Abstract
Trichophyton soudanense and Trichophyton mentagrophyte were isolated from the rice mill environment, cultured, and used singly and as co-culture in the treatment of measure quantities of pre-heated rice husk. Carboxymethyl cellulase activity of 57.61 μg/ml/min was optimum for T. mentagrophyte -treated rice husk crude enzymes at 50 °C and 80 °C, respectively. The duration of 120 h gave the highest CMCellulase activity of 75.84 μg/ml/min for crude enzyme of T. mentagrophyte heat-pretreated rice husk. The duration of 96 h gave maximum activity of 58.21 μg/ml/min for crude enzyme of T. soudanense heat-pretreated rice husk. Highest CMCellulase activities of 67.02 μg/ml/min and 69.02 μg/ml/min at pH of 5 were recorded for crude enzymes of monocultures of T. soudanense (TS) and T. mentagrophyte (TM) heat-pretreated rice husk. Heating before treatment with T. mentagrophyte gave 44.50 ± 10.90 cellulose as maximum, total lignin value of 28.90 ± 1.80 from co-culture of T. soudanense and T. mentagrophyte (TS + TM), hemicellulose content of 30.50 ± 2.12 (% ± SEM) from pre-heated rice husk treated with T. soudanense (TS), carbohydrate content of 16.79 ± 9.14, and reducing and non-reducing sugar values of 2.66 ± 0.45 and 14.13 ± 8.69 were all obtained from for pre- heated rice husk treated with T. mentagrophyte (TM). The pre-heated rice husk treated with T. mentagrophyte (TM) fermented with palm wine yeast gave bioethanol value of 11.11 ± 0.21 (% ± standard deviation) as the highest yield.
Keywords
Trichophyton soudanense ; Trichophyton mentagrophyte ; Biomass ; Bioethanol ; Rice husk
Introduction
The most readily available biomass apart from the forest flora are the agro-processed wastes. Rice husk is a great example of readily available but underutilized agricultural waste from processed rice grains. This waste is known to be a good source of lignocelluloses, which could be exploited for use in the commercial production of many specialty chemicals, cellulose, sugar, and ethanol. Due to the composition of its biomass, much research is currently ongoing in a bid to maximize its use, which has a twofold advantage viz: the production of useful substances and the amelioration of its dumps to reduce environmental occupation. According to Ezeonu et al. (2014) , the occupation of large land mass by agro-wastes has been a great environmental challenge of the 21st century. Thus, the need to utilize bioproducts from agricultural wastes stem from the fact that the growing population of the world could be better served when those by-products otherwise considered as wastes are reintegrated for the generation of new product as well as enhancement of greener environments. Balat et al. (2008) explained that lignocellulosic biomass is the largest source of hexose and pentose sugars, which can be used for the production of bioethanol. Despite this fact, Srivastava et al. (2014) explained that unlike first-generation biomass, in second-generation lignocellulosic substrates, cellulose in the cell wall is encased within hemicellulose and lignin matrix, and thus accessibility of cellulose is a major problem in bioethanol production from such sources. This in turn affects the cost of production of bioethanol from rice husk as well as other agro-processed wastes. Most fungi are capable cellulose degraders, and such fungi produce active polymer degrading enzymes, including cellulases and xylanases (Hodrova et al ., 1998 ; Malherbe and Cloete, 2002 ). Their cellulases are among the most active reported to date and able to solubilise both amorphous and crystalline cellulose (Wubah et al ., 1993 ; Malherbe and Cloete, 2002 ). The purpose of this research is to examine the ability of Trichophyton soudanense and Trichophyton mentagrophyte to optimize the release of biomass and production of bioethanol from rice husk.
Materials and Method
Plant Sample (Rice Husk)
Samples of fresh processed rice husks and 8 months decomposing rice husk were collected from Adani Rice Integrated Resources Nig. Ltd., Adani in Uzo-Uwani Local Government Area of Enugu State, Nigeria. All samples were kept in air tight cellophane bags before use.
Isolation of Fungi
Isolation of fungi from decomposing rice husk was carried out using potato dextrose agar (PDA) as the inoculation medium. The method used was the dilution plate technique.
