Abstract
Biochar (BC), a by-product from the fast pyrolysis of rice husk, was activated by chemical and physical process. The activated biochar (ABC) showed graphite-like morphology and had a large amount of random pores with BET surface area of 1058 m2/g. The biochar properties observed by further characterization revealed its feasibility to be used as a catalyst support. Syngas methanation as a probe reaction was utilized to characterize biochar supported Ru catalyst. The catalytic performance of the Ru/ABC catalyst in methanation was superior or comparable to the conventional activated carbon (AC) supported Ru catalyst. A high CH4 selectivity of 98% and a CO conversion of 100% were obtained under the proper reaction conditions over the Ru/ABC catalyst.
Introduction
Biochar (BC), a residual by-product from fast pyrolysis of lingo-cellulosic biomass for bio-oil production, is rich in carbon, up to 60 wt%, and typically accounts for 15–40 wt% of the biomass feedstock [1]. At present, the majority of BC is discarded, utilized as process fuel or sold commercially for soil amendment and carbon sequestration [2-4]. In general, BC is similar to graphite. It is mainly composed of the aromatic carbon rings linked together. However, these rings are irregularly stacked and randomly arranged [5]. As with activated carbon (AC), the surface chemistry of BC can be varied using chemical methods. As a consequence, BC, which is an abundant and low-cost renewable carbon source, has great potential to be used as a catalyst or catalyst support. In addition, BC can be easily separated from catalysts by oxidation to recover precious metals [6]. Therefore, the utilization of BC as catalyst will not only enhance its utilization but also promote the development of a variety of catalysts. Yan et al. [7] applied BC in the synthesis of carbon-encapsulated iron nanoparticles, and its catalytic activity on the conversion of synthesis gas to liquid hydrocarbons was evaluated. It was found that the CO conversion was about 95% and the selectivity of liquid hydrocarbon was as high as 68%. Dehkhoda et al. and Yu et al. [5, 8] reported the similar results that the BC, activated with KOH and sulfonated using fuming H2SO4, had a high activity on the transesterification of canola oil with methanol because of its high surface area and large acid density. To date, there have been few reports on the utilization of activated biochar (ABC) as a support for a heterogeneous catalyst.
It is known that a well-developed pore structure and high surface area, which are likely to favor the uniform dispersion of active components and to stabilize these against sintering, are essential for a good catalyst support. Since the original BC has a low density of pores, an activation process is required before it can be used as a support [9].
The methanation reaction of carbon monoxide and hydrogen has received considerable attention since it was first reported by Sabatier and Senderens [10]. By methanation process, syngas, derived from coal and biomass gasification and other kinds of methods such as coke oven gas, can be converted to substitute natural gas (SNG). Besides used as fuel gas, SNG in liquefied or compressed form can be used in transport vehicles as a substitute for diesel and gasoline. In the past decades, the selective methanation of CO from syngas was developed and has been widely applied in the gas purification process, where the content of CO in a syngas can be reduced to a very low level [11, 12]. As for the many fuels and chemicals synthesis from syngas, methanation is one of the reactions which have simple products. Syngas can be converted to methanol by methanol synthesis process and further converted to gasoline using methanol-to-gasoline process. Syngas is also used as a feed in producing synthetic petroleum such as gasoline and diesel via Fischer–Tropsch process. Herein, methanation was studied as a probe reaction for the catalyst support application of ABC.
Over recent decades, considerable effort has been made to develop efficient catalysts for syngas methanation [13, 14]. Carbonaceous materials, such as activated carbon (AC) and carbon nanotubes, have recently received a great deal of attention as catalysts or supports for the production of hydrocarbons. They show excellent catalytic activity, due to their unique magnetic and electric properties and their high mechanical and chemical stabilities [1, 15, 16].
The Ru-based catalyst is the most active for syngas methanation [11, 13, 17]. Compared to cobalt and iron, ruthenium catalysts have some unique features in the methanation process, and are suitable for fundamental research [18]. In this study, the BC, produced by rice husk pyrolysis, was activated to make it a suitable catalyst support. Methanation of syngas was selected just as a probe reaction to investigate the properties of ABC for the catalyst support application. And methanation over AC supported Ru catalyst was studied for the comparison. We hope to develop the utilization of biochar in catalytic field in our future work based on this exploration.
Experimental
BC activation and catalyst preparation
The original BC was produced by the fast pyrolysis of rice husk at 773 K. It was activated before being used as a support, in order to increase the surface area and porosity. Under stirring, original BC was impregnated in 5 mol/L KOH aqueous solution for 4 h at ambient temperature, and then filtered, and dried at 383 K overnight. The sample was calcined in a tube furnace for 2 h under a flow of N2, while the temperature was raised to 973 K at 5 K/min. After the activation, the sample was thoroughly washed with deionized water, and impregnated with 0.1 mol/L HCl aqueous solution for 3 h. Finally, it was filtered and washed with deionized water to remove soluble salts until the neutral pH value was achieved, and then dried at 383 K overnight to obtain ABC.
The as-prepared ABC and commercial AC supports were separately refluxed in the 30% aqueous solution of HNO3 for 6 h at 373 K. Then, the mixture was filtered, washed, and dried as described above. The pretreated support (5 g) was added to 20 mL aqueous solution of RuCl3 with a concentration of 0.124 mol/L. The suspension was agitated ultrasonically for 1 h, and then stirred for another 5 h at ambient temperature. Subsequently, the samples were filtered and dried at 383 K overnight, to give the Ru/ABC and Ru/AC catalysts with a Ru loading of 5 wt%.
