(Created page with "<!-- metadata commented in wiki content <div class="center" style="width: auto; margin-left: auto; margin-right: auto;"> <big>Can Combined Treatment of Ginseng and Omega-3 E...") |
m (NZHNG moved page Draft Zhang 928330811 to Review 382597607607) |
(No difference)
| |
Revision as of 07:35, 20 February 2025
Abstract
This study aims to research, evaluate, and potentially optimize the efficacy of Omega-3 fatty acids and ginseng extract as Alzheimer's Disease (AD) treatments. AD, which is characterized by central nervous system deterioration and cognitive impairment, affects millions worldwide, with cases expected to quadruple by 2050. Due to the failure of single-target amyloid beta (Að½) drugs in trials, attention has gradually shifted to multi-target drugs. Notable promising candidates include omega-3 (an essential nutrient) and Panax ginseng (from the Araliaceae family). While prior research examines these treatments individually, their combined effects remain unexplored, warranting further investigation into AD causes and neuroprotective agents. The potential optimization of these treatments was tested on CL2355 (transgenic AD model) and a wild-type (N2) Caenorhabditis elegans (C. elegans) on account of its advantageous neuronal connectivity in modeling functions correlated to AD. Administration of treatments ranged from two controls without either treatment (in a wild type and AD model) and treatment variations of DHA Omega-3 (mM) to Ginsenoside (μg/ml), in 5:0, 0:10, and 5:10 ratios on AD models. Evaluation of the treatment's effects on Að½ toxicity and AD symptoms were gauged through paralysis, chemotaxis index, and population growth assays. Results indicate that combined treatments show potential as multi target therapeutic strategies against AD either enhancing individual effect or optimizing benefits.
1.0 Introduction
Purpose and Importance
Alzheimer’s disease (AD) is a neurodegenerative disease that is closely associated with increasing age. Alzheimer’s disease is the main cause of dementia, accounting for 75% of cases (Qiu et al., 2009). As of 2014, AD impacts more than 35.6 million people worldwide, and this number is estimated to quadruple by 2050 (Anstey et al., 2014; Zhou et al., 2019). Alzheimer’s disease deteriorates the central nervous system, mitigating language recognition and cognitive behavior (Sharma, 2019). AD damages the neocortex and hippocampus, brain areas that are associated with memory capacity and cognitive behaviors, overall reducing life expectancy and increasing the probability of physical impairment in the elderly (Qiu et al., 2009). Although the cause of AD has been an area of mass study it remains not fully understood. According to current studies, it is potentially caused by abnormal accumulation of amyloid beta (Að½) aggregation leading to decaying nerve receptors containing depositions of Að½ (Sadigh-Etaghad et al., 2015; Deng et al., 2020; Bredesen, 2016). Að½ is transported to the brain and attacks the neurons, reducing brain functions (Soni & Galluci, 2023).
Currently, Food and Drug Administration approved treatments to alleviate Alzheimer’s disease in patients are based on cholinergic and glutamatergic systems in the brain (Kim et al., 2018). Cholinesterase inhibitors (CHEI), an enzyme, are the main medicinal drug to mitigate Alzheimer’s disease in patients. CHEI regulates cognitive systems by reducing the failure of acetylcholine, a neurotransmitter communicating information through nerve cells; however, the effectiveness of CHEI is restricted as it induces side effects and lessens in impact as Að½ prevalence increases (Sharma, 2019). NMDA (N-methyl-D-aspartate) receptor antagonists, another class of drugs, have also been used to relieve the progression of AD. N-methyl-D-aspartate receptors play a significant factor in conjunctive transmission and conjunctive flexibility, a foundation of literacy and knowledge retention, central to neurotoxicity (Liu et al, 2019). Previous studies indicated that triggering NMDA receptors are involved in AD conjunctive abnormality (Paoletti, 2011; Kodis et al., 2018). Consequently, NMDA receptor antagonists attempt to reduce glutamate excitotoxicity, the excessive triggering of neuron amino acid receptors which leads to neuritic atrophy (Mark et al., 2001).
Due to the failure of drugs solely targeting Að½ to pass Stage 3 trials, attention is gradually shifting to multi-target drugs or stress protein-based solutions. Multi-target drugs often come in the form of plant extracts and homeopathic therapeutics and may better address AD causes present in its etiology. This study sought to evaluate the efficacy of two established AD treatments, omega-3 fatty acids, and ginseng extract, in combination to evaluate efficacy and synergy to contribute to finding new treatments and thus alleviate the burden of AD.
Omega-3 fatty acids (ω-3FAs)
Omega-3 fatty acids (ω-3FAs), especially in the form of DHA (docosahexaenoic acid), are an essential nutrient for brain function and make up 40-50% of total brain mass (Dighriri et al., 2022). Additionally, dietary consumption of ω-3FAs, especially in moderate or high doses, shows promise for slowing down AD progression and restoring executive, and hippocampal functioning. Previous studies show that ω-3FAs supplementation decreases AD risk by up to ~64%, and may help restore working memory, and executive function in select AD cohorts (Arellanes et al., 2020; Wei et al., 2023, Power et al., 2022). These outcomes can be largely attributed to ω-3FAs multi-target free radical scavenging, anti-inflammatory, brain barrier supporting, antiapoptotic, and neurotrophic properties (Ajith, 2018; Patrick, 2019). When broken down, ω-3FAs create anti-inflammatory specialized pro-resolving mediators, which facilitate brain barrier repair and improve the brain's ability to remove excess Að½ and fibrinogens (Valente et al., 2022; Francos-Quijorna et al., 2017). Furthermore, Mfsd2a (omega-3 neuroreceptor) plays an integral role in the blood-brain barrier (BBB) and pericyte health but decreases with age and cognitive decline. Maintaining a high ω-3FAs index may offset this progression's deleterious effects, further solidifying ω-3FAs’ role in AD prevention and brain health (Yang et al., 2020; Patrick, 2019). Unfortunately, epidemiological studies indicate that U.S. intake of ω-3FAs is largely inadequate, with the average person only consuming ~0.1-0.2g of DHA or EPA (eicosapentaenoic acid) per day (Kris-Etherton et al., 2000). Equipped with this knowledge, many dementia researchers argue ω-3FAs deficiency to be a leading driver of the current prevalence of AD in the U.S. (Wei et al., 2023). The present study sought to spread awareness of ω-3FAs deficiency and contribute to the existing literature on ω-3FAs research.
Outside of targeting hallmark features of AD (i.e., Að½ plagues, neurofibrillary tangles (NFTs), etc.), ω-3FAs may provide benefits that attenuate the cognitive and motor problems associated with AD. In a systematic review including eleven studies with 698 participants, ω-3FAs substantially increased serum brain-derived neurotrophic factor (BDNF) levels in moderate or high doses (Ziaei et al., 2024). For context, BDNF is a neurotrophin that contributes to the formation and maintenance of neuronal synapses. Renowned as a cognitive “substrate”, BDNF plays a vital role in learning, memory formation, emotion, sensory integration, decision making, reward perception, motor function, executive function, and stress response; all of which hold major implications and are negatively affected by AD pathogenesis (Brown et al., 2017; Miranda et al., 2019). Unfortunately, various studies found lower levels of BDNF in the brains of AD patients; an association that not only negatively affects cognition but also exacerbates symptoms of AD (Zhang et al., 2014). Moreover, lower BDNF levels may also contribute to AD risk; however, whether this relationship is causal remains unclear and is an area of future research (Weinstein et al., 2014).
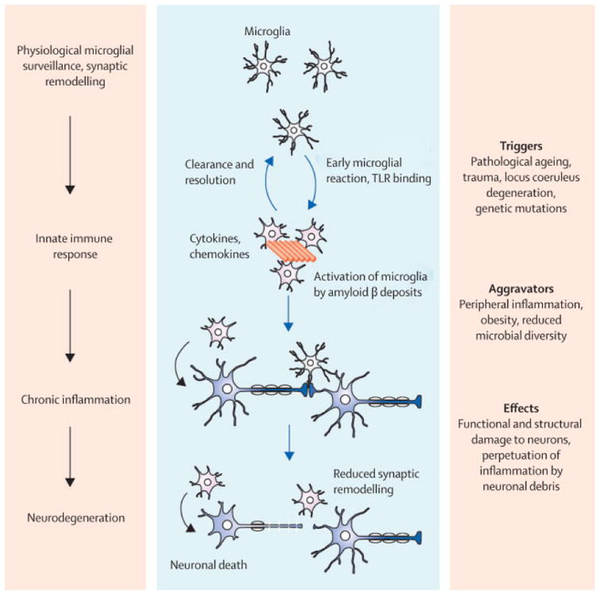
Outside of BDNF, ω-3FAs additionally possess anti-inflammatory properties; and more specifically, anti-neuroinflammatory effects as it relates to AD. In the aging brain, misfolded proteins, most readily in the form of Að½ aggregations and NFTs, trigger innate immune responses in the brain’s microglial leading to a cascade of neuro deterioration (Fig. 1). Additionally, ApoE-4, a gene that substantially raises AD risk, upregulates (relative to ApoE-2 or ApoE-3) glial cells through lipid modulation; thus, drastically increasing neuroinflammation (Parhizkar & Holtzman, 2022). Over the past decades, ω-3FAs were extensively studied and shown to possess a potent anti-inflammatory effect, especially in the field of cardiology and metabolic health (Wierenga & Pestka, 2021). However, recent discoveries show dietary ω-3FAs ability to traverse the BBB where it is not only used as a fuel substrate but also facilitates reduction in neuroinflammation. For instance, in a randomized controlled trial of 33 patients with mild AD, the supplementation of ω-3FAs (2.3 g; DHA-based), reduced levels of phosphorylated tau, and lowered neuroinflammatory biomarkers (interleukin-1 & interleukin-6); all within a relatively short period of 6 months (Freund et al., 2014).
