Abstract
Objectives
This study sought to investigate the clinical utility of aortic valve calcium score (AVCS) determined by using cardiac multislice computed tomography (MSCT).
Methods
Data of 1315 consecutive patients who underwent both conventional echocardiography and MSCT were reviewed. Degree of aortic stenosis (AS) was assessed according to mean pressure gradient (mPG) measured by echocardiography. Extent of coronary artery disease (CAD) derived by MSCT also was evaluated in 1173 patients who did not undergo prior coronary treatment. Both AVCS and coronary calcium score (CCS) were defined by Agatston units (AU) according to MSCT findings.
Results
A total of 613 of 1315 patients were defined as AVCS positive (mean, 100 AU [range, 31.0–380.0 AU]). AVCS showed significant correlations with mPG (Spearmans ρ = 0.81, p < 0.001), and CCS (ρ = 0.53, p < 0.001). Differential adequate cut-off values of AVCS were proved for predicting severe AS with mPG ≥ 40 mmHg (1596.5 AU; AUC, 0.88; sensitivity, 89.7%; specificity, 77.0%), and for predicting moderate AS with mPG ≥ 20 mmHg (886.5 AU; area under the curve [AUC], 0.91; sensitivity, 92.4%; specificity, 78.3%). Mean AVCS was higher with increased extent of CAD (none, 0 AU [range, 0–30 AU]; single vessel, 8.5 AU [range, 0–104 AU]; multivessel, 142 AU [range, 10–525 AU]; p < 0.001). The optimal cut-off value of AVCS for predicting multivessel disease was 49 AU (AUC, 0.77; sensitivity, 68.8%; specificity, 78.0%).
Conclusions
AVCS might be a surrogate marker not only for AS grading but also for CAD progression. Therefore, routine AVCS assessment could be useful for risk stratification.
Keywords
Aortic valve calcium score;Aortic stenosis;Coronary artery disease
1. Introduction
Cardiac multislice computed tomography (MSCT) is a noninvasive imaging modality that is widely used in the clinical field for screening patients with suspected cardiovascular disease [1]. MSCT findings provide information not only on coronary atherosclerotic burden but also on precise cardiac structure [2] ; [3]. Calcium score (CS) determined by using MSCT enables quantitative evaluation of the degree of calcification, as total volume of calcium deposition is considered to reflect atherosclerotic change in each coronary artery, aortic valve, or aorta [4] ; [5]. Several previous clinical studies have demonstrated the diagnostic utility of aortic valve calcium score (AVCS) for grading of aortic stenosis (AS) initially diagnosed by using echocardiography [6] ; [7]. Recent interesting data proved that AVCS assessment showed new insights regarding calcified AS based on discordant markers of severity between aortic valve area (AVA) and mean pressure gradient (mPG) when combined with standard Doppler echocardiography [8]. The above findings support that AVCS determined by using MSCT is useful for screening patients in daily practice, though AVCS assessment is not routinely recommended in the clinical field. Diagnostic MSCT could be more useful for detecting the severity of AS in addition to the existence of coronary artery disease (CAD). However, the relationship between CAD and AS based on CS derived by using MSCT has not been fully clarified. Therefore, in this study, the potential utility of AVCS assessment was investigated in patients who underwent both MSCT and echocardiography.
2. Methods
Between August 2013 and September 2014, 1315 consecutive patients underwent comprehensive MSCT and echocardiography within a 1-month period for evaluation of cardiac disease at our center. Reasons for undergoing diagnostic MSCT and echocardiography were as follows: (1) clinical symptoms, including chest pain, dyspnea on effort, palpitation, pretibial edema, or other physiologic abnormality; (2) examination findings, including electrocardiographic abnormality, chest radiographic abnormality, echocardiographic abnormality, elevation of laboratory data including cardiac biomarker, or other abnormal result; and (3) specific pretreatment preparation for percutaneous intervention or cardiac surgery. All 1315 patients were enrolled for the initial analysis concerning the relationship between AVCS and AS grading in the overall cohort. For the second analysis concerning CAD, 203 patients comprising 26 who underwent coronary artery bypass grafting and 177 who underwent percutaneous coronary intervention were excluded. The remaining 1112 patients who did not undergo prior coronary treatment were evaluated regarding the relationship between AVCS and CAD. This study had a retrospective, cross-sectional, observational design. The Medical Ethics Committee of Toyohashi Heart Center (Toyohashi, Japan) approved the study protocol. Informed consent was waived due to the retrospective nature of the study.