Catalyst characterization
The pore structures of the samples were determined from nitrogen adsorption–desorption isotherms obtained at 77 K using a Quantachrome Quadrasorb SI apparatus (Quantachrome Instruments, Boynton Beach, FL, USA). The specific surface area was calculated based on the multipoint BET method. Powder X-ray diffraction (XRD) analysis of the catalysts was carried out using a PANalytical X'Pert PRO X-ray diffractometer with a Cu Kα radiation source operating at 40 kV and 30 mA. The morphology of the samples was observed using a transmission electron microscope (TEM), Philips-FEI Tecnai G2F30 (Philips-FEI Corp., Amsterdam, Netherlands). Temperature-programmed reduction of hydrogen (H2-TPR) was conducted on a Micromeritics AutoChem II 2920 instrument (Micromeritics Instrument Corp., Norcross, GA, USA). The catalysts were pretreated with Ar at 473 K for 60 min and then cooled to 323 K. The TPR process was carried out by heating to 1173 K at a rate of 10 K/min in a mixture of 10% H2/Ar. Temperature-programmed desorption of carbon monoxide (CO-TPD) was also conducted on the AutoChem II 2920 instrument. Dispersion of Ru was calculated from the moles of CO desorbed by assuming a CO/Ru stoichiometry of 1.
Test of catalytic activity
The methanation reactions were carried out in a continuous-flow fixed-bed reactor. In each experiment, 3 mL(0.65 g for Ru/ABC, 1.95 g for Ru/AC) of catalyst was sandwiched in quartz sand, which was then packed into a steel tube reactor with an inner diameter of 8 mm. Before the reaction, the catalyst was activated with pure H2 at 723 K for 5 h, ramping at a rate of 3 K/min, then cooling to the reaction temperature. The flow rate of simulated syngas was controlled with a precise mass flowmeter (Brooks 5850E, Brooks Instrument, Hatfield, PA, USA). The reaction temperature was monitored with a K-type thermocouple inserted into the center of the catalyst bed. Typically, the methanation reaction was carried out at 2 MPa, H2/CO mole ratio of 3, and gas hourly space velocity (GHSV) of 1200 h−1, unless otherwise specified.
The products were analyzed using an on-line gas chromatograph (GC) (Agilent 6820, Agilent Technologies, Santa Clara, CA, USA). H2, CO, CO2 were separated using a TDX-01 packed column (0.5 m) and then detected by a thermal conductivity detector. The hydrocarbons were separated by a Propack-Q packed column (1 m) and then detected by a flame ionization detector.
The CO conversion and product selectivity were calculated relative to the number of moles of C, according to the following equations:
|
|
|
|
where XCO and refer to the CO conversion and the product selectivity. and represent the number of moles of CO in the feed and vent gas, respectively, and m represents the carbon number of the product Cm . refers to the number of moles of Cm .
Control experiments were carried out with simple ABC and AC using the condition 613 K, 2 MPa, H2/CO mole ratio of 3, and gas hourly space velocity (GHSV) of 1200 h−1. The conversion of CO over the ABC and AC was zero. Therefore, pure ABC or AC has no catalytic effect on the methanation.
Results and Discussion
Characterization of the support and catalyst
Nitrogen adsorption−desorption measurements were used to evaluate the textural properties of the samples. The BET surface area, pore volume and the average pore size of the BC before and after activation, the AC, as well as the Ru/ABC and Ru/AC catalysts are presented in Table 1.
| Catalyst | SBET (m2/g) | SExternal (m2/g) | SMicro (m2/g) | VPore (cm3/g) | DPore (nm) | dRu (nm)b | Ru dispersion (%)c |
|---|---|---|---|---|---|---|---|
| |||||||
| BC | 40 | 30 | 10 | 0.06 | 5.5 | – | – |
| ABC | 1058 | 225 | 833 | 0.59 | 2.3 | – | – |
| Ru/ABC | 806 | 240 | 566 | 0.58 | 2.9 | 3.3 | 36 |
| AC | 653 | 31 | 622 | 0.35 | 2.3 | – | – |
| Ru/AC | 558 | 24 | 534 | 0.34 | 2.4 | 1.9 | 41 |
The original BC showed a small BET surface area (40 m2/g), which was in accordance with the reported value [19]. It is indicated that only a few pores were generated during the fast pyrolysis, because of the incomplete pyrolysis of biomass in a short residence time and the entrapment of tar-like materials within the pores. During the chemical activation, further disruption of the carbon lattice and the release of the volatiles in the BC facilitated the enlargement of the existing pores and the generation of new ones [5]. The ABC showed a BET surface area of 1058 m2/g, some 25 times higher than that of the original BC. The average pore diameter of BC decreased from 5.5 to 2.3 nm after activation, which indicated that most of the new generated pores had smaller size than the original ones. In addition, it can be inferred from Table 1 that the surface area of the micropores accounts for ~78% of the total surface area in the ABC, and the value is 95% in the commercial AC. It indicated that microporous and mesoporous structures coexisted in ABC and its supported catalysts, while the AC and the Ru/AC catalyst showed the microporous structure. For a catalyst support, the coexistence is better than having a solely microporous structure, since any diffusion limitation in the reaction can be relieved and the mesopores can act as passages, allowing reactant and products to pass in and out of the interior of the catalyst [19, 20]. The BET surface area of the Ru/ABC and Ru/AC catalyst was lower than that of the ABC and AC, respectively, suggesting that some of the pores of the support had been blocked by active components. In addition, the BET surface area of the micropores was significantly reduced, while that of the mesopores remained almost unchanged. It is deduced that the size of the active components was small enough to enter the micropores of the support [21].
