Summary
Background/Objective
To investigate the factors associated with recurrence of differentiated thyroid cancer in children. We combined the clinical and pathological features to guide surgical treatment options, ensure efficacy, and reduce complications.
Methods
A prospective analysis of clinical data of 43 cases of pediatric differentiated thyroid cancer from March 2008 to June 2014 admitted in our department, including 38 cases of papillary cancers and five cases of follicular cancer; 40 cases were Stage I and three cases were Stage II (Union for International Cancer Control [UICC] Tumor Node Metastasis classification [TNM] staging). We performed the operations according to the condition of lesions and lymph nodes. Operations included subtotal resection in 36 cases, total resection in seven cases. We applied statistical methods to investigate the risk factors of recurrence and postoperative complications.
Results
The pathologic lymph node metastasis rate was 67.44% (29/43): Area VI lymph nodes metastases in 20 cases and Area III/IV or Area II/V lymph nodes metastases in nine cases. Postoperative hypocalcemia symptoms were seen in three cases and hoarseness in three cases, for a total rate of 13.95% (6/43). Until December 2015, patients were followed up from 1.5 years to 8.7 years, with a median of 4.9 years. There were three cases of cervical lymph node recurrence, one case of local recurrence, and one case of lung metastasis, for a total recurrence rate of 11.63% (5/43); all patients survived. Log-rank test of Kaplan-Meier curves and Cox stepwise regression analysis showed that lesion number, extrathyroidal extension, and lymph nodes metastases were the risk factors for postoperative recurrence; the relative risk values were, respectively, 3.117, 2.816, and 4.628 (p = 0.041, p = 0.048, and p = 0.031, respectively) and the 95% confidence intervals (CI) were, respectively, 1.094∼8.735, 1.046∼7.932, and 1.189∼10.205. However, the lesion excision approach was not a risk factor for postoperative recurrence (p = 0.107). The logistic stepwise regression model showed that lesion excision approach was a risk factor for postoperative hypocalcemia and hoarseness; the odds ratio value was 2.537 (p = 0.037) and the 95% CI was 1.034∼6.983.
Conclusion
Pediatric differentiated thyroid cancer has a high metastatic rate to lymph nodes and distant organs, but the total prognosis is good. Application of total resection cannot necessarily reduce the relapse rate of pediatric differentiated thyroid cancer, but it may increase the postoperative hypocalcemia and hoarseness. The authors propose strictly adhering to various operation indicators, and carrying out various operations with a full understanding of the local lesion and lymph nodes in order to reduce relapse and postoperative complications.
Keywords
complications;differentiated thyroid cancer;pediatrics;recurrence
1. Introduction
Pediatric thyroid cancer is rare, accounting for 2.4–9% of epithelial origin solid tumors in children,1 but there is an increasing trend in the occurrence of this cancer.2 The most common pathologies of differentiated thyroid cancers are papillary and follicular.3 Compared with adult thyroid cancer, pediatric differentiated thyroid cancer has different clinical features and there are different treatment strategies, with a lower malignancy, slower progress, longer course, and better prognosis. Currently, a multiple combined surgical-based therapy for the treatment of pediatric thyroid cancer is advocated, but the treatment standard is still not fully integrated, as Chinese and foreign approaches are not consistent. To find a better clinical treatment strategy, the authors explore the risk factors of postoperative recurrence and common complications in this article.
2. Methods
2.1. General data
We investigated 43 cases of children with pathologically confirmed differentiated thyroid carcinoma who were admitted to our department from March 2008 to June 2014. There were 19 males and 24 females, with a male to female ratio of 1:1.26. Patients were aged from 5 years old to 14 years old, with a median age of 8.7 years. The disease course was from 1 week to 3 months. The main complaints were a cervical area tumor in 25 cases, cervical lymphadenectasis in eight cases, or disease was determined by physical examination or laboratory examinations in 10 cases. (Table 1). We experienced no cases of hoarseness or difficulty breathing.
