Summary
Background and aims
Rapid null response (rNR), defined as less than one log decline of Hepatitis C virus (HCV-RNA) at Week 4 of treatment with pegylated interferon-α and ribavirin (PegIFN/RBV), is highly correlated with treatment failure in patients with chronic hepatitis C (CHC), genotype-1 (GT-1). In this study, we investigate the possible predictors of rNR.
Methods
We retrospectively analyzed a cohort of 199 GT-1 CHC naive patients who had been treated with a dual therapy of PegIFN/RBV. Clinical parameters and genotypes of rs12979860, the single nucleotide polymorphisms (SNPs) of interleukin-28B (IL28B ) were analyzed for their relationship with rNR.
Results
Of the patients analyzed, 41.7% did not exhibit a rapid virological response (RVR). Only 13.1% of patients who experienced a rNR showed an RVR. The treatment failure rate was 36.2%. High baseline viral load (OR: 5.74; p = 0.028), nonrapid virological response (non-RVR; OR: 4.32; p = 0.004) and rNR (OR: 51.82; p < 0.001) were the predictors of treatment failure. In addition, both the non-CC allele of rs12979860 (OR: 13.8; p < 0.001) and the Hb (hemoglobin) decline to < 3 g/dL within 4 weeks of treatment (no early anemia; OR: 4.6; p = 0.024) were predictors of rNR.
Conclusions
rNR predicted treatment failure early in GT-1 CHC patients treated with PegIFN/RBV. Non-CC genotype of rs12979860 and no early anemia were significant predictors of rNR.
Keywords
Anemia ; Chronic hepatitis C ; Early null-response ; Interleukin-28B polymorphism
Introduction
The development of direct-acting antiviral agents (DAAs) for the treatment of chronic hepatitis C (CHC) has revolutionized the treatment strategies [1] ; [2] . Although DAAs are now incorporated into the standard of care in Western countries [1] , dual therapy of pegylated interferon-α and ribavirin (PegIFN/RBV) is still a popular and effective treatment in countries where DAAs are not available or not affordable [3] ; [4] ; [5] . The goals of treatment for CHC are to eradicate the hepatitis C virus (achieving sustained viral response, SVR) and to decrease the risks of cirrhosis, hepatocellular carcinoma, and hepatic decompensation [6] ; [7] . According to previous studies, PegIFN/RBV could achieve 42–79% of the SVR rate in HCV genotype 1 (GT-1) [4] ; [8] . However, almost all patients treated with PegIFN/RBV experienced one or more adverse events during the course of therapy. Adverse reactions are a major reason that patients decline or stop therapy altogether. In the registration trials of peginterferon-α-2a and 2b plus RBV, 10–14% of patients had to discontinue therapy due to an adverse event. Moreover, these undesirable side effects may increase morbidity [8] . Therefore, predictors of treatment failure, to avoid unnecessary treatment and treatment-related side effects, are as important as predictors of successful treatment.
The new reagents (DAAs) have been introduced into the treatment protocol to reduce the treatment failure rate and to rescue the patients who were failed by PegIFN/RBV [2] . Therefore, the factors contributing to treatment failure with dual therapy in GT-1 patients became an interesting and important issue to explore. No early virological response (EVR; null response) or positivity of HCV-RNA at Week 24 of treatment is the stopping rule. Less than one log HCV-RNA decay at Week 4 has been reported to predict treatment failure accurately and is the single strongest predictor for treatment failure [9] . Therefore, rapid null response (rNR) was named. However, in the triple therapy of PegIFN/RBV/boceprevir or telaprevir, viral response at Week 4 after a lead-in phase can also predict treatment outcomes [1] ; [10] . This highlights the role of rNR not only in dual therapy, but also in the era of triple therapy.
In addition, interleukin-28B (IL28B ) single nucleotide polymorphisms (SNPs) rs12979860 are powerful predictors for SVR [11] ; [12] ; [13] ; [14] . Lindh et al. [15] reported that IL28B SNPs influence the early viral kinetics in GT-1 CHC patients treated with PegIFN/RBV. Therefore, we conducted this study to clarify the relationship between rNR and treatment failure, and the association between IL28B SNP and rNR.