Figure 1: Pathomechanism sequelae of immune activation (Heneka et al., 2015)
Panax ginseng
Panax ginseng is a perennial plant from the Araliaceae family and was used since 1500 CE in East Asian regions such as Chinese, Korea, and Japan where ginseng is commonly consumed in liquid forms such as tea or wine. Consumption of ginseng is also emphasized due to its health, beauty, and dietary benefits. The indicated restorative impacts of ginseng in tea include invigorating the body and uplifting mood (Kim, 2018). In recent years, there is also an increase in recognition of ginseng in Western countries (Lin et al., 2020). Ginseng is one of the earliest Chinese herbal medicines for the treatment of dementia, such medicines were utilized for the treatment of neurological disorders, showing a low risk for negative interaction when used in medicinal blends (Wang et al., 2023).
Ginseng contains multi-target effects and is effective in AD prevention and amelioration in multiple human trials (Lee et al., 2023). Researchers emphasize the need for further research on ginseng, especially those concerning transport vehicles and drug combinations in AD (Wang et al., 2023). Ginsenosides, the main biologically active components isolated from Panax ginseng, act as a neuroprotective agent with potential benefits for AD patients (Zhang et al., 2023). Moreover, ginseng exhibits anticholinergic activity, neuroplasticity and memory promotion, anti-Að½, anti-tau, neuroprotective, anti-inflammatory, antioxidant, anti-apoptosis, insulin resistance alleviating, and BBB strengthening properties in in-vitro and animal studies; all of which have major implications for AD patients (Wang et al., 2023).
Blood-brain barrier (BBB) and nutrient transport
Although previous research shows that ApoE-4 increases the risk of AD exponentially, ApoE’s evolutionary function is less conventionally known. On the fundamental level, ApoE in the brain serves as a regulator of astrocytes and glial cells, helping to modulate inflammation and thus the brain’s ability to fight pathogens and repair. At the beginning of human history, ApoE-4 existed as the sole isoform. Alternate isoforms which increase inflammation less dramatically, namely, ApoE-2 and ApoE-3, appeared only in the presence of a more glucose-based diet facilitated by agriculture. Although ApoE-4 was highly evolutionarily advantageous before the advent of modern medicine; it now poses a leading risk factor for AD pathogenesis, raising AD risk by up to 15-fold in double allele cohorts (Belloy et al., 2019; Yamazaki et al., 2019).
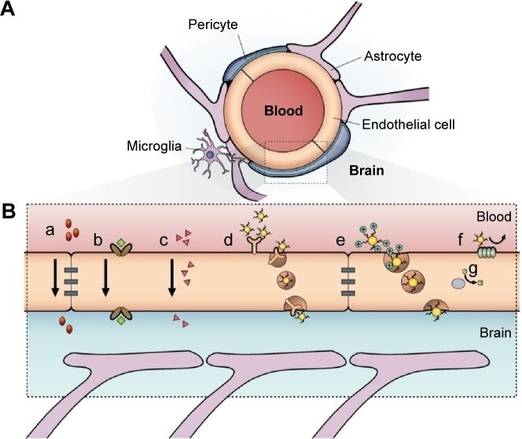
Many factors attributable to ApoE-4 underpin the drastic increase in risk. For instance, mechanistic research suggests that ApoE-4 binds to receptor LRP1, responsible for Að½ clearance, and thereby elevates levels of Að½ oligomers in the brain by up to 2.7 times (Ba et al., 2016). Furthermore, as ApoE-4 binds to LRP1, proteins such as MMP9 and cyclophilin A are released, further augmenting the BBB-degrading consequences of Að½. This breakdown of the BBB makes the brain vulnerable to neurotoxic proteins (such as plasminogen, auto-antibodies, albumin, fibrinogen, etc.) from the bloodstream, leading to a cascade of neuroinflammation (Fig. 2).
Perhaps more directly pertaining to the present study is the BBB’s role in ω-3FAs transport (Platt et al., 2017; Levine, 2016; Valente et al., 2022). Although ω-3FAs supplementation shows immense efficacy in the prevention of AD, contradictory outcomes regarding efficacy in post-onset cohorts impede the recommendation of ω-3FAs for neurodegenerative disease on a larger scale. For instance, in a randomized controlled trial including 174 patients with mild to moderate AD, a moderate dose of ω-3FAs was unable to improve cognitive outcomes relative to placebo in a six-month period (Freund et al., 2006). Although the short study time likely played a part in facilitating inconclusive results, the existing BBB damage of the AD patients may also be a factor. When the BBB becomes compromised, the ability of ω-3FAs transporters to utilize and uptake ω-3FAs drastically deteriorates. For instance, although the brains of young adults readily uptake ω-3FAs, regardless of ApoE status; older adults, especially those with ApoE-4 and/or AD, fail to uptake ω-3FAs at the same rate (Arellanes et al., 2020). This pattern is observed in a randomized controlled trial that concluded supplementation with DHA was significantly less effective at mitigating AD symptoms in ApoE-4 cohorts compared to those with ApoE-3 (Quinn et al., 2010).
However, multiple methods were proposed to increase ω-3FAs bioavailability in the brain. One such approach is prescribing ω-3FAs synergistically with applicable antioxidants and/or polyphenols. For instance, a recent study looking to improve cognitive function in older but otherwise healthy adults combined standard ω-3FAs with carotenoid and vitamin E and attributed their positive results to the addition of antioxidants inducing superior bioavailability (Power et al., 2022). Although antioxidants likely work synergistically with ω-3FAs in the aforementioned study, combining ω-3FAs with ginseng may have even greater effects. Outside of neurodegenerative disease, ginseng and by extension ginsenosides also have strong implications in terms of lipid metabolism. Preliminary studies suggest that ginseng modulates lipid metabolism and thus reduces adipose disposition in the C. elegans model (Shi et al., 2023). Human trials also support this claim, albeit to a limited extent as current studies are subject to substantial limitations and show contradictory results (Delui et al., 2013). Regarding ω-3FAs specifically, ginseng extract shows extended health span by upregulating ω-3FAs (in the form of lipoic acid) metabolism signaling pathway in C. elegans; however, it is unclear whether this effect translates to DHA and EPA. Together, these results suggest that ginseng extract positively modulates the usage of ω-3FAs through upregulating metabolism and preventing adipose deposition which by extension increases ambient ω-3FAs levels and transport to the brain (Yu et al., 2021).
Figure 2. Schematic representation of the blood–brain barrier (Abbott et al., 2010)
Caenorhabditis elegans
Caenorhabditis elegans are commonly used experimental organisms for genetic and genomic analysis research. This is attributed to their cellular simplicity, genetic amenability, inexpensiveness, and homology to humans possessing complex biochemical pathways and genes in which mutations are correlated with AD (Sofela et al., 2021). The nervous system of C. elegans consists of 302 neurons that control behaviors such as locomotion, allowing processes and mechanisms to be traced at the level of individual neurons (Alvarez et al., 2022). This neuronal connectivity in C. elegans is advantageous in modeling learning and memory impairments seen during AD (Alexander et al., 2014). The short life span of 2-3 weeks allows for the observation of neurodegeneration in nematode cohorts during the entire adult life of the organism (Alvarez et al., 2022). Previous studies utilized its transparent body for visualization of all cell types and expression of fluorescent proteins in specific cells and tissues, useful for in vivo investigation of neuronal death or protein aggregation processes (Alvarez et al., 2022). Although previous research uses ginseng extract and Omega-3 supplements separately on the C. elegans Alzheimer’s model, the combination of both factors remains unknown.
Previous research
Previous research indicates that ginseng may be a suitable therapeutic to mitigate the spread of AD. In transgenic mice models influenced by Alzheimer’s disease, fermented ginseng extract treatment improved memory dysfunction. Repaired cognitive functions are fundamental to reduced Aβ42 brain accumulation, indicating that fermented ginseng ameliorated Aβ42 buildup (Kim et al., 2013). Red, black, and white ginseng extracts show an ability to hinder acetylcholinesterase, resulting in reduced hippocampus Aβ oligomer injection-influenced memory anomaly (Lee et al., 2011). In this study, ginseng was added in the form of Korean red ginseng (KRG). Compared to other ginseng types, KRG has the most ginsenosides. Korean red ginseng treatment shows improved frontal lobe functions and positive influences on quantitative electroencephalography (a method to record the electrical activity in the brain) (Heo et al., 2016). Studies also indicate that KRG benefits cognitive functions and dependable clinical efficiency in patients impacted with AD. Additionally, KRG extract application indicates that cognitive function results can be held up for over two years (Heo et al., 2011).
