Abstract
The characteristics of four stages of demutational succession of a valley Korean pine–broadleaf forest are provided according to the parameters most vividly capturing the structure of the plant community and influencing the renewal and mycorrhization of Korean pine (Pinus koraiensis ) seedlings. It was found that Korean pine seedlings grow in a competitive environment on the fresh burned site; hence the mycorrhization occurs from specialized symbiotes that provide competitive advantages and adaptation to stress factors. Mycorrhiza forms the least successful in the 50–60-year-old larch–birch–spiraea association during the seral stages of succession. The 90–100-year-old birch–broadleaf association offers the most suitable soil and cenotic conditions for the development of Korean pine seedlings that can successfully generate mycorrhiza from both the spores present in the thick soil layer, and through mycelia of shared mycorrhizal networks of seral and primary (including Korean pine) tree species. A 230–250-year-old climax community comprises all patterns to ensure that pine seedlings encounter a fungal component; in this community the greatest abundance of species and a balanced composition of ectomycorrhizal fungi communities of Korean pine are observed.
Keywords
Pinus koraiensis ; Demutational succession ; Fires ; Cenopopulation ; Ectomycorrhiza ; Central Sikhote-Alin
Introduction
The increase of the number and the area of fires, both of man-made and natural origin, on the territory of one of the richest forest regions of Russia — the Far East leads to a sharp reduction of valuable forest ecosystems, including primary pine–broadleaf forests (Astafiev et al., 2004 and Sukhomlinov, 2006 ; Petropavlovsky and Astafiev, 2010 ). Natural regeneration of pine forests after the fires is a complex, lengthy and understudied process. So far data was obtained on demutational successions in pine forests after the fires, mainly concerning the changes in the composition and structure of communities, the early stages of succession, soil banks of seeds and morphogenesis of species (Kolesnikov, 1938 , Solovyov and Solodukhin, 1957 , Shemetova, 1970 , Komarova, 1986 , Komarova, 1992 and Komarova et al., 2008 ; Gromyko et al., 2010 ). In the study of primary forests restoration process it is important to understand the development of tree species dominant in the original community.
Korean pine (Pinus koraiensis Siebold et Zucc.) is an edificator of pine and pine–broadleaf forests within the boundaries of its range — south of Russian Far East, northeastern China, Korea, and Japanese Honshu island ( Koropachinsky and Vstovskaya, 2002 ). This is the key species in the organization and functioning of ecosystems and a valuable tree species for economic use. But at the same time, Korean pine is a species highly susceptible to fire, pine forests belonging to the second class of fire hazard (Gromyko, 2010 ). In order to understand and control the processes of reforestation after the fires one of the key objectives is to study the growth and dynamics of Korean pine and the factors determining the success of its restoration.
Korean pine, similar to all tree species of nemoral and boreal forests, forms an obligate symbiosis with ectomycorrhizal (ECM) fungi, which was revealed both in the study of ground variety of fungi by collection of fruit bodies (Vasilyeva, 1973 ; Vasilyeva, 1982 ), and in morphological–anatomical and molecular analysis of mycorrhizal endings (Zengpu et al., 1995 , LePage et al., 1997 , Choi et al., 2005 and Malysheva et al., 2014 ; Yamada et al., 2014 ). The symbiosis of representatives of genus Pinus and ECM fungi has formed over 50 million years ago (LePage et al., 1997 ). The symbiosis that appeared in different clades of plants can be seen as an adaptation to changes in climate towards pronounced seasonality and dryness (Wang and Qui, 2006 ). Thanks to mycorrhiza a plant benefits in availability of nutrients and resistance to pathogens, which is reflected in its growth and development (Smith and Read, 2008 ). In this regard, one of the most important factors in the study of regenerative successions of communities is the mycorrhization of seedlings of the host plant and its impact on the direction of development of the ecosystem ( Grogan et al., 2000 , Guariguata and Ostertag, 2001 , Nara, 2006 , Ishida et al., 2007 , Bent et al., 2011 and Peay et al., 2011 ).
The goal of our study is to examine the process of demutational succession after the fires in pine–broadleaf forests and features of formation of Korean pine ectomycorrhizas.
Among the main issues that we addressed in the research were the following: What changes occur in the process of succession of valley pine forest under different scenarios of its restoration after the fires? What components or features of the community may affect the mycorrhization of Korean pine seedlings? What sources of fungal inoculum are present in the community at different successional stages after different types of fires? Does the presence of mycorrhiza impact the success of regeneration of Korean pine?
Materials and Methods
Study Area
The research was carried out on the territory of the Sikhote-Alin Biosphere Reserve located in the southern part of the Russian Far East (44°49′13″–45°41′25″N, 135°48′46″–136°34′23″E), in the central part of the Sikhote-Alin mountain ridge. Climate-wise the Reserve is located in the Pacific region of the temperate zone of the Far East, in the coastal climate area (Vitvitskiy, 1961 ). The climate has a pronounced monsoon character, manifested in a sharp change of wind direction towards the opposite in winter and summer. The total annual temperature during the growing season is approx. 2000–2500 °C, frost-free season is 105–120 days long, growing season is about 160 days long, and total annual precipitation is about 700 mm. The major amount of precipitation (78–85%) falls during frost-free, wet season — from April to November. Climatic differences are mostly manifested on the eastern and western macroslopes of the Sikhote-Alin. The eastern macroslope is under the constant influence of the Sea of Japan and the Pacific Ocean, so its climate is characterized by high humidity and smoothness of most hydrothermal phenomena. The western slopes, being under constant “protection” of the mountain system of Sikhote-Alin, are isolated from the direct influence of the sea. The average monthly temperature in January on the eastern slopes is − 12.3 °C, while on the western slopes it reaches − 22.2 °C; the average temperature in July is + 15.6 °C and + 19.2 °C, respectively. A similar pattern is observed with respect to the annual precipitation: on the western slopes the average is 680.0 mm, while on the eastern ones — 818.7 mm (the calculation of average-multiannual indices was made according to data from meteorological stations Terney and Melnichnoe for the period from 1940 to 2013).
