Summary
Morbid obesity is a common health problem worldwide. Laparoscopic adjustable gastric banding has been used extensively around the world for the treatment of morbid obesity. Life-threatening hemorrhage as a late complication of laparoscopic adjustable gastric banding is extremely rare. We report a case of massive upper gastrointestinal bleeding due to intragastric erosion of a gastric band 6 years postoperatively and review the English literature on this life-threatening rare condition. Initially, the patient was aggressively resuscitated and treated conservatively. After 1 year, the band was removed laparoscopically following an unsuccessful attempted removal by endoscopy. Clinicians who follow up patients with gastric banding should be aware of this condition. The early detection of erosion is important to avoid this serious complication.
Keywords
bariatric surgery;hemorrhage;laparoscopy;postoperative complication
1. Introduction
Morbid obesity is a common health problem worldwide. Nonsurgical approaches are usually ineffective for long-term weight reduction.1 Laparoscopic adjustable gastric banding (LAGB) has been extensively used around the world for the treatment of morbid obesity. It is a minimally invasive procedure, effective, and reversible. Life-threatening hemorrhage as a late complication of LAGB is extremely rare.2 ; 3 We report a case of massive upper gastrointestinal bleeding due to intragastric erosion of an adjustable gastric band. A complete review of the English literature on this topic was performed through PubMed search. We have also directly contacted the authors of the reported cases by e-mail to collect missing data of some patients.
2. Case report
A 38-year-old woman had undergone LAGB for treatment of morbid obesity 6 years before presenting to our hospital. The gastric band was a Swedish Adjustable Gastric Band (2002), which is a high-volume, low-pressure band. She was not regularly followed up. She lost 35 kg during that period. Except for obesity, the patient did not have any serious chronic medical illness. She was admitted to the emergency department (Al-Ain Hospital) complaining of epigastric pain, dizziness, and fainting. She had fresh red blood per rectum and melena for the past 6 days with no hematemesis. On examination, she was pale, her pulse was 133 beats/min, and her blood pressure dropped to 80/50 mmHg. The abdomen was soft with tenderness in the epigastrium. Her hemoglobin was 6.1 g/dL. The patient received aggressive resuscitation with 5 L of crystalloids, 5 units of packed red blood cells, and 4 units of fresh frozen plasma.
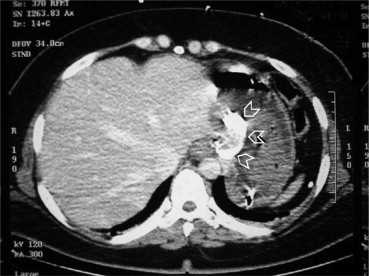
Upper gastrointestinal endoscopy showed an eroding gastric band protruding into the proximal stomach and fresh blood clot without active bleeding. The patient became hemodynamically stable, and the urine output improved to 100 mL/h. Computed tomographic scan of the abdomen with intravenous contrast showed migration of the gastric band into the stomach. There was no intraperitoneal fluid or free gas (Fig. 1). The patient stayed in the intensive care unit for 2 days and was transferred to the surgical ward where she was managed conservatively and did not require surgery. The patient was discharged to be followed up at the Morbid Obesity Unit at Tawam Hospital. She was advised to wait until the erosion is complete so as to remove the band endoscopically.
|
|
|
Figure 1. Computed tomographic scan of the abdomen with intravenous contrast showing the gastric band (arrowheads) eroding through the stomach wall. No free air or fluid in the peritoneum was detected. |
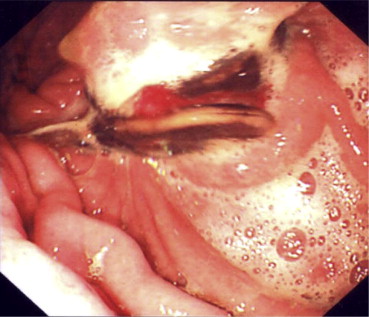
After 1 year, the patient complained of continuous abdominal pain which was not relieved with usual analgesia. An attempt to remove the band endoscopically under general anesthesia was performed (Fig. 2). Because of lack of special endoscopic instruments, the intraluminal endoscopic removal of the band has failed. The patient underwent laparoscopic removal of the band through four ports. The band was eroding the anterior wall of the stomach and protruded into the lumen. Adhesiolysis was performed. The posterior wall of the stomach was intact. Incision through a healthy scar-free area in the anterior wall of the stomach was performed to access the gastric lumen. The band was cut and removed. The incision of the anterior wall of the stomach, and the defect produced by the eroding band were closed in two layers using 00 nonabsorbable sutures. The integrity of the repair was examined by endoscopic air insufflation of the stomach and by methylene blue test. The postoperative period was uneventful. Three months after surgery, the patient was free of pain and tolerated normal diet. The patient’s weight was within average levels. She preferred to be kept on diet control rather than undergo a gastric bypass operation as a secondary weight-reducing operation.