| Demographic data | n (%) |
|---|---|
| Patient sex | |
| Male | 19 (44.19) |
| Female | 24 (55.81) |
| Patient age (y) | 9.1 ± 0.83 |
| Disease course (mo) | 1.2 ± 0.49 |
| Main complaint | |
| Cervical area tumor | 25 (58.14) |
| Cervical lymphadenectasis | 8 (18.60) |
| Medical examination (physical and ultrasonic) | 10 (23.26) |
| Lesion location | |
| Left lobe | 17 (39.53) |
| Right lobe | 23 (53.49) |
| Bilateral lobes | 3 (6.98) |
| Lesion size | |
| <2.0 cm | 24 (55.81) |
| 2.0∼4.0 cm | 19 (44.17) |
| Lesion number | |
| Solitary nodule | 38 (88.37) |
| Multiple nodules | 5 (11.63) |
| Extrathyroidal extension | |
| Occur | 6 (13.95) |
| None | 37 (86.05) |
| Pathologic type | |
| Papillary | 38 (88.37) |
| Follicular | 5 (11.63) |
| Tumor stage | |
| Stage I | 40 (93.02) |
| Stage II | 3 (6.98) |
| Lesion excision | |
| Subtotal resection | 34 (79.07) |
| Total resection | 9 (16.28) |
| Lymph node metastasis | |
| None | 14 (32.56) |
| Area VI | 20 (46.51) |
| Area III/IV or II/V | 9 (20.93) |
| Postoperative 131I therapy | |
| Occur | 8 (18.60) |
| None | 35 (81.40) |
SD = standard deviation.
2.2. Preoperative examination
All cases underwent neck ultrasonography, thyroid function, systemic radionuclide scans, and other tests before the operation. Ultrasonography showed lesion located in the left lobe in 17 cases, the right lobe in 23 cases, bilateral lobes in three cases, solitary nodules in 38 cases, multiple nodules in five cases, nodules with calcification in 17 cases, and cervical lymphadenectasis in 29 cases. Thyroid function tests showed that thyroid-stimulating hormone (TSH) was increased in five cases and antithyroid peroxidase autoantibody (TPoAb) was elevated in two cases, while the rest were normal. An ultrasound-guided fine needle aspiration biopsy was performed in 13 cases, including 11 cases of thyroid cancer, one case where follicular cells were found, and in one case a few atypical cells were identified and could not be ruled out as cancer. A systemic radionuclide scan revealed pulmonary nodules in three cases, which were used with lung enhanced computed tomography (CT) to confirm disease, but no bone metastasis was identified.
2.3. Surgical approach and pathology
According to local lesions, lymph nodes, and intraoperative frozen pathology samples, we selected the appropriate lesion excision approach, including subtotal resection (refers to the ipsilateral lobe + isthmus + contralateral partial lobectomy) for 34 cases and total resection for nine cases. The pathology included 38 cases of papillary cancer, five of follicular cancer, and 29 cases of lymph node metastasis. The total lymph node metastasis rate was 67.44%, and Area VI lymph node metastasis was seen in 20/29 cases, accounting for 68.97% of lymph node metastasis cases. Using the Union for International Cancer Control (UICC) Tumor Node Metastasis classification (TNM) staging (2002 Edition) we saw 40 cases with Stage I disease and three cases with Stage II disease (Table 1).