Patients and methods
Patients
We retrospectively analyzed a cohort of treatment naive GT-1 CHC patients who had been treated with a combination therapy of PegIFN/RBV at Chang Gung Memorial Hospital, Linkou Medical center and agreed to receive genomic study of IL28B between June 2007 and May 2011. All had received liver biopsies that were evaluated using the Metavir and Ishak scoring system. Patients with other concomitant liver diseases, such as hepatitis B virus, human immunodeficiency virus, alcoholic liver disease, and autoimmune hepatitis, were excluded. PegIFN/RBV included either peginterferon-α-2a (180 μg/week) or peginterferon-α-2b (1.5 μg/kg/week) subcutaneously plus weight-based ribavirin (1000 mg/day for weight < 75 kg and 1200 mg/day for weight > 75 kg). Patients who did not fulfill the 80/80/80 adherence rule were also not enrolled. A total of 213 GT-1 CHC patients were identified. Fourteen were excluded because of no viral data at Week 4 or Week 12 in nine patients and no hemoglobin (Hb) data before Week 4 in five patients. Baseline characters of these 14 patients were comparable with those of the total cohort. There were 199 patients remaining in the study.
Definitions of treatment responses by serum level of HCV-RNA were: undetectable HCV-RNA 24 weeks after the cessation of treatment as sustained virological response (SVR), undetectable HCV-RNA at Week 4 of treatment as rapid virological response (RVR), and at least two log reduction of HCV-RNA from baseline at treatment Week 12 as EVR. The rNR was defined as less than one log decline of HCV-RNA at treatment Week 4. Those who failed to achieve SVR were defined as treatment failures. Patients who did not achieve EVR and therefore terminated PegIFN/RBV early were also counted as treatment failures.
Because of changing reimbursement policies by the Bureau of National Health Insurance according to professor Yus study [16] . For analytical convenience, patients were divided into two groups according to treatment duration. The standard treatment regimen included a 48-week treatment for patients with no RVR, patients with RVR but high baseline viral load (HCV-RNA > 0.4 × 106 IU/ml), and a 24-week treatment for patients with RVR and low baseline viral load (HCV-RNA < 0.4 × 106 IU/ml). Another group received a shortened treatment regimen which was a 24-week treatment for both patients with no RVR and patients with RVR but with high baseline viral load. No EVR was applied as the stop rule to both standard and shortened treatment regimens. Treatment was terminated if HCV-RNA was detectable at 24 weeks. During the treatment, early anemia was defined as a decline of Hb more than 3 g/dL before or at Week 4.
The HCV-RNA levels in this study were measured using a commercial quantitative polymerase chain reaction (PCR) assay, either VERSANT HCV RNA 3.0. Assay (HCV 3.0 bDNA assay, Bayer Diagnostics, Berkeley, CA, USA, lower limit of detection: 5.2 × 102 IU/mL) or COBAS TaqMan HCV Test (TaqMan HCV; Roche Molecular Systems Inc., Branchburg, NJ, USA, lower limit of detection: 15 IU/mL). If nondetection of HCV-RNA using the VERSANT HCV RNA 3.0 assay occurred, it would be tested further using the COBAS AMPLICOR HCV test, v2.0 (CA V2.0, Roche Diagnostic Systems, Pleasanton, CA, USA, lower limit of detection: 50 IU/mL). The HCV genotype was determined using a genotype-specific probe-based assay in the 5′ untranslated region (LiPA; Innogenetics, Ghent, Belgium).
Genomic DNA extraction and IL28 B genotyping
Anticoagulated peripheral blood was obtained from HCV patients. Genomic DNA was isolated from EDTA anticoagulated peripheral blood using the Puregene DNA isolation kit (Gentra Systems, Minneapolis, MN, USA) as previously described [12] . The oligonucleotide sequences flanking 10 IL28B polymorphisms were designed as primers for Taqman allelic discrimination. The allele specific primers for rs12979860 were labeled with a fluorescent dye (FAM and VIC) and used in the PCR reaction. Aliquots of the PCR product were genotyped with allele specific probes of SNPs using real-time PCR (ABI).
Ethics statements
All patients in this study provided written informed consent. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the ethical committees of Chang Gung Memorial Hospital.