|
|
|
Figure 2. Preoperative upper gastrointestinal endoscopy showing erosion of the gastric band into the lumen of the stomach. |
3. Discussion
Delayed massive upper gastrointestinal bleeding presenting after LABG is a very rare complication. Our patient had LAGB for treatment of obesity 6 years before developing severe upper gastrointestinal bleeding. The bleeding complication of our patient resulted from chronic intragastric migration and erosion of gastric wall by the adjustable band. To the best of our knowledge, this is the sixth case in the English literature having such delayed presentation (Table 1). Most of the reported cases (5 patients) were females who were 30–50 years old. Patients developed massive upper gastrointestinal bleeding 1.5–72 months after surgery. Two patients died.4 ; 5
| Author | Age (y) | Sex | Postoperative duration (mo) | BMI (kg/m2) | Clinical presentation | Treatment | Outcome |
|---|---|---|---|---|---|---|---|
| Chelala et al 1997 | 18 | M | 1.5 | 42 | Massive upper GI bleeding from Mallory–Wiess syndrome | Died soon after arrival to hospital | Died |
| Angrisani et al 2003 | 47 | F | 1.5 | 37.4 | Arrived at Emergency Department in coma and massive upper GI bleeding | Died soon after arrival to hospital. Postmortem showed that the stomach and small bowel were full of blood. | Died |
| Rao et al 2006 | 40 | F | 6 | 35.9 | Retrosternal pain, sudden syncope, bleeding per rectum | Band removed by laparotomy | Survived |
| Campos et al 2007 | 30 | F | 13 | 37 | Epigastric pain, sudden syncope, melena, hypotension | Band removal by gastroscopy | Survived |
| Iqbal et al 2008 | 49 | F | 4 | — | Upper abdominal pain, melena | Band removed by laparotomy | Survived |
| Torab et al 2010 (This article) | 38 | F | 72 | 40 | Epigastric pain, fainting, bleeding per rectum, melena | Conservative management followed by band removal after 1 y by laparoscopy | Survived |
Intragastric migration of the gastric band is an underreported complication. Its incidence may reach up to 11% after long-term follow-up.6 It may cause serious complications including severe infection and sepsis, gastric bleeding, and even hypovolemic shock.7; 8 ; 9 Late intragastric penetration is usually asymptomatic and occurs slowly over time. Epigastric pain is the most common symptom in symptomatic patients.9 Other symptoms may include weight gain, increasing need for band adjustment, and spontaneous port infection.10 These symptoms should raise early suspicion of intragastric band migration. The patients who are complaining of abdominal pain may receive analgesics like nonsteroidal anti-inflammatory drugs. This can mislead the treating physician when the patient starts to develop gastric bleeding.1 Erosion was not initially suspected in our patient, so a plain abdominal X-ray which possibly could have raised the suspicion of the accurate diagnosis of our case was not performed. Although upper gastrointestinal endoscopy can detect the presence of migrating gastric band in the gastric lumen, it may be unable to determine the site of bleeding during the acute phase of hemorrhage. In one of the reported cases, the bleeding was due to erosion into the celiac axis which was detected during laparotomy.1
Intragastric migration of the band may be caused by chronic ischemia due to pressure applied to the gastric wall or due to chronic inflammation resulting from the reaction of the tissues to the silicon gastric band.9 ; 11 This may be avoided by using different types and brands of bands as high-volume, low-pressure bands.3; 11 ; 12 Continuous care for patients with LAGB should include regular follow-up gastroscopy and barium swallow. This can help in the early diagnosis and management of migrating gastric bands especially in asymptomatic cases.9 ; 10 Our patient did not have regular follow-up and presented with delayed acute severe upper gastrointestinal bleeding that responded to aggressive resuscitation and blood transfusion. When the patient became hemodynamically stable, abdominal computed tomography scan was performed to exclude the presence of intraperitoneal free air, fluid, or abscess formation.13
Another cause for delayed life-threatening upper gastrointestinal bleeding following gastric banding was Mallory–Weiss syndrome, which had been reported in one of the patients.5
The treatment of intragastric migration of a gastric band is controversial. Some surgeons prefer the conservative approach with repeated follow-up gastroscopy until the band becomes accessible to be removed endoscopically.4 Laparoscopic removal or even laparotomy is indicated in the presence of acute uncontrolled bleeding caused by the eroding band.1 In our patient, we decided to wait until erosion is complete. Because of the severe intolerable continuous pain that prevented the patient from having a normal lifestyle, the patient asked for a removal of the band. Accordingly, we have chosen to remove the band. Initially, we tried to remove it endoscopically. Finally, it was successfully removed with laparoscopy. The decision to remove or replace the band or to convert to another bariatric procedure should be made preoperatively if possible.3 We advised the patient to undergo gastric bypass operation as a secondary weight-reducing operation, but the patient preferred to wait because her weight was within average limits.