Omega-3 is another previously established therapeutic strategy to mitigate cognitive impairment, and progression to Alzheimer’s disease. Evidence from 33 studies suggests that dietary and supplemental LC ω3 PUFAs have a protective effect against cognitive impairment, demonstrating profounder effects as a preventative treatment or in the early stages of AD (Wood et al., 2021). Treatments with omega-3 can reduce Aβ accumulation being effective against factors such as the typical hallmarks of AD including Aβ accumulation, neuroinflammation, and neuronal loss and hippocampal neurogenesis impairment by protecting against mitochondrial dysfunction in AD pathogenesis (Park et al., 2020). Although, nutritional or dietary do not function as a total remedy for AD they greatly aid in alleviating symptoms. In fact, in another study it was determined that subjects who adhered to higher omega-3 based diets possessed a relatively lower risk of developing dementia, in particular Alzheimer’s disease (Avallone et al., 2019).
Research question/hypothesis
Based on evidence from previous studies, the researchers of this study hypothesize that a combination of ginseng extract and omega 3s would exert a synergistic effect on alleviating Að½ toxicity, and other AD symptoms in CL2355 (transgenic AD model); while also prolonging the life span of both wild type (WT) and CL2355 C. elegans. To date, there are no AD studies combining ω-3FAs (in DHA or EPA form) with ginseng; however, a previous study looking at the aforementioned combination was conducted on ADHD children; and found that the combination significantly alleviated ADHD symptoms with benefits beyond the seldom use of either ginseng or omega 3; possibly indicating a synergistic effect (Lee et al., 2020). Treatments solely using either ginseng extract or ω-3FAs would likely have much of the same positive outcomes as the combination, albeit to a lesser extent. This hypothesis is built on multiple studies of these compounds on C. elegans. Examples of this can be seen in C. elegans studies showing ginsenosides and omega-3 to be successful at protecting against Að½-induced toxicity and extending lifespan through lowering oxidative stress, respectively (Zhang et al., 2017; Qi et al., 2017).
2.0 Materials and Methods
Creating M9 buffer
Using a top loading balance, 1.5 g of KH2PO4, 3 g of Na2HPO4, and 2.5 g of NaCl was dissolved in 500 mL of distilled water in a graduated cylinder. The solution was sealed with aluminum foil and autoclaved for 20 minutes. After, 1 mL of 1 M MgSO4 was added to the solution. The final solution was stored at room temperature and sterilized using an autoclave before each use.
Creating ginsenoside stock solution (10 mg/ml)
Five grams of ginseng extract was measured using a top loading balance. Next, 50 mL of distilled water was measured into a graduated cylinder mixing it with the ginseng extract in the graduated cylinder until fully dissolved. The solution was then sterilized by autoclaving it at 120°C for 2 hours before being transferred to the test tube and stored in a dark and cool environment. If necessary, the solution was repeatedly mixed before each use.
Creating Omega-3 stock solution (500 mM)
Utilizing a top-loading balance 8.2g of DHA omega-3 was measured in an Erlenmeyer flask using a top-loading balance. In a graduated cylinder, 50 mL of sterilized M9 solution was measured. Using 1.5 mL Tween 80, the measured omega-3 was dissolved. The water and fatty acid emulsion was transferred into a vial and vortex until it was fully homogeneous. The stock solution was stored in a light blocking tinted vial and placed in a dark and cool environment until its use. Before each use, a vortex was used.
Creating Nematode Growth Media (NGM) agar plates
The C. elegans were cultured in the Nematode Growth Media (NGM) agar plates with various concentrations of the tested treatments. The Petri dishes were labeled as a group from A-E separately per distinct treatment concentration and strain according to Table 1. The NGM (provided by Carolina Biological) in solid form was melted into liquid form for usage. First, the bottle was uncapped about halfway to release steam and pressure. The NGM agar was then added into the microwave for 30 seconds in intervals of 15 seconds checking each time for according to if adequately melted (alternatively a water bath at approximately 100℃ was used when time permitted). Due to caution of the heat gloves were used when taking the agar out of the microwave, properly shaking it to ensure no solids remain (agar should be a dark-yellow liquid). Then, the agar was allowed to cool for a few minutes with caution of re-solidification if excessive cooling.
Half of the NGM agar was dedicated to ginseng groups by transferring them to a separate graduated cylinder. A 1000x dilution of ginsenoside solution (0.18 mL) was then performed using 180 mL of NGM agar as the solvent. Then the agar was poured into the Petri dish very quickly, covering the lid of the Petri dish and tapping the bottle to avoid possible contamination. The aforementioned procedure was repeated for all Petri dishes with a waiting time of 15-20 minutes until agar solidified in the dish prior to storage in the refrigerator for 24 hours before usage (keeping the Petri dish upside down to prevent condensation from falling on the agar).
Each plate was then spotted with E. coli OP50 grown for 24 hours in LB Media. Upon transferring, plates were dried for an additional 24 hours. The previously made omega-3 stock solution was diluted from 1mL to 5 mM with sterilized water to a final volume of 100 mL (100x dilution). Then, a dispersion of 0.1 mL of DHA omega-3 aliquot to the solidified and spotted NGM in Groups C, and E was done using a spreader. Upon administration of treatment in each Petri dish, the mediums were allowed to dry for another 24 hours.
Table 1: Experimental Setup
| Group Name | C. elegans strain | DHA Omega-3 (mM) | Ginsenoside (μg/ml) |
| A | N2 (wild type) | 0 | 0 |
| B | CL2355 (AD) | 0 | 0 |
| C | CL2355 (AD) | 5 | 0 |
| D | CL2355 (AD) | 0 | 10 |
| E | CL2355 (AD) | 5 | 10 |
Toothpick technique C. elegans Transfer
A blunt dissecting probe, acting as a pick for C. elegans transfer, was sterilized on a flame. A bead of OP50 was collected from an empty place to make the point sticky. Gently touch the worm selected using the OP50 coated probe. When the worm disappeared, it was on the probe. The worms were placed on the destination place by gently rolling the probe's tip until it became free. The worm was zoomed in on using the microscope to ensure that it was not damaged after the transfer and only the selected worm or egg was transferred.
Creating bacteria agar
The table was cleaned before beginning and preparing the materials for the study. All safety precautions were followed as well. A weighing paper was folded to make it easier to handle. On a weighing boat on a scale, 17.5 g of premix LB Agar powder was weighed. This powder was transferred to an Erlenmeyer flask and distilled water was added until reaching a final volume of 500 mL. The bottle cap and the top were sealed with aluminum foil tightly. The solution was then autoclaved in the liquid setting for 20 minutes. Following the autoclaving, the agar was cooled to about 55℃. A thin layer of LB agar was poured into each Petri dish, ensuring the liquid was properly and evenly spread. After putting on the lid of the Petri dishes, each plate was left to cool down until solidification. After 24 hours of the solidification process, the dish was flipped to avoid the condensation falling onto the agar. After this, the plates were placed in plastic bags in the refrigerator at 4℃.
Bacterial aseptic technique
E. coli OP50 (Source: https://cgc.umn.edu/strain/OP50) was used to feed C. elegans
(Sourced: CL2355 strain: https://cgc.umn.edu/strain/CL2355 and N2 strain: https://cgc.umn.edu/strain/N2) and placed in all Petri dishes. A rubber tube was connected to the valve of the gas and the valve of a Bunsen burner. Once the gas ignited, a match was lit and placed over the gas. The inoculation loop was sterilized by passing the tip and the neck of the inoculation loop through the flame. After the inoculation loop cooled, it was used to scoop the agar from a bacterial colony. The bacteria were streaked across the surface of a new Petri dish, ensuring that there was no excessive pressure put on the agar to refrain from breaking it. After, the lid was placed back on to the Petri dish and flipped upside down in the incubator to ensure that the accumulated condensation does not fall on the agar. Following two days of the Petri dishes being incubated, parafilm was wrapped around the sides of the Petri dish and placed into the fridge to store for later use. Before and after each process, diluted bleach was used to sterilize all surfaces.
Spotting Petri dishes with E. coli OP50
A 0.5 cm x 0.5 cm square chunk of agar with E. coli OP50 was cut using forceps or other sterilized metal. Sterilized forceps were used to grab a piece of agar. The agar was placed into the middle of the premade NGM plates. Each chunk was gently placed into its respective Petri dish. Before and after cutting and transferring each chunk, disinfectant tools were used to prevent contamination.
Conducting the paralysis assay
After 48 hours of C. elegans culturing, the temperature was either adjusted to 27℃ (for Trial 1) or 30℃ (for Trial 2). To ensure that the plates reached the temperature at the same time, the plates were not stacked on each other. After 24 hours of temperature upshift, the scoring of paralysis was performed for every two hours until all nematodes were paralyzed. Paralyzed nematodes were transferred to a part of the plate that was not spotted to refrain from scoring them again. In C. elegans, the head of nematodes is last to become paralyzed while the tail is first, restricting its movement. The C. elegans were considered paralyzed when they were unable to move, excluding its head. Nematodes that rarely voluntarily move were poked using a worm picker. If C. elegans did not show full movement, they were considered paralyzed.
Testing for Population Growth
A 24 well plate’s first column was labeled “Group A”, the second column “Group B”, the third column “Group C”, the fourth column “Group D”, and the fifth column “Group E”. Four wells were used for each group. First, 1.0 mL of ginsenoside NGM was poured into the columns of Group D and Group E. Then, 0.1 mL of DHA omega-3 aliquot was pipetted to the solidified and spotted NGM in the columns Group C and Group E. Then, C. elegans were chunked into each column containing NGM. Every day for one week, the amount of C. elegans was recorded under a compound microscope. For Trial 1 of the assay, 10 μL ginseng was spread on top of the NGM for the treatment groups that required it. For Trial 2, ginseng was diluted into the NGM following the previous procedure.