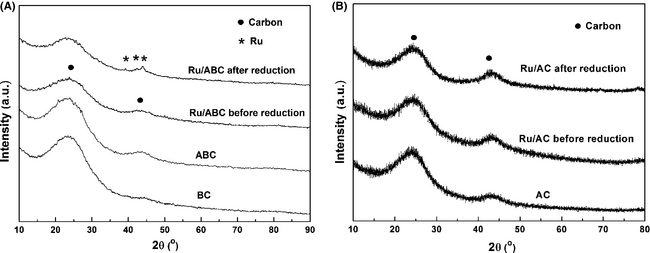
The XRD patterns of the BC before and after activation, the AC, as well as the catalysts before and after reduction are depicted in Figure 1. All the XRD patterns showed two wide diffraction peaks corresponding to the characteristic peaks of carbon. The strongest diffraction peaks, located at 2θ = 20°−30°, are indicative of the amorphous carbon structure. The weaker diffraction peak at around 2θ = 40°−50° is assigned to the reflection from graphite. The presence of more obvious graphite-like structure in the ABC indicates that this is the more stable state for the carbonaceous material [5, 22]. No diffraction peaks of active components were detected on the Ru/ABC and Ru/AC catalysts before reduction. After reduction, the diffraction peaks emerged at 2θ = 38.4°, 42.2°, and 44.0° over the Ru/ABC catalyst. These are assigned to the crystal planes of Ru (1 0 0), (0 0 2), and (1 0 1), respectively, indicating that the RuCl3 precursor has been transformed to metallic Ru [16]. However, there was no Ru diffraction peak observed over the reduced Ru/AC catalyst, mostly because of their small particle size and uniform distribution. The stronger Ru diffraction peaks on the reduced Ru/ABC catalyst than that on the Ru/AC catalyst indicated the larger Ru crystallite size on the ABC.
|
|
|
Figure 1. XRD patterns of (A) BC and BC supported catalysts and (B) AC and AC supported catalysts. |
The structure of the supports and the distribution of active components were analyzed by TEM, shown in Figure 2. TEM images showed that large numbers of random pores were observed on the ABC, owing to the release of volatiles and the reaction between the BC and the activation agent [23]. The presence of the metallic Ru, Ru (0 0 2), and Ru (1 0 0) on the Ru/ABC and Ru/AC catalysts, respectively, was verified by its interplanar crystal spacing on the high-resolution transmission electron microscope (HRTEM) images. Ru particles were uniformly distributed on both of the reduced Ru/ABC and Ru/AC catalysts. The average particle size of the metallic Ru was 3.3 nm for the Ru/ABC catalyst, while a smaller value of 1.9 nm was observed on the Ru/AC catalyst (Table 1). This was calculated from the TEM images by , where ni is the number of particles having a characteristic diameter di (within a given diameter range) [24].
|
|
|
Figure 2. TEM and HRTEM images for ABC, Ru/ABC, AC, and Ru/AC. |
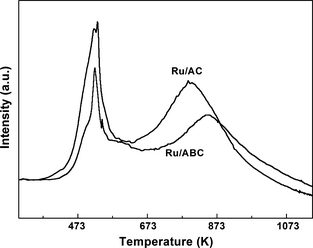
The TPR profiles of the Ru/ABC and the Ru/AC catalyst are depicted in Figure 3. The wide reduction peaks at about 873 K and 800 K for Ru/ABC and Ru/AC catalysts, respectively, were caused by the gasification of carbon [25]. The intense reduction peaks at 523 K was assigned to the reduction of the Ru species on both of the catalysts [26]. It was reported that the reduction temperature of Ru species on Ru/Al2O3 catalyst was 380 K [27]. The higher reduction temperature in this work suggests the presence of a relatively strong interaction between Ru and the supports, which might decrease the possibility of sintering [28].
|
|
|
Figure 3. TPR profiles of the Ru/ABC and Ru/AC catalysts. |
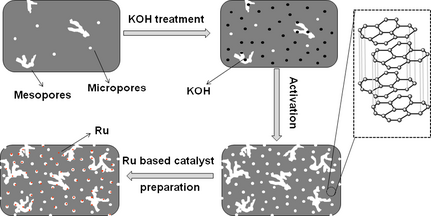
Based on the above results, a possible mechanism of the BC activation and the Ru/ABC catalyst preparation was proposed (Fig. 4). Firstly, the original BC was treated with KOH aqueous solution. Then, most of the pore structure of the BC was developed during activation, widening the existing pores and generating new ones, partly forming graphite-like morphology. Finally, the ABC can provide a much larger surface area for the dispersion of the active components during preparation of the catalyst. It is observed in Table 1 that the Ru/ABC catalyst showed a high Ru dispersion of 36%, which was close to the value of Ru/AC catalyst (41%).
|
|
|
Figure 4. A schematic illustration of the preparation of ABC and Ru/ABC catalyst. |
Syngas methanation
The catalytic performance of the prepared catalysts was carried out in the syngas methanation, to evaluate the catalytic activity of the Ru/ABC catalyst, compared with the Ru/AC catalyst. The effect of different reaction conditions on the catalytic activity was investigated.