Screening, Isolation, and Characterization of Fungi
Decomposing rice husk (1 g) was added to 9 ml of sterile distilled water in a beaker and mixed thoroughly. This served as the stock for the isolation of the fungi. The serial dilution of the sample was carried out by pipetting 1 ml of the stock solution into another 9 ml of distilled water. The sample suspension was further diluted to 10− 6 ml. From 10− 6 serial diluted (fungal sources) stock, 0.1 ml was pipetted into five different Petri dishes containing freshly prepared potato dextrose agar with inclusion of streptomycin/chloramphenicol at 50 °C on an alcohol sterilized bench. Spreading of inoculum was done by the pour plate method followed by gentle agitation to enable uniform spread. This was carried out using standard sterilization techniques in the presence of gentle Bunsen flame. The inoculated plates were incubated in a microbial laboratory incubator at room temperature of 38 ± 0.06 °C for 5 days. Growth was monitored daily and identification of the various fungal colonies carried out by microscopic cell observation, colony morphology, and biochemical tests. Characteristics such as size, surface appearance, texture, sporing structure, and color of the colonies (picture of the various colonies were also taken and compared with standards for identification) were carried out. Subcultures (3 times for each identified colony) from the various plates were carried out by aseptically transferring each independently identified colony isolate into other potato dextrose agar slants (containing antibiotics) until pure fungal strains were obtained (for any batch the incubation was at room temperature recorded for 5 days). Pure fungal isolates were stored in culture tubes plugged with cotton wool in a refrigerator at 4 °C for further use. The subsequent culturing of the identified pure fungal strains was carried out using PDA agar in Petri dishes with the inclusion of streptomycin/chloramphenicol after autoclave to be followed by incubation at room temperature for 6 days before use.
Preparation of Culturing and Fermentation Medium
For the cultivation of the fungi and hydrolysis of the rice husk, Mendle’s medium was prepared as reported by Patel et al. (2007) . One liter of Mandle’s medium consists of the following mineral salt ingredients: (NH4 )2 SO4 (1.4 g), KH2 PO4 (2.0 g), urea (0.3 g), CaCl2 ·2H2 O (0.4 g), MgSO4 ·7H2 0 (0.6 g), MnSO4 ·H2 O (1.0 mg), ZnSO4 7H2 O (1.4 mg), FeSO4 ·7H2 O (5.0 mg), CoCl2 ·6H2 O (3.7 mg), protease-peptone (0.75 g), and Tween 80 (2.0 mg). The medium (rice husk in Mandles medium) was sterilized at 121 °C for 20 min and the pH adjusted to 5.5.
Experimental Design for the Fungal Treatment of Rice Husk
The design of the experiment involved modification of the method reported by Patel et al. (2007) . Into each 500 ml conical flask used in the experiment, 20 g of rice husks were weighed (total of 5 samples) and 400 ml of Mandle’s medium introduced. Sterilization of the various conical flasks plugged with cotton and covered with aluminum foil was carried out using an autoclave at 121 °C for 20 min. The mixtures were allowed to cool and each conical flask except the controls (C1 = non-fungal but heat-treated sample; C2 = non-fungal, non-heat-treated sample) was inoculated with the fungi by addition of 10 ml of 0.1% Tween 80 into PDA Petri dishes of pure fungal isolates both as monoculture: T. soudanense (TS), T. mentagrophyte (TM) and co-culture (inoculates): combination of T. soudanense and T. mentagrophyte (TS + TM) were prepared followed by proper labeling and aseptic transfer of their conidia and spores into sterile tubes with the aid of sterilized cotton swabs. From each sterile tube, 1 ml fungal suspension was used for the inoculation of treated rice husk where appropriate. The flasks were incubated at room temperature for 7 days with 90 min daily agitation. The mycelia were separated by filtration through Whatman filter paper No. 1 and discarded. The filtrate was recovered while the treated rice husk residues were dried on filter paper using an oven temperature of 105 °C for 10 min. From each treatment, 1 g was used in each determination of carbohydrate, reducing sugar and non-reducing sugar, cellulose, total sugar (carbohydrate), hemicelluloses, lignin, and protein content in triplicates. The remaining rice husks filtrate (treatment and control samples) measure quantities were inoculated with baker’s yeast and yeast from palm wine separately and left to ferment for 7 days. Thereafter, ethanol yield was estimated.
