Abstract
The applications of synchrotron X-ray powder diffraction (XRPD) and laser micro-Raman techniques in an examination of the dissolution, transformation, and gasification of pure cellulose and models for biomass residue under hydrothermal conditions in a diamond-anvil cell are reported. The results contribute to the measurement of in situ time-resolved profiles of biomass reactions, catalyst stability, and residue formation that occur in aqueous fluids at near- and supercritical conditions.
Introduction
The study of hydrothermal systems for combustion and related processes has been an active research area for many years, with there even being some limited examples of industrial application [1-3]. The results presented here represent an advancement of our efforts to develop the hydrothermal diamond-anvil cell (HDAC) as a valuable experimental tool for in situ measurements under hydrothermal or supercritical water (SCW) conditions. Our particular interest is focused on the use of SCW as a reaction medium for biomass conversion and combustion [4-10]. The potential for hydrothermal processing as a unique and efficient method for biomass conversion has previously been demonstrated for various other experimental setups and biomass varieties [11-14]. Synchrotron radiation offers great potential for fundamental and applied study of hydrothermal processes. We report here on our successful efforts to perform in situ synchrotron X-ray powder diffraction (XRPD) and laser micro-Raman spectrometry analyses of cellulose and biomass residue under hydrothermal conditions using the HDAC.
In this communication, we will describe a new approach and discuss our results, which agree with established mechanisms for cellulose and biomass evolution, and allow us to propose, for the first time, a more detailed hypothesis for biomass transformation within a SCW-dominated environment in terms of particulate behavior. We first discuss the physicochemical data of cellulose and biomass materials. Next, we explain why microstructural and crystallographic evidence suggests that cellulosic biomass loses crystallinity with an increase in temperature. We then discuss a plausible mechanism which we believe satisfactorily accounts for both the chemical and structural data. In conclusion, we suggest some practical implications of the proposed approach.
Experimental
All of the work described here was performed using a Bassett-type HDAC (Foxwood Instruments, Ithaca, NY, USA) [15]. The HDAC permits in situ measurements at high temperature and pressure, and has been described in detail elsewhere [16, 17]. All chemicals were purchased from Sigma-Aldrich (Burlington, ON, Canada) and were used without further purification. A stainless-steel gasket, 250 μm in thickness, with sample chamber of 250 μm o.d., was used in all the experiments. The sample chamber was loaded with distilled water and analyte under a constant flow of nitrogen, which was maintained during the heating cycle of the cell.
Raman microscopy setup and data acquisition/processing
Raman spectra were recorded on a Renishaw inVia Raman microscope system using either 514.5- or 785-nm laser excitation. Calibration of the Raman spectrometer was performed prior to each set of measurements with a standard Si wafer. The proprietary software Renishaw WiRE™ (version 3.2, Wotton-under-Edge, Gloucestershire, UK), was used for data acquisition and processing. Focusing was achieved with a super long-working distance 20× objective (Olympus SLMPLN20X; Olympus, Waltham, MA, USA).
XRPD setup
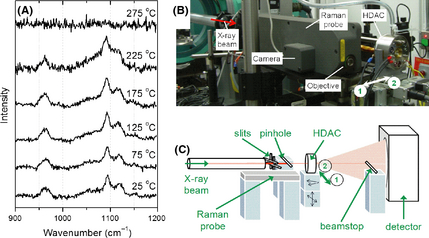
XRPD was performed using the hard X-ray micro analysis (HXMA, 06ID-1) beamline at the Canadian Light Source (Saskatoon, Saskatchewan), Canadas national synchrotron radiation facility. The cell was mounted on a motorized swivel stage (SA05B-RL; Kohzu Precision, Kawasaki, Japan), allowing for angular rotation of the cell, which was itself mounted on a second stage to provide vertical and horizontal motion. The cell was fixed in position on the stage using an insulating U-shaped mount, machined from Grade A lava stone (Maryland Lava, Bel Air, MD, USA), to prevent heat dissipation from the cell to sensitive components. A 250 μm pinhole was used to prevent interferences from beam divergence and scattering. The layout of the associated equipment in the experimental hutch has been described in detail earlier [18]. Shown in Figure 1B and C is an image and sketch of the setup used in our experiments.