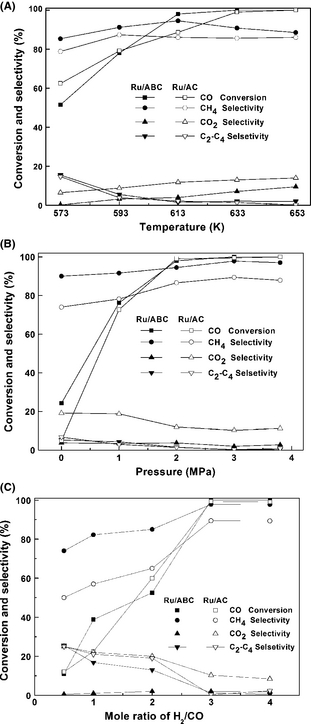
The effect of reaction temperature on the activity of Ru/ABC and Ru/AC catalysts was conducted under the conditions of H2/CO = 3, P = 2 MPa, gas hourly space velocity (GHSV) = 1200 h−1, and the results are presented in Figure 5A. The CO conversion increased significantly with elevated temperature, because the mobility of the H atom on the catalyst surface was promoted at higher temperature [29]. The Ru/ABC catalyst showed a high CO conversion of 98% at 613 K, while a higher temperature of 633 K was needed to obtain the same CO conversion over the Ru/AC catalyst. For the two catalysts, the main different properties are dispersed Ru crystallite size, the support pore structure, and may the electronic state. The Ru particle size is 3.3 nm for the Ru/ABC catalyst and 1.9 nm for the Ru/AC catalyst, as shown in Table 1. The effect of Ru crystallite size on CO and CO2 hydrogenation reactions has been reported in several works. The extent of the impact was depended on the nature of support. It was found that the TOF of CO over Ru/TiO2 catalyst increased by a factor of 40, with increasing dRu from 2.1 to 4.5 nm. While on Ru/Al2O3 catalyst, the TOF value increased only by a factor of 3.5, with increasing dRu from 1.3 to 13.6 nm [30]. Highly dispersed Ru–Zr/CNTs catalyst in amorphous form with the particle size ranging from 4 to 8 nm exhibited more active in CO methanation than the corresponding catalyst with the larger particle size in crystallized form [31]. The effect of Ru crystallite size on the catalytic activity and product selectivity was found be insensitive in CO2 methanation over SiO2 supported catalyst [32]. The catalytic behavior of Ru can be modified by the metal oxide supports through electron transfer. CO concentration decreased to below 10 ppm over ZrO2 promoted CNTs supported Ru catalyst, since Zr has high electronegativity and its charge transfer weaken C=O bond of the adsorbed CO molecules [31, 33]. The coexistence of mesopores and micropores over the Ru/ABC catalyst was the superiority to the Ru/AC catalyst in terms of mass transfer, especially in the reaction of large molecule synthesis. Therefore, the slightly high catalytic activity of ABC supported catalyst may be ascribed to the relative large Ru crystallite size, the special structure, and the probably appropriate electron state of catalyst.
|
|
|
Figure 5. Catalytic performance of the Ru/ABC and Ru/AC catalysts: (A) effect of reaction temperature; (B) effect of reaction pressure; (C) effect of H2/CO mole ratio. |
The selectivity of CH4 reached the maximum value of 95% and a CO conversion of 98% at 613 K over the Ru/ABC catalyst. However, further increase of the temperature reduced the selectivity of CH4. The CO conversion reached 100%, and the selectivity of CH4 decreased to 91% at 633 K over the Ru/ABC catalyst. While the selectivity of CH4 over the Ru/AC catalyst remained stable at around 87% at the temperature higher than 613 K, which was lower than that over the Ru/ABC catalyst. The results indicate that the structure and electron state in ABC supported catalyst are beneficial for methanation while inhibiting the side reaction, as the physical effect, such as Ru particle size, governs the selectivity for CO methanation [34]. CO hydrogenation over Al2O3 supported Ru catalyst was reported by Tajammul HS and his coworker. The promotion of Mn led to increase methane selectivity from 67% to 89% [35]. The selectivities of methane between 49.2% and 79.6% were obtained in CO methanation on Ni/Al2O3 catalysts with transition metals and rare-earth metals improved [36].
CO2 and C2–C4 hydrocarbons are the main by-products of the methanation reaction. It is known that CO2 is the product of water–gas shift (WGS) reaction in CO hydrogenation. At higher CO conversion, the increased production of water provided higher water concentration for WGS reaction [37]. Small amount of C2–C4 hydrocarbons was produced, and their selectivity fell close to zero at temperatures higher than 613 K. Thus, 613 K and 633 K appeared to be the optimum temperatures for syngas methanation using the Ru/ABC and Ru/AC catalysts, respectively, considering both of the high CO conversion and CH4 selectivity.