Analysis with Filtrate
Cellulase Enzyme Assay
The carboxymethyl cellulase (CMC) activities of day 7 supernatants of cultures from the Mandel’s fungal-treated rice husk were determined using the DNS method by Miller (1959) . The carboxymethyl cellulase activities were carried out by measuring the amount of reducing sugar released from CMcellulose. A reaction mixture containing 0.2 ml supernatant (crude enzyme) and 1.3 ml 2% (w/v) CMcellulose in 0.1 M acetate buffer pH 5.0 was incubated at 30 °C, 40 °C, 50 °C, and 60 °C for 60 min. The reactions were quenched by cooling on ice and the amount of reducing sugar measured by dinitrosalicylic (DNS) acid procedure (Miller, 1959 ). The mixtures were then incubated at 100 °C for 5 min. Subsequently, the reactions were stopped by cooling on ice. Distilled water was also added into the final volume to make up a total of 16 ml and the color intensity measured at wavelength of 550 nm. The non-enzymatic release of sugar was corrected by setting up a separate blank for each sample. One unit (U or μg/ml/min) of CMcellulose activity is defined as the amount of enzyme that produced 1 μg/ml of reducing sugar as glucose per minute under the assay condition.
Determination of Incubation Time on Carboxymethyl Cellulase Activity
Test for carboxymethyl cellulase activity was carried out on days 1 (24 h), 2 (48 h), 3 (72 h), 4 (96 h), 5 (120 h), 6 (144 h), and 7 (168 h) at optimal temperature of 30 °C and pH 5.
Determination of Optimal Incubation Temperature for Carboxymethyl Cellulase Activity
The effect of incubation temperature on activity of carboxymethyl cellulase produced by the fungal isolated monocultures (T. soudanense (TS) and T. mentagrophyte (TM)) were studied by determining the released sugar using the DNS method by Miller (1959) at various temperature ranges of 30 °C, 40 °C, 50 °C, and 60 °C for 60 min.
Determination of Optimal Incubation pH for Carboxymethyl Cellulase Activity
Carboxymethyl cellulase activity by the two fungal isolates (fungal co-culture) was determined at various pH range 4, 5, 6, 7, 8, 9, and 10 using 0.1 M acetate buffer with the pH adjustments carried out using HCl (0.1 N) and 0.1 N NaOH to achieve acidity and alkalinity, respectively, for 60 min at temperature of 30 °C.
Protein Assay
The protein contents of the samples were determined using the method of biuret as applied by Ezeonu (2010) ; Oyeleke et al. (2010) and described by Jayaraman (1981) . To 4 ml of each fungal-treated rice husk filtrate was added 6 ml of Biuret’s reagent in a test tube. The contents were properly mixed and the tubes kept at 37 °C for 10 min for color development (purple color formation). The optical density of each tube was measured at 540 nm, adjustment were carried out using the reagent blank. The concentrations of protein in the enzyme samples were determined with reference to standard bovine serum albumin.
Fermentation for Ethanol Production using Baker’s Yeast and Yeast from Palm Wine
The fermentation method using both Baker’s yeast and yeast from palm wine as carried out in this research is a modification of the methodology reported elsewhere (Ezeonu et al., 2014 ). Culture filtrates of the fungal-treated rice husk were inoculated with yeast (Saccharomyces cerevisiae ) and allowed to ferment for seven days was as reported by Sandhu et al. (1998) . Spectrophotometric estimation of percentage ethanol produced was determined.