|
|
|
Figure 1. (A) Raman spectra (785 nm laser line) for cellulose with increasing temperature showing the loss of crystallinity at temperatures >225°C, (B) image and (C) sketch (not to scale) of synchrotron setup for XRPD measurements in the HDAC at the HXMA beamline. The cell was shuttled between position 1 (Raman/camera) and position 2 (X-ray) for these measurements. XRPD, X-ray powder diffraction; HDAC, hydrothermal diamond-anvil cell; hard X-ray micro analysis. |
XRPD and Raman probe data acquisition and processing
X-ray diffraction images were recorded on a MAR345 image plate detector (Marresearch, Norderstedt, Germany) at the HXMA beamline. Exposure times were 60 s unless otherwise noted, with λ = 0.509175 Å. Calibration was performed using a NIST lanthanum hexaboride (LaB6) standard. The raw image files were processed and integrated with Fit2D (V12.077) software [19]. Baseline correction (spline interpolation) and final processing of the resultant powder patterns were performed with FullProf Suite (1.10) software [20].
Raman spectroscopic measurements at the beamline were conducted using a Renishaw RM 2000 Raman microscope system (Renishaw, Wotton-under-Edge, Gloucestershire, UK) coupled to a Renishaw RP20 fiber optic probehead with 514.5-nm laser excitation. The probe head also incorporated a Philips ToUcam PRO II (1.2 Megapixel) CCD camera, which was used to obtain images of the sample in the HDAC. A super long-working distance 20× objective (Olympus SLMPLN20X) was mounted on the probehead. The probe was positioned parallel to the X-ray beam path and the HDAC was moved between the two positions as shown in Figure 1B and C to measure images (position 1) and X-ray patterns (position 2).
Discussion
We have previously reported on the decomposition of glucose, cellulose, and lignocellulose under hydrothermal conditions [4, 21, 22]. Our approach in the present work has been to extend the study of high temperature high-pressure aqueous systems with the use of more advanced analytical techniques. In the initial stage, we measured the micro-Raman spectra of microgranular cellulose in the HDAC. As this organic material is a rather weak Raman scatterer, ideally a prolonged measurement must be performed (e.g., 30 min) to achieve a reasonable signal-to-noise ratio. In this work, the approach taken to avoid long data acquisition times was to select small spectral windows in an attempt to identify chemical changes as they occurred. Having identified two key regions of interest (namely 300–500 and 900–1200 cm−1, see Fig. S1), we proceeded with an investigation of the changes in the spectra with increasing temperature in the HDAC, as shown in Figure 1A. In the 900–1200 cm−1 region, we observed a clear loss of cellulose crystallinity or dissolution of cellulose at temperatures greater than 225°C.
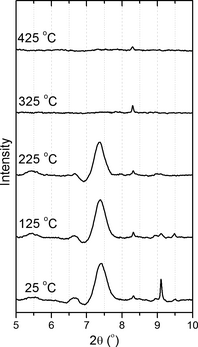
Our initial synchrotron experiments, using the setup shown in Figure 1B and C, employed these Raman data as a basis for experimental validation. Further details of the experimental methods are provided in the Supporting Information. Figure 2 shows an analogous experiment for measurement of X-ray diffraction of cellulose with increasing temperature in the HDAC. Heating rates for both the Raman and diffraction measurements were ~10°C/min. Again, we observed a clear loss of signal at temperatures greater than 225°C, corresponding to a loss of crystallinity. Notably, the initial diffraction pattern (and indeed the initial Raman spectrum) was not recovered upon cooling of the sample, implying that the change is irreversible. This is in agreement with the mechanisms we proposed earlier for cellulose conversion under hydrothermal conditions, with an initial step of cellulose dissolution and depolymerization (hydrolysis), followed by reaction of the component sugars [7-10, 23-25]. The significance of this measurement relates to defining a means to identify the point of loss of crystallinity, presumably immediately preceding dissolution of the cellulosic component of biomass, and possible use in refining existing kinetic models for this transformation. We note that the identification and quantification of the products of the cellulose decomposition, widely reported in the literature, is outside of the scope of this manuscript.
|
|
|
Figure 2. XRPD patterns for cellulose with increasing temperature showing the loss of crystallinity at temperatures >225°C. XRPD, X-ray powder diffraction. |
Our subsequent work has dealt with the measurement of XRPD data for other important solid materials that are present in biomass residue, measurement of pressure, and the ongoing development of the Raman system for simultaneous (same spot) XRPD and micro-Raman measurements. A brief discussion on the limitation of the combined technique, in particular with regard to the reliable measurement of pressure under reaction conditions is provided in the Supporting Information.