2.4. Postoperative follow-up, recurrence, and complications
We used a combination of outpatient service and telephone follow-up for the patients. All cases were followed up successfully. Three cases of lung metastasis (UICC Stage II) received 131I therapy after the total resection surgery, and the lung lesions disappeared. All cases were reviewed with neck ultrasonography, thyroid function, thyroglobulin, calcium, and chest examination every 3 months for 2 years, every 6 months from 2∼5 years, then annually after 5 years. Conventional postoperative oral levothyroxine tablets were used for TSH suppression therapy for 5 years and the TSH levels were maintained below 0.2 mU/L. If a review displayed a suspicion of recurrence or metastasis, oral levothyroxine tablets were stopped for 2 weeks until the TSH level increased before systemic radionuclide scans and fine needle aspiration biopsy were used to confirm. Follow-up was continued until the end of December 2015, from 1.5 years to 8.7 years with a median of 4.9 years, and no deaths occurred. There were a total of five cases of recurrence with a recurrence rate of 11.63% (5/43); cervical lymph node recurrence in three cases, local recurrence in one case, and pulmonary metastasis and recurrence in one case. The local recurrence and two cases of cervical lymph node recurrence were treated with reoperation and postoperative 131I therapy. One case of cervical lymph node recurrence received 131I therapy because of undergoing total resection in the first operation and his family’s unwillingness to undergo another operation. The lung recurrence patient also received 131I therapy. The lung recurrence patient improved after 131I therapy with the pulmonary lesion becoming significantly reduced without completely disappearing. Follow-up for 6 years in this patient showed that the lesion did not progress. The other four cases were cured, with the lesions disappearing after treatment, and follow-up found no evidence of recurrence. Postoperative hypocalcemia and hoarseness both occurred in three cases for a total rate of 13.95% (6/43). Following treatment with calcium, Methycobal supplements and acupuncture, the symptoms were relieved to varying degrees.
2.5. Statistical methods
We used SPSS 19.0 statistical software (SPSS 19.0 version: IBM Corporation, 1 New Orchard Road, Armonk, New York, United States) for data analysis to investigate the influence of factors such as lesion location, lesion size, lesion number, extrathyroidal extension, pathologic type, tumor stage, lesion excision, lymph node metastasis, and postoperative 131I therapy on the postoperative recurrence and complication. A log-rank test of Kaplan-Meier curves was used to filter variables and the Cox regression model was used to identify risk factors of the postoperative recurrence. A logistic regression model was used to filter variables and identify risk factors of the postoperative complication. A p value <0.05 was considered statistically significant and relative risk or odds ratio values were obtained.
3. Results
3.1. Risk factors of recurrence
A log-rank test of Kaplan-Meier curves showed that variables such as lesion number, extrathyroidal extension, lesion excision, and lymph node metastasis were significant variables (p = 0.029, p = 0.040, p = 0.049, and p = 0.016, respectively). Then Cox regression analysis showed that lesion number, extrathyroidal extension, and lymph node metastasis were the risk factors for postoperative recurrence; the relative risk values were 3.117, 2.816, and 4.628, respectively (p = 0.041, p = 0.048, and p = 0.031, respectively) and the 95% CI were 1.094∼8.735, 1.046∼7.932, and 1.189∼10.205, respectively. However, lesion excision approach was not a risk factor for postoperative recurrence (p = 0.107) (Table 2).
| Characteristics | Log-rank test | Cox regression analysis | |
|---|---|---|---|
| p | RR (95% CI) | p | |
| Lesion location | 0.318 | — | — |
| Lesion size | 0.206 | — | — |
| Lesion number | 0.029* | 3.117 (1.094∼8.735) | 0.041 |
| Extrathyroidal extension | 0.040* | 2.816 (1.046∼7.932) | 0.048 |
| Pathologic type | 0.190 | — | — |
| Tumor stage | 0.130 | — | — |
| Lesion excision | 0.049* | 1.169 (0.517∼2.845) | 0.107 |
| Lymph node metastasis | 0.016* | 4.628 (1.189∼10.205) | 0.031 |
| Postoperative 131I therapy | 0.252 | — | — |
- significant variables into cox regression model.
CI = confidence interval; RR = relative risk.
3.2. Risk factors for postoperative complications
Logistic regression analysis (univariate) showed that lesion number and lesion excision approach were significant variables (p = 0.043 and p = 0.015). Then, logistic regression analysis (multivariate) showed that lesion excision approach was the risk factor for postoperative hypocalcemia and hoarseness; the odds ratio value was 2.537 (p = 0.037) and the 95% CI was 1.034∼6.983 (Table 3).
| Characteristics | Univariate | Multivariate | |
|---|---|---|---|
| p | OR (95% CI) | p | |
| Lesion location | 0.436 | — | |
| Lesion size | 0.450 | — | |
| Lesion number | 0.043* | — | |
| Extrathyroidal extension | 0.114 | — | |
| Pathologic type | 0.271 | — | |
| Tumor stage | 0.082 | — | |
| Lesion excision | 0.015* | 2.537 (1.034∼6.983) | 0.037 |
| Lymph node metastasis | 0.183 | — | |
| Postoperative 131I therapy | 0.214 | — | |
- Significant variables into multivariate logistic regression model.