Analysis of Treated Rice Husk
Estimation of Reducing Sugar
This was determined by the dinitrosalicylic acid (DNS) method as described by Miller (1959) using glucose in establishing the standard curve. The DNS method is simple, sensitive, and adoptable during handling of a large number of samples at a time. In preparation of dinitrosalicylic reagent (DNS reagent), a solution was formed consisting of 3,5-dinitrosalicylic acid (1 g), 200 mg crystalline phenol, and 500 mg sodium sulfite dissolved by stirring in 100 ml 1% NaOH. The reagent was stored at 4 °C for at least 2 h before use. Since the reagent deteriorates due to presence of sodium sulfite, sodium sulfite was added at the time of use. A solution of Rochelle salt (potassium sodium tartrate) was also prepared. From each of the rice husks (both Trichophyton spp.-treated and -untreated rice husk), 100 mg was weighed and sugar extracted from each using hot 80% ethanol twice (5 ml each time). The supernatant (top/decanted liquid) was collected after the sugar extraction and evaporated by being kept on a water bath at 80 °C. Distilled water (10 ml) was added to each test tube to dissolve the sugar by agitation. From the extracted sugar, 3 ml was pipetted out into test tubes, and 3 ml of distilled water was added to each test tube containing the extracted sugar. To each of these solutions were added 3 ml of DNS reagent and heated in a boiling water bath for 5 min. While the contents of the tubes were still warm after boiling for 5 min, 1 ml of the prepared 40% Rochelle salt solution was added to each treatment. The reaction was cooled, and the intensity of dark red color read at 510 nm spectrophotometrically. In readiness for the standard curve, standard glucose solution stock was prepared by dissolving 100 mg glucose in 100 ml distilled water. A series of standards were determined using glucose (0.2–1 ml) and a graph plotted. The amount of reducing sugar present in each treatment was extrapolated and calculated.
Calculation
|
|
|
|
Estimation of Total Sugar (Carbohydrate)
The carbohydrate content was determined by the phenol sulfuric acid method as described by Dubois et al. (1956) . The method is as reported by Ezeonu et al. (2014) .
Calculation
|
|
|
|
Non-reducing sugar: this was obtained by subtracting the values of the reducing sugar from the total sugar.
Measurement of Cellulose Content of Fungal and Non-fungal Treated Rice Husk
The cellulose content of fungal and non-fungal-treated rice husk was determined according to the modified gravimetric method of Marzieh and Marjan (2010) as earlier reported by Ezeonu et al. (2014) . The cellulose content was calculated from the following equation (Oakley, 1984 ; Ritter and Fleck, 1924 ).
|
|
Determination of Lignin Contents of Heat-treated and Fungal Hydrolyzed Rice Husk
Methodology adopted in determining lignin contents is earlier reported by Ezeonu et al. (2014) .
The lignin content was calculated by the following formula:
|
|
where IL = insoluble lignin content (%); W lignin = oven dry weight of the insoluble lignin or Klason lignin (g); W fiber = oven dry weight of rice husks.
Spectrophotometric Method for Determination of Soluble Lignin
The filtrate obtained by the insoluble lignin was used to determine the soluble lignin content in sulfuric acid by the spectrophotometric method. In this method, 5 ml of 3% sulfuric acid was added to 5 ml of the filtrate. A spectrophotometer UV was used to measure the absorbance of the solution at a wavelength of 330 nm. Therefore, the soluble lignin content was calculated by the following formula:
|
|
where SL = soluble lignin content (%); C = concentration of soluble lignin in the filtrate (g/l); V = total volume of the filtrate (ml); W fiber = oven dry weight of rice husk (g).
The concentration (C) of soluble lignin in the filtrate is given by
|
|
where A = absorbance at a wavelength of 330 nm; Vfinal = final volume of the solution (ml); Vinitial = initial volume of the solution (ml).
The total lignin content was obtained by the addition of insoluble and soluble lignin obtained by both methods.
Thus, total lignin (TL) % = insoluble lignin (IL) (%) + soluble lignin (SL) %.
Estimation of Hemicellulose by Neutral Detergent Fiber (NDF)
The estimation of hemicellulose was done according to the method of Goering and Vansoest (1975) and reported by Ezeonu et al. (2014) .
