Highlights
- The active fraction of the gut microbiome is changed during HIV infection.
- Changes are accentuated in immunological responders.
- Alterations are both a consequence and a cause of immune recovery.
- Immune system-bacteria synergism mediates solutions to immune recovery.
With active antiretroviral therapy, changes in the active fraction of the gut microbiome may be both a consequence and a driver of the recovery of mucosal and systemic immunity in HIV-infected patients. Immune system-bacteria synergism likely exists to mediate solutions to inflammation and immune recovery. There is a need to assess the metabolic contributions of active gut bacteria to the recovery and maintenance of, or persistent defects in, innate and adaptive immunity across different diseases.
Abstract
While changes in gut microbial populations have been described in human immuno-deficiency virus (HIV)-infected patients undergoing antiretroviral therapy (ART), the mechanisms underlying the contributions of gut bacteria and their molecular agents (metabolites and proteins) to immune recovery remain unexplored. To study this, we examined the active fraction of the gut microbiome, through examining protein synthesis and accumulation of metabolites inside gut bacteria and in the bloodstream, in 8 healthy controls and 29 HIV-infected individuals (6 being longitudinally studied). We found that HIV infection is associated to dramatic changes in the active set of gut bacteria simultaneously altering the metabolic outcomes. Effects were accentuated among immunological ART responders, regardless diet, subject characteristics, clinical variables other than immune recovery, the duration and type of ART and sexual preferences. The effect was found at quantitative levels of several molecular agents and active bacteria which were herein identified and whose abundance correlated with HIV immune pathogenesis markers. Although, we cannot rule out the possibility that some changes are partially a random consequence of the disease status, our data suggest that most likely reduced inflammation and immune recovery is a joint solution orchestrated by both the active fraction of the gut microbiota and the host.
Graphical Abstract
Keywords
Antiretroviral therapy ; Gut microbiota ; HIV ; Immune recovery ; Metabolome ; Metaproteome
1. Introduction
Human immunodeficiency virus (HIV) is a chronic inflammatory disease, in which numerous structural (enterocyte apoptosis and the loss of epithelial integrity), functional (errors in the production of mucin/IgA), and immunological (loss of sub-mucosal lymphocytes) defects contribute to an excess risk of mortality (Buzón et al., 2010 , Mutlu et al., 2014 and McGrath et al., 2015 ). It has recently been shown that HIV infection also induces a compositional shift in the gut microbiota, with enrichment for bacterial populations, genes and functional capabilities that are either pro-inflammatory or potentially pathogenic, and whose abundance correlated with immune status (McHardy et al., 2013 , Vujkovic-Cvijin et al., 2013 , Mutlu et al., 2014 and Vázquez-Castellanos et al., 2015 ). However, imbalances in gut bacteria have been found to be different in patients undergoing highly active antiretroviral therapy (ART) compared to untreated patients (Vázquez-Castellanos et al., 2015 ). This raises the question of whether the gut ecosystem contributes to chronic inflammation and disease progression (Vujkovic-Cvijin et al., 2013 and Vázquez-Castellanos et al., 2015 ) but also to immune recovery, although these patients are still characterized by residual inflammation and excess mortality (Vujkovic-Cvijin et al., 2013 , Somsouk et al., 2014 and Vázquez-Castellanos et al., 2015 ).
Here, we determine whether gut microbiota contribute to inflammation and immune recovery in HIV-infected individuals, and, if they do, which molecular agents are implicated. The present investigation demonstrates an association between an optimal immunovirological ART response and the active fraction of the gut microbiome, as gut bacteria in immunological responders were found to be metabolically distinct from those in viremic untreated and immunological-non responders. We showed that this occurs both at the level of microbes´ protein synthesis and microbes´ metabolic activity. It is plausible that immune recovery might induce alterations in metabolic profiles in the gut environment. However, our findings provide also evidences suggesting the contribution of gut bacteria to ART-mediated immune recovery. Changes in the gut ecosystem in HIV-infected patients undergoing ART may thus be both a consequence and a potential cause of the recovery of mucosal and systemic immunity. Here, we provide an explanation of the causes that contribute to this association, such as that active gut bacteria are involved in controlling mucosal epithelial integrity and the accumulation of pro-inflammatory mediators and molecules that reduce viral infection and increase immune recovery. This was demonstrated by their association with adaptive immune and inflammatory markers involved in HIV immune-pathogenesis. Furthermore, plasma metabolome analyses demonstrated that human cells respond reciprocally by increasing the biosynthesis of anti-inflammatory and pro-solving lipid mediators that circulate in the bloodstream. Altogether, it is plausible that immune system-bacteria synergism mediates solutions to inflammation and promotes immune recovery.