CI = confidence interval; OR = odds ratio.
4. Discussion
Pediatric thyroid cancer is rarely seen in the clinic. At present, the most recognized cause is exposure to radiation.4 After the 1986 Chernobyl nuclear accident, the incidence of thyroid cancer in children who were exposed to radioactive fallout increased significantly, especially for children under the age of 4 years.5 Studies have shown that the RET/PTC3 gene rearrangement frequently occurred in children after radiation exposure, and this gene rearrangement is the most common in pediatric thyroid papillary cancer.6
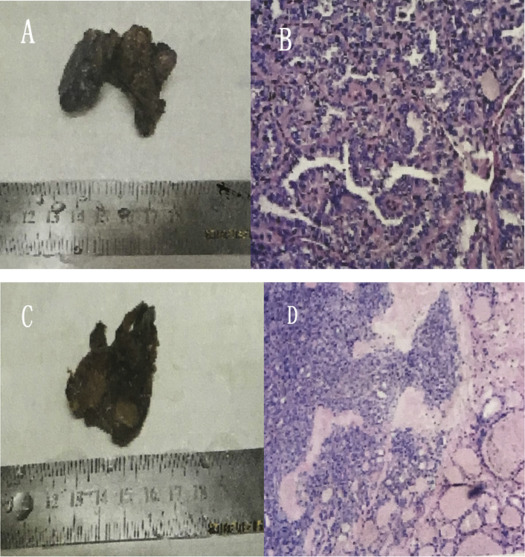
The clinical features of thyroid cancer in children and adults are different. The features of pediatric thyroid cancer are mainly: (1) solid nodules are relatively common but cystic or solid-cystic lesions have also been found; (2) the pathologic type is mainly based on differentiated cancer, and papillary cancer (Figs. 1A and B) is the most common, while follicular cancer (Figs. 1C and D) is relatively less common than in adults, and undifferentiated cancer is extremely rare; (3) compared with adults, the invasion rate of pediatric thyroid cancer is higher and lymph node and distant organ metastases are more common. According to the literature, the rate of lymph node metastasis is about 60–80% and the rate of lung metastasis is around 10%7; and (4) invasion and lymph nodes and distant organ metastases in children are more common than in adults, but their biological effects are moderate, progression is slow, and the final prognosis is good after active treatment.8 In this study, 38 cases of pediatric thyroid papillary cancer, five cases of follicular cancer, and no medullary cancer or undifferentiated cancer were seen. The rate of local lymph node metastasis was 67.44%, including Area VI lymph nodes metastasis that accounted for 68.97% of lymph node metastasis. There were also three cases of lung metastasis. All patients were alive after treatment with no deaths during follow up, and the results of treatment were satisfactory.
|
|
|
Figure 1. Thyroid papillary cancer in children. (A) gross specimen; (B) microscopic view; (C) gross specimen; and (D) microscopic view. |
At present, pediatric differentiated thyroid cancers are mostly treated with surgical resection combined with 131I treatment and TSH suppression treatment to form comprehensive treatment strategies. Most oncologists believe that external radiation therapy and chemotherapy is ineffective and it is now used infrequently. Although most agree with surgical treatment, the lesion excision and lymphadenectomy approach is still controversial.
To predict the lesion excision approach, some oncologists advocate bilateral thyroidectomy with the main reasons as follows: (1) the bilateral lobes of the thyroid are not separate and are linked to each other into an organ; you must remove the entire organ in order to reduce the cancer relapse; (2) pediatric differentiated thyroid cancer has the characteristics of multifocal lesions. Amarasinghe et al9 reported that the incidence of residual cancer may increase up to 30% without total thyroidectomy; and (3) pediatric differentiated thyroid cancer has the characteristics of a high metastatic rate to lymph nodes and lung, and the total thyroidectomy may create favorable conditions for 131I therapy after the operation. Most Chinese oncologists advocate subtotal instead of total resection and the main considerations are as follows: (1) children undergo growth and development and a total thyroidectomy may cause permanent thyroid or parathyroid dysfunction, affecting growth and development, even threatening life; (2) a long-term follow-up review shows that the difference in local recurrence and distant organ metastatic rates between subtotal resection and total resection are not significant10; and (3) the total prognosis for pediatric differentiated thyroid cancer is good.