Results and Discussion
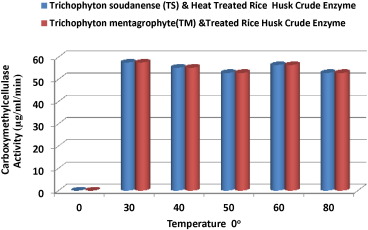
In investigating the optimized condition for better yield of biomass and bioethanol from rice husk, the best temperature, pH, and duration were ascertained. The highest carboxymethyl cellulase (CMCellulase) activity in each optimized condition was used as index for choosing each acceptable temperature, pH, and duration as the best or otherwise. Carboxymethyl cellulase activity of 57.61 μg/ml/min was the highest activity recorded for crude enzyme of T. soudanense heat pre-treated rice husk at 30 °C. This value is in agreement with research by Hallesmeersch and Vandamme (2003) and Srivastava et al. (2014) . However, carboxymethyl cellulase activity of 57.61 μg/ml/min was optimum for T. mentagrophyte heat-pretreated rice husk crude enzyme at 50 °C and 80 °C, respectively. This is a departure from the maximum crude enzyme values obtained at 30 °C as reported by Hallesmeersch and Vandamme (2003) and Srivastava et al. (2014) . Fig. 1 showed the detailed CMCellulase activities at varied temperatures. All the temperatures had CMCellulase activities in the range of 51.73–57.61 μg/ml/min except for crude enzyme of T. mentagrophyte heat-pretreated rice husk at 40 °C with CMCellulase activity of 49.38 μg/ml/min, which was the least. Thus, temperature variation of the range 30 °C to 80 °C has little or no effect in the reaction condition of the enzymes examined.
|
|
|
Fig. 1. Effect of temperature on carboxymethyl cellulase activity of crude enzyme of Aspergillus spp -treated and non-fungal-treated rice husk. |
The duration of 120 h (5 days) gave the highest carboxymethyl cellulase activity of 75.84 μg/ml/min for crude enzyme of T. mentagrophyte heat-pretreated rice husk. However, 96 h (4 days) duration gave maximum activity of 58.21 μg/ml/min for crude enzyme of T. soudanense heat-pretreated rice husk as shown in Fig. 2 . These results differed with the studies carried out by Ezeonu et al. (2014) in which Aspergillus fumigatus showed the highest duration of maximum carboxymethyl cellulase activity (65.26 ± 7.12 μg/ml/min) to have occurred at 168 h (7 days). Comparison between the crude enzyme activities of fungal-treated rice husk and the non-fungal, non-heat-treated rice husk showed great disparity as higher activities were recorded for the crude enzyme of fungal and heat-treated rice husk.
|
|
|
Fig. 2. Effect of incubation duration on carboxymethyl cellulase activities of crude enzymes of Trichophyton soudanense and Trichophyton mentagrophyte heat-pretreated rice husk. |
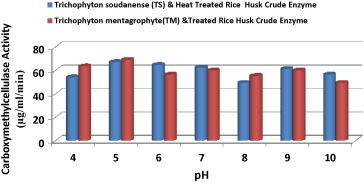
There was no significant difference in the pH range of 4 to 10 (Fig. 3 ) in terms of the CMCellulase activities of the crude enzyme of the two fungal and heat-treated rice husk. However, highest CMCellulase activities of 67.02 μg/ml/min and 69.02 μg/ml/min at pH of 5 were recorded for crude enzymes of T. soudanense (TS) and T. mentagrophyte (TM) heat-pretreated rice husk, respectively. Similarly, crude enzyme from A. fumigatus as well as Aspergillus niger -treated rice husk showed highest carboxymethyl cellulase activities of 61.14 ± 5.38 μg/ml/min and 59.96 ± 8.45 μg/ml/min at pH 5 as reported by Ezeonu et al. (2014) .