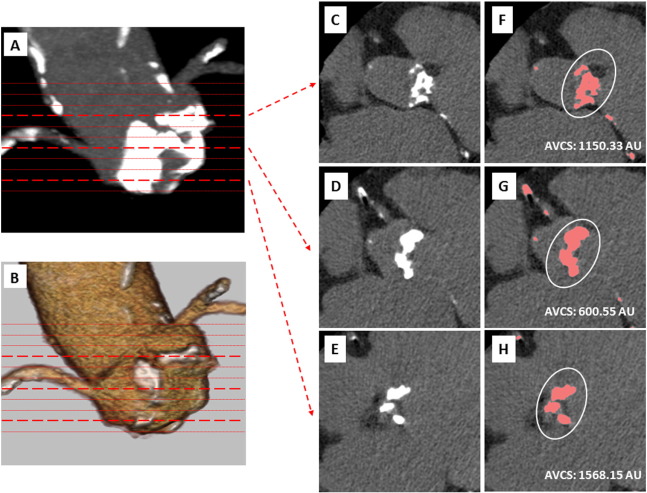
All patients were scanned using a 256-slice scanner (Brilliance iCT; Philips Medical Systems, Eindhoven, The Netherlands) at the Toyohashi Heart Center. Before MSCT angiography, a noncontrast MSCT scan was performed to measure CS according to the modified Agatston method [9] ; [10]. Prospective electrocardiogram triggering (75% of R–R interval) was used with a slice thickness of 2.5 mm. The scan was performed between the tracheal bifurcation and the diaphragm with the following parameters: collimation width, 32 × 0.625 mm; rotation time, 330 ms; tube voltage, 120 kV; and maximum effective tube current, 64 mA. Image reconstruction was gated prospectively to 75% of R–R interval. MSCT images were reconstructed using a cardiac standard filter with a slice thickness of 2.5 mm. MSCT data sets were transferred to an offline workstation (Intelli Space Portal; Philips Medical Systems) for image analysis. MSCT data sets were transferred to an offline workstation (Aquarius NetStation; Terarecon Inc., San Mateo, CA, USA) for image analysis. AVCS and coronary calcium score (CCS) were determined 2-dimensionally by using the CS data sets on the workstation, and defined by Agatston units (AU). The AVCS area of interest comprised the sinotubular junction (STJ), valve cusp, aortic annulus, and left ventricle outflow tract; the start of acquisition was placed from the bottom of the valve to the level of the STJ. A representative example of AVCS assessment is shown in Fig. 1. CCS also was determined by summing the individual lesion scores of each of the main coronary arteries based on modified American Heart Association classification; segments with a diameter > 2.0 mm were analyzed. Luminal diameter stenosis was assessed by visual estimation, with significant stenosis defined as > 50% stenosis. Structures > 2 mm2 adjacent to the lumen that could be distinguished from the lumen and the surrounding pericardial tissue were defined as coronary plaque, as we have previously shown. All MSCT angiographic images were analyzed by 2 independent observers. In the case of different observer readings, a consensus reading was performed and used in the final analysis.
|
|
|
Fig. 1. A whole image of AVCS calculation by using MSCT was presented in panels A–B. Corresponding with each thin slice images, the AVCS area of interest included from STJ to aortic annulus basal plane between the red lines. Three dotted lines correlated with each section were given (panels C–E) and the AVCS calculated the other aorta or coronary calcification was excluded from the analysis (panels F–H). |
A comprehensive M-mode echocardiogram was performed in all patients by an experienced echocardiographic technician using a high-quality commercially available ultrasonic system; 2-dimensional imaging was performed using integral pulsed and continuous wave Doppler. Severity of AS (none, mild, moderate, severe) was classified based on the American clinical guidelines for valvular heart disease [11]. The current study defined severe AS as mPG ≥ 40 mmHg or peak velocity ≥ 4.0 m/s, and moderate AS as mPG ≥ 20 mmHg or peak velocity ≥ 3.0 m/s. Several other parameters, including ejection fraction, indexed AVA (AVA/body surface area), and peak velocity, also were calculated.