Conducting the Chemotaxis Index Assay
Seven days before conducting the chemotaxis, C. elegans from each group were transferred to similar treatment plates to allow the C. elegans to culture. These plates were regarded as the treatment plates. Two days before the chemotaxis, a marker was used to divide each of 5 NGM Petri dishes into 4 equal quadrants, and a circle was created from the origin with a 1 cm radius. Then, another circle was created from the origin with a 2 cm radius. 10 µL of E. coli, acting as an attractant, was pipetted onto quadrant 1 and quadrant 3 of the chemotaxis plate. These 5 Petri dishes with the absence of treatment were regarded as the chemotaxis plates.
A micropipette was used to pipette 2 mL M9 on the treatment plates. The treatment plate was gently angled to cover all surface area to ensure that all nematodes were cleansed. A pipette was used to pipette 1 mL of the previously made worm solution in a microcentrifuge tube. The solution was centrifuged for 6,600 rpm for 10 seconds. After the centrifugation, the remaining M9 was removed. One mL M9 was pipetted in the centrifuge tube and the tube was turned to ensure that all nematodes were cleansed. The process was repeated three times. After, the volume of the supernatant was reduced to 100 μl and centrifuged for another 10 seconds. A pipette was used to transfer 2 μl of nematodes onto the chemotaxis plates.
After the 60 minute cleansing period, the nematodes were used. A micropipette was used to transport 2 μL of the nematode solution from the centrifuged solution to the center of the chemotaxis plate, or the intersection of the perpendicular lines drawn. Immediately after, a recording device was set directed towards the chemotaxis plates in a compound microscope, and a 60 minute video was recorded. After 60 minutes, the chemotaxis plates with the nematodes were placed in an incubator with a controlled temperature of 4 °C. The number of nematodes that passed through the circle with a 1 cm radius and moved towards the attractant zone or control zone and the time it took was recorded in Microsoft Excel corresponding with their respective quadrants. The process was repeated, replacing the treatment solution with sole control treatment. This Petri dish was regarded as the control plate. The chemotaxis assay was performed in quadruplicate.
Safety
Proper lab safety, including wearing gloves, goggles, and tied hair during the experimentation process, was fulfilled especially when conducting bacterial works. The tabletop working area was cleansed with a diluted bleach solution before and after experimentation along with the sterilization of all instruments at the end of the trial period. All Petri plates were sealed with parafilm during experimentation and autoclaved at 120 ℃ and 100 kPa for 20 minutes as part of the disposal procedure. Tools in contact with cultures were also soaked in 10% bleach and sterilized with heat as a segment of the disposing process. Hands were washed after any bacterial work. Three days of safety training specifically for bacterial works and the presence of experienced supervisors were ensured whilst dealing with microorganisms and the flame or gas of a Bunsen burner required for sterilization.
Three days of safety training specifically for bacterial works and the presence of experienced supervisors were ensured while dealing with microorganisms and the flame or gas of a Bunsen burner required for sterilization of tools. Most substances utilized in the study possessed a biosafety level BSL-1 and deemed “not hazardous” under the criteria of the federal OSHA Hazard Communication Standard 29CFR 1910.1200, and Regulation (EC) No 1272/2008 (GHS). Nonetheless, substances specifically can be dangerous when ingested, inhaled, or especially in cases of in contact with eyes (skin contact and inhalation do not pose serious harm unless it is done excessively or if the researcher feels unwell after exposure) so precautionary safety such as protective gloves, clothing, and above all else eye protection was adhered to.
Data Analysis
Data was collected, stored, and analyzed using Microsoft Excel. Data was shown using scatter plots and bar graphs in which the x values were time in hours, time in days, and group names and the y values were not paralyzed C. elegans as a percentage, number of C. elegans, and chemotaxis index. The median paralysis time was conducted using a Kaplan–Meier survival analysis. In the paralysis assay, the number of non-paralyzed C. elegans on each plate was converted into a percentage. The standard mean of error was calculated and represented as error bars added using Microsoft Excel. To calculate significance, one-way ANOVA followed by Tukey HSD was done using https://astatsa.com/OneWay_Anova_with_TukeyHSD/. Significance was determined using p-values in which a p-value less than 0.05 was considered significant. Chemotaxis index was determined using the following formula:
CI = (number of worms in attractant zone - number of worms in control zone)/total number of scored worms
Results
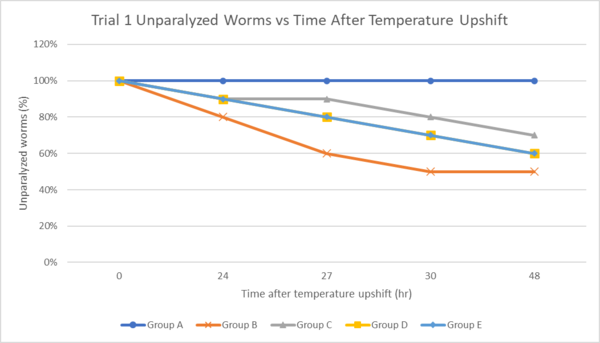
Figure 3. The effect of ginseng and omega-3 on the percentage of unparalyzed worms following temperature upshift to 27 ℃ in Trial 1.
In C. elegans strain CL2355, when temperature rises, Aꞵ peptide is excessively produced in the muscles of nematodes, causing paralysis. Group B (the negative control group) had the lowest percentage of unparalyzed worms 48 hours following the temperature upshift. Group C had the highest percentage of unparalyzed worms compared to the negative control group with a percentage of 70% 48 hours following temperature upshift. Additionally, Group D and Group E displayed similar effects compared to each other with the percentage of unparalyzed worms being identical throughout all time. Throughout the assay, all treatment groups showed a higher percentage of unparalyzed nematodes, showing the alleviation of the impact of amyloid-beta aggregation. The wildtype C. elegans (Group A) do not produce Aꞵ peptides so the nematodes did not display any paralysis following temperature upshift.
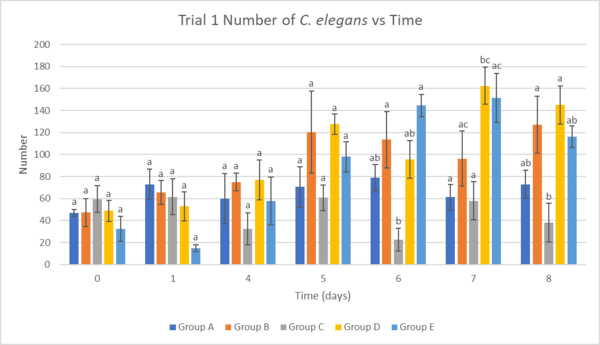
Figure 4. The effect of ginseng and omega-3 on the average population growth of transgenic and N2 C. elegans in Trial 1. Error bar values represent mean ±SEM. Statistically significant comparisons (p<0.05) are represented with different letters.
Figure 4 shows population growth throughout the whole assay with Group D and Group E showing higher populations compared to Group A (wild type control) from Day 5. The increasing population of Group B (negative control) can be attributed to a result of a compensatory mechanism or lack of immediate significant symptoms in the trial's time span. At Day 6, Group E (combined treatments) had a statistically significant higher population than Group C (sole omega-3 treatment). Similarly, at Day 7, Group D had statically higher growth than Group A along with Day 8, demonstrating a significantly higher population compared to Group C.
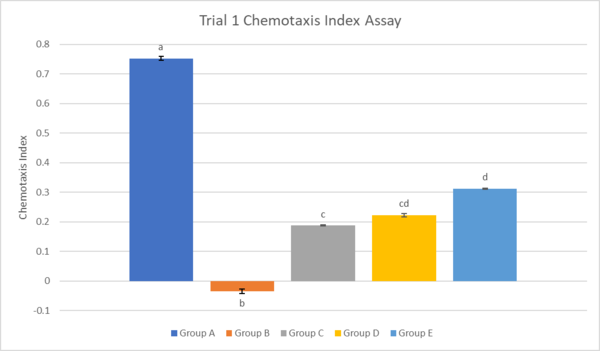
Figure 5. The effect of ginseng and omega-3 on chemotaxis index values in Trial 1. Error bar values represent mean ±SEM. Statistically significant comparisons (p<0.05) are represented with different letters.
The results shown in Figure 5 indicate that Group A (N2 wildtype) had the greatest chemotaxis index compared to all other groups. Group B, the negative control group, had the lowest chemotaxis index, which was statistically significant when compared to Group C, Group D, and Group E. Out of the groups expressing Aꞵ peptides, Group E had the greatest chemotaxis index, with statistical significance when compared to Group B and Group C. Group D had the next greatest chemotaxis index (0.222), followed by Group C (0.188).
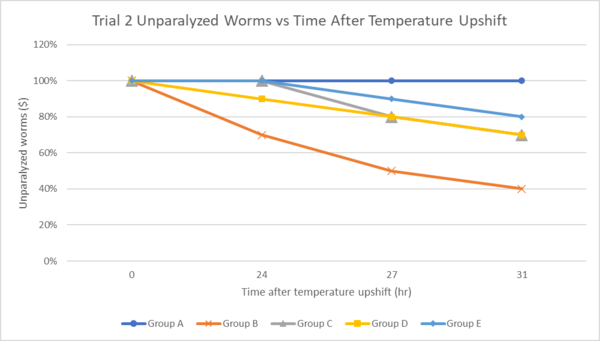
Figure 6. The effect of ginseng and omega-3 on the percentage of unparalyzed worms following temperature upshift to 30 ℃ in Trial 2.