2. Materials and Methods
2.1. Study Design, Participants, Setting and Eligibility
We recruited HIV-infected individuals, including 9 ART-naïve untreated (VU) individuals, 12 immunological ART responders (IR), 8 immunological ART non-responders (INR) (IR and INR, ≥ 350 and < 350 CD4 + T-cell counts/μL after > 2 years of viral suppression, respectively), and 8 unmatched HIV-uninfected (HIV− ) individuals. HIV-infected individuals on ART were representative of a middle-aged long-term treated population without metabolic abnormalities (Table 1 ). Subjects were recruited from the HIV clinics of two University hospitals in Madrid, Spain (University Hospital Clínico San Carlos and University Hospital Ramón y Cajal). The inclusion criteria were serologically documented HIV infection and an age of 18 years old or older. Controls (VIH− ) were healthy HIV-uninfected volunteers (most of them were staff working at either institution) that were recruited with the aim of obtaining a group of similar age. We additionally included longitudinal samples for 6 individuals to assess intra-individual variation. (VU: 1; IR: 3; VIH− : 2). The following exclusion criteria were used: concomitant medications, the use of systemic antibiotics during the previous 3 months, and any acute or chronic condition other than chronic HIV infection, including gastrointestinal symptoms (constipation, bloating or diarrhoea) or co-infections by hepatitis B or C viruses. This study conformed to the principles of the Declaration of Helsinki and Good Clinical Practice Guidelines and was approved by the Independent Ethics Committees of both recruiting institutions (approval number 11/284). Table 1 summarizes the characteristics of the individuals, which are detailed in Table S1 . Table S2 shows the type of ART regimen in each group.
| Control (n = 8 + 2)a | VU (n = 9 + 1)a | IR (n = 12 + 3)a | INR (n = 8 + 0) 2 | |
|---|---|---|---|---|
| Age (years, IQR)b | 31 (44.25–28.75) | 35 (39–33.25) | 44 (47.5–35) | 48 (51.25–41) |
| Male gender (No., %) | 8 (80%)a | 9 (90%)a | 12 (80%)a | 8 (100%)a |
| CD4 + T-cell count (cells/mm3 , IQR) | 748 (813.5–647) | 577.5 (835–445) | 600 (830–522) | 291 (322.25–242.25) |
| CD4/CD8 ratio (IQR) | 1.22 (1.45–1.09) | 0.60 (0.93–0.46) | 1.00 (1.21–0.91) | 0.71 (0.89–0.36) |
| HIV RNA Level (copies/mL, IQR) | – | 22,198 (9955–40,621) | < 20 | < 20 |
| Nadir CD4 + T-cell count (cells/mm3, IQR) | – | 577.5 (835–445) | 225 (256.5–116.5) | 132.5 (184.5–103.5) |
| Time since HIV diagnosis (years, IQR) | – | 5.4 (2.9–9.9) | 8.0 (4.5–13.9) | 11.8 (5.6–20.8) |
| Duration of ART (years, IQR)c | – | – | 6.1 (4.3–11.7) | 8.9 (4.6–12.7) |
| Framingham Risk Score (%, IQR) | 3.63 (2.5–1) | 2.73 (2–1) | 5.23 (7–2) | 5.29 (6–4) |
| Body mass index (kg/m2 ) | 21.89 (23.53–20.43) | 22.98 (25.88–22.58) | 22.45 (24.53–23.37) | 25.56 (26.63–24.61) |
| Glucose (mg/dL) | 92 (95–88.25) | 86.5 (91–83.25) | 91 (96–82) | 92 (97.5–88.25) |
| Creatinine (mg/dL) | 0.99 (1.04–0.93) | 0.95 (1.02–0.93) | 0.98 (1.08–0.89) | 0.98 (1.05–0.95) |
| Total cholesterol (mg/dL) | 173 (213–149.5) | 155 (173.75–152) | 190 (211.5–165) | 151.5 (199.75–144.25) |
| HDL cholesterol (mg/dL) | 64 (75.75–51.25) | 47.5 (53–42.25) | 54 (61–48.5) | 50 (58.5–43.25) |
| LDL cholesterol (mg/dL) | 92.5 (118–78.75) | 90.5 (99.5–87.25) | 106 (131–94.5) | 86.5 (118.5–70) |
| Triglycerides (mg/dL) | 68 (83.75–59.5) | 107 (136.5–70.75) | 102 (140.5–80.5) | 122 (148–81.25) |
a. Data are included for all individuals, including those for the 6-weeks longitudinal samples (initial + longitudinal).