All statistical analyses were performed using SPSS version 22 (SPSS Inc., Chicago, IL, USA). AVCS and CCS values were expressed as median (interquartile range) because of their skewed distribution. Other continuous variables were expressed as average ± SD, while categorical data were expressed as percentage of the total. Between-group comparisons were performed using Pearsons bivariate test and the χ2 test for categorical covariates, and one-way analysis of variance for continuous covariates. Comparisons of each parameter (e.g. AVCS, AVA, CCS) determined by using MSCT and echocardiography were analyzed by using Spearmans correlation. Receiver operating characteristic (ROC) curve analysis also was performed to examine the ability of each parameter to identify subjects according to AS or CAD classification. All statistical tests were 2-sided, and p values of < 0.05 were considered significant.
3. Results
Baseline clinical, echocardiographic, and MSCT findings of 1315 patients are presented in Table 1. Average estimated glomerular filtration rate (eGFR) was 66.8 ± 15.6 mL/min/1.73 m2; 388 patients (29.5%) were classified as having chronic kidney disease (eGFR < 60 mL/min/1.73 m2). A total of 142 patients (10.8%) were diagnosed as having AS (mild to severe) on echocardiography. Prevalence of patients who were defined as AVCS positive (minimum, 1 AU; maximum, 6757 AU) on MSCT was 46.6% (n = 613). Of the 1112 patients in whom extent of CAD was evaluated, 620 (55.8%) were defined as CCS positive (minimum, 0.2 AU; maximum, 4662 AU). A total of 157 (15.9%) of the 1112 patients were suspected to have CAD (single- to 3-vessel disease) based on MSCT findings. There was only a small number of patients with 3-vessel disease; thus, patients with 2-vessel disease (n = 51, 4.6%) and 3-vessel disease (n = 13, 1.2%) were categorized as having multivessel disease (n = 64, 5.8%).
| Patients | n = 1315 |
|---|---|
| Baseline clinical characteristics | |
| Age, years | 65.3 ± 13.1 |
| Gender (male) | (52.2%) |
| Body mass index (kg/m2) | 23.4 ± 3.8 |
| Body surface area (m2) | 1.6 ± 0.045 |
| Diabetes mellitus, n | 184 (14.0%) |
| Dyslipidemia, n | 305 (23.2%) |
| Hypertension, n | 550 (41.8%) |
| Smoking, n | 258 (19.6%) |
| Chronic kidney disease, n | 388 (29.5%) |
| Estimated glomerular filtration rate (mL/min/1.73 m2) | 66.8 ± 15.6 |
| Echocardiographic data | |
| Ejection fraction, % | 63.3 ± 7.8 |
| Degree of aortic stenosis | |
| None-mild, n | 1201 (91.3%) |
| Moderate, n | 46 (3.5%) |
| Severe, n | 68 (5.2%) |
| Aortic valve area (cm2) | 1.2 ± 0.56 |
| Indexed aortic valve area (cm2/m2) | 0.78 ± 0.36 |
| Mean gradient (mmHg) | 26.1 ± 22.1 |
| Peak velocity (m/s) | 3.2 ± 1.4 |
| Cardiac multidetector computed tomography data | |