Statistical analysis
The Chi-square test was used to compare the categorical variables of the groups. Continuous variables were compared using the Student t test or Mann-Whitney U test. Logistic regression analyses for predictors of treatment failure and rNR were conducted using patients' demographics, clinical variables, and IL28B SNPs. The clinical variables included gender, age, viral load of HCV-RNA, stage of fibrosis, body mass index (BMI), alanine transaminase (ALT), γ-glutamyl transpeptidase (γ-GT), Hb, and rs12979860 SNPs. The odds ratios (OR) and 95% confidence intervals (95% CI) were also calculated. All p values < 0.05 by the two-tailed test were considered statistically significant. Variables that achieved a statistical significance < 0.05 on univariate analysis were entered into multivariate logistic regression analysis to identify the significant independent predictive factors. All statistical analyses were performed with statistical software, SPSS for Windows (version 16, SPSS. Inc., Chicago, IL, USA) and SAS version 9.2 software (The SAS Institute, Cary, NC, USA).
Results
rNR had significant impact on treatment failure
The demographic characteristics of the 199 GT-1-CHC patients are shown in Table 1 . Their mean age was 54.0 ± 11.2 years and 57.3% (n = 114) were male. Forty-two (21.1%) patients had the CT or TT allele of IL28B . Standard treatment was received by 119 (59.8%) patients and 80 (40.2%) patients had shortened treatment. Sixty-nine (34.7%) patients developed early anemia. There were 116 (58.3%) patients who could not reach RVR and 26 (13.1%) patients demonstrated a rNR. The treatment failure rate in these patients was 36.1% (n = 72). The factors of older age, cirrhosis, baseline Hb level, high baseline viral load, CT/TT allele of rs12979860, no RVR and rNR were significantly associated with treatment failure ( Table 1 ). Multivariate logistic regression revealed high baseline viral load (OR: 5.74; p = 0.028), non-RVR (OR: 4.32; p = 0.004) and rNR (OR: 51.82; p < 0.001) were the independent significant predictors of treatment failure ( Table 2 ).
| Baseline characteristic | Overall n = 199 | Treatment | p | rNR | p | ||
|---|---|---|---|---|---|---|---|
| Failure n = 72 | Success n = 127 | Yes n = 26 | No n = 173 | ||||
| Male, n (%) | 114 (57.3) | 35 (48.6) | 79 (62.2) | 0.062 | 15 (57.7) | 99 (57.2) | 0.964 |
| Age, y, mean (SD) | 54.0 (11.2) | 56.9 (10.0) | 52.3 (11.5) | 0.011 | 54.0 (9.4) | 53.9 (11.5) | 0.890 |
| Necroinflammatory score, medium (range) | 5 (1–13) | 5 (1–10) | 5 (1–13) | 0.639 | 6 (2–9) | 5 (1–13) | 0.803 |
| Cirrhosis, n (%) | 50 (25.1) | 26 (36.1) | 24 (18.9) | 0.007 | 8 (30.8) | 42 (24.3) | 0.477 |
| BMI, kg/m2 , mean (SD) | 24.5 (3.4) | 24.7 (2.9) | 24.4 (3.6) | 0.450 | 25.2 (3.0) | 24.4 (3.4) | 0.215 |
| Baseline HCV RNA > 0.4mIU/mL, n (%) | 158 (79.4) | 66 (91.7) | 92 (72.4) | 0.001 | 22 (84.6) | 136 (78.6) | 0.480 |
| Baseline HCV RNA, log IU/mL, mean (SD) | 6.2 (0.9) | 6.4 (0.6) | 6.1 (1.0) | 0.340 | 5.8 (0.6) | 6.2 (0.9) | 0.414 |
| ALT, IU/L, mean (SD) | 113 (81) | 102 (68) | 120 (87) | 0.090 | 94 (51) | 116 (85) | 0.390 |
| γ-GT, IU/L, mean (SD) | 76.3 (85.7) | 81 (81) | 74 (92) | 0.128 | 96.2 (55.8) | 73.2 (89.2) | 0.004 |
| rs12979860 of CT + TT, n (%) | 42 (21.1) | 29 (40.3) | 13 (10.2) | < 0.001 | 18 (69.2) | 24 (13.9) | < 0.001 |
| Baseline Hb, g/dL, mean (SD) | 14.5 (1.6) | 14.2 (1.6) | 14.7 (1.5) | 0.028 | 14.4 (1.7) | 14.5 (1.6) | 0.867 |
| Hb decline < 3g/dL at Week 4, n (%) | 130 (65.3) | 51 (70.8) | 79 (62.2) | 0.219 | 23 (88.5) | 107 (61.8) | 0.003 |
| rNR, n (%) | 26 (13.1) | 26 (36.1) | 0 (0) | < 0.001 | |||
| No RVR, n (%) | 116 (58.3) | 62 (86.1) | 54 (42.5) | < 0.001 | 26 (100) | 90 (52.0) | < 0.001 |
| Treatment failure, n (%) | 26 (100) | 46 (26.6) | < 0.001 | ||||
ALT = alanine transaminase; BMI = body mass index; cirrhosis = Ishak fibrosis stages 5–6 or Metavir F4; γ-GT = gamma-glutamyl transpeptidase; Hb = hemoglobin; necroinflammatory score = Ishak modified HAI (histological activity index) score; PegIFN/RBV = pegylated interferon-α and ribavirin; rNR = rapid null response; RVR = rapid virological response.