b. IQR, interquartile range.
c. The ART regime described in Table S2.
2.2. Metabolite Measurements in Gut Bacteria and Plasma Samples
Fresh stool samples were collected from each subject, frozen immediately and stored until processing at − 80 °C. After thawing, the gut bacteria were separated from faecal material, and the metabolites that had accumulated intracellularly were extracted from equal amounts of microbial cells per sample by adapting a previously reported method (Rojo et al., 2015a and Rojo et al., 2015b ) in which a two-step extraction method produces the optimal extraction efficiency for both polar and hydrophobic metabolites. Briefly, microbial cells were separated from the faecal matrix by mixing 0.4 g of faecal sample with 1.2 mL of phosphate-buffered saline (PBS) (1:3 w /v faeces to PBS ratio). Following re-suspension (by 1 min of vigorous vortexing), the samples were then centrifuged at 1,000 g at 4 °C for 1 min to remove faecal debris. The supernatant (1.2 mL) was transferred to a 2-mL Eppendorf tube and centrifuged at 13,000 g at 4 °C for 5 min to pellet the cells. This protocol was repeated 3 times. The cell counts in the bacterial pellets were then immediately analysed, as previously described (Bargiela et al., 2015 ), so that the same amount of bacterial cells was used in each extraction experiment. A total of 108 gut bacterial cells per sample were used for methanol extractions, in which 1.2 mL of cold (− 80 °C) high performance liquid chromatography (HPLC)-grade methanol was added. The samples were then vortex-mixed (for 10 s) and sonicated for 30 s (in a Sonicator® 3000; Misonix) at 15 W in an ice cooler (− 20 °C). This protocol was repeated twice with a 5-min storage period at − 20 °C between each cycle, and the final pellet was removed following centrifugation at 12,000 g for 10 min at 4 °C. Immediately after the methanol extracts were obtained, the methanol solution was stored at − 80 °C, and the remaining cell pellet was re-suspended in 1.2 mL of cold (4 °C) HPLC-grade H2 O and subjected to 3 cycles of sonication for 20 s (in a Sonicator® 3000; Misonix) at 15 W in ice water. The samples were incubated on ice for 2 min between cycles. The final pellet was removed following centrifugation at 12,000 g for 10 min at 4 °C. Immediately after the H2 O and methanol extracts were obtained, a mixture was prepared by combining equal amounts (1 mL) of each of the extracts. Once prepared, the final solution was stored in 20-mL penicillin vials at − 80 °C until they were analysed. Prior to analysis, a pre-processing step was performed to eliminate impurities and particles by mixing the samples with (HPLC)-grade acetonitrile (1:1) and subsequently centrifuging them (13,000 g at 4 °C, 10 min). Then, the supernatant was divided into two aliquots one of them immediately analysed by liquid chromatography electrospray ionization quadrupole time-of-flight mass spectrometry (LC-ESI-QTOF-MS) and the other evaporated and reconstituted in an equal volume of MilliQ water containing an internal standard (0.2 mM l -methionine sulfone) and 0.1 M formic acid and subsequently analysed by capillary electrophoresis time-of-flight mass spectrometry (CE-TOF-MS).