| Aortic valve calcium score (Agatston unit) | 100 (31.0–380.0) |
| Coronary calcium score (Agatston unit) | 307.2 (31.5–379.0) |
| Number of coronary artery disease (n = 1112) | |
| None, n | 921/1112 (82.8%) |
| Single vessel disease, n | 113/1112 (10.2%) |
| Two vessel disease, n | 51/1112 (4.6%) |
| Three vessel disease, n | 13/1112 (1.2%) |
| Multi-vessel disease, n | 64/1112 (5.8%) |
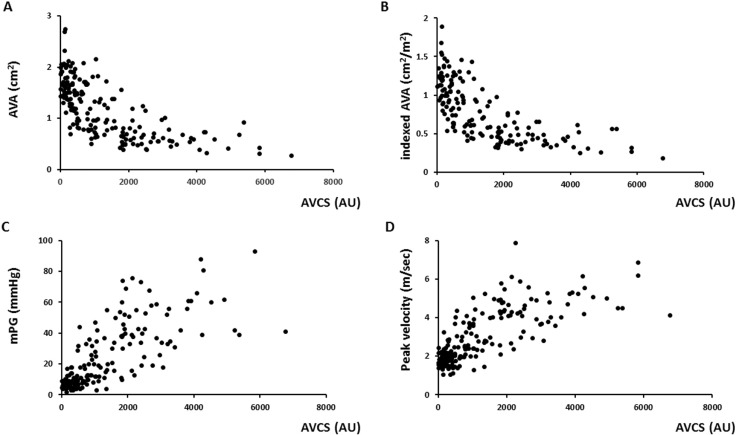
There were significant correlations between AVCS and AVA (Spearmans ρ = − 0.78, p < 0.001), indexed AVA (ρ = 0.81, p < 0.001), mPG (ρ = 0.81, p < 0.001), and peak aortic valve velocity (ρ = 0.81, p < 0.001). Those scatter plots are shown in Fig. 2. AVCS also showed a mild positive correlation with CCS (ρ = 0.53, p < 0.001). When patients were divided into 3 groups according to AS grade (none/mild [n = 1201], moderate [n = 46], and severe [n = 68]), mean AVCS was higher with increased severity of AS (0 AU [range, 0–40.3 AU], 498 AU [range, 276–1318 AU], 2143 AU [range, 1577–3275 AU], respectively; p < 0.001). When patients were divided into 3 groups according to extent of CAD (none [n = 921], single-vessel disease [n = 113], and multivessel disease [n = 64]), mean AVCS also was higher with increased extent of CAD (3.2 AU [range, 0–134.6 AU], 255.5 AU [range, 0–1097.2 AU], 481.4 AU [range, 0.25–967.1 AU], respectively; p < 0.001).
|
|
|
Fig. 2. The scatter plots presented the relationship between the AVCS and each echocardiographic parameters AVA (panel A), indexed AVA (panel B), mPG (panel C), and peak velocity (panel D). |
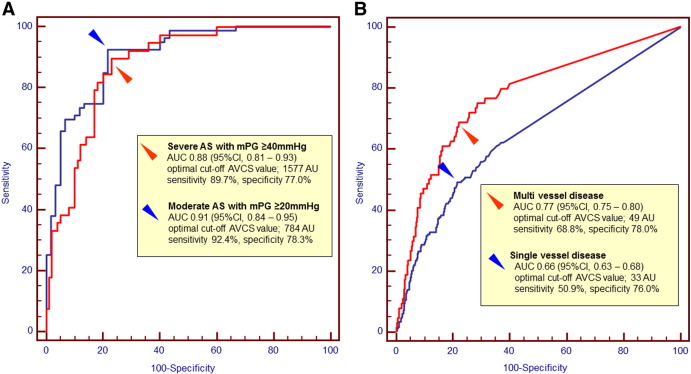
ROC curve analysis indicated that the optimal threshold of AVCS for predicting severe AS with mPG ≥ 40 mmHg was 1596.5 AU (area under the curve [AUC], 0.88; 95% confidence interval [CI], 0.81–0.93; sensitivity, 89.7; specificity, 77.0; p < 0.001), and for predicting moderate AS with mPG ≥ 20 mmHg was 886.5 AU (AUC, 0.91; 95% CI, 0.84–0.95; sensitivity, 92.4%; specificity, 78.3%; p < 0.001) (Fig. 3A). Another optimal thresholds of AVCS for predicting severe AS with peak velocity ≥ 4.0 m/s were 1574.0 AU (AUC, 0.91; 95% CI, 0.87–0.96; sensitivity, 85.5%; specificity, 88.0%; p < 0.001), and for predicting moderate AS with peak velocity ≥ 3.0 m/s was 793.0 AU (AUC, 0.90; 95% CI, 0.86–0.95; sensitivity, 90.4%; specificity, 75.6%; p < 0.001), and for predicting AVA < 1.0 cm2 was 785.5 AU (AUC, 0.92; 95% CI, 0.88–0.96; sensitivity, 92.4%; specificity, 81.6%; p < 0.001). In addition, the cut-off value of AVCS for predicting single-vessel disease was 33 AU (AUC, 0.66; 95% CI, 0.63–0.68; sensitivity, 50.9%; specificity, 76.0%; p < 0.001), and for predicting multivessel disease was 49 AU (AUC, 0.77; 95% CI, 0.75–0.80; sensitivity, 68.8%; specificity, 78.0%; p < 0.001) (Fig. 3B).