| Responses | Variables | OR | 95% CI | p | |
|---|---|---|---|---|---|
| rNR | |||||
| rs12979860 | CC | 1 | |||
| CT or TT | 13.8 | 5.3–36.2 | <0.001 | ||
| Hb decline, n (%) | >3 g/dL | 1 | |||
| < 3 g/dL | 4.6 | 1.2–17.4 | 0.024 | ||
| Treatment failure | |||||
| rNR | No | 1 | |||
| Yes | 51.8 | 7.9–infinity | <0.001 | ||
| Baseline HCV RNA | < 0.4 mIU/mL | 1 | |||
| >0.4 mIU/mL | 5.7 | 1.2–57.0 | 0.028 | ||
| RVR | Yes | 1 | |||
| No | 4.3 | 1.5–13.9 | 0.004 | ||
CI = confidence interval; Hb = hemoglobin; OR = odds ratio; rNR = rapid null response; RVR = rapid virological response.
Both IL28B polymorphism and no early anemia predict the rNR
The current analysis highlights the importance of rNR in predicting treatment failure due to high OR. Differences between patients who demonstrated rNR and those who did not were examined. As shown in Table 1 , patients demonstrating rNR had higher levels of GGT (96.2 ± 55.8 IU/L vs. 73.2 ± 89.2 IU/L, p = 0.004), were more likely to display the nonfavorable alleles (CT/TT) of rs12979860 (69.2% vs. 13.9%, p < 0.001), and were less likely to have early anemia (88.5% vs.61.8%, p = 0.003). Therefore, the possible predictors for rNR were analyzed. As shown in Table 2 , by multivariate logistic analysis, only the CT/TT allele of rs12979860 (OR: 13.8, p < 0.001) and no early anemia (OR: 4.6, p = 0.024) were predictors of rNR. The factors associated with early anemia including gender, age, cirrhosis, baseline Hb level, BMI and rNR, as shown in Table 3 , were also analyzed. Age (OR: 1.04, p = 0.004) and rNR (OR: 5.56, p = 0.007) were the two factors associated with early anemia. An age of ≥ 55 years with AUROC (Area Under Receiver Operating Characteristic) of 0.62 (p = 0.004, 95% CI: 0.54–0.71) was found to increase the likelihood of early anemia.
| Variables | Univariate | Multivariable | |||||
|---|---|---|---|---|---|---|---|
| OR | 95% CI | p | OR | 95% CI | p | ||
| Gender | Male | 1 | |||||
| Female | 1.095 | 0.61–1.96 | 0.760 | ||||
| Age | 1.040 | 1.01–1.07 | 0.006 | 1.042 | 1.01–1.07 | 0.006 | |
| Cirrhosis | Yes | 1 | |||||
| No | 1.139 | 0.59–2.20 | 0.697 | ||||
| Baseline Hb | 1.196 | 0.99–1.44 | 0.061 | ||||
| BMI | 1.015 | 0.93–1.11 | 0.737 | ||||
| rNR | Yes | 1 | 1 | ||||
| No | 5.46 | 1.59–20.0 | 0.007 | 5.65 | 1.59–20.0 | 0.007 | |
BMI = body mass index; CI = confidence interval; Hb = hemoglobin; OR = odds ratio; rNR = rapid null response.
