 459–464
- Sova et al., 2013 V.V. Sova, M.S. Pesentseva, A.M. Zakharenko, S.N. Kovalchuk, T.N. Zvyagintseva; Glycosidases of marine organisms; Biochemistry, 78 (7) (2013), pp. 746–759
- Stonik and Orlova, 1998 I.V. Stonik, T.Y. Orlova; Summer-autumn phytoplankton in Amursky Bay, the Sea of Japan; Russ. J. Mar. Biol., 24 (1998), pp. 205–211
- Whyte, 1987 J.N.C. Whyte; Biochemical composition and energy content of six species of phytoplankton used in mariculture of bivalves; Aquaculture, 60 (1987), pp. 231–241
- Wong et al., 2003 W.H. Wong, J.S. Levinton, B.S. Twining, N.S. Fisher; Assimilation of micro- and mesozooplankton by zebra mussels: a demonstration of the food web link between zooplankton and benthic suspension feeders; Limnol. Oceanogr., 48 (2003), pp. 308–312
- Zolotarev, 1989 V.N. Zolotarev; Sclerochronology of Marine Bivalves; Naukova dumka, Kiev (1989), pp. 1–112
- Zvyagintseva et al., 1999 T.N. Zvyagintseva, N.M. Shevchenko, I.V. Popivnich, V.V. Isakov, A.S. Scobun, E.V. Sudukova, L.A. Elyakova; A new procedure for the separation of water-soluble polysaccharides from brown seaweeds; Carbohydr. Res., 322 (1999), pp. 32–39
Document information
Published on 27/03/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?