|
|
|
Fig. 3. The ROC curve used to determine the optimal cut-off value for severe or moderate to severe AS (panel A), and single or multi vessel disease (panel B). |
4. Discussion
Although AVCS assessment is not routinely recommended during MSCT examination, the current study demonstrated that it might be useful for risk stratification of both CAD and AS. Abundant calcium proliferation is associated with progressive atherosclerotic stage. Local calcium quantification according to CS of a coronary artery or aortic valve is thought to be related to precise diagnosis or subsequent disease in an individual patient [13] ; [14]. There is general agreement that diagnosis of AS should be based on echocardiographic findings, while previous clinical studies have demonstrated the additional diagnostic value of AVCS for AS grading [6]; [7] ; [8]. In this study, the thresholds of AVCS for predicting severe AS with mPG ≥ 40 mmHg and with peak velocity ≥ 4 m/s were defined as 1596.5 AU and 1574.0 AU, respectively. Previous cut-off values of AVCS for predicting severe AS have ranged from approximately 1200 to 2000 AU in similar to our data. The AVCS value of 785.5 AU was also optimal for reflecting AVA < 1.0 cm2, while this cut-off value was found to be a little bit overestimated when considered as severe AS with AVA < 1.0 cm2 compared with other findings of mPG ≥ 40 mmHg or peak velocity ≥ 4.0 m/s. In addition, we showed cut-off values of AVCS of 886.5 AU and 793.0 AU were optimal for including moderate AS with mPG ≥ 20 mmHg and with peak velocity ≥ 3.0 m/s. Recent interesting data proved that AVCS classification showed new insights regarding calcified AS based on discordant markers of severity between AVA and mPG (or peak velocity) when combined with standard Doppler echocardiography [9]. It meant the difference between moderate AS or low-flow, low-gradient severe AS in the same category with AVA < 1.0 cm2 and mPG ≤ 40 mmHg (or peak velocity < 4.0 m/s). In general, dobutamine stress echocardiography is useful for distinguishing between moderate AS and low-flow, low-gradient severe AS. However, the abovementioned cut-off values are only a guide and should not be overstated compared with standard diagnostic echocardiographic criteria for AS grading. Nonetheless, patients with an AVCS approximately ranging 750 to 1500 AU might be considered to have borderline moderate-to-severe AS. Especially in patients with calcified AS, AVCS assessment may support diagnosis of AS in addition to conventional echocardiographic findings.
This study also clarified the positive correlation between AVCS and CCS, as well as the higher AVCS with increased extent of CAD. Our results indicate that a possible threshold for detecting single-vessel disease is 33 AU, while that for detecting multivessel disease is 40 AU. It is best to screen for both AS and CAD before invasive therapy, such as percutaneous coronary intervention or cardiac surgery. For example, calcified plaque in a coronary artery frequently requires rotational atherectomy, and surgeons can be aware of any subsequent or coexisting AS during the procedure to avoid unexpected hemodynamic instability. Patients who have scheduled aortic valve replacement or coronary artery bypass grafting should be screened for CAD and AS, though they are not routinely evaluated during MSCT examination. Calcium deposition in an aortic valve or coronary artery is thought to reflect systemic atherosclerosis [4] ; [5]. Thus, quantitative AVCS and CCS assessment would be more informative for evaluating the degree of AS and CAD derived by using MSCT. Quantitative AVCS assessment during cardiac MSCT is easily and rapidly evaluated not specific technique as we shown. The above results may encourage physicians to evaluate not only coronary calcium proliferation but also aortic valve calcium deposition in the general population, though quantitative calcium scoring is difficult in non-cardiac MSCT examination.