We employed several different calcium-based compounds as models for the residue that may be produced from the hydrothermal decomposition reactions of lignocellulosic materials. These model compounds, viz., calcium oxalate, calcium hydroxyapatite, and calcium carbonate, were selected based on earlier reports of their transformation under hydrothermal or high-pressure conditions, the prevalence of calcium-containing deposits from SCW biomass conversion [5] and the high proportion of calcium in biomass, particularly in certain woody species [26-28]. No data are actually reported for calcium carbonate as, unfortunately, this substance proved to be too weak a Raman scatterer to produce reliable data.
Calcium oxalate is a species known to be present in a wide variety of types of biomass and plays a key role in structural and biochemical processes. In some cases, it is present in crystalline form in plant tissues. Figure 3 shows the visible changes occurring in the cell during the heating experiment. With a suitable choice of heating rate (~10°C/min), it was possible to collect the images at position 1 (see Fig. 1B and C regarding the location of position 1 and 2), move the cell while heating to position 2, perform the XRPD measurement and finally return the cell to position 1 while reading the detector image plate. Thus, the data collection can be performed in a relatively short time period (i.e., a total heating time of 45–60 min).
|
|
|
Figure 3. Images of calcium oxalate in HDAC with increasing temperature showing qualitatively the change in the material during heating, and the N2 bubble dissolution used for pressure determination (Th = 150°C, ρ = 917.01 kg/m3). HDAC, hydrothermal diamond-anvil cell. |
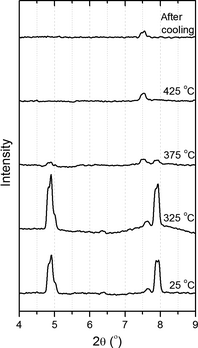
The XRPD data measurements performed for calcium oxalate under hydrothermal conditions show a dramatic change in the powder pattern at a temperature close to the critical temperature of pure water (Tc = 374°C), as shown in Figure 4. Similar to the earlier observations with cellulose, the crystalline component of the sample appears to react and is not present in the pattern obtained after cooling. Moreover, there was an increased amount of gaseous products present in the sample volume after cooling (see Fig. 3). We suggest that calcium oxalate under these conditions reacts irreversibly via a decarboxylation or decarbonylation pathway, producing CO2 or CO, respectively.
|
|
|
Figure 4. XRPD patterns for calcium oxalate with increasing temperature showing the change in structure at around 375°C (patterns between 25 and 325°C omitted for clarity). XRPD, X-ray powder diffraction. |
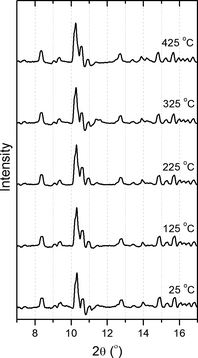
Calcium hydroxyapatite and related materials are known to undergo structural transformations at hydrothermal conditions and at high pressures [29, 30]. In our XRPD study of this material, however, we observed no significant changes with increasing temperature, as shown in Figure 5. It should be emphasized that our study was principally concerned with the understanding of rapid processes for application in chemical reactors, whereas the structural changes reported in the literature are most likely due to less rapid transformations. The stability of this material under the tested conditions provides an indication of the types of material that may be present in the residue following prolonged hydrothermal treatment.
|
|
|
Figure 5. XRPD patterns for hydroxyapatite with increasing temperature (Th = 225°C, ρ = 833.75 kg/m3) showing little change over the studied range. XRPD, X-ray powder diffraction. |
Conclusions
SCW remains a promising alternative to conventional chemical and materials processing technologies. We have demonstrated in this work some key analyses to aid in our understanding of the fundamental processes that occur during the hydrothermal transformations of biomass models. Our results concur with established mechanisms for cellulose reactivity and provide new insights into the chemical pathways involved in the production of residues in these experiments. We have also demonstrated the usefulness of synchrotron science for biomass conversion research. Specifically, the use of XRPD as an in situ measurement tool is invaluable and, more generally, synchrotron science offers a wealth of opportunities for fundamental and applied research for hydrothermal processes.
The data we have presented here will be greatly enhanced by the proposed simultaneous (i.e., same spot) XRPD/micro-Raman facility at the Canadian Light Source. Such a setup, demonstrating the advantages of simultaneous measurement, has already been developed at several synchrotron facilities [31, 32], and the addition of this technique will be advantageous in developing the HDAC experimental capabilities. In this work, we have primarily employed both Raman spectroscopic measurements and XRPD to determine the crystallinity of relevant moieties. Although the current work has not fully highlighted the advantages, in future work, the complementary nature of these two techniques will be fully employed, that is, to measure the functional group transformations via Raman spectroscopy and the consequent crystalline sample changes using XRPD.