In this study the log-rank test of Kaplan-Meier curves and Cox stepwise regression analysis the author used showed that lesion number, extrathyroidal extension, and lymph node metastasis were the risk factors for the recurrence of pediatric differentiated thyroid cancer without lesion excision approach. We offer the following analysis as reasons: (1) the study of pediatric differentiated thyroid cancer, of which papillary or follicular cancer are the most common pathological types, show that its biological behavior is better than this cancer in adults. There were no undifferentiated tumors that relapsed easily in the study, so the factor of pathologic type has little effect on the recurrence of this kind of thyroid cancer; (2) UICC staging was used in the study and there is only Stage I or Stage II papillary and follicular cancer in patients under the age of 45 years; no Stage III or Stage IV disease was seen. In this study, distant organ metastasis mainly focuses on lung without bone metastasis, and even in the presence of distant lung metastasis, the treatment is still satisfactory. So the tumor stage may also have little effect on the recurrence of this kind of thyroid cancer in the study, but it requires a larger sample size for further study; (3) for the extrathyroidal extension, the authors recommend cleanly cutting the tumor-infiltrating tissue under the naked eyes as far as possible, since it is a risk factor for postoperative recurrence. If the tumor- infiltrating tissue cannot be completely removed, postoperative 131I therapy is necessary for the removal of residual cancer; and (4) for the lesion excision approach, although Hay et al11 suggested that the local recurrence rate after total resection was lower than after unilateral lobectomy for the children with thyroid cancer (p < 0.05), the American Thyroid Association in 2006 made no clear provision for surgical excision standards for children with differentiated thyroid cancer. In addition, some oncologists believe that the influence of subtotal and total resection on the recurrence was not significantly different,10 but there may be some impact on postoperative complications and growth and development.12 In this study, the results showed that the relation between lesion excision approach and postoperative recurrence was not strong (p = 0.107), but it is a risk factor for postoperative hypocalcemia and hoarseness (p = 0.037). In addition, as an important endocrine organ in children, the entire thyroid function is not fully understood; the ability of levothyroxine tablets to completely replace the entire thyroid function or not after total resection lacks a long-term follow-up study and endocrine-related complications in children after total resection are more common.12 Therefore, the authors believe that the lesion excision approach should be based on the specific condition of the lesion and we consider the relevant literature as follows: (1) if the lesion lies in the side of lobes without invasion, it is appropriate to apply subtotal resection, but the contralateral lobe needs to be carefully explored with a preoperative contralateral lobe fine-needle biopsy or intraoperative frozen pathology; (2) when contralateral lobe invasion or multiple lesions are found, a total resection should be considered, since lesion number was a risk factor for postoperative recurrence in this study; (3) when lesions near the isthmus are found without foreign invasion, a subtotal resection should be considered; and (4) for the cases with distant organ metastases, positive surgical options are appropriate, and a total resection should be applied together with postoperative 131I therapy to achieve a favorable efficacy.
The lymphadenectomy approach remains somewhat controversial for many oncologists. Most suggest that for a central section lymphadenectomy (Area VI), the main consideration is that the incidence of pediatric differentiated thyroid carcinoma lymph node metastasis is greater than in adults.9 Hay et al11 have reported an incidence up to 50∼80%. The central section lymph nodes are the most likely site and are mainly composed of the paratracheal, pretracheal, and prelarynx lymph nodes. In this study, the lymph node metastasis rate was 67.44%; the Area VI lymph node metastasis accounted for 68.97% of the total lymph node metastasis, therefore the authors believe that the central section lymphadenectomy is necessary, even for children with cN0, because while less than half of the central section lymph node metastasis are discovered by preoperative physical and imaging examination, a significant number are not discovered. Since deep cervical lymph nodes have a wide range that includes Area II, Area III, Area IV, and Area V, etc., and the metastasis rate is relatively lower than for the central section, it is currently controversial to routinely perform lymphadenectomy for the deep cervical lymph nodes. In this study, lymph node metastasis was a risk factor for postoperative recurrence, so the authors consistently advise applying lymphadenectomy for deep cervical lymph nodes that are positive or suspect from a preoperative ultrasonography, fine needle aspiration biopsy, or intraoperative frozen pathology. A modified lymphadenectomy which is mainly based on the central section should be recommended, both to reduce the rate of residual cancer in the lymph nodes to the maximum extent, and to protect the neck function in children as far as possible.