The soil cover of the Reserve is complex and highly diverse, characterized by latitude, altitude and expository bioclimatic changes and a variety of lithological and geomorphological conditions (Gracheva and Utenkova, 1982 ). Fires and post-fire successions, which manifest themselves differently in different ecosystems, also have an impact on soil formation, further complicating the composition and structure of soil (Shemetova, 1970 ; Sapozhnikov et al., 2001 ).
Humic-illuvial brown soils are widespread under pine–broadleaf forests in the study area. In the profile of these soils a 5–7 cm thick mixed coarse-humus and soft-humus layer gives place to brown or dark-brown loamy-detritus mass. The soils are acidic and unsaturated. The processes and inherent properties of cold alpha-humic and warm brown-soil formation processes are combined here (Gracheva, 2005 ).
The dominant formations in the Reserve are: pine–broadleaf, spruce–fir and oak forests covering 120.2, 94.7, and 44 thousand ha, respectively. Rather large areas of the forest-covered territory are located in fire sites (14.3 thous. ha), secondary small-leaf forests (32.4 thous. ha) and larch forests (45.4 thous. ha). There were 70 fires during the evaluation period from 1977 to 2013, and the area covered by the fire was more than 23.1 thous. ha. Korean pine is one of the main forest-forming species of the Reserve, with the volume of 701 thousand m3 .
Geobotanical Description
The monitoring of the dynamics of primary and derivative communities is being carried out in the Sikhote-Alin Reserve since the 1950s (Anon, 1954 ). A significant expansion of the network of permanent sample plots was carried out in the 1970s and 1980s Flyagina (1982) . The study of successions of pine–broadleaf forests was carried out mainly by selecting different age stages in similar conditions and modeling a development series of a community (Shemetova, 1970 ; Flyagina, 1982 ). The first study area for the monitoring of the regeneration of pine forest after the fires was organized in 1978. Several more study areas were added subsequently in connection with the problems of forest fires on the territory of the Russian Federation reserves (Kuleshova and Korotkov, 1998 and Kuleshova and Korotkov, 2002 ).
Based on the results of long-term studies of regenerative successions in the Reserve (Petropavlovsky and Astafiev, 2010 ), a successional series in a valley pine–broadleaf forest with several regeneration scenarios was selected for this project. In order to assess the successional processes we made observations at permanent sample plots (PSP), with the dimensions of 50 × 50 m and 50 × 100 m in the primary community and late successional stages, and transects of 50 × 10 m and 50 × 1 m on fresh burned sites. Additional 20 × 20 m sample plots were established in August 2013 for the sampling of root tips of Korean pine and study of the patterns of mycorrhization of seedlings.
The first sample plot (PSP 1) is located in Ust–Shanduy area at the territory of a 10-year long restoration after a steady ground fire in a forest with linden, yellow birch and dark coniferous species (45°19.642′ N, 136°29.629′ E, 267 m above sea level). The second sample plot (PSP 2) is located within the primary valley pine forest with linden, yellow birch and dark coniferous species, hazel and mock orange, miscellaneous herbs and sedges, not affected by the fires and selected for control (the plot is located at a distance of 50 m from PSP 1, 45°19.666′ N, 136°29.650′ E, 264 m above sea level). Two more sample plots of the same size were established in Yasnaya area tract within two plant associations of the same successional series, at the stages of 50–60 years and 90–100 years old recovery after the fire: a birch and broadleaf species sample plot with pine, mixed shrubs and sedges (PSP 3 45°14.088′ N, 136°30.508′ E, 154 m above sea level), and a birch–larch–spirea sample plot with sedges and ferns (PSP 4, 45°14.350′ N, 136°30.350′ E, 158 m above sea level).
Geobotanical descriptions of vegetation were made for all sample plots with full enumeration of components of the communities: tree layers, undergrowth, shrub and grass canopies. The quantitative characteristics of the stand were measured in the full-sized sample plots; the enumeration of undergrowth, shrub and grass layers was done on 5 × 5 m sites and 50 × 1 m transects. A detailed description of associations is presented in Table 1 .
| Association | Succession stage (in: Sukachev, 1938 ) | Age of community, years | The depth of the soil layer to the mineral level, cm | Store of litter, tones per ha | Composition of the stand | Composition of regrowth | The quantity of Korean pine regrowth, ind. per ha | The quantity of undergrowth, ind. per ha | Herb canopy, % | The variety of vascular plants, number of species per 100 m2 | The quantity of registered OTU mycorrhizal fungi |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Fresh burned site in a valley pine forest (PSP 1) | Closed forming phytocenosis | 10 | 21 | 24.1 | Un. Ta | 3Pk3Bc2Ta1Am1(At + Pa) | 2800 | 28,400 | 100 | 37 | 21 |
| Valley pine forest with linden, yellow birch, hazel and mock orange (PSP 2) | Closed constant (primary) phytocenosis | 230–250 | 18 | 19.1 | 4Pk3Ta2Bc1Ulun.Am | 3Au2Pk2Am1An1At1Pav un.Bc,Ta,Fm,La | 2200 | 71,600 | 90 | 40 | 18 |
| Birch–broadleaved with pine, misc. herbs and sedges (PSP 3) | Closed constant phytocenosis | 90–100 | 23 | 11.9 | 2Bp2Qm2Pk1Ta1Am + Bc,Pa,Ld,Pt un.La | 5At2Pk1An1Am1Pa + Pt un.Au | 2700 | 61,600 | 20 | 28 | 23 |
| Birch–larch–spirea with sedges and ferns (PSP 4) | Closed constant phytocenosis | 50–60 | 16 | 14.2 | 4Ld2Bp2Qm2Ta un.Am,La | 5Pmax4Am1Pk un.La | 600 | 29,200 | 80 | 29 | 11 |
Fn: Am — Acer mono ; At — Acer tegmentosum ; Au — Acer ukurunduense ; An — Abies nephrolepis ; Bc — Betula costata ; Bp — Betula platyphylla ; Fm — Fraxinus mandshurica ; La — Ligustrina amurensis ; Ld — Larix dahurica ; Qm — Quercus mongolica ; Ta — Tilia amurensis ; Pa — Picea ajanensis ; Pk — Pinus koraiensis ; Ul — Ulmus laciniata ; Pav — Padus avium ; Pm — Padus maackii ; Pmax — Padus maximowiczii ; Pt — Populus tremula .