Figure 5B illustrates the effect of reaction pressure on the catalytic performance of the Ru/ABC and Ru/AC catalysts under their optimal temperatures. It is observed that the CO conversion increased rapidly with increasing pressure and maintained at 100% at the pressure higher than 3 MPa over both of the catalysts. Syngas methanation is a stoichiometric number reducing reaction, so high pressure would facilitate the reaction, leading to high CO conversion [13]. The selectivity of CH4 maintained the values higher than 90% at all the testing pressures, and reached the maximum value of 98% at 3 MPa over the Ru/ABC catalyst. However, the Ru/AC catalyst showed lower CH4 selectivity. In addition, further increase in the reaction pressure had little effect on either the CO conversion or CH4 selectivity.
Under the conditions of 3 MPa, 613 K, and 633 K for the Ru/ABC and Ru/AC catalysts, respectively, the effect of H2/CO mole ratio was studied and the results are displayed in Figure 5C. The CO conversion was as low as 11% at the H2/CO ratio of 0.5, while it sharply increased to 100% at a H2/CO ratio of 3 and the selectivity to CH4 simultaneously increased to its highest value of 98% over the Ru/ABC catalyst. For the Ru/AC catalyst, CO conversion increased from 12% to 99% with increasing H2/CO from 0.5 to 3, companied by the increasing of CH4 selectivity. The high concentration of H2 favors the formation of CH4 and might suppress the WGS reaction, resulting in a reduced selectivity for CO2 with the increase of the H2/CO ratio. Thus, a H2/CO mole ratio of 3 is proper for the complete conversion of CO, with high selectivity of CH4.
The statistical analysis on process parameters was performed using design expert 8.0 software with response surface methodology to evaluate the effect of reaction conditions on the methanation [38]. According to the preliminary analysis, the process variables multiple linear regression model was used for the subsequent variance analysis, and P-value of the model is 0.0004. Generally, parameters with P-values less than 0.05 were considered statistically significant with 95% confidence. The effects of process variables on syngas conversion and CH4 selectivity were evaluated by the following equations:
|
|
|
|
where A, B, and C represent the process parameters of temperature, pressure, and H2/CO molar ratio, respectively. The parameter coefficients show that the effect of H2/CO molar ratio on CO conversion and CH4 selectivity is of most significant. The further analysis of catalyst parameter revealed that in the present work, the effect of ABC catalyst on CH4 selectivity was more significant than CO conversion.
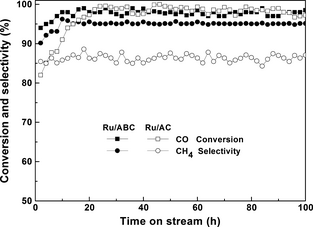
In addition, there was no deactivation observed over the ABC supported Ru catalyst for syngas methanation after as long as 100 h on stream (Fig. 6). It is indicated that the Ru/ABC catalyst showed an excellent catalytic activity and stability, which is comparable or superior to the Ru/AC catalyst, in the syngas methanation reaction. Furthermore, syngas methanation over ABC supported Ru catalyst was a gas–solid heterogeneous catalysis reaction. It is quite different with the liquid phase reaction. Hanson et al. pointed out that the catalysts would be deactivated due to the leaching of active sites in the sulfonated carbon during the esterification of free fatty acids for biodiesel production [39]. However, the leaching of active metals could not be observed in the gas–solid phase reaction, as was also confirmed by the fact that no deactivation was observed over the ABC supported Ru catalyst during the 100-h catalytic stability test. There is reason to believe that the BC is a promising substitute to the AC in the syngas methanation reaction. Furthermore, using the insights herein, BC can also be used as catalyst or support into other kind of reactions.
|
|
|
Figure 6. Catalytic stability of the Ru/ABC (613 K, 2 MPa, H2/CO = 3, GHSV = 1200 h−1) and Ru/AC catalysts (633 K, 2 MPa, H2/CO = 3, GHSV = 1200 h−1). |
Conclusions
The porosity of BC was significantly developed with the BET surface area increasing from 40 to 1058 m2/g after activation, and the structure of which became more graphite-like, which made the ABC feasible to be used as catalyst support. The catalytic activity of Ru/ABC catalyst was evaluated in syngas methanation compared with the Ru/AC catalyst. The proper Ru particles and the coexistence of microporous and mesoporous structure of the Ru/ABC catalyst showed advantages over the Ru/AC catalyst with smaller Ru particles and solely microporous structure in syngas methanation. The Ru/ABC catalyst exhibited excellent catalytic activity in syngas methanation reaction that CO conversion and the selectivity of CH4 were 100% and 98%, respectively. Additionally, the Ru/ABC catalyst showed excellent stability for a period of at least 100 h on stream.
Acknowledgments
We acknowledge financial support from the National Natural Science Foundation of China (51276166), the National Science and Technology supporting plan through contract (2015BAD15B06), and the International Science and Technology Cooperation Program of China (2011DFR60190).
Conflict of Interest
None declared.
References
- Brown, T. R., M. M. Wright, and R. C. Brown. 2011. Estimating profitability of two biochar production scenarios: slow pyrolysis vs fast pyrolysis. Biofuels, Bioprod. Biorefin.5:54–68.
- Abdullah, H., K. A. Mediaswanti, and H. Wu. 2010. Biochar as a fuel: 2. Significant differences in fuel quality and ash properties of biochars from various biomass components of Mallee trees. Energy Fuels24:1972–1979.
- Kim, Y., and W. Parker. 2008. A technical and economic evaluation of the pyrolysis of sewage sludge for the production of bio-oil. Bioresour. Technol.99:1409–1416.