Our observations suggest possible explanations for a number of important phenomena. For example, the loss of crystallinity of the cellulose during dissolution of the cellulose prior to reaction of component sugars and the processes involved in the formation of calcium-containing residues. However, the general scheme we have proposed for biomass rearrangements in hydrothermal environment is as yet incomplete. It is clear from our analyses that many questions remain to be answered before the picture of biomass structure evolution can be described in detail. What stimulates loss of crystallinity of cellulose other than thermochemical interaction? What role is played by catalysts and volatilization of selected species? Are hydrolysis and dissolution alone responsible for biomass conversion, or do they interact with other processes (e.g., pyrolysis)? The results of this study also provide the possibility for measurement of in situ time-resolved profiles of biomass reaction, catalyst stability, and residue formation in hydrothermal biomass processing.
Acknowledgments
We thank Jason Maley, Sophie Brunet, and Ramaswami Sammynaiken (Saskatchewan Structural Sciences Centre, University of Saskatchewan), Kelly Akers (ProSpect Scientific Inc.), and Richard Bormett (Renishaw Inc.) for their kind support with Raman instrumentation. We are grateful for the assistance of our colleagues Gaëlle Dupouy (York University) and Chang-Yong Kim (beamline scientist, Canadian Light Source) in developing our beamline experiments. We also thank John Tse (University of Saskatchewan) and William Bassett (Cornell University) for their helpful suggestions related to diamond-anvil cells and measurement. We are grateful to the assistance given by technical staff at the College of Engineering, University of Saskatchewan. The research was supported by Discovery, Strategic and Research Tools and Instrumentation grants to I. S. B. and J. A. K. from the Natural Sciences and Engineering Research Council of Canada and as part of the Agricultural Biorefineries Innovation Network (Canada).
Conflict of Interest
None declared.
References
- Hodes, M., P. A. Marrone, G. T. Hong, K. A. Smith, and J. W. Tester. 2004. Salt precipitation and scale control in supercritical water oxidation – Part A: fundamentals and research. J. Supercrit. Fluids29:265–288.
- Marrone, P. A., S. D. Cantwell, and D. W. Dalton. 2005. SCWO system designs for waste treatment: Application to chemical weapons destruction. Ind. Eng. Chem. Res.44:9030–9039.
- Bermejo, M. D., and M. J. Cocero. 2006. Supercritical water oxidation: a technical review. AIChE J.52:3933–3951.
- Fang, Z., T. Minowa, C. Fang, R. L. Smith, H. Inomata, and J. A. Kozinski. 2008. Catalytic hydrothermal gasification of cellulose and glucose. Int. J. Hydrogen Energy33:981–990.
- Bocanegra, P. E., C. Reverte, C. Aymonier, A. Loppinet-Serani, M. M. Barsan, I. S. Butler, et al. 2010. Gasification study of winery waste using a hydrothermal diamond anvil cell. J. Supercrit. Fluids53:72–81.
- Calahoo, C., M. M. Barsan, M. L. Thomas, J. A. Kozinski, and I. S. Butler. 2011. Hydrothermal Raman microscopy studies of manganese carbonyls. Vib. Spectrosc.57:2–7.
- Sobhy, A., I. S. Butler, and J. A. Kozinski. 2007. Selected profiles of high-pressure methanol-air flames in supercritical water. Proc. Combust. Inst.31:3369–3376.
- Fang, Z., H. Assaaoudi, A. Sobhy, M. M. Barsan, I. S. Butler, R. I. L. Guthrie, et al. 2008. Use of methanol and oxygen in promoting the destruction of deca-chlorobiphenyl in supercritical water. Fuel87:353–358.
- Sobhy, A., R. I. L. Guthrie, I. S. Butler, and J. A. Kozinski. 2009. Naphthalene combustion in supercritical water flames. Proc. Combust. Inst.32:3231–3238.
- Xu, S. K., I. Butler, I. Gokalp, and J. A. Kozinski. 2011. Evolution of naphthalene and its intermediates during oxidation in subcritical/supercritical water. Proc. Combust. Inst.33:3185–3194.
- Matsumura, Y., T. Minowa, B. Potic, S. R. A. Kersten, W. Prins, van Swaaij W. P. M., et al. 2005. Biomass gasification in near- and super-critical water: Status and prospects. Biomass Bioenergy29:269–292.
- Kruse, A., P. Bernolle, N. Dahmen, E. Dinjus, and P. Maniam. 2010. Hydrothermal gasification of biomass: consecutive reactions to long-living intermediates. Energy Environ. Sci.3:136–143.