Plasma samples were also collected from each subject from fasting venous blood. Samples were centrifuged and stored at − 80 °C until use. After thawing, the plasma was subjected to a pre-processing step to eliminate impurities and particles by mixing it with (HPLC)-grade acetonitrile (1:1) followed by centrifugation (13,000 g at 4 °C, 10 min), and analysed immediately by LC-ESI-QTOF-MS.
Bacterial and plasma samples were metabolome-typed using a combination of untargeted mass spectrometry and two different and complementary separation techniques: LC-ESI-QTOF-MS (LC: 1290 infinity, Agilent, QTOF-MS: Agilent 6550 iFunnel) in positive and negative mode and CE-TOF-MS (CE: 7100 Agilent, TOF-MS 6224 Agilent) in positive mode. The full details for chemical and reagents, methods, and preparation of quality controls (QCs) are available in Rojo et al., 2015a and Rojo et al., 2015b .
2.3. Metabolite Data Treatment, Statistical Analysis and Identification
For metabolomic data treatments, the Molecular Feature Extraction tool in the Mass Hunter Qualitative Analysis software (B.06.00, Agilent) was used. The alignment of the raw data (from bacteria and plasma datasets) was performed using Mass Profiler Professional software (version 13.0, Agilent). The variables were then filtered according to the filtering criteria proposed by Godzien et al. (2015) : variables that were present in at least 50% of the samples of each group and i) present in at least 80% of the QCs that obtained a coefficient of variation < 30% or ii) present in < 20% of the QCs. Models were subsequently built using SIMCA-P + software (12.0.1.0, Umetrics). The full details for data treatment and statistical analysis can be seen in Rojo et al., 2015a and Rojo et al., 2015b . Briefly, after missing values replacement, Mann–Whitney U tests were used to perform all of the comparisons, followed by Benjamini–Hochberg post hoc correction (P < 0.05). The resulting list of accurate masses that significantly differed between groups was searched using the CEU Mass Mediator search tool (http://biolab.uspceu.com/mediator; error ± 5 Da) to obtain tentative identifications. This procedure was performed independently for each analytical platform.
The identity of the compounds selected according to their significance in the class separation was further confirmed using LC-MS/MS in the same LC-ESI-QTOF-MS. Ions were targeted for collision-induced dissociation (CID) fragmentation based on the previously determined accurate masses and retention times. Their identities were confirmed by comparing the fragments that were obtained with the structure of the proposed compound in the MS/MS spectra in a public database (METLIN: https://metlin.scripps.edu/metabolites_list.php ) or against commercially available standards.
2.4. Protein Extraction, Mass Spectrometry and Data Analysis
Protein extracts were isolated as previously described (Hernández et al., 2013 ), with small modifications. Briefly, equal amounts of enriched gut bacteria samples (108 cell in total per sample), obtained as described above, were re-suspended in 1.2 mL of BugBuster® Protein Extraction Reagent (Novagen, Darmstadt, Germany) for 30 min with shaking (250 rpm). Then, the samples were disrupted by sonication using a pin Sonicator® 3000 (Misonix, New Highway Farmingdale, NY, USA) for a total time of 2 min (10 W) on ice (4 cycles × 0.5 min with 1.0 min ice-cooling between each cycle). The extracts were then centrifuged for 10 min at 12,000 g and 4 °C to separate cellular debris and intact cells, and the supernatants were then carefully aspirated (to avoid disturbing the pellet), transferred to new tubes, and stored at − 80 °C until use.
Protein extracts were thawed on ice, and 50 μg of each sample was precipitated using 1 ml 20% trifluoroacetic acid at 4 °C for 40 min followed by 15 min of centrifugation at 12,500 g and 4 °C. Protein pellets were washed twice in ice-cold acetone and dried using vacuum centrifugation at 30 °C. Prior to 1D-SDS-PAGE (12%, 60 min at 20 mA), pellets were solubilized in 15 μL Laemmli-buffer by 5 min of sonication followed by vortexing. Samples were incubated for 8 min at 95 °C to reduce disulphide bonds, and 12 μL of each sample was run on a gel. Proteins were separated by < 1 cm, cut, and subjected to in-gel trypsin digestion overnight at 37 °C (Shevchenko et al., 2006 ). The obtained peptides were purified and desalted using C18 StageTips ( Rappsilber et al., 2003 and Rappsilber et al., 2007 ), dried using vacuum centrifugation, and prior to LC-MS/MS measurements, they were reconstituted in 20 μL 0.1% trifluoroacetic acid/2% acetonitrile.