The age range of Korean pine in the described area was from 1 to 250 years; the absolute age was taken into account, without reference to ontogenetic stages. We singled out the following provisional classes of the absolute age of undergrowth and mature trees: 1–5 years, 7–20 years, 40–70 years, 120–150 years, and > 200 years.
A procedure for collection of soil samples and study of ectomycorrhizal fungi with molecular-genetic methods used in our research is described in detail earlier Malysheva et al. (2014) .
Data Processing and Statistical Analysis
In order to address the stated issues, we evaluated such parameters as the store of soil litter, the store of soil layer, the presence or absence of mature living pine trees and trees of other species in the stand, the abundance of undergrowth and richness of shrub and herbaceous species, and the number of Korean pine by age categories. For comparative characteristics of successional stages we examined in detail cenopopulation groups of plant species (Korchagin, 1964 ) and their role in the formation of uneven-aged communities, as well as the structure of these plant groups, taking into account their ecological and cenotic affiliation, life form and geographical range. In order to select the cenopopulation classes we used the concept of initial, seral and climax species Clements (1928) . The calculation of the composition of elements of different classes was conducted using data obtained in the Southern Sikhote Alin (Komarova, 1986 , Komarova, 1989 and Komarova, 1992 ; Komarova et al., 2008 ), and with the results of our own research (Gromyko et al., 2010 ). To analyze the ecological and cenotic structure we used the introduced classification Smirnova (1990) with amendments (Pimenova, 2005a ). Geographical groups were singled out using chorological elements of the flora (Pimenova, 2005b ). Eco-bio-morphological analysis was performed using the existing classification of seed plants Bezdelev and Bezdeleva (2006) .
For comparison of successional stages we evaluated the species richness, diversity and common elements in the four described communities of valley pine forest at different stages of the age range, based on the slice of data collected in August 2013. To assess diversity we used four indices calculated using a variety of approaches: the Shannon diversity index based on the relative abundance of species; Jacknife2 and Chao 2 indices based on the analysis of rare species (registered once or twice); and ICE (incidence-based coverage estimator) calculated with species found in ≤ 10 sampling units (Chazdon et al., 1998 ; Colwell, 2013 ). The number of randomizations for the calculation of the indices was 50. In order to assess the similarity of species we used Jaccard and Sørensen indices. The indices were calculated in EstimateS software, version 9.1.0 (Colwell, 2013 ; Colwell and Elsensohn, 2014 ) available at: http://purl.oclc.org/estimates .
Results and Discussion
In order to characterize the succession periods we grouped the species into three cenopopulation classes: initial (pioneer), seral and climax (indigenous) ones. The initial cenopopulation class comprises of pioneers growing on fire sites, reaching maximum abundance in the first year after the fire. Exposure to fire initiates mass germination of their seeds, dormant in the soil. This class includes herb (Chelidonium asiaticum , Corydalis pallida , Chamerion angustifolium ) and shrub (Sambucus racemosa ) species. The seral cenopopulation class includes species massively developing 3–5 years after the fire and providing a series of transitional stages of development of pre-fire community. In the presence of repeated fires they form long-standing derivative secondary forests. This group includes mostly tree (Salix caprea , Betula platyphylla , Populus tremula, Larix dahurica ) and shrub (Lespedeza bicolor ) species. The climax cenopopulation class includes species of primary communities most typical historically for this type of natural and geographical conditions. These species are present in all successional stages with varied abundance. Tree and shrub representatives of this cenopopulation class (P. koraiensis , Picea ajanensis and Acer mono ) appear on fire sites on the 3rd–5th year after the fire, and in the absence of subsequent fires reach maximum abundance in 20–50 year-old communities.