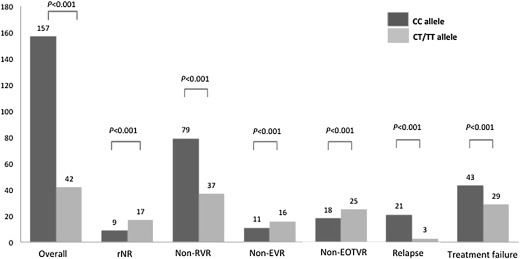
Patient groups were divided according to the genotype of rs12979860 and their treatment responses analyzed separately. As shown in Fig. 1 , significantly higher rates of rNR (40.5% vs. 5.7%, p < 0.001), non-EVR (38.1% vs. 7.0%, p < 0.001), non-RVR (88.1% vs. 50.3%, p < 0.001), nonend of treatment virological response (EOTVR) (59.5% vs. 11.5%, p < 0.001), relapse rate (17.6% vs. 15.1%, p < 0.001) and treatment failure (69.0% vs. 27.4%, p < 0.001) were observed in patients with the CT/TT allele than in those with the CC allele. In addition, because no early anemia also had significant impact on the rNR, this effect was analyzed in patients with different genotypes of rs12979860. In patients with the CC allele, those with no early anemia were significantly more likely to demonstrate rNR than patients with early anemia (100% vs. 0%, p = 0.027, Table 4 ). No such correlation was observed in patients with the CT/TT allele (83.3% vs. 16.7%, p = 0.299, Table 4 ).
|
|
|
Figure 1. The correlation between treatment responses and rs12979860. non-EOTVR = non-end of treatment virological response; non-EVR = non-early virological response; non-RVR = non-rapid virological response; rNR = rapid null response. |
| Rs12979860 | ||||||
|---|---|---|---|---|---|---|
| Early | CC allele | Non-CC allele | ||||
| Anemia | rNR | p | rNR | p | ||
| Yes (n = 8) | No (n = 149) | Yes (n = 18) | No (n = 24) | |||
| Yes, n (%) | 0 (0) | 58 (38.9) | 0.027 | 3 (16.7) | 8 (33.3) | 0.299 |
| No, n (%) | 8 (100) | 91 (61.1) | 15 (83.3) | 16 (66.7) | ||
rNR = rapid null response.
Discussion
This study strongly supports the notion that rNR is a powerful predictor of treatment failure in patients with GT-1 CHC receiving PegIFN/RBV. CT/TT genotypes of rs12979860 and Hb decline ≤ 3 g/dL by Week 4 (no early anemia) were the only two predictors of rNR.
Previous studies have shown that rNR has a high positive predictive value for treatment failure [9] ; [16] . Reau et al. [9] reported that 24% of all patients treated with PegIFN/RBV demonstrated rNR and 92% of patients could not achieve SVR. In addition, Yu et al. [16] reported that a poor Week 4 response, defined as a reduction of HCV RNA reduction of less than one log IU/mL or HCV RNA > 10,000 IU/mL (in non-TT genotype), has a high negative predictive value (95%) for treatment response. In this study, all patients demonstrating an rNR (13.1%) failed to achieve sustained viral response to the PegIFN/RBV regimen. These results support the proposal to use the simple viral kinetic measurement, rNR, to identify patients unlikely to have an early SVR during treatment.
Single nucleotide polymorphism of IL28B has been acknowledged as an important host predictor for SVR in patients with GT-1 CHC treated with PegIFN/RBV [11] ; [12] ; [13] ; [14] ; [17] . In this study, rs12979860 is not only a predictor for SVR, but also a predictor for rNR. In another large Taiwanese cohort study, Huang et al. [18] showed that host IL-28B polymorphisms play a determinant role in patients with moderate viral reduction at Week 4. Hsu et al. [19] constructed a unique hepatitis C viral kinetic model and showed that different SNPs in IL28B have different therapeutic responses. Lindh et al. [15] reported that the IL28B SNP of rs12979860 has significant influence on early viral kinetics. The extent of decrease of HCV RNA level at Week 4 of treatment of patients with the CC allele of rs12979860 treated with PegIFN/RBV is higher than those in patients with the CT or TT allele of rs12979860. This study shows that patients with the CT/TT allele of rs12979860 demonstrate a higher proportion of rNR, non-EVR, non-RVR, non-EOTVR, relapse rate and treatment failure. These results again emphasize the significant impact of genotype of IL28B on the viral kinetics after PegIFN/RBV treatment.