The results of Figure 6 are consistent with that of Trial 1 in which all treatment groups had a higher percentage of unparalyzed C. elegans compared to Group A (negative control group). Group A had the lowest percentage of unparalyzed worms throughout the whole trial apart from Hour 0. Out of all the treatment groups with C. elegans exhibiting amyloid-beta aggregation, Group E (combined treatment) had the highest percentage of unparalyzed C. elegans of 80% 31 hours following temperature upshift. Group C and Group D both had 70% of unparalyzed C. elegans 31 hours following temperature upshift. Therefore, the treatment of sole ginseng, sole omega-3, and a combination of ginseng and omega-3 all show alleviation of amyloid-beta aggregation influences, with the combination of treatments exhibiting the greatest effect within Trial 2 of the paralysis assay.
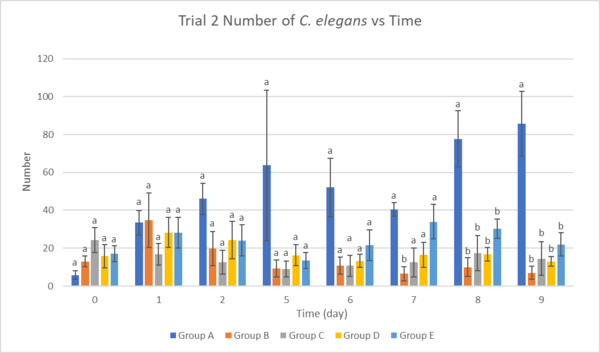
Figure 7. The effect of ginseng and omega-3 on the average population growth of transgenic and N2 C. elegans in Trial 2. Error bar values represent mean ±SEM. Statistically significant comparisons (p<0.05) are represented with different letters.
Figure 7 results show that Group A (N2 wildtype) had the highest population number throughout the whole assay. All groups exhibiting amyloid-beta aggregation had a significantly lower population compared to Group A on Day 8 and Day 9. Group B (the negative control group) decreased in population number as the assay proceeded. Within the groups exhibiting amyloid-beta aggregation, Group E (combined treatments) displayed the highest population throughout the whole trial other than Day 5.
Discussion
The results of this study suggest that a multitarget treatment consisting of Panax ginseng extract and ω-3FAs confers a synergistic, or at minimum additive, effect in alleviating Að½ toxicity. This conclusion is supported by the outcomes in Fig. 6, where Group E (combined treatment) displayed 40% and 10% lower rates of paralysis relative to untreated CL2355 worms (Group B) and sole treatment groups (Group C & Group D), respectively. Lower rates of paralysis in AD worms indicate mitigation of Að½ oligomer production and/or attenuation of its deleterious effects (Tangrodchanapong et al., 2023). Although researchers proposed the amyloid cascade theory in 1992 as the guiding framework of AD research, many now deviate from or modify this original hypothesis, forming numerous schools of thought, each backed by tumultuous evidence regarding the causes of AD, such as blood-brain barrier breakdown, mitochondrial dysfunction, and the Aβ Oligomer Hypothesis (Bhatia et al., 2022; Sweeney et al., 2018; Cline et al., 2018). The latter directly relates to interpreting paralysis in the C. elegans model of AD. Put simply, the Aβ Oligomer Hypothesis identifies Aβ oligomers, instead of Aβ plaques, as the key neurotoxic species driving neuroinflammation, neuron damage, and eventually AD pathogenesis. In C. elegans models of AD, researchers typically observe paralysis without visible deposition of Aβ plaques, concluding Aβ oligomers as the primary cause of paralysis (Tangrodchanapong et al., 2023). Over the years, hundreds of trials have identified drugs that work to clear Aβ, but none, with the exception of Donanemab, has improved outcomes to a clinically significant extent. Researchers attribute the relative effectiveness of Donanemab to its unique ability to specifically target neurotoxic forms of Aβ, unlike treatments that target harmless or even protective Aβ species indiscriminately (Rashad et al., 2022). However, with the drug currently costing $32,000 per year and proving effective only in the early stages of AD, it has a long way to go before widespread clinical adoption (Sims et al., 2023). As a combination of Panax ginseng extract and ω-3FAs similarly targets neurotoxic forms of Aβ (i.e., Aβ oligomers), these treatments may serve as a viable alternative with benefits beyond Aβ clearance due to their multitarget nature.
However, even with promising preliminary results, the paralysis assays in the present study are not without their limitations. Firstly, Trial 1 of the paralysis assay (Fig. 3) displays results that contradict those of Trial 2 (Fig. 6). Specifically, Fig 3. shows that the combined treatment of ginseng and ω-3FAs (30% less paralysis relative to Cl2355 control) exerts weaker anti-paralytic effects than the sole treatment of omega-3 (40% less paralysis relative to Cl2355 control). These contradictory results are likely due to the method of ginseng delivery. In the first trial, ginseng was infused within NGM (nematode growth medium) agar instead of being spread across the surface (the delivery method used for omega-3). Initially, this approach aimed to control the treatment volume (i.e., amount of water) across groups and address concerns that volatile ginsenosides might denature upon contact with tween 80 (a surfactant and emulsifier used in the omega-3 treatment solution). Since this delivery system likely impeded proper ginseng absorption, it was replaced with a spreading method in Trial 2, as per prior research. With this change, Trial 2 displayed markedly higher efficacy in both the sole ginseng group and combined treatment (10% and 10% reduction in paralysis in Trial 1, compared to 30% and 40%, respectively). Moreover, this theorized improvement in absorption correlates with the increased population in the Trial 2 ginseng group (Group D; relative to groups within each trial) compared to Trial 1.
Even with this limitation addressed, additional concerns arise regarding the C. elegans strain. Although researchers used Cl2355 for both trials, which demonstrates temperature-induced pan-neuronal expression of Aβ, it ultimately proves nonoptimal for paralysis testing. Initially, researchers of this study chose Cl2355 due to its incomplete sterility (suitable for population assays) and its prior use in a chemotaxis assay (Tangrodchanapong et al., 2020). However, Aβ formation in Cl2355 only partially depends on temperature and thus cannot achieve complete paralysis solely through temperature upshift. In contrast, the CL4176 strain is strictly temperature-dependent and has undergone extensive use in previous studies for testing paralysis. Therefore, future research should use CL4176 to address the limitations of Cl2355.
For both paralysis trials, data significance (p<0.05) could not be determined due to each group having only one sample (n=10/group). Although this experimental design aligns with some previous research, demonstrating significance can allow the experiment to undergo more rigorous scrutiny, thereby improving the validity of findings. To address this concern, future research involving C. elegans paralysis should be performed in triplicate and subject to data analysis through ANOVA.
Similar to paralysis, results of the population assay (Fig. 4 & Fig. 7) also notably differed between trials. Likewise, Trial 1 (Fig. 4) was subject to similar oversights as Trial 1 of paralysis, such as the delivery method of ginseng treatment (Groups D & E). Further exacerbating these limitations, nematodes lacked a consistent food source throughout the experimental duration. Although C. elegans was transferred from agar spotted with OP50, this limited food source may have been depleted through the trial. Considering that both Omega-3 and ginseng are extensively researched to increase metabolism in both human and animal models, OP50 may have been depleted sooner in treatment groups, thereby inducing a starved state (Yu et al., 2021; Yarizadeh et al., 2021). Additionally, group A became contaminated at around day 4, which resulted in a final population lower than that of group B (Cl2355 without treatment). As extensive research shows Cl2355 to possess highly impaired reproductive capabilities, this result ultimately invalidates the outcomes of Trial 1 (Tangrodchanapong et al., 2020). Trial 2 addressed these limitations by changing ginseng administration to spreading and implementing more rigorous sterilization. To that end, the results of Trial 2 (Fig. 6) suggest interventions to have marginal, insignificant effects on restoring reproductive capability and preventing embryonic lethality of Cl2355. Moreover, results regarding groups A & B (Fig. 6) were now consistent with prior research (Tangrodchanapong et al., 2020).
A chemotaxis index assay was performed to further assess the validity of the paralysis assay and, by extension, the neuroprotective properties of ginseng & ω-3FAs. The C. elegans strain CL2355 produces Aβ in the neurons through the synaptobrevin orthologous (snb-1) promoter, leading to flaws in 5-Hydroxytryptamine (5-HT) sensitivity, neuronal manners, and chemotaxis. Figure 5 shows that the nematodes treated with a combined application of ginseng and ω-3FAs had the greatest increase in chemotactic ability out of the groups containing nematodes expressing Aꞵ. The sole ginseng treatment showed similar effects when compared to Group B, the CL2355 group, with no external treatment. These findings indicate that the combined treatment of ginseng and ω-3FAs strengthens the proteostasis of transgenic C. elegans expressing Aꞵ, mitigating the neurodegenerative effects of excessive amyloid-ꞵ deposits.