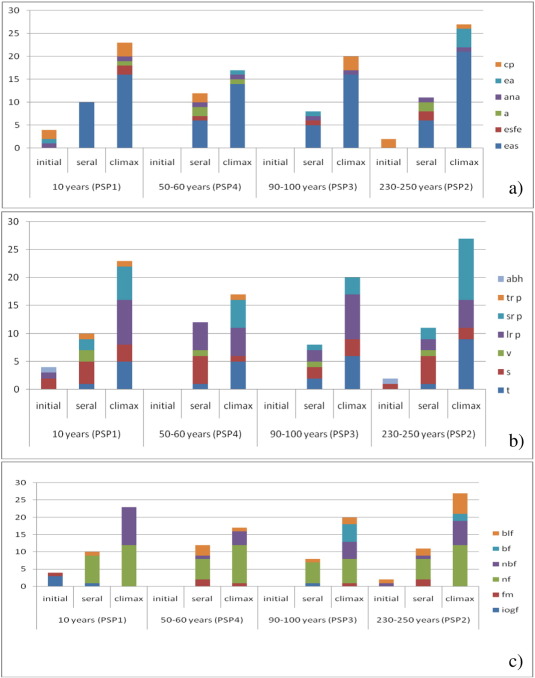
The research of demutational changes after the fires and identification of the impact of ECM fungi communities on the regeneration of Korean pine was carried out on a model succession series in the valley Korean pine forest with hazel and mock orange. With the results of long-term observations we created the age range of a community with two development scenarios depending on the type of fire. The first stage in the selected range is a fresh 10-year-old burned site. A steady ground fire which happened in the primary Korean pine forest in 2003 led to almost total destruction of all layers of vegetation and scorching of soil litter, especially in the areas of its aggregation. Accumulative humus layers of brown and soddy soil were slightly damaged by fire, burning only together with deadwood. Five years after the fire a new 1–3 cm thick soil litter is formed, but up to 90% of its fractional composition consists of branches and bark of dead trees, with the volume of 26.1 tons per ha. By the age of 10, the volume decreased slightly and amounted to 24.1 tons per ha, which exceeds the amount of soil litter compared to later stages in the successional series (Table 1 ). Individual Tilia amurensis trees, 35–40 cm in diameter, and A. mono trees are present in the tree canopy. Besides the deciduous species, solitary Korean pine trees remained on the borders of burned forest. The presence of mature generative individuals of Korean pine in the vicinity of fire site during seed years after the fire (in 2004 the seed yield reached 4 points) provided good sowing of the territory. Seedlings of Korean pine of the first two age groups (1–5 years and 7–10 years) reached 2800 individuals per ha. Abelia coreana , Ribes maximoviczianum , and Rubus sachalinensis form closed groups in the undergrowth. Among the large shrubs Philadelphus tenuifolius and Corylus mandshurica dominate. The sprouts of vines of Actinidia kolomikta and Vitis amurensis are distributed evenly. Grass cover reaches 100% mainly due to the growth of nemoral-forest sedges and ferns. Cenopopulation structure analysis shows that the group of initial species most active in the early years after the fire constitutes only 10% of the total number of species, while seral and climax cenopopulations reach considerable diversity ( Fig. 1 ). Initial cenopopulations are represented by annual and biennial herbs and shrubs with circumpolar, Eurasian or Asia–North American range. Seral cenopopulations are formed predominantly by woody species widespread in East Asia. Cenopopulations of primary species exceed seral ones twofold; they are composed of main forest-forming species that were present in the pre-fire community, and related species of shrubs and herbaceous perennial plants. The variety of vascular plants registered in the field studies was 37 species per 100 m2 . The Shannon index value of over two reflects high species diversity on the fresh 10-year-old burned site (Table 2 ). However, indices aimed at identifying the true diversity (Jacknife 2, Chao 2, ICE) show the undercount of species on the fresh burned site by 20–30%. The ecosystem after the fire is open to the regeneration of the representatives of all cenopopulation classes, and the diversity reaches the highest values by the 10th year. In this case, according to the values of valuation indices, combinations of 54 species per 100 m2 are possible.
|
|
|
Fig. 1. The distribution of eco-cenotic (a), eco-bio-morphologic (b), and chorologic (c) elements in cenopopulation classes at different stages of post-fire succession of the valley pine forest. Legend: blf — bottom-land forest; bf — boreal forest; nbf — nemoral boreal forest; nf — nemoral forest; fm — forest–meadow; iogf — initial overgrowth forest; abh — annual and biennial herbs; tr p — tap root polycarpics; sr p — short root polycarpics; lr p — long root polycarpics; v — vines; s — shrubs; t — trees; cp — circumpolar; ea. — Eurasian; ana — Asia–North American; a — Asian; esfe — East Siberian — Far Eastern; eas — East Asian. |
| Age stage | Sobs | Shannon | Jack 2 | Chao 2 | ICE |
|---|---|---|---|---|---|
| 10 years (PSP 1) | 37 | 2.42 | 53.3 | 46.0 | 53.8 |
| 50–60 years (PSP 4) | 29 | 1.80 | 35.7 | 30.9 | 34.1 |
| 90–100 years (PSP 3) | 28 | 2.66 | 35.1 | 30.6 | 32.6 |
| 230–250 years (PSP 2) | 40 | 3.18 | 46.9 | 41.9 | 45.4 |
Under what conditions does Korean pine appear, grow and develop; and ECM fungal communities obligately connected to it are formed? During the succession stage described above the regrowth of Korean pine is in the conditions of disturbed primary community without tree canopy, with formed layer of seral species regrowth, with lush undergrowth and thick ground cover. There is a high diversity of species of vascular plants, different in composition from the subsequent successional stages. Phytocenosis is formed on low-disturbed soil under conditions of high concentrations of organic matter as soil litter. The regrowth of only two age categories of Korean pine (1–5 years old and 7–10 years old) is present on the fire site, while seedstalking trees have remained at its border. Under the new conditions initiated by fire a certain fungal community is formed which may affect the growth processes and lead to a shift in succession by the symbiosis with particular species or a number of species, as shown by the studies (Visser, 1995 and Nara, 2006 ; Bent et al., 2011 ). Mycorrhization of pine seedlings is possible both with fungal spores resistant to fire and from the surviving climax species of trees that are able to develop shared mycorrhizal networks. However, mycorrhization in the early stages of succession takes place in the conditions of high competition both for ground and soil space, with a large number of plants in need of fungal component for nutrition. As shown in our earlier studies (Malysheva et al., 2014 ), in this area the seedlings are colonized mainly by specialized symbiotes (species of the genus Suillus ) that can form mycorrhiza out of spores. The specialization of fungal symbiote provides young pine regrowth with a competitive advantage for the sources of nitrogen and phosphorus and contributes to avoiding optional epiparasitism in the early stages of development of woody plants. In addition, ectomycorrhizal symbiote Cenococcum geophilum was found in this area, which allows the seedlings to adapt successfully to stressful environmental factors and plays an important role in the regeneration of pine forests ( Ashkannejhad and Horton, 2006 ; Matsuda et al., 2009 ). In this successional stage 21 operational taxonomic units (OTU) of ectomycorrhizal fungi were identified. Their distribution in the soil mass showed their association to the upper layers, including the semi-decomposed litter. In summary, ECM fungal community on early stages fully satisfies the prevailing soil and cenotic conditions, in which the Korean pine seedlings develop.