Nonetheless, the following limitations apply to the current study. First, this study had a retrospective, cross-sectional design, and did not focus on prognosis. Second, the majority of patients with AS were categorized as having degenerated calcified AS, reflecting the aging of society; however, the etiology of AS is known to be multifactorial. The diagnostic value of AVCS for AS grading would be attenuated when considering rheumatic or non-calcified origins of AS. Finally, although the total sample size of the current study was relatively large, prevalence of CAD and AS was low because MSCT examinations were performed for several clinical purposes including screening.
Disclosures
The authors have no conflicts of interest to disclose.
References
- [1] L.J. Shaw, J. Hausleiter, S. Achenbach, et al.; CONFIRM Registry Investigators. Coronary computed tomographic angiography as a gatekeeper to invasive diagnostic and surgical procedures: results from the multicenter CONFIRM (Coronary CT angiography evaluation for clinical outcomes: an international multicenter) registry; J. Am. Coll. Cardiol., 60 (2012), pp. 2103–2114
- [2] T. Ito, M. Terashima, H. Kaneda, et al.; In vivo assessment of ergonovine-induced coronary artery spasm by 64-slice multislice computed tomography; Circ. Cardiovasc. Imaging, 5 (2012), pp. 226–232
- [3] T. Ito, Y. Suzuki, M. Ehara, et al.; Impact of epicardial fat volume on coronary artery disease in symptomatic patients with a zero calcium score; Int. J. Cardiol., 167 (2013), pp. 2852–2858
- [4] N.D. Wong, H. Gransar, L. Shaw, et al.; Thoracic aortic calcium versus coronary artery calcium for the prediction of coronary heart disease and cardiovascular disease events; JACC Cardiovasc. Imaging, 2 (2009), pp. 319–326
- [5] C.R. Walsh, M.G. Larson, M.J. Kupka, et al.; Association of aortic valve calcium detected by electron beam computed tomography with echocardiographic aortic valve disease and with calcium deposits in the coronary arteries and thoracic aorta; Am. J. Cardiol., 93 (2004), pp. 421–425
- [6] D. Messika-Zeitoun, M.C. Aubry, D. Detaint, et al.; Evaluation and clinical implications of aortic valve calcification measured by electron-beam computed tomography; Circulation, 110 (2004), pp. 356–362
- [7] J.P. Laissy, D. Messika-Zeitoun, J.M. Serfaty, et al.; Comprehensive evaluation of preoperative patients with aortic valve stenosis: usefulness of cardiac multidetector computed tomography; Heart, 93 (2007), pp. 1121–1125
- [8] C. Cueff, J.M. Serfaty, C. Cimadevilla, et al.; Measurement of aortic valve calcification using multislice computed tomography: correlation with haemodynamic severity of aortic stenosis and clinical implication for patients with low ejection fraction; Heart, 97 (2011), pp. 721–726
- [9] M.A. Clavel, D. Messika-Zeitoun, P. Pibarot, et al.; The complex nature of discordant severe calcified aortic valve disease grading: new insights from combined Doppler echocardiographic and computed tomographic study; J. Am. Coll. Cardiol., 62 (2013), pp. 2329–2338
- [10] A.S. Agatston, W.R. Janowitz, F.J. Hildner, N.R. Zusmer, M. Viamonte Jr., R. Detrano; Quantification of coronary artery calcium using ultrafast computed tomography; J. Am. Coll. Cardiol., 15 (1990), pp. 827–832
- [11] J. Shemesh, S. Apter, J. Rozenman, et al.; Calcification of coronary arteries: detection and quantification with double-helix CT; Radiology, 197 (1995), pp. 779–783
- [13] G.S. Pressman, V. Crudu, A. Parameswaran-Chandrika, A. Romero-Corral, B. Purushottam, V.M. Figueredo; Can total cardiac calcium predict the coronary calcium score?; Int. J. Cardiol., 146 (2011), pp. 202–206
- [14] H. Utsunomiya, H. Yamamoto, E. Kunita, et al.; Combined presence of aortic valve calcification and mitral annular calcification as a marker of the extent and vulnerable characteristics of coronary artery plaque assessed by 64-multidetector computed tomography; Atherosclerosis, 213 (2010), pp. 166–172
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?