Despite this, the chemotaxis assay possessed several limitations. Previous studies have suggested that C. elegans at different life stages can respond differently to certain stressors (Margie et al., 2013). Previous research recommends using C. elegans in the young adult stage when analyzing chemotactic ability. Although the nervous system is fully developed in C. elegans at the end of the L1 life stage, there are some intermediate changes within the chemosensory neurons in nematodes at the L4 life stage (Hart, 2006). Earlier studies have used bleach to synchronize C. elegans' life stages; however, bleach synchronization was implemented in this study due to constraints of laboratory equipment available to student researchers. However, variance in nematode life stages was minimized by having a relatively high sample size in each quadrant . Additionally, most studies performing chemotaxis assay apply an anesthetic (i.e. sodium azide) to paralyze C. elegans upon entering the quadrant. However, most, if not all, anesthetics, such as sodium azide, are highly volatile and are quickly absorbed by the skin within a few seconds of exposure, leading to potential poisoning. To mimic the results of anesthetics, a 60-minute video was taken of the chemotaxis plates where the first quadrant that each C. elegans entered was recorded as their final quadrant, and the recorded nematodes were transferred to refrain from rescoring.
C. elegans, as a model of AD, possesses several advantages and disadvantages. Firstly, C elegans lack many hallmark AD factors such as the APOE gene, complex mammalian neurophysiology affected by AD pathogenesis (namely, hippocampus, BBB, cortex, and secretion/utilization of molecules such as BDNF), tau phosphorylation (in models expressing Aβ) and microbiota profile similar to humans (Alvarez et al., 2022). For instance, a recent study from the Journal of Ginseng Research found that restored lactobacillus dominance (a probiotic essential to human health that is not present in C. elegans) plays a critical role in ginseng's ability to ameliorate cognitive defects in mice model of AD (Lee et al., 2022). As mentioned previously, one of the main mechanisms of ω-3FAs' unique ability to prevent AD is its role in strengthening the BBB through mitigating age-related loss of the DHA transporter, MFSD2a. In C. elegans, this mechanism is absent as nematodes lack a BBB.
Nevertheless, C. elegans still possesses valuable features, making it a valuable model of AD. For instance, researchers generally agree that the short lifespan and versatility (ex, temperature-induced Aβ secretion) of C. elegans make it a perfect candidate for rapid testing of novel treatments (Alvarez et al., 2022). When sequenced, 83% of the C. elegans proteome has human homologous genes (Lai et al., 2000). Although C. elegans lacks mammalian-specific genes such as BDNF, it still processes similar evolutionary conserved functions such as klotho production, whose expression is upregulated by ginseng intervention (lim et al., 2019; Château et al., 2010). Additionally, the present study is the first to evaluate the efficacy of ω-3FAs' in the C. elegans model of AD, thereby establishing viability and educating future research. Together, these strengths and weaknesses of C. elegans as a model for Alzheimer's are important considerations when determining the potential of a specific treatment and underscore the importance of further research in higher-order organisms in the case of promising results. To make use of the versatility of C. elegans, future research should explore the effect of combined treatment (Panax ginseng extract and ω-3FAs) on AD models expressing tau protein and gene knockout C. elegans to uncover underlying mechanisms. Additionally, western blot assessing Anti-Ab 6E10 & anti-oligomer A11 antibodies should be performed to confirm the ability of combined treatment to specifically target Aβ oligomers. Conclusion
The results of this study suggest promising preliminary evidence that combined treatments consisting of Panax ginseng extract and ω-3FAs may enhance individual effects and optimize benefits. Together, findings suggest that the synergistic or at least additive effects of the treatments can potentially lead to better managing of Að½ toxicity, damage, and the harmful effects of Að½ oligomers. The present study also was the first to confirmed the efficacy of ω-3FAs' in the mitigating Að½ toxicity in the C. elegans model of AD, however mechanisms behind its effects has yet to be uncovered. As millions of worldwide cases of AD are expected to grow; with expensive drugs only effective at the early stages of AD, exploring treatments' potential as viable therapeutic strategies are of utmost importance. Future research should focus on performing similar studies on additional C. elegans strains, and the use of these treatments on more complex models prior to widespread clinical adoption.
References
Alvarez, J., Alvarez-Illera, P., Santo-Domingo, J., Fonteriz, R. I., & Montero, M. (2022).
Modeling Alzheimer's disease in Caenorhabditis elegans. Biomedicines, 10(2), 1-27. https://doi.org/10.3390/biomedicines10020288
Alexander, A. G., Marfil, V., & Li, C. (2014). Use of caenorhabditis elegans as a model to study
Alzheimer’s disease and other neurodegenerative diseases. Frontiers in Genetics, 5, 1-21 https://doi.org/10.3389/fgene.2014.00279
Anstey, K. J., Cherbuin, N., Herath, P. M., Qiu, C., Kuller, L. H., Lopez, O. L., ... & Fratiglioni, L. (2014). A self-report risk index to predict occurrence of dementia in three independent cohorts of older adults: the ANU-ADRI. PloS one, 9(1), 1-8.
Ajith T. A. (2018). A Recent Update on the Effects of Omega-3 Fatty Acids in Alzheimer's
Disease. Current clinical pharmacology, 13(4), 252–260. https://doi.org/10.2174/1574884713666180807145648
Arellanes, I. C., Choe, N., Solomon, V., He, X., Kavin, B., Martinez, A. E., Kono, N.,
Buennagel, D. P., Hazra, N., Kim, G., D'Orazio, L. M., McCleary, C., Sagare, A., Zlokovic, B. V., Hodis, H. N., Mack, W. J., Chui, H. C., Harrington, M. G., Braskie, M. N., Schneider, L. S., Yassine, H. N. (2020). Brain delivery of supplemental docosahexaenoic acid (DHA): A randomized placebo-controlled clinical trial. EBioMedicine, 59, 1-15. https://doi.org/10.1016/j.ebiom.2020.102883
Avallone, R., Vitale, G., & Bertolotti, M. (2019). Omega-3 fatty acids and neurodegenerative
diseases: New evidence in clinical trials. International Journal of Molecular Sciences,
20(17), 4256. https://doi.org/10.3390/ijms20174256
Ba, M., Kong, M., Li, X., Ng, K. P., Rosa-Neto, P., & Gauthier, S. (2016). Is ApoE ɛ 4 a good
biomarker for amyloid pathology in late onset Alzheimer's disease?. Translational neurodegeneration, 5, 20. https://doi.org/10.1186/s40035-016-0067-z
Brown, S. P., Mathur, B. N., Olsen, S. R., Luppi, P. H., Bickford, M. E., & Citri, A. (2017).
New Breakthroughs in Understanding the Role of Functional Interactions between the Neocortex and the Claustrum. Journal of neuroscience, 37(45), 10877–10881.
https://doi.org/10.1523/JNEUROSCI.1837-17.2017
Belloy, M. E., Napolioni, V., & Greicius, M. D. (2019). A Quarter Century of APOE and
Alzheimer's Disease: Progress to Date and the Path Forward. Neuron, 101(5), 820–838. https://doi.org/10.1016/j.neuron.2019.01.056
Bredesen D. E. (2016). Inhalational Alzheimer's disease: an unrecognized - and treatable -
epidemic. Aging, 8(2), 304–313. https://doi.org/10.18632/aging.100896
Bhatia, S., Rawal, R., Sharma, P., Singh, T., Singh, M., & Singh, V. (2022). Mitochondrial
Dysfunction in Alzheimer's Disease: Opportunities for Drug Development. Current neuropharmacology, 20(4), 675–692. https://doi.org/10.2174/1570159X19666210517114016
Cline, E. N., Bicca, M. A., Viola, K. L., & Klein, W. L. (2018). The Amyloid-β Oligomer
Hypothesis: Beginning of the Third Decade. Journal of Alzheimer's Disease, 64(1), 567–610. https://doi.org/10.3233/JAD-179941
Château, M. T., Araiz, C., Descamps, S., & Galas, S. (2010). Klotho interferes with a novel
FGF-signalling pathway and insulin/Igf-like signalling to improve longevity and stress resistance in Caenorhabditis elegans. Aging, 2(9), 567–581. https://doi.org/10.18632/aging.100195
Dighriri, I. M., Alsubaie, A. M., Hakami, F. M., Hamithi, D. M., Alshekh, M. M., Khobrani, F.
A., Dalak, F. E., Hakami, A. A., Alsueaadi, E. H., Alsaawi, L. S., Alshammari, S. F., Alqahtani, A. S., Alawi, I. A., Aljuaid, A. A., & Tawhari, M. Q. (2022). Effects of Omega-3 Polyunsaturated Fatty Acids on Brain Functions: A Systematic Review. Cureus, 14(10), 1-13. https://doi.org/10.7759/cureus.30091
Deng, X., Zhao, S., Liu, X., Han, L., Wang, R., Hao, H., Jiao, Y., Han, S., & Bai, C. (2020).
Polygala tenuifolia: a source for anti-Alzheimer's disease drugs. Pharmaceutical biology, 58(1), 410–416. https://doi.org/10.1080/13880209.2020.1758732.