- Letellier, S., F. Marias, P. Cezac, and J. P. Serin. 2010. Gasification of aqueous biomass in supercritical water: a thermodynamic equilibrium analysis. J. Supercrit. Fluids51:353–361.
- Hammerschmidt, A., N. Boukis, E. Hauer, U. Galla, E. Dinjus, B. Hitzmann, et al. 2011. Catalytic conversion of waste biomass by hydrothermal treatment. Fuel90:555–562.
- Bassett, W. A., A. H. Shen, M. Bucknum, and I. M. Chou. 1993. A new diamond-anvil cell for hydrothermal studies to 2.5 GPa and from 190°C to 1200°C. Rev. Sci. Instrum.64:2340–2345.
- Smith, R. L., and Z. Fang. 2009. Techniques, applications and future prospects of diamond anvil cells for studying supercritical water systems. J. Supercrit. Fluids47:431–446.
- Syassen, K.2008. Ruby under pressure. High Pressure Res.28:75–126.
- Desgreniers, S., and C.-Y. Kim. 2011. La matière condensée sous conditions extrêmes: un programme de recherche au Centre canadien de rayonnement synchrotron. Physics in Canada / La Physique au Canada67:3–6.
- Hammersley, A. P., S. O. Svensson, M. Hanfland, A. N. Fitch, and D. Häusermann. 1996. Two-Dimensional Detector Software: From Real Detector to Idealised Image or Two-Theta Scan. High Pressure Res.14:235–248.
- Rodríguez-Carvajal, J.1993. Recent advances in magnetic structure determination by neutron powder diffraction. Physica B.192:55–69.
- Hashaikeh, R., Z. Fang, I. S. Butler, and J. A. Kozinski. 2005. Sequential hydrothermal gasification of biomass to hydrogen. Proc. Combust. Inst.30:2231–2237.
- Hashaikeh, R., Z. Fang, I. S. Butler, J. Hawari, and J. A. Kozinski. 2007. Hydrothermal dissolution of willow in hot compressed water as a model for biomass conversion. Fuel86:1614–1622.
- Fang, Z., and J. A. Kozinski. 2000. Phase behavior and combustion of hydrocarbon-contaminated sludge in supercritical water at pressures up to 822 MPa and temperatures up to 535 degrees C. Proc. Combust. Inst.28:2717–2725.
- Fang, Z., and J. A. Kozinski. 2001. Phase changes of benzo(a)pyrene in supercritical water combustion. Combust. Flame124:255–267.
- Fang, Z., T. Minowa, R. L. Smith, T. Ogi, and J. A. Kozinski. 2004. Liquefaction and gasification of cellulose with Na2CO3 and Ni in subcritical water at 350 degrees C. Ind. Eng. Chem. Res.43:2454–2463.
- Meerts, P.2002. Mineral nutrient concentrations in sapwood and heartwood: a literature review. Ann. For. Sci.59:713–722.
- Misra, M. K., K. W. Ragland, and A. J. Baker. 1993. Wood ash composition as a function of furnace temperature. Biomass Bioenergy4:103–116.
- Hudgins, J. W., T. Krekling, and V. R. Franceschi. 2003. Distribution of calcium oxalate crystals in the secondary phloem of conifers: a constitutive defense mechanism?New Phytol.159:677–690.
- Xu, J., D. F. R. Gilson, I. S. Butler, and I. Stangel. 1996. Effect of high external pressures on the vibrational spectra of biomedical materials: calcium hydroxyapatite and calcium fluoroapatite. J. Biomed. Mater. Res.30:239–244.
- Parthiban, S. P., K. Elayaraja, E. K. Girija, Y. Yokogawa, R. Kesavamoorthy, M. Palanichamy, et al. 2009. Preparation of thermally stable nanocrystalline hydroxyapatite by hydrothermal method. J. Mater. Sci. Mater. Med.20:S77–S83.
- Davies, R. J., M. Burghammer, and C. Riekel. 2005. Simultaneous microRaman and synchrotron radiation microdiffraction: tools for materials characterization. Appl. Phys. Lett.87:264105.
- Boccaleri, E., F. Carniato, G. Croce, D. Viterbo, W. van Beek, H. Emerich, et al. 2007. In situ simultaneous Raman/high-resolution X-ray powder diffraction study of transformations occurring in materials at non-ambient conditions. J. Appl. Crystallogr.40:684–693.
Document information
Published on 01/06/17
Submitted on 01/06/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?