- Bird, C. M., C. M. Wurster, P. H. Paula Silva, A. M. Bass, and de Nys R.2011. Algal biochar – production and properties. Bioresour. Technol.102: 1886–1891.
- Yu, J. T., A. M. Dehkhoda, and N. Ellis. 2011. Development of biochar-based catalyst for transesterification of canola oil. Energy Fuels25:337–344.
- Xiong, H, M. A. M. Motchelaho, M. Moyo, L. L. Jewell, and N. J. Coville. 2011. Correlating the preparation and performance of cobalt catalysts supported on carbon nanotubes and carbon spheres in the Fischer–Tropsch synthesis. J. Catal., 278:26–40.
- Yan, Q., C. Wan, J. Liu, J. Gao, F. Yu, J. Zhang, et al. 2013. Iron nanoparticles in situ encapsulated in biochar-based carbon as an effective catalyst for the conversion of biomass-derived syngas to liquid hydrocarbons. Green Chem.15:1631–1640.
- Dehkhoda, A. M., A. H. West, and N. Ellis. 2010. Biochar based solid acid catalyst for biodiesel production. Appl. Catal. A382:197–204.
- Azargohar, R. and A. K. Dalai2006. Biochar as a precursor of activated carbon. Appl. Biochem. Biotechnol.131:762–773.
- Kopyscinski, J., T. J. Schildhauer, F. Vogel, S. M. A. Biollaz, and A. Wokaun. 2010. Applying spatially resolved concentration and temperature measurements in a catalytic plate reactor for the kinetic study of CO methanation. J. Catal.271:262–279.
- Chen, A., T. Miyao, K. Higashiyama, H. Yamashita, and M. Watanabe. 2010. High catalytic performance of ruthenium-doped mesoporous nickel–aluminum oxides for selective CO methanation. Angew. Chem. Int. Ed.49:9895–9898.
- Kimura, M., T. Miyao, S. Komori, A. Chen, K. Higashiyama, H. Yamashita, et al. 2010. Selective methanation of CO in hydrogen-rich gases involving large amounts of CO2 over Ru-modified Ni–Al mixed oxide catalysts. Appl. Catal. A379:182–187.
- Zhang, J., Z. Xin, X. Meng, and M. Tao. 2013. Synthesis, characterization and properties of anti-sintering nickel incorporated MCM-41 methanation catalysts. Fuel109:693–701.
- Liu, Z., B. Chu, X. Zhai, Y. Jin, and Y. Cheng. 2012. Total methanation of syngas to synthetic natural gas over Ni catalyst in a micro-channel reactor. Fuel95:599–605.
- Malek Abbaslou, R. M., J. Soltan, and A. K. Dalai. 2011. Iron catalyst supported on carbon nanotubes for Fischer–Tropsch synthesis: effects of Mo promotion. Fuel90:1139–1144.
- Wu, Z., Y. Mao, X. Wang, and M. Zhang. 2011. Preparation of a Cu-Ru/carbon nanotube catalyst for hydrogenolysis of glycerol to 1,2-propanediol via hydrogen spillover. Green Chem.13:1311–1316.
- Panagiotopoulou, P., D. I. Kondarides, and X. E. Verykios. 2008. Selective methanation of CO over supported noble metal catalysts: effects of the nature of the metallic phase on catalytic performance. Appl. Catal. A344:45–54.
- Kang, J., K. Cheng, L. Zhang, Q. Zhang, J. Ding, W. Hua, et al. 2011. Mesoporous zeolite-supported ruthenium nanoparticles as highly selective Fischer-Tropsch catalysts for the production of C5–C11 isoparaffins. Angew. Chem.123:5306–5309.
- Özçimen, D., and A. Ersoy-Meriçboyu. 2010. Characterization of biochar and bio-oil samples obtained from carbonization of various biomass materials. Renew Energ35:1319–1324.
- Surisetty, V. R., A. K. Dalai, and J. Kozinski. 2011. Influence of porous characteristics of the carbon support on alkali-modified trimetallic Co–Rh–Mo sulfided catalysts for higher alcohols synthesis from synthesis gas. Appl. Catal. A393:50–58.
- Muthu Kumaran, G., S. Garg, K. Soni, M. Kumar, L. D. Sharma, G. Murali Dhar, et al. Effect of Al-SBA-15 support on catalytic functionalities of hydrotreating catalysts: I. Effect of variation of Si/Al ratio on catalytic functionalities. Appl. Catal. A, 2006, 305: 123–129.
- Patrick, J. W. 1995. Pp. 1–48inPorosity in carbons: characterization and applications. Halsted Press, John Wiley & Sons, New York.
- Yang, K., J. Peng, C. Srinivasakannan, L. Zhang, H. Xia, and X. Duan. 2010. Preparation of high surface area activated carbon from coconut shells using microwave heating. Bioresour. Technol.101:6163–6169.
- Wang, S., Q. Yin, J. Guo, B. Ru, and L. Zhu. 2013. Improved Fischer-Tropsch synthesis for gasoline over Ru, Ni promoted Co/HZSM-5 catalysts. Fuel108:597–603.
- Xiong, J., X. Dong, and L. Li. 2012. CO selective methanation in hydrogen-rich gas mixtures over carbon nanotube supported Ru-based catalysts. J. Nat. Gas Chem.21:445–451.