Treatment related anemia has long been a significant adverse event. Compliance of 80/80/80 has been the golden rule of treatment. In 2010, Sulkowski et al. [20] reported that anemia resulting from chronic hepatitis C treatment with PegINF/RBV was associated with a higher SVR rate. Subsequently, several reports also demonstrated that a higher SVR rate was evident in patients who had treatment-related anemia. Sievert et al. [21] found that anemia (Hb < 10 g/dL) and a significant decline of Hb (> 3 g/dL) during Weeks 5–48 of treatment are associated with a higher rate of SVR. In another retrospective study, both early anemia with hemoglobin < 11.5 g/dL within 4 weeks of treatment and hemoglobin decline > 3 g/dL were associated with higher rates of SVR [22] . Based on these studies, the relationship between SVR and the timing and extent of hemoglobin decline is still controversial. In this study, hemoglobin decline of ≦ 3 g/dL before Week 4 was not a significant predictor for treatment failure (p = 0.219) but was a significant predictor for rNR (p = 0.008). Two associated factors with early anemia were old age (≧ 55 years) and rNR. Therefore, the relationship between early Hb decline and rNR was further analyzed in different IL28B SNPs. In patients with the CC allele of rs12979860, Hb decline of ≦ 3g/dL within 4 weeks of treatment was a significant (P = 0.027) predictor for rNR but similar analysis was not significant (P = 0.299) in patients with a non-CC allele of rs12979860. Due to the small case number in the non-CC allele group further studies with larger populations are required to validate these findings.
The reason why anemia is related to treatment response is still unclear but the theory of individual pharmacokinetic response to ribavirin is supported by the study findings. According to previous studies, ribavirin reaches a steady state of serum concentration after 3–12 weeks of continued dosing and the drug is well known for its hemolytic effect [21] ; [22] ; [23] . Therefore, no early anemia after treatment possibly represents low serum concentration of ribavirin and may be associated with rNR. It is also reported that inosine triphosphate pyrophosphatase (ITPA) polymorphism plays a role in ribavirin induced anemia [24] . It would be interesting to examine the relationship between ITPA polymorphisms, rNR, and early anemia. However, ITPA polymorphisms were beyond the scope of this study and therefore are not included in this discussion.
In the new era of using DAAs to treat GT-1 CHC patients, rNR should also be considered when developing a treatment strategy. Based on SPRINT-2 and RESPOND-2 studies, less than one log of viral load decline after a 4-week lead-in treatment is strongly associated with failure of SVR in GT-1 CHC patients who received boceprevir/PegIFN/RBV treatment [10] ; [25] ; [26] . Current practice guidelines suggest that treatment with telapravir/PegIFN/RBV should be stopped if the HCV RNA level is > 1000 IU/mL at treatment Week 4 [27] . Therefore, early viral kinetics still play an important role in the era of DAAs. Although the viral load decline of less than one log after a 4-week lead-in treatment is associated with a relatively poor response of triple therapy with boceprevir/PegIFN/RBV, combination treatment containing DAAs still leads to SVR in about one third of the patients who respond poorly to interferon. According to the Phase III trial of boceprevir (SPRINT-2), the addition of boceprevir significantly improved SVR rates from 4% with a conventional treatment regimen to 28–38% with a boceprevir containing treatment regimen [25] ; [26] . Thus, current guidelines suggest that a poor response during the lead-in phase should not preclude patients from using boceprevir [27] . Even though DAAs should be made available to this group of patients, it is important to consider the treatment timing. As most patients with rNR cannot attain SVR either with or without boceprevir, especially patients with the non-CC allele of IL28B SNP, it may be advisable to defer treatment with DAAs until more potent forms are developed. This becomes particularly relevant when the side effects and cost of DAAs and the potential for HCV resistant variants that may jeopardize treatment response of future regimens are considered.