Each sample of 8 μL was injected using an autosampler and concentrated on a trapping column (Pepmap100, C18 , 100 μm × 2 cm, 5 μm, Thermo Fisher Scientific) with water containing 0.1% formic acid and 2% acetonitrile at flow rates of 4 μL min− 1 . After 10 min, the peptides were eluted into a separation column (PepmapRSLC, C18 , 75 μm × 50 cm, 2 μm, Thermo Fisher Scientific). Chromatography was performed using 0.1% formic acid in solvent A (100% water) and B (100% acetonitrile). The solvent B gradient was set from 4 to 8% for the first 15 min and subsequently increased to 20% for the next 110 min. After this, solvent B was increased from 20% to 30% over 15 min, from 30% to 40% over 10 min, and finally switched to 90% solvent B for an additional 10 min using a nano-high pressure liquid chromatography system (Ultimate3000 UHPLC, Thermo Fisher Scientific). Ionized peptides were measured and fragmented using a Q Exactive mass spectrometer (Thermo Fisher Scientific). For an unbiased analysis, continuous scanning of the eluted peptide ions was performed between 400 and 1200 m /z and then automatically switched to MS/MS higher energy collisional dissociation mode with twelve MS/MS events per survey scan. For MS/MS higher energy collision conditions (HCD) measurements, a dynamic precursor exclusion of 30 s per peptide match and an apex trigger of 2 to 30 s were enabled.
To achieve a more precise evaluation of protein abundances and to decrease the false discovery rate of peptide identifications, a two-step database search was used (Jagtap et al., 2013 and Hansen et al., 2014 ). Raw MS and MS/MS data were first processed using Thermo Proteome Discoverer software (v.1.4.1.14) and Mascot Server (v. 2.4.1) and then independently searched against the NCBInr databases (v. April 25, 2015) for bacteria. Oxidation of methionine was set as the variable modification, and carbamidomethylation of cysteine was set as the fixed modification. Precursor ion tolerance was defined at 10 ppm, and fragment ion tolerance was set to 0.02 Da. Furthermore, all peaks except for the top 12 peaks per 100 Da in each MS/MS were removed to remove noise from the spectra before identification. To extract a more specific protein Fasta database, the default settings of Thermo Proteome Discoverer were kept, including protein grouping with a minimum peptide confidence set to “medium” and a delta Cn of 0.15. A strict maximum parsimony principle was used.
At the second step, the obtained in-house database was used for label-free protein quantification by applying the Label Free Quantification (LFQ) modality of the MaxQuant software (v. 1.5.3.8). Cysteine carbamidomethylation was set as the fixed modification, and methionine oxidation was set as the variable modification. Re-quantification was enabled. Two missed cleavage sites were allowed for protease digestion, and peptides were required to be fully tryptic. Other parameters of the software were kept at the default settings. These included a peptide and protein False Discovery Rate (FDR) below 1%, at least 1 peptide per protein, a precursor mass tolerance of 4.5 ppm and a fragment ion mass tolerance of 20 ppm. The LFQ abundance values were subjected to statistical analyses using Primer 6 software (v. 6.1.16). Principal coordinate analysis (PCO) was calculated on the basis of the Bray–Curtis dissimilarity matrix (Bray and Curtis, 1957 ). Significant differences between treatment groups were determined by performing one-way Analysis of Similarities (ANOSIM) and Similarity Percentages (SIMPER) analyses.
Phylogenetic analyses based on the identified peptides were performed using Unipept (Mesuere et al., 2012 and Mesuere et al., 2015 ).
2.5. Histone Acetylation
Thawed peripheral blood mononuclear cells (PBMC) were analysed by western blot using cell lysates (equal protein concentration) and antibodies to acetylated (Ac) histone (H)3 as described previously (Elliott et al., 2014 ).