In 10 years after the fire the regrowth starts to close and thin out, and in 20 years a part of large regrowth of fast-growing species (seed-bearing Betula platyphylla , P. tremula , S. caprea and sprouting T. amurensis ) goes into the stand, reaching a height of 6–7 m and a diameter of 6–7 cm (Gromyko et al., 2010 ). In 20 years after the fire shrubs in the closed layer amount to 67.6 thousand shoots per ha, and the majority of the species reach their maximum abundance. Among the shrubs R. maximoviczianum , A. coreana and C. mandshurica dominate. The thick ground cover is dominated by sedges, Thalictrum tuberiferum , Lathyrus humilis , Galium davuricum ; and among the ferns — nemoral boreal species Pseudocystopteris spinulosa .
Subsequently, in 50–60 years after the fire, depending on the degree of burning-out and the presence of areas unaffected by the fire nearby in the pine forest, the succession process goes in several directions. In our model series a birch–larch–spirea association with sedge and fern (PSP 4) is an example of the development of a community with completely destroyed primary tree stand, located at a distance from the primary forest. The stand is dominated by seral species: Larix dahurica and Betula platyphylla with a diameter of 30–35 cm. A significant part of primary tree species consists of T. amurensis and A. mono . Coniferous species are represented poorly. Pine regrowth is present in small quantities ( Table. 1 ). The undergrowth is dominated by seral species as well: Lonicera chrysantha and Spiraea flexuosa . The herbaceous canopy is less diverse compared to the primary community, covering up to 80% of the territory with the dominance of Maianthemum dilatatum and nemoral sedges. At this stage initial cenopopulations are absent, and the diversity of seral and climax groups is approximately equal. Seral cenopopulations consist of nemoral-forest and bottom-land forest species, with significant participation of forest–meadow flora ( Fig. 1 ). Shrubs are the dominant group among the life forms, which is typical for the seral cenopopulations class in general.
In the areas where the destruction of the tree stand was gradual due to the ground fire, intensive reforestation with pine, spruce and fir began in the first years after the fire and continued throughout the period of succession. The case under study represents another pattern of primary forest development as exemplified by 90–100 years old birch–broadleaf association with pine, miscellaneous herbs and sedges (PSP 3). Thickness of the soil in this community is maximal, and the stock of ground litter is minimal (Table 1 ). Pine regrowth is abundant and multiple-aged. The undergrowth is dominated by species typical of primary pine forests: C. mandshurica , R. maximoviczianum , Euonymus pauciflora etc. Ground cover is underdeveloped, dominated by ferns and short grasses typical of coniferous forests. Initial cenopopulations are absent at this stage, while seral ones are represented mainly by tree species (Betula platyphylla , P. tremula ), their life cycle near completion. Climax species dominate more than twofold over the seral ones ( Fig. 1 ).
In the intermediate stages of regenerative succession described above seral and climax cenopopulation groups are the leading ones. The complex of seral cenopopulations is the most complicated, since assigning a number of species to this group may have ambiguous interpretation, as in the case of Aralia elata or C. mandshurica . However, information on the successional status of a number of the seral tree cenopopulations (in the studied demutational series these are Betula platyphylla , Larix dahurica , P. tremula ) and their role as intermediate dominants in communities may be found in the works of other researchers ( Komarova, 1992 ; Komarova et al., 2008 ). Seral cenopopulations are most often composed of species with hard-to-define eco-cenotic status. Typically, they are considered nemoral-forest species, or forest–meadow ones, or accompanying plants to larch, birch or oak. Shannon index, based on the assessment of all the species present in the community, shows rather high species diversity for the late 90–100-year-old successional stage and the lowest diversity for the 50–60-year-old birch–larch–spirea association (Table 2 ). Jacknife 2, Chao 2 and ICE indices show roughly the same undercount of species in seral stages — by 10–20%.
The estimations of the similarity of species diversity (Jaccard and Sørensen indices) showed the greatest similarity of the later seral stages. There are significant differences between succession stages separated from each other by large time intervals (Table 3 ). This contrast of the species composition on multiple-aged burned sites was identified in the study of post-fire succession of larch forests (Lytkina and Mironova, 2009 ).
| 10 years (PSP 1) | 50–60 years (PSP 4) | 90–100 years (PSP 3) | 230–250 years (PSP 2) | |
|---|---|---|---|---|
| 10 years (PSP 1) | 1.000 | 0.333 | 0.308 | 0.338 |
| 50–60 years (PSP 4) | 0.200 | 1.000 | 0.456 | 0.319 |
| 90–100 years (PSP 3) | 0.182 | 0.295 | 1.000 | 0.324 |
| 230–250 years (PSP 2) | 0.203 | 0.190 | 0.193 | 1.000 |
The development of cenopopulations of Korean pine occurs in different ways during the described late-succession stages characterized by the formation of the tree canopy. In the 50–60-year-old larch–birch–spirea association formed after a major fire under the circumstances of the complete destruction of primary phytocenosis and the absence of generative Korean pine trees nearby, the number of Korean pine regrowth is only 600 individuals per ha. The tree canopy of the first layer is formed by seral cenopopulations, the primary deciduous species are present only in the subordinate canopy, undergrowth is quite thick, and ground cover is up to 100% of the cover. The soil layer is thin in this community, and the store of soil litter is low. Species diversity is small, seral cenopopulations still retaining some activity. These conditions are least favorable for successful mycorrhization and development of Korean pine. Pine seedlings can obtain fungal inoculum only through the spores of a limited group of ECM fungi or mycelium of shared mycorrhizal networks of seral leaf species. The competition for thin soil layer and ground space is quite high. The least number of ECM fungi in Korean pine was found in this association — 11 OTU (Malysheva et al., 2014 ).