|
|
|
Fig. 3. Effect of pH on carboxymethyl cellulase activities of crude enzymes of Trichophyton soudanense and Trichophyton mentagrophyte heat-pretreated rice husk. |
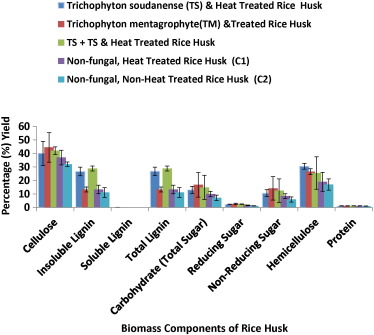
The biomass component studied included cellulose, lignin, carbohydrate, reducing and non-reducing sugar, hemicellulose and protein content in heat and fungal-treated rice husk both as single and co-culture-treatments, and controls involving heat-treated rice husk only (C1) and non-treated rice husk (C2). Thus, rice husk cooled after heating followed by treatment with T. mentagrophyte gave 44.50 ± 10.90 (% ± standard error of mean) cellulose as the highest yield followed by 42.00 ± 2.83 from rice husk cooled after heating followed by treatment with co-culture of T. soudanense and T. mentagrophyte as seen in Fig. 5 , both controls; heated rice husk (C1) and non-treated rice husk (C2) showed cellulose values of 37.00 ± 5.44 and 32.00 ± 1.63, respectively. The values of the fungal treatments and those of the controls differed significantly as seen in the result of Fig. 5 . This is in agreement with the works carried out by Belewu and Babalola (2009) , Lawal and Ugheoke (2010) , and Srivastava et al. (2014) in which the values of cellulose from their work ranged from 33% to 43%.
Highest total lignin values of 28.90 ± 1.80 and 26.77 ± 3.14 (% ± SEM) were obtained from pre-heated rice husk-treated with di-culture of T. soudanense and T. mentagrophyte and pre-heated rice husk treated with T. soudanense , respectively. The above lignin values obtained gave significant values compared to the heat-treated rice husk (C1) and non-treated rice husk (C2) with values of 13.33 ± 3.14 and 11.11 ± 3.63, respectively. A. niger and co-culture combination of A. fumigatus and A. niger with insoluble lignin yield of 26.67 ± 6.26% and 33.33 ± 8.32%, respectively, from treated rice husk as reported by Ezeonu et al. (2014) gave value which are in concordance with the result of the current research. However, pre-heated rice husk treated with T. mentagrophyte gave lignin value of 13.34 ± 1.90 (% ± SEM) showed no significant difference to the value of the controls (C1 and C2).
The hemicelluloses contents of 30.50 ± 2.12, 26.50 ± 2.12 and 25.50 ± 12.02 (% ± SEM) were obtained from pre- heated rice husk treated with T. soudanense (TS), pre-heated rice husk treated with T. mentagrophyte (TM), and pre-heated rice husk treated with co-culture of T. soudanense and T. mentagrophyte. The treatments showed increased yield of hemicelluloses when compared to the values (19.00 ± 7.07 and 17.00 ± 4.24) obtained from the heat-treated rice husk (C1) and non-treated rice husk (C2). These results are similar to the values obtained by Ezeonu et al. (2014) in which A. fumigatus (AF)-treated rice husk yielded 29.00 ± 6.00% as the maximum hemicelluloses value obtained, followed by 28.00 ± 0.00% from A. fumigatus and A. niger (AF + AN)-treated rice husk and 28.50 ± 1.50% hemicellulose yield from A. niger -treated rice husk. These values are different from those obtained by Belewu and Babalola (2009) , who recorded 19.05% hemicellulose from rice husk treated with Rhizopus oligosporum as against the initial hemicellulose content of 14.67% of the untreated rice husk, but similar to the work by Srivastava et al. (2014) with values of 20–35% from rice husk treated with Trichoderma reesei (MTCC-4876).
The carbohydrate content did not show any significant difference between the fungal-treated rice husk and the controls. The highest carbohydrate content of 16.79 ± 9.14 (% ± SEM) was recorded for pre-heated rice husk treated with T. mentagrophyte (TM); the other carbohydrate values are shown in Fig. 5 . There was no significant difference in the values of the reducing sugar between the various fungal-treated rice husks and also between fungal-treated rice husks and the controls. However, the values of the non-reducing sugar obtained differed significantly between the fungal-treated rice husk and the control (Fig. 5 ). Rice husk heated and aseptically cooled followed by treatment with T. mentagrophyte (TM) gave the highest reducing and non-reducing sugar values of 2.66 ± 0.45 and 14.13 ± 8.69.
There was no significant difference in protein content values of both the treatments and the controls, although pre- heated rice husk treated with T. mentagrophyte (TM) and heat-treated rice husk (C1) had the highest values of 1.23% each ( Fig. 5 .).