Francos-Quijorna, I., Santos-Nogueira, E., Gronert, K., Sullivan, A. B., Kopp, M. A., Brommer,
B., David, S., Schwab, J. M., & López-Vales, R. (2017). Maresin 1 Promotes Inflammatory Resolution, Neuroprotection, and Functional Neurological Recovery After Spinal Cord Injury. The Journal of neuroscience : the official journal of the Society for Neuroscience, 37(48), 11731–11743. https://doi.org/10.1523/JNEUROSCI.1395-17.201
Freund Levi, Y., Vedin, I., Cederholm, T., Basun, H., Faxén Irving, G., Eriksdotter, M., Hjorth,
E., Schultzberg, M., Vessby, B., Wahlund, L. O., Salem, N., Jr, & Palmblad, J. (2014). Transfer of omega-3 fatty acids across the blood-brain barrier after dietary supplementation with a docosahexaenoic acid-rich omega-3 fatty acid preparation in patients with Alzheimer's disease: the OmegAD study. Journal of internal medicine, 275(4), 428–436. https://doi.org/10.1111/joim.12166
Freund-Levi, Y., Eriksdotter-Jönhagen, M., Cederholm, T., Basun, H., Faxén-Irving, G.,
Garlind, A., Vedin, I., Vessby, B., Wahlund, L. O., & Palmblad, J. (2006). Omega-3 fatty acid treatment in 174 patients with mild to moderate Alzheimer disease: OmegAD study: a randomized double-blind trial. Archives of neurology, 63(10), 1402–1408. https://doi.org/10.1001/archneur.63.10.1402
Hart, A.C., ed. Behavior (2006). WormBook, ed. The C. elegans Research
Community, WormBook, doi/10.1895/wormbook.1.87.1, http://www.wormbook.org.
Heo, J. H., Lee, S. T., Oh, M. J., Park, H. J., Shim, J. Y., Chu, K., & Kim, M. (2011). Improvement of cognitive deficit in Alzheimer’s disease patients by long term treatment with Korean red ginseng. Journal of ginseng research, 35(4), 457.
Heo, J. H., Park, M. H., & Lee, J. H. (2016). Effect of Korean red ginseng on cognitive function and quantitative EEG in patients with Alzheimer's disease: a preliminary study. The Journal of Alternative and Complementary Medicine, 22(4), 280-285.
Heneka, M. T., Carson, M. J., El Khoury, J., Landreth, G. E., Brosseron, F., Feinstein, D. L.,
Jacobs, A. H., Wyss-Coray, T., Vitorica, J., Ransohoff, R. M., Herrup, K., Frautschy, S. A., Finsen, B., Brown, G. C., Verkhratsky, A., Yamanaka, K., Koistinaho, J., Latz, E., Halle, A., Petzold, G. C.,Kummer, M. P. (2015). Neuroinflammation in Alzheimer's disease. The Lancet. Neurology, 14(4), 388–405. https://doi.org/10.1016/S1474-4422(15)70016-5
Kim, H. J., Jung, S. W., Kim, S. Y., Cho, I. H., Kim, H. C., Rhim, H., ... & Nah, S. Y. (2018).
Panax ginseng as an adjuvant treatment for Alzheimer’s disease. Journal of ginseng research, 42(4), 401-411.
Kim, J., Kim, S. H., Lee, D. S., Lee, D. J., Kim, S. H., Chung, S., & Yang, H. O. (2013). Effects of fermented ginseng on memory impairment and β-amyloid reduction in Alzheimer’s disease experimental models. Journal of ginseng research, 37(1), 100.
Kodis, E. J., Choi, S., Swanson, E., Ferreira, G., & Bloom, G. S. (2018). N-methyl-D-aspartate
receptor–mediated calcium influx connects amyloid-β oligomers to ectopic neuronal cell cycle reentry in Alzheimer's disease. Alzheimer's & Dementia, 14(10), 1302-1312.
Kris-Etherton, P. M., Taylor, D. S., Yu-Poth, S., Huth, P., Moriarty, K., Fishell, V., Hargrove, R. L., Zhao, G., & Etherton, T. D. (2000). Polyunsaturated fatty acids in the food chain in the United States. The American journal of clinical nutrition, 71(1), 179–188. https://doi.org/10.1093/ajcn/71.1.179S
Lee, B. C., Choe, Y. M., Suh, G. H., Choi, I. G., Kim, H. S., Hwang, J., Yi, D., Jhoo, J. H., &
Kim, J. W. (2023). Ginseng intake and Alzheimer disease-specific cognition in older adults according to apolipoprotein ε4 allele status. Frontiers in aging neuroscience, 15, 1-11. https://doi.org/10.3389/fnagi.2023.1152626
Lee, J., Lee, A., Kim, J. H., Shin, Y. M., Kim, S. J., Cho, W. D., & Lee, S. I. (2020). Effect of
Omega-3 and Korean Red Ginseng on Children with Attention Deficit Hyperactivity Disorder: An Open-Label Pilot Study. Clinical psychopharmacology and neuroscience, 18(1), 75–80. https://doi.org/10.9758/cpn.2020.18.1.75
Lee, M. R., Yun, B. S., In, O. H., & Sung, C. K. (2011). Comparative study of Korean white, red, and black ginseng extract on cholinesterase inhibitory activity and cholinergic function. Journal of Ginseng research, 35(4), 421-428. 10.5142/jgr.2011.35.4.421
LeVine, S.M. (2016). Albumin and multiple sclerosis. BMC Neurol, 16(47) . https://doi.org/10.1186/s12883-016-0564-9
Liu, H., Lu, X., Hu, Y., & Fan, X. (2020). Chemical constituents of Panax ginseng and Panax
Notoginseng explains why they differ in therapeutic efficacy. Pharmacological research, 161, 1-17. https://doi.org/10.1016/j.phrs.2020.105263
Liu, J., Chang, L., Song, Y., Li, H., & Wu, Y. (2019). The role of NMDA receptors in
Alzheimer’s disease. Frontiers in neuroscience, 13(43), 1-22. https://doi.org/10.3389/fnins.2019.00043
Lai, C. H., Chou, C. Y., Ch'ang, L. Y., Liu, C. S., & Lin, W. (2000). Identification of novel
human genes evolutionarily conserved in Caenorhabditis elegans by comparative proteomics. Genome research, 10(5), 703–713. https://doi.org/10.1101/gr.10.5.70
Lee, M., Lee, S.-H., Kim, M.-S., Ahn, K.-S., & Kim, M. (2022). Effect of Lactobacillus
dominance modified by Korean Red Ginseng on the improvement of Alzheimer’s disease in mice. Journal of Ginseng Research, 46(3), 464–472.[1]https://doi.org/10.1016/j.jgr.2021.11.001
Lim, S. W., Shin, Y. J., Luo, K., Quan, Y., Cui, S., Ko, E. J., Chung, B. H., & Yang, C. W.
(2019). Ginseng increases Klotho expression by FoxO3-mediated manganese superoxide dismutase in a mouse model of tacrolimus-induced renal injury. Aging, 11(15), 5548–5569. https://doi.org/10.18632/aging.102137
Margie, O., Palmer, C., & Chin-Sang, I. (2013). C. elegans chemotaxis assay. Journal of
visualized experiments: JoVE, (74), 50069.
Mark, L. P., Prost, R. W., Ulmer, J. L., Smith, M. M., Daniels, D. L., Strottmann, J. M., ... &
Hacein-Bey, L. (2001). Pictorial review of glutamate excitotoxicity: fundamental concepts for neuroimaging. American journal of neuroradiology, 22(10), 1813-1824.
Miranda, M., Morici, J. F., Zanoni, M. B., & Bekinschtein, P. (2019). Brain-Derived
Neurotrophic Factor: A Key Molecule for Memory in the Healthy and the Pathological Brain. Frontiers in cellular neuroscience, 13, 1-25.
https://doi.org/10.3389/fncel.2019.00363
Power, R., Nolan, J. M., Prado-Cabrero, A., Roche, W., Coen, R., Power, T., & Mulcahy, R.
(2022). Omega-3 fatty acid, carotenoid and vitamin E supplementation improves working memory in older adults: A randomized clinical trial. Clinical nutrition, 41(2), 405–414. https://doi.org/10.1016/j.clnu.2021.12.004
Patrick R. P. (2019). Role of phosphatidylcholine-DHA in preventing APOE4-associated
Alzheimer's disease. FASEB journal, 33(2), 1554–1564. https://doi.org/10.1096/fj.201801412R
Parhizkar, S., & Holtzman, D. M. (2022). APOE mediated neuroinflammation and
neurodegeneration in Alzheimer's disease. Seminars in immunology, 59, 1-10. https://doi.org/10.1016/j.smim.2022.101594
Park, Y. H., Shin, S. J., Kim, H. soo, Hong, S. B., Kim, S., Nam, Y., Kim, J.-J., Lim, K., Kim,
J.-S., Kim, J., Jeon, S. G., & Moon, M. (2020). Omega-3 fatty acid-type docosahexaenoic acid protects against AΒ-mediated mitochondrial deficits and pathomechanisms in Alzheimer's disease-related animal model. International Journal of Molecular Sciences, 21(11), 1-21. https://doi.org/10.3390/ijms21113879
Paoletti, P. (2011). Molecular basis of NMDA receptor functional diversity. European Journal
of Neuroscience, 33(8), 1351-1365.
Platt, M. P., Agalliu, D., & Cutforth, T. (2017). Hello from the Other Side: How Autoantibodies Circumvent the Blood-Brain Barrier in Autoimmune Encephalitis. Frontiers in immunology, 8, 1-15. https://doi.org/10.3389/fimmu.2017.00442
Qiu, C., Kivipelto, M., & Von Strauss, E. (2009). Epidemiology of Alzheimer's disease:
occurrence, determinants, and strategies toward intervention. Dialogues in clinical neuroscience, 11(2), 111-128.
Qi, W., Gutierrez, G. E., Gao, X., Dixon, H., McDonough, J. A., Marini, A. M., & Fisher, A. L.