The formation of a new pine cenopopulation is rather intense in another line of the successional series with the gradual destruction of the burned primary forest stand and the close proximity of generative individuals of Korean pine, which can be observed on the 90–100-year-old burned site. This site features multiple-aged regrowth of Korean pine, a part of which has already entered the stand and is capable of having seeds. The tree canopy is complex, with the dominance of seral species in the first layer, and primary cenopopulations — in the lower tree layer. The undergrowth is dense, and the ground cover in coniferous parcels is hardly expressed. The soil layer is quite thick, and the store of soil litter is low. Species diversity of vascular plants decreases at this stage due to the loss of initial species and the reduction of diversity of the seral ones. In our opinion, these are the most suitable conditions for the development of Korean pine seedlings that can successfully generate mycorrhiza from both the spores present in the thick soil layer, and through mycelia of shared mycorrhizal networks of seral and primary (including Korean pine) tree species. The number and abundance of competing species are markedly decreased. For this association, the number of identified mycorrhizal fungi was the highest — 23 OTU (Malysheva et al., 2014 ), which confirms the conclusions drawn on the basis of the characteristics of the soil and cenotic conditions of this successional stage.
As a model of the primary (initial and final) community we consider valley pine forest with linden, yellow birch, dark coniferous species, hazel and mock orange, miscellaneous herbs and sedges. The stand of phytocenosis is characterized as complex, three-layer, with the dominance of Korean pine aged 230–250 years, 50–60 cm in diameter. The regrowth of pine is small in number, up to 2200 individuals per ha (Table 1 ). The age range of Korean pine is represented by all stages. The primary community is the most balanced with regard to the composition of cenopopulations, and all three groups are represented at a ratio of 1:6:13. Climax cenopopulations reach maximum diversity here, which indicates the stability of the community. Their composition is dominated by tree species and polycarpic short root grasses belonging to nemoral forest, boreal forest and bottom-land forest eco-cenotic groups and East Asian chorologic group, which is typical in general for indigenous flora (Pimenova, 2005b ). The greatest diversity of vascular plants (40 species per 100 m2 ) is found in the primary community, which is indicated by the high value of the Shannon diversity index (3.18). According to the evaluation using Jacknife 2, Chao 2 and ICE indices, the undercount of species was only 5–15% (Table 2 ). Given the specifics of calculating the indices, we can assume that rare species with low abundance and occasional occurrence are undercounted.
The successful regeneration of Korean pine, and thus, the formation of mycorrhiza with suitable fungi are already rooted in a 230–250 year old climax community with the balanced structure and composition of all components and established mechanisms of maintaining a sustainable balance. A primary community comprises all patterns to ensure that pine seedlings encounter a fungal component: domination of Korean pine in the stand, universal age structure of pine cenopopulation, high diversity of plant species mostly of the climax class of cenopopulations, sufficiently thick soil layer undisturbed by fire, and regular flow of organic matter in the form of leaf fall and litter. The development of pine regrowth in a primary community occurs mainly in the gaps of the canopy where other species may provide competition at the initial stages, but pine receives benefits in gaining the ground in case of successful mycorrhization. A total of 18 taxa of ECM fungi (Malysheva et al., 2014 ) were identified for this phytocenosis, some of them appearing only in the primary community.
The distribution of mycorrhizal endings of pine in all soil layers, including the deeper ones, and the presence of specialized groups of fungi in relation to organic substrates from which they can extract nitrogen, lead to the greatest abundance of species and balanced composition of ectomycorrhizal fungi communities of Korean pine in the primary phytocenosis.
Acknowledgment
The research was supported by the Russian Foundation for Basic Research (grants no. 13-04-00110 , no. 14-04-10040 ).
References
- Anon, 1954 Anon; The Draft Long-term Plan of Forestry Management in the Sikhote-Alin State Reserve; Explanatory Note., Vol. I. (1954) L. (268 pp. in Russian)
- Petropavlovsky and Astafiev, 2010 B.S. Petropavlovsky, A.A. Astafiev (Eds.), Fires and Their Impact Influence on the Natural Ecosystems of the Central Sikhote-Alin, Dalnauka, Vladivostok (2010) (264 pp., in Russian)
- Ashkannejhad and Horton, 2006 S. Ashkannejhad, T.R. Horton; Ectomycorrhizal ecology under primary succession on coastal sand dunes: interactions involving Pinus contorta , suilloid fungi and deer ; New Phytol., 169 (2006), pp. 345–354
- Astafiev et al., 2004 A.A. Astafiev, Y.M. Dunishenko, V.N. Dyukarev, D.F. Efremov, V.G. Kryukov, A.N. Kulikov, V.A. Kulikov, N.A. Marchenko, V.P. Negodiaev, G.I. Sukhomirov, V.I. Trush, V.A. Chelyshev, A.S. Sheyngauz, M.A. Sheshukov; Fire Management in Biologically Valuable Forests of the Amur–Sikhote-Alin Eco-region; Scientific and Technical Basis of the Project, ZHASO-Amur, Khabarovsk (2004) (130 pp., in Russian)
- Bent et al., 2011 E. Bent, P. Kiekel, R. Brenton, D.L. Taylor; Root-associated ectomycorrhizal fungi shared by various boreal forest seedlings naturally regenerating after a fire in interior Alaska and correlation of different fungi with host growth responses; Appl. Environ. Microbiol., 77 (10) (2011), pp. 3351–3359
- Bezdelev and Bezdeleva, 2006 A.B. Bezdelev, T.A. Bezdeleva; Life Forms of Seed Plants of the Russian Far East; Vladivostok, Dalnauka (2006) (296 pp., in Russian)
- Chazdon et al., 1998 R.L. Chazdon, R.K. Colwell, J.S. Denslow, M.R. Guariguata; Statistical Methods for Estimating Species Richness of Woody Regeneration in Primary and Secondary Rain Forests of NE Costa Rica; F. Dallmeier, J.A. Comiskey (Eds.), Forest Biodiversity Research, Monitoring and Modeling: Conceptual Background and Old World Case Studies, Parthenon Publishing, Paris (1998), pp. 285–309
- Choi et al., 2005 D.S. Choi, M. Kayama, D.J. Chung, H.O. Jin, A.M. Quoreshi, Y. Maruyama, T. Koike; Mycorrhizal activities in Pinus densiflora , P. koraiensis and Larix kaempferi native to Korea raised under high CO2 concentrations and water use efficiency ; Phyton. Horn., 45 (4) (2005), pp. 139–144
- Clements, 1928 F.E. Clements; Plant Succession and Indicators; Wilson, N.Y. (1928) (452 pp.)