In conclusion, rNR has high negative predictive value for sustained virologic response as confirmed by this study and previous reports. The genotype of CT/TT alleles of rs12979860 are important predictors for rNR. In addition, Hb decline of ≦ 3 g/dL is also a predictor of rNR in GT-1 CHC patients treated with a combination regimen of PegIFN/RBV. The findings from this study highlighted both genotype of IL28B and early anemia had significant impact on the rNR that in turn lead to the treatment failure.
In summary, this study clearly demonstrates that both the genotype of rs12979860 and no early anemia are predictors for rNR.
Conflicts of interest
The authors declare no conflicts of interest.
Acknowledgements
We greatly appreciate the secretarial and clerical help from Hui-Chuan Cheng and Pei-Ling Lin.
References
- [1] A.S. Barritt IV, M.W. Fried; Maximizing opportunities and avoiding mistakes in triple therapy for hepatitis C virus; Gastroenterology, 142 (2012), pp. 1314–1323
- [2] P.S. Belperio, E.W. Hwang, I.C. Thomas, L.A. Mole, R.C. Cheung, L.I. Backus; Early virologic responses and hematologic safety of direct-acting antiviral therapies in veterans with chronic hepatitis C; Clin Gastroenterol Hepatol, 11 (2013), pp. 1021–1027
- [3] T. Asselah, P. Marcellin; New direct-acting antivirals' combination for the treatment of chronic hepatitis C; Liver Int, 31 (2011), pp. 68–77
- [4] M.L. Yu, W.L. Chuang; Treatment of chronic hepatitis C in Asia: when East meets West; J Gastroenterol Hepatol, 24 (2009), pp. 336–345
- [5] A. Mangia, A. Andriulli; Tailoring the length of antiviral treatment for hepatitis C; Gut, 59 (2010), pp. 1–5
- [6] M.G. Swain, M.Y. Lai, M.L. Shiffman, W.G. Cooksley, S. Zeuzem, D.T. Dieterich, et al.; A sustained virologic response is durable in patients with chronic hepatitis C treated with peginterferon alfa-2a and ribavirin; Gastroenterology, 139 (2010), pp. 1593–1601
- [7] P.J. Pockros, F.M. Hamzeh, P. Martin, E. Lentz, X. Zhou, S. Govindarajan, et al.; Histologic outcomes in hepatitis C-infected patients with varying degrees of virologic response to interferon-based treatments; Hepatology, 52 (2010), pp. 1193–1200
- [8] M.G. Ghany, D.B. Strader, D.L. Thomas, L.B. Seeff; Diagnosis, management, and treatment of hepatitis C: an update; Hepatology, 49 (2009), pp. 1335–1374
- [9] N. Reau, R. Satoskar, H. Te, A. DeVoss, C. Elsen, G. Reddy, et al.; Evaluation of early null response to pegylated interferon and ribavirin as a predictor of therapeutic nonresponse in patients undergoing treatment for chronic hepatitis C; Am J Gastroenterol, 106 (2011), pp. 452–458
- [10] B.R. Bacon, S.C. Gordon, E. Lawitz, P. Marcellin, J.M. Vierling, S. Zeuzem, et al.; Boceprevir for previously treated chronic HCV genotype 1 infection; N Engl J Med, 364 (2011), pp. 1207–1217
- [11] C.Y. Lin, J.Y. Chen, T.N. Lin, W.J. Jeng, C.H. Huang, C.W. Huang, et al.; IL28B SNP rs12979860 is a critical predictor for on- treatment and sustained virologic response in patients with hepatitis C virus genotype-1 infection; PLoS One, 6 (2011), p. e18322
- [12] J.Y. Chen, C.Y. Lin, C.M. Wang, Y.T. Lin, S.N. Kuo, C.F. Shiu, et al.; IL28B genetic variations are associated with high sustained virological response (SVR) of interferon-[alpha] plus ribavirin therapy in Taiwanese chronic HCV infection; Genes Immun, 12 (2011), pp. 300–309
- [13] D. Ge, J. Fellay, A.J. Thompson, J.S. Simon, K.V. Shianna, T.J. Urban, et al.; Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance; Nature, 461 (2009), pp. 399–401