In summary, delayed massive life-threatening upper gastrointestinal bleeding is a serious complication of LAGB. Chronic migration of the band through the stomach is usually asymptomatic. Clinicians should be aware of such a complication, and regular follow-up of patients is essential for its early detection and management.
Acknowledgments
We thank Professor Guy-Bernard Cadière, European School of Laparoscopic Surgery, Brussels, Belgium, and Professor Giovanni Lesti, Surgical Department, General Hospital of Lanciano, Italy, for sending us the details of the first and second patients.
References
- 1 M. Iqbal, S. Manjunath, M. Seenath, A. Khan; Massive upper gastrointestinal hemorrhage: an unusual presentation after laparoscopic adjustable gastric banding due to erosion into the celiac axis; Obes Surg, 18 (2008), pp. 759–760
- 2 M. Gagner, L. Milone, E. Yung, A. Broseus, A.A. Gumbs; Causes of early mortality after laparoscopic adjustable gastric banding; J Am Coll Surg, 206 (2008), pp. 664–669
- 3 A.D. Rao, G. Ramalingam; Exsanguinating hemorrhage following gastric erosion after laparoscopic adjustable gastric banding; Obes Surg, 16 (2006), pp. 1675–1678
- 4 L. Angrisani, F. Furbetta, S.B. Doldi, et al.; Lap band adjustable gastric banding system: the Italian experience with 1863 patients operated on 6 years; Surg Endosc, 17 (2003), pp. 409–412
- 5 E. Chelala, G.B. Cadiére, F. Favretti, et al.; Conversions and complications in 185 laparoscopic adjustable silicone gastric banding cases; Surg Endosc, 11 (1997), pp. 268–271
- 6 A. Westling, K. Bjurling, M. Ohrvall, S. Gustavsson; Silicone-adjustable gastric banding: disappointing results; Obes Surg, 8 (1998), pp. 467–474
- 7 J. Campos, A. Ramos, M. Galvão Neto, et al.; Hypovolemic shock due to intragastric migration of an adjustable gastric band; Obes Surg, 17 (2007), pp. 562–564
- 8 S.L. Brown, M.H. Reid, H.J. Duggirala; Adjustable silicone gastric banding adverse events reported to the Food and Drug Administration; J Long Term Eff Med Implants, 13 (2003), pp. 509–517
- 9 B. Hainaux, E. Agneessens, E. Rubesova, et al.; Intragastric band erosion after laparoscopic adjustable gastric banding for morbid obesity: imaging characteristics of an underreported complication; AJR Am J Roentgenol, 184 (2005), pp. 109–112
- 10 W. Brown, A. Korin, P. Burton, P.E. O'Brien; Laparoscopic adjustable gastric banding; Aust Fam Physician, 38 (2009), pp. 972–976
- 11 Akova A. Doğan ÜB, S. Solmaz, M. Aydin; Gastroscopic removal of a migrated adjustable gastric band: a case report; Turk J Gastroenterol, 21 (2010), pp. 297–301
- 12 J. Ponce, B. Lindsey, S. Pritchett, M. Bleech, K. Marlowe; New adjustable gastric bands available in the United States: a comparative study; Surg Obes Relat Dis, 7 (2011), pp. 74–79
- 13 H. Prosch, R. Tscherney, S. Kriwanek, D. Tscholakoff; Radiographical imaging of the normal anatomy and complications after gastric banding; Br J Radiol, 81 (2008), pp. 753–757
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?