2.6. Experimental Validations of Key Active Metabolites
The ability of Succinivibrio dextrinosolvens (type strain DSM -3072; DSMZ, Braunchweig, Germany), Pseudomonas aeruginosa PAO, Escherichia coli K12, Staphylococcus aureus and Enterococcus faecalis to transport and assimilate biliverdin (BV) and oleamide (Sigma Aldrich, Madrid, Spain), was confirmed as follows. First, pure cultures (in triplicates) of S. dextrinosolvens were setup in medium 330 (anaerobic, 37 °C, 2 weeks) in the presence of both metabolites at a final concentration of 10 mg/L. Cultures of the control strains were setup in Luria Bertani medium as described previously (Kubacka et al., 2007 ). Control cultures without the addition of BV and oleamide were set up. The extent of assimilation in test and control samples was quantified as follows. Briefly, bacterial cells (from 500 ml culture) were separated by centrifugation at 13,000 g at room temperature for 10 min. After supernatant separation, bacterial pellet was used for methanol extraction and water extraction as described above. Metabolite solutions were stored at − 80 °C until they were analysed by mass spectrometry as described above. The abundance levels of mass signatures of BV and oleamide were determined in both supernatants and cells.
2.7. Biomarker Discovery
The linear discriminative analysis (LDA) effect size (LEfSe) algorithm (Segata et al., 2011 ) was used to identify the species that were overrepresented in each of the four groups of the cohort. The p -value cut-off was set to be below to 0.001 for the Kruskal–Wallis test and 0.01 for each of the Wilcoxon test parwise comparisons between the groups. The resulting species with significant differences between samples were used to build the LDA model and to estimate its effect as a discriminant feature among them. The threshold used to consider a discriminative feature for the logarithmic LDA score was set to > 2.
2.8. Clinical Associations between the Active Microbiota and the Immune Activation Markers
The association between the active microbiota and innate immune activation and bacterial translocation markers was determined by applying a generalized linear regression model, using glmnet package (library “glment” function “cv.glmnet ”) ( Friedman et al., 2010 ). As response variables, we took the log10 transformation of all the immune activation markers measured for each of the participants of the cohort. As predictors, we considered the active taxa abundance matrix, transformed by the Arcsin square root normalization. The Least Absolute Shrinkage and Selection Operator was used as the regression method to penalize the absolute size of the regression coefficients. The performance of each of the predictive model was validated by means of the k-mean cross-validation using cv. glmnet function in the “glmnet ” R package. In order to increase the confidence of the predicted associations only those edges that possess a significative Spearman-correlation p -value (< 0.05) were included into the model. All the models were represented as a network manner using the igparh package (library “igraph” function “graph.data.frame”). Markers were determined as described previously (Vázquez-Castellanos et al., 2015 ).
3. Results
3.1. Methodology and Study Population
A compositional survey of proteins and metabolites accumulated inside gut bacteria, previously separated from faecal samples, was used to identify changes in microbes´ protein synthesis and microbes´ metabolic activity, respectively. In parallel, metabolites accumulated in plasma samples were used as estimation of alterations in human cell metabolism and translocation of microbial products. To capture results for a wide spectrum of HIV immune-pathogenesis, we recruited and investigated viremic untreated HIV-infected individuals (VU; n = 9) and immunological antiretroviral therapy (ART) responders (IR; n = 12) and non-responders (INR; n = 8) (IR and INR, ≥ 350 and < 350 CD4 + T-cell counts/μL after > 2 years of viral suppression, respectively). The comparator group was composed of healthy controls (HIV− ; n = 8). A full description of the methods is available in the Materials and Methods section.