- [14] J.S. Doyle, M.E. Hellard, A.J. Thompson; The role of viral and host genetics in natural history and treatment of chronic HCV infection; Best Pract Res Clin Gastroenterol, 26 (2012), pp. 413–427
- [15] M. Lindh, M. Lagging, B. Arnholm, A. Eilard, S. Nilsson, G. Norkrans, et al.; IL28B polymorphisms determine early viral kinetics and treatment outcome in patients receiving peginterferon/ribavirin for chronic hepatitis C genotype 1; J Viral Hepat, 18 (2011), pp. e325–e331
- [16] M.L. Yu, C.H. Liu, C.F. Huang, T.C. Tseng, J.F. Huang, C.Y. Dai, et al.; Revisiting the stopping rule for hepatitis C genotype 1 patients treated with peginterferon plus ribavirin; PLoS One, 7 (2012), p. e52048
- [17] M. Kurosaki, Y. Tanaka, N. Nishida, N. Sakamoto, N. Enomoto, M. Honda, et al.; Pre-treatment prediction of response to pegylated-interferon plus ribavirin for chronic hepatitis C using genetic polymorphism in IL28B and viral factors; J Hepatol, 54 (2011), pp. 439–448
- [18] C.F. Huang, M.L. Yu, J.H. Kao, T.C. Tseng, M.L. Yeh, J.F. Huang, et al.; Profound week 4 interferon responsiveness is mandatory for hepatitis C genotype 1 patients with unfavorable IL-28B genotype; J Clin Virol, 56 (2013), pp. 293–298
- [19] C.S. Hsu, S.J. Hsu, H.C. Chen, T.C. Tseng, C.H. Liu, W.F. Niu, et al.; Association of IL28B gene variations with mathematical modeling of viral kinetics in chronic hepatitis C patients with IFN plus ribavirin therapy; Proc Natl Acad Sci USA, 108 (2011), pp. 3719–3724
- [20] M.S. Sulkowski, M.L. Shiffman, N.H. Afdhal, K.R. Reddy, J. McCone, W.M. Lee, et al.; Hepatitis C virus treatment-related anemia is associated with higher sustained virologic response rate; Gastroenterology, 139 (2010), pp. 1602–1611
- [21] W. Sievert, G.J. Dore, G.W. McCaughan, M. Yoshihara, D.H. Crawford, W. Cheng, et al.; Virological response is associated with decline in hemoglobin concentration during pegylated interferon and ribavirin therapy in hepatitis C virus genotype 1; Hepatology, 53 (2011), pp. 1109–1117
- [22] A. Amanzada, S. Schneider, F. Moriconi, A. Lindhorst, T. Suermann, D.H. van Thiel, et al.; Early anemia and rapid virological response improve the predictive efficiency of IL28B-genotype for treatment outcome to antiviral combination therapy in patients infected with chronic HCV genotype 1; J Med Virol, 84 (2012), pp. 1208–1216
- [23] A. Bruchfeld, K. Lindahl, R. Schvarcz, L. Stahle; Dosage of ribavirin in patients with hepatitis C should be based on renal function: a population pharmacokinetic analysis; Ther Drug Monit, 24 (2002), pp. 701–708
- [24] T.M. Scherzer, A.F. Stattermayer, R. Stauber, A. Maieron, M. Strasser, H. Laferl, et al.; Effect of gender and ITPA polymorphisms on ribavirin-induced anemia in chronic hepatitis C patients; J Hepatol, 59 (2013), pp. 964–971
- [25] F. Poordad, J. McCone Jr., B.R. Bacon, S. Bruno, M.P. Manns, M.S. Sulkowski, et al.; Boceprevir for untreated chronic HCV genotype 1 infection; N Engl J Med, 364 (2011), pp. 1195–1206
- [26] M.P. Manns, J. McCone Jr., M.N. Davis, L. Rossaro, E. Schiff, M.L. Shiffman, et al.; Overall safety profile of boceprevir plus peginterferon alfa-2b and ribavirin in patients with chronic hepatitis C genotype 1: a combined analysis of 3 phase 2/3 clinical trials; Liver Int, 34 (2014), pp. 707–719
- [27] M.G. Ghany, D.R. Nelson, D.B. Strader, D.L. Thomas, L.B. Seeff; An update on treatment of genotype 1 chronic hepatitis C virus infection: 2011 practice guideline by the American Association for the Study of Liver Diseases; Hepatology, 54 (2011), pp. 1433–1444
Document information
Published on 15/05/17
Submitted on 15/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?