- Colwell, 2013 R.K. Colwell; EstimateS: statistical estimation of species richness and shared species from samples. Version 9. Users guide and application at; http://purl.oclc.org/estimates (2013)
- Colwell and Elsensohn, 2014 R.K. Colwell, J.E. Elsensohn; EstimateS turns 20: statistical estimation of species richness and shared species from samples, with non-parametric extrapolation; Ecography, 37 (2014), pp. 609–613
- Flyagina, 1982 I.A. Flyagina; Reforestation of Pine Forests on the Eastern Slopes of the Sikhote-Alin; Far Eastern Scientific Center of the USSR Academy of Sciences, Vladivostok (1982) (180 pp., in Russian)
- Gracheva, 2005 R.G. Gracheva; The Soil Cover//Structural Organization and Dynamics of the Natural Complexes of the Sikhote-Alin Biosphere Reserve; OAO Primpoligrafkombinat, Vladivostok (2005), pp. 5–21 (in Russian)
- Gracheva and Utenkova, 1982 R.G. Gracheva, A.P. Utenkova; Soils; Flora and Fauna of the Sikhote-Alin Reserve. M. (1982), pp. 22–27 (in Russian).
- Grogan et al., 2000 P. Grogan, J. Baar, T.D. Bruns; Below-ground ectomycorrhizal community structure in a recently burnet bishop pine forest; J. Ecol., 88 (2000), pp. 1051–1062
- Gromyko, 2010 M.N. Gromyko; The Characteristics of Ecosystems in Relation to Forest Fires; Fires and Their Impact Influence on the Natural Ecosystems of the Central Sikhote-Alin, Dalnauka, Vladivostok (2010), pp. 86–103 (in Russian)
- Gromyko et al., 2010 M.N. Gromyko, E.A. Pimenova, E.A. Smirnova; Post-fire Successions in Pine and Pine–Broadleaf Forests; Fires and Their Impact Influence on the Natural Ecosystems of the Central Sikhote-Alin, Dalnauka, Vladivostok (2010), pp. 121–173 (in Russian)
- Guariguata and Ostertag, 2001 M.R. Guariguata, R. Ostertag; Neotropical secondary forest succession: chances in structural and functional characteristics; For. Ecol. Manag., 148 (2001), pp. 185–206
- Ishida et al., 2007 T.A. Ishida, K. Nara, T. Hogetsu; Host effects on ectomycorrhizal fungal communities: insight from eight host species in mixed conifer–broadleaf forests; New Phytol., 174 (2007), pp. 430–440
- Kolesnikov, 1938 B.P. Kolesnikov; The Vegetation of the Eastern Slopes of the Central Sikhote-Alin; Transactions of the Sikhote-Alin Reserve. M., Vol. 1 (1938), pp. 25–207 (in Russian).
- Komarova, 1986 T.A. Komarova; Seed Renewal on Fresh Burned Areas (Forests of the Southern Sikhote-Alin); FESC, Vladivostok (1986) (222 pp., in Russian)
- Komarova, 1989 T.A. Komarova; Seed Renewal of Plants on Fresh Burned Areas of the Southern Sikhote-Alin; For. Sci. (2) (1989), pp. 51–59 (in Russian)
- Komarova, 1992 T.A. Komarova; Post-fire Successions in the Forests of the Southern Sikhote-Alin; Dalnauka, Vladivostok (1992) (224 pp., in Russian)
- Komarova et al., 2008 T.A. Komarova, L.A. Sibirina, D.K. Lee, H.S. Kang; Demutational successions after fires in the broadleaf and pine forests with vines and mixed shrubs of the southern Sikhote-Alin; For. Sci. (4) (2008), pp. 10–19 (in Russian)
- Korchagin, 1964 A.A. Korchagin; Intraspecific (Population) Composition of Plant Communities and Methods of its Study; Field Geobotany, Vol. 3, M., L.: Publishing House of the USSR Academy of Sciences (1964), pp. 63–125 (in Russian)
- Koropachinsky and Vstovskaya, 2002 I.Y. Koropachinsky, T.N. Vstovskaya; Woody Plants of the Asian Russia; Publishing House of SB RAS, Geo subsidiary, Novosibirsk (2002) (707 pp., in Russian)
- Kuleshova and Korotkov, 1998 L.V. Kuleshova, V.N. Korotkov; Fires in the Reserves of the Russian Federation: The Long-term Dynamics and Geographic Characteristics; Anthropogenic Impacts on Natural Complexes of Reserves, Problems of Reserve Management, Vol. 9. (1998), pp. 4–36 M., (in Russian)
- Kuleshova and Korotkov, 2002 L.V. Kuleshova, V.N. Korotkov; On the Strategy of Forest Fires Management on the Territories of Reserves; Monitoring of Communities in the Burned Areas and Fire Management in the Reserves. M. (2002), pp. 158–173 (in Russian).