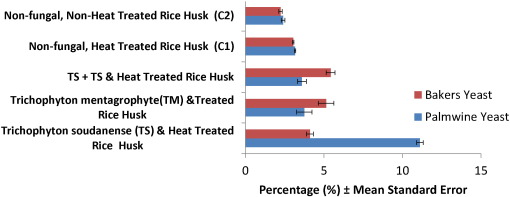
Values of Fig. 4 on bioethanol yields showed that the pre-heated rice husk treated with T. mentagrophyte (TM) fermented with palm wine yeast gave bioethanol value of 11.11 ± 0.21 (% ± Standard Deviation), which showed the highest yield (this corresponded with highest carbohydrate, reducing and non-reducing sugar yields as seen in the values discussed) and has significant value compared to the other treatments as well as the controls. Ezeonu et al. (2014) reported that bioethanol yield from rice husk treated with A. fumigatus and fermented with yeast from palm wine gave a yield of 6.60 ± 0.48% and that from A. niger -treated rice husk fermented with same yeast (from palm wine) gave a yield of 6.46 ± 0.39%. These values are less than the bioethanol value obtained using pre-heated rice husk treated with T. mentagrophyte (TM) fermented with palm wine yeast.
|
|
|
Fig. 4. Bioethanol yield of rice husk treatments fermented with Bakers and Palmwine Yeasts. |
|
|
|
Fig. 5. Effect of Trichophyton soudanense and Trichophyton mentagrophyte heat-pretreated rice husk and controls on biomass of rice husk. |
Bakers yeast fermented rice husk gave maximum values 5.43 ± 0.55 and 5.14 ± 0.13 obtained from pre-heated rice husk treated with di-culture of T. soudanense and T. mentagrophyte and pre- heated rice husk treated with T. mentagrophyte (TM), respectively, which have no significant difference in value compared to other values obtained in the research. However, the value of bioethanol obtained in this work is higher than the values of 3.20 ± 0.36 g/l or 0.27 g/g total sugar reported by Srivastava et al. (2014) in rice husk treated with Trichoderma reseei before fermenting with S. cerevisiae for 7 days.
Conclusion
The optimized condition for the performance of the fungal crude enzyme on carboxymethyl cellulase activities from T. soudanense heat pre-treated rice husk showed maximum activity at 30 °C, pH 5, and duration of 96 h (4 days), while T. soudanense showed maximum activities at 50 °C and 80 °C, respectively, also at pH of 5 and duration of 120 h (5 days). Rice husk aseptically cooled after heating followed by treatment with T. mentagrophyte gave 44.50 ± 10.90 (% ± SEM) cellulose as the highest yield; highest total lignin value of 28.90 ± 1.80 was obtained from pre-heated rice husk treated with co-culture of T. soudanense and T. mentagrophyte ; hemicelluloses contents of 30.50 ± 2.12 were obtained from pre-heated rice husk treated with T. soudanense (TS); rice husk heated and cooled followed by treatment with T. mentagrophyte (TM) gave the highest carbohydrate, reducing and non-reducing sugar values of 16.79 ± 9.14, 2.66 ± 0.45, and 14.13 ± 8.69 (% ± SEM) as well as highest bioethanol yield of 11.11 ± 0.21 (% ± standard deviation) when fermented using palm wine yeast. There were slight differences between the values of the fungal treatments and controls in all the parameters tested except for the protein content.
Acknowledgment
The authors acknowledge the management of Godfrey Okoye University Enugu, Nigeria, for their support in making available the laboratory facilities which facilitated this research work.