(2017). The ω-3 fatty acid α-linolenic acid extends Caenorhabditis elegans lifespan via NHR-49/PPARα and oxidation to oxylipins. Aging cell, 16(5), 1125–1135. https://doi.org/10.1111/acel.12651
Quinn, J. F., Raman, R., Thomas, R. G., Yurko-Mauro, K., Nelson, E. B., Van Dyck, C., Galvin, J. E., Emond, J., Jack, C. R., Jr, Weiner, M., Shinto, L., & Aisen, P. S. (2010). Docosahexaenoic acid supplementation and cognitive decline in Alzheimer disease: a randomized trial. JAMA, 304(17), 1903–1911. https://doi.org/10.1001/jama.2010.1510
Rashad, A., Rasool, A., Shaheryar, M., Sarfraz, A., Sarfraz, Z., Robles-Velasco, K., &
Cherrez-Ojeda, I. (2022). Donanemab for Alzheimer's Disease: A Systematic Review of Clinical Trials. Healthcare, 11(1), 1-17. https://doi.org/10.3390/healthcare11010032
Sweeney, M., Sagare, A. & Zlokovic, B. (2018). Blood–brain barrier breakdown in Alzheimer
disease and other neurodegenerative disorders. Nature Reviews Neurology, 14, 133–150.
https://doi.org/10.1038/nrneurol.2017.188
Sims, J. R., Zimmer, J. A., Evans, C. D., Lu, M., Ardayfio, P., Sparks, J., Wessels, A. M.,
Shcherbinin, S., Wang, H., Monkul Nery, E. S., Collins, E. C., Solomon, P., Salloway, S., Apostolova, L. G., Hansson, O., Ritchie, C., Brooks, D. A., Mintun, M., Skovronsky, D. M., & TRAILBLAZER-ALZ 2 Investigators. (2023). Donanemab in Early Symptomatic Alzheimer Disease: The TRAILBLAZER-ALZ 2 Randomized Clinical Trial. JAMA, 330(6), 512–527. https://doi.org/10.1001/jama.2023.13239
Sadigh-Eteghad, S., Sabermarouf, B., Majdi, A., Talebi, M., Farhoudi, M., & Mahmoudi, J. (2015). Amyloid-beta: a crucial factor in Alzheimer's disease. Medical principles and practice, 24(1), 1-10.
Shi, H., Zhao, J., Li, Y., Li, J., Li, Y., Zhang, J., Qiu, Z., Wu, C., Qin, M., Liu, C., Zeng, Z.,
Zhang, C., & Gao, L. (2023). Ginsenosides Rg1 regulate lipid metabolism and temperature adaptation in Caenorhabditis elegans. Journal of ginseng research, 47(4), 524–533. https://doi.org/10.1016/j.jgr.2022.11.005
Sharma, K. (2019). Cholinesterase inhibitors as Alzheimer's therapeutics. Molecular medicine reports, 20(2), 1479-1487.
Sofela, S., Sahloul, S., & Song, Y.-A. (2021). Biophysical analysis of drug efficacy on C.
elegans models for neurodegenerative and neuromuscular diseases. PLOS ONE, 16(6). https://doi.org/10.1371/journal.pone.0246496
Soni, D., & Galluci, L. (2023). The Causes and Effects of Alzheimer's Disease. Journal of Student Research, 12(2). 41-68. https://doi.org/10.47611/jsrhs.v12i2.4168
Tangrodchanapong, T., Sobhon, P., & Meemon, K. (2020). Frondoside A Attenuates Amyloid-β
Proteotoxicity in Transgenic Caenorhabditis elegans by Suppressing Its Formation. Frontiers in pharmacology, 11, 1-12. https://doi.org/10.3389/fphar.2020.553579
Valente, M., Dentoni, M., Bellizzi, F., Kuris, F., & Gigli, G. L. (2022). Specialized
Pro-Resolving Mediators in Neuroinflammation: Overview of Studies and Perspectives of Clinical Applications. Molecules, 27(15), 1-25. https://doi.org/10.3390/molecules27154836
Wei, B. Z., Li, L., Dong, C. W., Tan, C. C., Alzheimer’s Disease Neuroimaging Initiative, & Xu, W. (2023). The Relationship of Omega-3 Fatty Acids with Dementia and Cognitive Decline: Evidence from Prospective Cohort Studies of Supplementation, Dietary Intake, and Blood Markers. The American journal of clinical nutrition, 117(6), 1096–1109. https://doi.org/10.1016/j.ajcnut.2023.04.001
Wood, A. H., Chappell, H. F., & Zulyniak, M. A. (2021). Dietary and supplemental long-chain
omega-3 fatty acids as moderators of cognitive impairment and Alzheimer's disease. European Journal of Nutrition, 61(2), 589–604. https://doi.org/10.1007/s00394-021-02655-4
Wang, Z., Zhang, Z., Liu, J., Guo, M., & Li, H. (2023). Panax Ginseng in the treatment of
Alzheimer's disease and vascular dementia. Journal of ginseng research, 47(4), 506–514. https://doi.org/10.1016/j.jgr.2023.03.001
Weinstein, G., Beiser, A. S., Choi, S. H., Preis, S. R., Chen, T. C., Vorgas, D., Au, R., Pikula,
A., Wolf, P. A., DeStefano, A. L., Vasan, R. S., & Seshadri, S. (2014). Serum brain-derived neurotrophic factor and the risk for dementia: the Framingham Heart Study. JAMA neurology, 71(1), 55–61. https://doi.org/10.1001/jamaneurol.2013.4781
Wierenga, K. A., & Pestka, J. J. (2021). Omega-3 Fatty Acids And Inflammation - You Are
What You Eat!. Frontiers for young minds, 9, 601068.
https://doi.org/10.3389/frym.2021.601068
Yang, A. C., Stevens, M. Y., Chen, M. B., Lee, D. P., Stähli, D., Gate, D., Contrepois, K., Chen,
W., Iram, T., Zhang, L., Vest, R. T., Chaney, A., Lehallier, B., Olsson, N., du Bois, H., Hsieh, R., Cropper, H. C., Berdnik, D., Li, L., Wang, E. Y., … Wyss-Coray, T. (2020). Physiological blood-brain transport is impaired with age by a shift in transcytosis. Nature, 583, 425–430. https://doi.org/10.1038/s41586-020-2453-z
Yu, X., Li, H., Lin, D., Guo, W., Xu, Z., Wang, L., & Guan, S. (2021). Ginsenoside Prolongs the Lifespan of C. elegans via Lipid Metabolism and Activating the Stress Response Signaling Pathway. International journal of molecular sciences, 22(18), 1-18. https://doi.org/10.3390/ijms22189668
Yamazaki, Y., Zhao, N., Caulfield, T. R., Liu, C. C., & Bu, G. (2019). Apolipoprotein E and Alzheimer disease: pathobiology and targeting strategies. Nature reviews. Neurology, 15(9), 501–518. https://doi.org/10.1038/s41582-019-0228-7
Yarizadeh, H., Hassani, B., Nosratabadi, S., Baharlooi, H., Asadi, S., Bagherian, S. A., Islam,
S., Djafarian, K., & Mirzaei, K. (2021). The Effects of Omega-3 Supplementation on Resting Metabolic Rate: A Systematic Review and Meta-Analysis of Clinical Trials. Evidence-based complementary and alternative medicine, 2021(1), 1-9. https://doi.org/10.1155/2021/6213035
Zhang, M., Qian, F., Liu, Q., Qian, C., Thu, P. M., Wang, Y., ... & Xu, X. (2017). Evaluation of
structure–activity relationships of ginsenosides against amyloid β induced pathological behaviors in transgenic Caenorhabditis elegans. RSC advances, 7(64), 40095-40104. DOI: 10.1039/c7ra05717b
Zhang, S., Liu, F., Li, J., Jing, C., Lu, J., Chen, X., Wang, D., Cao, D., Zhao, D., & Sun, L. (2023). A 4.7-KDA polysaccharide from Panax ginseng suppresses AΒ pathology via mitophagy activation in cross-species Alzheimer's disease models. Biomedicine & Pharmacotherapy, 167, 1-15. https://doi.org/10.1016/j.biopha.2023.115442
Zhou, L., Huang, P. P., Chen, L. L., & Wang, P. (2019). Panax notoginseng saponins ameliorate
Aβ-mediated neurotoxicity in C. elegans through antioxidant activities. Evidence-Based Complementary and Alternative Medicine, 1, 1-8.
https://doi.org/10.1155/2019/7621043
Zhang, Z., Liu, X., Schroeder, J. P., Chan, C. B., Song, M., Yu, S. P., Weinshenker, D., & Ye, K. (2014). 7,8-dihydroxyflavone prevents synaptic loss and memory deficits in a mouse model of Alzheimer's disease. Neuropsychopharmacology, 39(3), 638–650. https://doi.org/10.1038/npp.2013.243
Ziaei, S., Mohammadi, S., Hasani, M., Morvaridi, M., Belančić, A., Daneshzad, E., Saleh, S. A.
K., Adly, H. M., & Heshmati, J. (2024). A systematic review and meta-analysis of the omega-3 fatty acids effects on brain-derived neurotrophic factor (BDNF). Nutritional neuroscience, 27(7), 715–725. https://doi.org/10.1080/1028415X.2023.2245996
Document information
Published on 09/03/26
Submitted on 20/02/25
Volume 8, 2026
Licence: CC BY-NC-SA license
Share this document
Keywords
claim authorship
Are you one of the authors of this document?