- LePage et al., 1997 B.A. LePage, R.S. Currah, R.A. Stockey, G.W. Rothwell; Fossil ectomycorrhizae from the middle Eocene; Am. J. Bot., 84 (1997), pp. 410–412
- Lytkina and Mironova, 2009 L.P. Lytkina, S.I. Mironova; Post-fire succession in the forests of the cryolithic zone (through the example of Central Yakutia); Ecology (3) (2009), pp. 168–173 (in Russian)
- Malysheva et al., 2014 V.F. Malysheva, E.F. Malysheva, A.E. Kovalenko, E.A. Pimenova, M.N. Gromyko, S.N. Bondarchuk; Ectomycorrhizal symbiotes fungal diversity of Pinus koraiensis in the forests of the Central Sikhote-Alin, identified on the basis of the analysis of based on the rDNA sequence analysis of mycorrhizal endings tips ; Mycol. Phytopathol., 48 (6) (2014), pp. 372–385 (in Russian)
- Matsuda et al., 2009 Y. Matsuda, Y. Noguchi, S. Ito; Ectomycorrhizal fungal community of naturally regenerated Pinus thunbergii seedlings in a coastal pine forest ; J. For. Res., 14 (2009), pp. 335–341
- Nara, 2006 K. Nara; Ectomicorrhizal networks and seedling establishment during early primary succession; New Phytol., 169 (2006), pp. 169–178
- Peay et al., 2011 K.G. Peay, P.G. Kennedy, T.D. Bruns; Rethinking ectomycorrhizal succession: are root density and hyphal exploration types drivers of spatial and temporal zonation?; Fungal Ecol., 4 (2011), pp. 233–240
- Pimenova, 2005a Pimenova E.A. Ecological and Cenotic Structure of Flora of the Sikhote-Alin Reserve // Status of Protected Natural Areas: Proceedings of Research and Practical Conference Dedicated to the 70th Anniversary of the Lazovsky Nature Reserve, Lazo, April 19–20, 2005. Vladivostok: Russkiy Ostrov. 2005a. P. 138–142. (in Russian).
- Pimenova, 2005b E.A. Pimenova; Flora of the Sikhote-Alin Biosphere Reserve (Taxonomic Composition, Ecological and Geographical Characteristics, Conservation of Rare Plant Complexes): Dis. Cand. of Biol. Sci.; BSI FEB RAS, Vladivostok (2005) (462 pp., in Russian)
- Sapozhnikov et al., 2001 N.P. Sapozhnikov, L.O. Karpachevsky, L.S. Ilyina; Post-fire soil formation in pine–broadleaf forests; Forest Bull. (No. 1 (16)) (2001), pp. 132–164 (in Russian)
- Shemetova, 1970 N.S. Shemetova; Pine–Broadleaf Forests and Their Burned Sites on the Eastern Slopes of the Central Sikhote-Alin; (Vladivostok) (1970) (104 pp., in Russian)
- Smirnova, 1990 E.A. Smirnova; Ecological and Phyto-cenotic Characteristics of Derivative Forests of the Sikhote-Alin Reserve; Ecological Research in the Sikhote-Alin Reserve. (Features of the Ecosystems of Oak Forests Belt): Transactions of the Central Research Laboratory of Glavokhoty RSFSR. M. (1990), pp. 5–17 (in Russian).
- Smith and Read, 2008 S.E. Smith, D.J. Read; Mycorrhizal Symbiosis; (third ed.)San Diego, Calif (2008)
- Solovyov and Solodukhin, 1957 K.P. Solovyov, E.D. Solodukhin; Reforestation of Burned Areas in Coniferous–Broadleaf Forests of the Far East; Coll. of Papers on Forestry (1957), pp. 5–12 M., L. (in Russian)
- Sukachev, 1938 V.N. Sukachev; The Main Concepts of the Plant Cover Doctrine; Vegetation of the USSR, Vol. 1, Publishing House of the USSR Academy of Sciences (1938), pp. 15–37 M., L. (in Russian)
- Sukhomlinov, 2006 N.R. Sukhomlinov; Fires as a Factor in the Degradation of the Far Eastern Forests: Social–Historical View of the Problem; Forest Ecosystems of Northeast Asia and Their Dynamics: Transactions of Intern. Conf. Vladivostok: Dalnauka (2006), pp. 134–137 (in Russian)
- Vasilyeva, 1973 L.N. Vasilyeva; Agaric Pileate Fungi (Agaricales) of Primorsky Krai; L.: Nauka (1973) (331 pp., in Russian)
- Vasilyeva, 1982 L.N. Vasilyeva; Macromycetes; Flora and Fauna of the Sikhote-Alin Reserve, Nauka (1982), pp. 38–48 M. (in Russian)
- Visser, 1995 S. Visser; Ectomicorrhizal fungal succession in jack pine stands following wildfire; New Phytol., 129 (1995), pp. 389–401
- Vitvitskiy, 1961 G.N. Vitvitskiy; Climate; Far East. M., USSR Academy of Sciences (1961), pp. 93–117 (in Russian).
- Wang and Qui, 2006 B. Wang, Y.-L. Qui; Phylogenetic distribution and evolution of mycorrhizas in land plants; Micorrhiza, 16 (2006), pp. 299–363
- Yamada et al., 2014 A. Yamada, N. Endo, H. Murata, A. Ohta, M. Fukuda; Tricholoma matsutake Y1 strain associated with Pinus densiflora shows a gradient of in vitro ectomycorrhizal specificity with Pinaceae and oak hosts ; Mycoscience, 55 (1) (2014), pp. 27–34
- Zengpu et al., 1995 L. Zengpu, J. Junran, W. Chanweng; Antagonism Between Ectomycorrhizal Fungi and Plant Pathogens in Mycorrhizas for Plantation Forestry in Asia; Proceedings of an International Symposium and Workshop. Kaiping, Guangdong Province (1995), pp. 77–81
Document information
Published on 27/03/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?