References
- Balat et al., 2008 M. Balat, H. Balat, C. Öz; Progress in bioethanol processing; Prog. Energy Combust. Sci., 4 (2008), pp. 551–573
- Belewu and Babalola, 2009 M.A. Belewu, F.T. Babalola; Nutrient enrichment of some waste agricultural residues after solid state fermentation using Rhizopus oligosporus; J. Appl. Biosci., 13 (2009), pp. 695–699
- Dubois et al., 1956 M. Dubois, K.A. Gilles, P.A. Rebers, F. Smith; Phenol sulphuric acid method for total carbohydrate; Anal. Chem., 25 (1956), p. 350
- Ezeonu, 2010 C.S. Ezeonu; Impact of soluble metal salts and crude oil contaminants on water retention capacity of soil and protein content of zea mays; Elec. J. Env. Agricult. Food Chem., 9 (5) (2010), pp. 885–891
- Ezeonu et al., 2014 C.S. Ezeonu, O. Otitoju, I.N.E. Onwurah, C.M. Ejikeme, O.C. Ugbogu, E.N. Anike; Enhanced availability of biofuel and biomass components in Aspergillus niger and Aspergillus fumigatus treated Rice Husk ; Eur. Sci. J., 10 (18) (2014), pp. 97–117
- Goering and Vansoest, 1975 H.D. Goering, P.J. Vansoest; Forage Fibre Analysis; Dept. of Agriculture, Agricultural Research Service, Washington (1975)
- Hallesmeersch and Vandamme, 2003 I. Hallesmeersch, E.J. Vandamme; Grass cell wall degradation by fungal cellulases and hemicellulases; Fungal Divers., 13 (2003), pp. 13–27
- Hodrova et al., 1998 B. Hodrova, J. Kopecny, J. Kas; Cellulolytic enzymes of rumen anaerobic fungi Orpinomyces joyonii and Caecomyces communis; Res. Microbiol., 149 (1998), pp. 417–427
- Jayaraman, 1981 J. Jayaraman; Laboratory Manual in Biochemistry; H.S. Poplai for Wilely Eastern Ltd, New-Delhi (1981), p. 78
- Lawal and Ugheoke, 2010 S.A. Lawal, B.I. Ugheoke; Investigation of alpha-cellulose content of agro-waste products as alternatives for paper production; Aust. J. Technol., 13 (4) (2010), pp. 258–260
- Malherbe and Cloete, 2002 S. Malherbe, T.E. Cloete; Lignocellulose biodegradation: fundamentals and applications; Rev. Environ. Sci. Biotechnol., 1 (2) (2002), pp. 105–114
- Marzieh and Marjan, 2010 M.N. Marzieh, M.N. Marjan; Utilization of sugar beet pulp as a substrate for the fungal production of cellulase and bioethanol; Afr. J. Microbiol. Res., 4 (23) (2010), pp. 2556–2561
- Miller, 1959 G.L. Miller; Use of Dinitrosalicylic acid reagent for determination of reducing sugars; Anal. Chem., 31 (1959), pp. 426–429
- Oakley, 1984 E.T. Oakley; Determination of cellulose index of tobacco; Am. Chem. Soc., 32 (1984), pp. 1192–1194
- Oyeleke et al., 2010 S.B. Oyeleke, E.C. Egwim, S.H. Auto; Screening of Aspergillus flavus and Aspergillus fumigatus strains for extracellular protease enzyme production ; J. Microbiol. Antimicrob., 2 (7) (2010), pp. 83–87
- Patel et al., 2007 S.J. Patel, R. Onkarappa, K.S. Shobha; Comparative study of ethanol production from microbial pretreated agricultural residues; J. Appl. Sci. Environ. Manag., 11 (4) (2007), pp. 137–141
- Ritter and Fleck, 1924 H. Ritter, L.C. Fleck; Determination of cellulase in wood; Ind. Eng. Chem., 16 (1924), pp. 147–148
- Sandhu et al., 1998 H. Sandhu, K.L. Bajaj, J.S. Arneja; Biochemical Analysis; Kalyani Publishers, New Delhi (1998)
- Srivastava et al., 2014 A.K. Srivastava, P. Agrawal, A. Rahiman; Delignification of rice husk and production of bioethanol; Int. J. Innov. Res. Sci. Eng. Technol., 3 (3) (2014), pp. 10187–10194
- Wubah et al., 1993 D.A. Wubah, D.E. Akin, W.S. Borneman; Biology, fiber-degradation, and enzymology of anaerobic zoosporic fungi. Crit; Rev. Microbiol., 19 (1993), pp. 99–115
Document information
Published on 27/03/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?