In summary, the use of total resection in an attempt to significantly reduce the recurrence rate is still debatable, but it may increase the postoperative hypocalcemia and hoarseness. Pediatric differentiated thyroid cancer has many clinical and biological characteristics that are different from adult disease, the final prognosis is good, and even with recurrence you can still achieve good results after surgery or 131I therapy.13 Therefore, the authors suggest considering the various surgical indications in the growth and development of children to ensure efficacy and reduce complications.
References
- 1 A.M. Samuel, S.M. Sharma; Differentiated thyroid carcinomas in children and adolescents; Cancer, 67 (1991), pp. 2186–2190
- 2 A.Y. Chen, A. Jemal, E.M. Ward; Increasing incidence of differentiated thyroid cancer in the United States, 1988–2005; Cancer, 115 (2009), pp. 3801–3807
- 3 J.B. Gorlin, S.E. Sallan; Thyroid cancer in childhood; Endocrinol Metab Clin N Am, 19 (1990), p. 649
- 4 S.J. Rybakov, I.V. Komissarenko, N.D. Tronko, et al.; Thyroid cancer in children of Ukraine after the Chernobyl accident; World J Surg, 24 (2000), pp. 1446–1449
- 5 C. Reiners, Y.E. Demldchlk, V.M. Drozd, J. Biko; Thyroid cancer in infants and adolescents after Chernobyl; Minerva Endocrinol, 33 (2008), pp. 381–395
- 6 S.A. Rivkees, E.L. Mazzaferri, F.A. Verburg, et al.; The treatment of differentiated thyroid cancer in children: emphasis on surgical approach and radioactive iodine therapy; Endocr Rev, 32 (2011), pp. 798–826
- 7 R. Feinmesser, E. Lubin, K. Segal, A. Noyek; Carcinoma of the thyroid in children-a review; J Pediatr Endocrinol Metab, 10 (1997), pp. 561–568
- 8 P. Miccoli, M.N. Minuto, C. Ugolini, et al.; Papillary thyroid cancer: pathological parameters as prognostic factors in different classes of age; Otolaryngol Head Neck Surg, 138 (2008), pp. 200–203
- 9 I.Y. Amarasinghe, N.M. Perera, N. Bahinathan, H.H. Marzook, A.K. Peiris; Review of distribution of nodal disease in differentiated thyroid cancers in an oncosurgical center in Sri Lanka; Ann Surg Oncol, 14 (2007), pp. 1560–1564
- 10 Tang Zhaoyou (Ed.), Modern Oncology, Shanghai Medical University Press, Shanghai (1996), p. 824 (Chinese)
- 11 I.D. Hay, T. Gonzalez-Losada, M.S. Reinalda, J.A. Honetschlager, M.L. Richards, G.B. Thompson; Long-term outcome in 215 children and adolescents with papillary thyroid cancer treated during 1940 through 2008; World J Surg, 34 (2010), pp. 1192–1202
- 12 J.A. Sosa, C.T. Tuggle, T.S. Wang, et al.; Clinical and economic outcomes of thyroid and parathyroid surgery in children; J Clin Endocrinol Metab, 93 (2008), pp. 3058–3065
- 13 R. Vassilopoulou-Sellin, H. Goepfert, B. Raney, P.N. Schultz; Differentiated thyroid cancer in children and adolescents: Clinical outcome and mortality after long-term follow-up; Head Neck, 20 (1998), pp. 549–555
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?