- Perkas, N., J. Teo, S. Shen, Z. Wang, J. Highfield, Z. Zhong, et al. 2011. Ru supported catalysts prepared by two sonication-assisted methods for preferential oxidation of CO in H2 . Phys. Chem. Chem. Phys.13:15690–15698.
- Rynkowski, J., T. Paryjczak, A. Lewicki, M. Szynkowska, T. Maniecki, and W. Jóźwiak. 2000. Characterization of Ru/CeO2-Al2O3 catalysts and their performance in CO2 Methanation. React. Kinet. Catal. Lett.71:55–64.
- Li, X., S. Wang, Q. Cai, L. Zhu, Q. Yin, and Z. Luo. 2012. Effects of preparation method on the performance of Ni/Al2O3 catalysts for hydrogen production by bio-oil steam reforming. Appl. Biochem. Biotechnol.168:10–20.
- Trépanier, M., and A. Tavasoli, A. K Dalai, and N. Abatzoglou. 2009. Fischer-Tropsch synthesis over carbon nanotubes supported cobalt catalysts in a fixed bed reactor: Influence of acid treatment. Fuel Process. Technol.90:367–374.
- Panagiotopoulou, P., D. I. Kondarides and X. E. Verykios2009. Selective methanation of CO over supported Ru catalysts. Appl. Catal. B88:470–478.
- Xiong, J., X. Dong, and L. Li. 2012. CO selective methanation in hydrogen-rich gas mixtures over carbon nanotube supported Ru-based catalysts. J. Nat. Gas Chem.21:445–451.
- Scir‘e, S., C. Crisafulli, R. Maggiore, S. Minic‘o, and S. Galvagno. 1998. Influence of the support on CO2 methanation over Ru catalysts: an FT-IR study. Catal. Lett., 51:41–45.
- Perkas, N., J. Teo, S. Shen, Z. Wang, J. Highfield, Z. Zhong, et al. 2011. Ru Supportedcatalysts prepared by two sonication-assisted methods for preferential oxidation of CO in H2 . Phys. Chem. Chem. Phys.13:15690–15698.
- Eckle, S., M. Augustin, and H.-G. Anfang, and R. J. Behm. 2012. Influence of the catalyst loading on the activity and the CO selectivity of supported Ru catalysts in the selective methanation of CO in CO2 containing feed gases. Catal. Today181:40–51.
- Tajammul, H. S., and A. M. Ashraf. 1997. Carbon monoxide hydrogenation over Ru-Mn-K/Al2O3 catalysts. Turk. J. Chem.21:77–83.
- Hwang, S., J. Lee, U. G. Hong, J. C. Jung, D. J. Koh, H. Lim, et al. 2012. Hydrogenation of carbon monoxide to methane over mesoporous nickel-M-alumina (M=Fe, Ni, Co, Ce, and La) xerogel catalysts. J. Ind. Eng. Chem.18:243–248.
- Abbaslou, R. M. M., J. Soltan, and A. K. Dalai. 2010. Effects of nanotubes pore size on the catalytic performances of iron catalysts supported on carbon nanotubes for Fischer-Tropsch synthesis. Appl. Catal. A379:129–134.
- Bezerra, M. A., R. E. Santelli, E. P. Oliveira, L. S. Villar, and L. A. Escaleira. 2008. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta76:965–977.
- Deshmane, C. A., M. W. Wright, A. Lachgar, M. Rohlfing, Z. Liu, J. Le, et al. 2013. A comparative study of solid carbon acid catalysts for the esterification of free fatty acids for biodiesel production. Evidence for the leaching of colloidal carbon. Bioresour. Technol.147:597–604.
References
- Jäger-Waldau, A.2004. Status of thin film solar cells in research, production and the market. Sol. Energy77:667–678.
- Shah, A., P. Torres, R. Tscharner, N. Wyrsch, and H. Keppner. 1999. Photovoltaic technology: the case for thin-film solar cells. Science285:692–698.
- Staebler, D. L., and C. R. Wronski. 1977. Reversible conductivity changes in discharge-produced amorphous Si. Appl. Phys. Lett.31:292–294.
- Okamoto, S., Y. Hishikawa, and S. Tsuda. 1996. New interpretation of the effect of hydrogen dilution of silane on glow-discharged hydrogenated amorphous silicon for stable solar cells. Jpn. J. Appl. Phys.35:26–33.
- Tsu, D. V., B. S. Chao, S. R. Ovshinsky, S. Guha, and J. Yang. 1998. Effect of hydrogen dilution on the structure of amorphous silicon alloys. Appl. Phys. Lett.71:1317–1318.
- Koh, J., Y. Lee, H. Fujiwara, C. R. Wronski, and R. W. Collins. 1998. Optimization of hydrogenated amorphous silicon p-i-n solar cells with two-step I layers guided by real-time spectroscopic ellipsometry. Appl. Phys. Lett.73:1526–1528.
- Ahn, J. Y., K. H. Jun, M. Konagai, and K. S. Lim. 2003. Stable protocrystalline silicon and unstable microcrystalline silicon at the onset of a microcrystalline regime. Appl. Phys. Lett.82:1718–1720.
- Myong, S. Y., S. W. Kwon, M. Konagai, and K. S. Lim. 2005. Highly stabilized protocrystalline silicon multilayer solar cell using a silicon-carbide double p-layer structure. Sol. Energy Mater. Sol. Cells85:133–140.
- Myong, S. Y., and S. W. Kwon. 2013. Unique features of alternately hydrogen-diluted protocrystalline silicon multilayers as wide bandgap and highly stable absorbers of thin-film silicon solar cells. Sol. Energy Mater. Sol. Cells116:182–186.
- Matsuda, A.2004. Thin-film silicon – growth process and solar cell application. Jpn. J. Appl. Phys.43:7909–7920.
- Myong, S. Y., S. S. Kim, and K. S. Lim. 2004. Improvement of pin-type amorphous silicon solar cell performance by employing double silicon-carbide p layer structure. J. Appl. Phys.95:1525–1530.
- Myong, S. Y., and K. S. Lim. 2005. Natural hydrogen treatment effect during formation of double amorphous silicon-carbide p-layer structures producing high-efficiency pin-type amorphous silicon solar cells. Appl. Phys. Lett.86:033506, 3pp.
- Myong, S. Y., J. M. Pearce, and K. S. Lim. 2005. Double amorphous silicon-carbide p-layer structures producing highly stabilized pin-type protocrystalline silicon multilayer solar cells. Appl. Phys. Lett.87:193509, 3pp.
- Myong, S. Y., K. Sriprapha, Y. Yashiki, S. Miyajima, A. Yamada, and M. Konagai. 2008. Silicon-based thin-film solar cells fabricated near phase boundary by VHF-PECVD technique. Sol. Energy Mater. Sol. Cells92:639–645.
- Myong, S. Y., J. Steinhauser, R. Schlüchter, S. Faÿ, E. Vallat-Sauvain, A. Shah, et al. 2007. Temperature dependence of the conductivity in large-grained boron-doped ZnO films. Sol. Energy Mater. Sol. Cells91:1269–1274.
- Myong, S. Y., K. Sriprapha, S. Miyajima, M. Konagai, and A. Yamada. 2007. High efficiency protocrystalline silicon/microcrystalline silicon tandem cell with zinc oxide intermediate layer. Appl. Phys. Lett.90:263509, 3pp.
- Haas, S., A. Gordijn, and H. Stiebig. 2008. High speed laser processing for monolithical series connection of silicon thin-film modules. Prog. Photovoltaics Res. Appl.16:195–203.
- Kurnik, J., M. Jankovec, K. Brecl, and M. Topic. 2011. Outdoor testing of PV module temperature and performance under different mounting and operational conditions. Sol. Energy Mater. Sol. Cells95:373–376.
- Klaver, A., and R. A. C. M. M. van Swaaij. 2008. Modeling of light-induced degradation of amorphous silicon solar cells. Sol. Energy Mater. Sol. Cells92:50–60.
- Takatsuka, H., M. Noda, Y. Yonekura, Y. Takeuchi, and Y. Yamauchi. 2004. Development of high efficiency large area silicon thin film modules using VHF-PECVD. Sol. Energy77:951–960.
- Ishii, T., K. Otani, and T. Takashima. 2010. Effects of solar spectrum and module temperature on the outdoor performance of photovoltaic modules induced in round-robin measurements in Japan. Prog. Photovoltaics Res. Appl.19:141–148.
- del Cueto, J., and B. von Roedern. 1999. Temperature-induced changes in the performance of amorphous silicon multi-junction modules in controlled light soaking. Prog. Photovoltaics Res. Appl.7:101–112.
- Fecioru-Morariu, M., B. Mereu, S. Bakehe, J. Kalas, O. Kluth, and T. Eisenhammer. 2012. High quality amorphous Si solar cells for large area mass production Micromorph tandem cells. J. Non-Cryst. Solids358:2264–2267.
- Morrison, S., U. Das, and A. Madan. 2003. Deposition of thin film silicon using the pulsed PECVD and HWCVD techniques. Sol. Energy Mater. Sol. Cells76:281–291.
- Kondo, M., and A. Matsuda. 2004. Novel aspects in thin film silicon solar cells-amorphous, microcrystalline and nanocrystalline silicon. Thin Solid Films457:97–102.
- Akhmad, K., H. Okamoto, F. Yamamoto, and A. Kitamura. 1997. Long-term performance modeling of amorphous silicon photovoltaic module. Jpn. J. Appl. Phys.36:629–632.
- Ishii, T., T. Takashima, and K. Otani. 2011. Long-term performance degradation of various kinds of photovoltaic modules under moderate climatic conditions. Prog. Photovoltaics Res. Appl.19:170–179.
- Ishii, T., K. Otani, T. Takashima, and K. Ikeda. In press. Change in I-V characteristics of thin-film photovoltaic (PV) modules induced by light soaking and thermal annealing effects. Prog. Photovoltaics Res. Appl. (in press), doi:10.1002/pip.2346.
- Söderström, T., F.-J. Haug, V. Terrazzoni-Daudrix, and C. Ballif. 2010. Flexible micromorph tandem a-Si/μc-Si solar cells. J. Appl. Phys.107:014507-1–014507-7.
- Klein, S., M. Rohde, S. Buschbaum, and D. Severin. 2012. Throughput optimized a-Si/μc-Si tandem solar cells on sputter-etched ZnO substrates. Sol. Energy Mater. Sol. Cells98:363–369.
Document information
Published on 01/01/2017
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?