3.2. Associations between Gut Bacteria Metabolism, HIV, ART and CD4 + T Cell Recovery
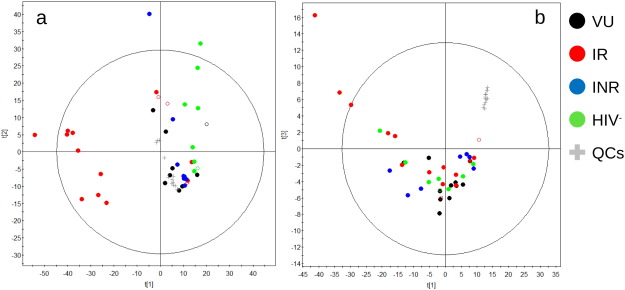
We first asked whether gut bacteria metabolism might be representative of the HIV immune-pathogenesis disease status. To prove this, gut bacteria metabolomes were examined in three groups of HIV-infected patients (VU, IR and INR) and a set of HIV-uninfected controls. A total of 336,502 mass features associated to microbes' metabolic activity were detected. After treatment of the data (Table S3 ; P < 0.05), an unsupervised scatter plot generated using Principal Component Analysis (PCA) scores revealed differences between HIV subjects and controls (Fig. 1 a; Fig. S1), which is in agreement with previous evidences at the level of community composition ( Dillon et al., 2014 , Mutlu et al., 2014 and Noguera-Julian et al., 2016 ). Further, we observed clusters in which the VU and INR groups of patients were the most similar and were more similar to the HIV-uninfected group. It also revealed a clear separation of the IR group. The fact that INR patients who received ART were clustered separately from IR individuals suggests that the observed changes were not due to the ART treatment itself, but they were instead due to a biological cause or the consequence of immune recovery. We further analysed the possible confounding effect of other relevant factors. Since a major immunological driver of impaired CD4 + T cell recovery is late presentation (Girardi et al., 2007 ), as expected, INR patients were at more advanced disease at diagnosis and showed a statistically significant lower Nadir CD4 + T-cells (Table 1 ; P = 0.041). However, neither diet, subject characteristics such as age, gender, race, and body mass index, clinical variables such as glucose, creatinine, triglyceride and cholesterol levels, and adaptive immune markers such as CD4 + T-cells, Nadir CD4 + T-cells, CD4/CD8 ratio and %CD8 + HLA-DR + CD38 + T cells, nor the duration and the type of ART and sexual preferences seem to have exerted a confounding effect in the results (for details see Supplementary Results and Fig. S2).
|
|
|
Fig. 1. Changes in metabolite profiles. Principal Component Analysis (PCA) plots for the models were built using the set of LC-MS (−) filtered data that were present in at least 50% of the samples of each group and i) present in at least 80% of the Quality Controls (QCs) that obtained a coefficient of variation < 30% or ii) present in < 20% of the QCs (for details see Materials and Methods ). (a) Gut microbiota (left panel): 5 principal components, R2 : 0.310, Q2: 0.084. (b) Plasma (right panel): 5 principal components, R2 : 0.483, Q2: 0.308. The empty circle labels the placebo cohort. |
These results demonstrate an association between adequate CD4 + T cell recovery (to > 350 cells/mm3 ) in HIV-infected individuals treated with ART and gut bacteria metabolism. However, the fact that HIV non-infected individuals with CD4+ T cell counts > 350/mm3 displayed different metabolic profiles than ART responders who also had CD4+ T cell counts > 350/mm3 indicates that the CD4+ T cell level itself is not a marker of the metabolic status of the gut ecosystem.
3.3. Bacterial Metabolomics
3.3.1. Comparisons of Metabolic Deficits: Influence of HIV, ART and Immune Recovery
We explored the metabolic alterations (P < 0.05) associated to HIV infection per se, ART use and immune recovery (Fig. S3). Among 336,502 mass features associated to bacterial activity detected, a total of 2716 (or 0.8%) exclusively distinguished uninfected and HIV-untreated groups, suggesting that the impact of HIV infection per se on microbes' metabolic activity is moderately low. When comparing HIV-untreated and ART treated groups, so that the impact of ART is evaluated, we found additional alterations in 974 (or 0.29%) mass features, suggesting that the impact of ART per se on the gut microbiota metabolite-wide flux distribution is also moderately low. These findings may contradict with the major imbalances in the gut bacterial composition in HIV-infected patients compared to controls and patients undergoing highly active ART compared to untreated patients ( McHardy et al., 2013 , Vujkovic-Cvijin et al., 2013 , Mutlu et al., 2014 and Vázquez-Castellanos et al., 2015 ). However, it should be noticed that albeit our gut microbiome is always changing, its also remarkably stable, and that the bacteria of the gut have evolved specialized core functions that are maintained under different conditions (Moya and Ferrer, 2016 ). We finally explored the alterations associated to the immune recovery by comparing IR and INR groups, and found 1542 mass features as biomarkers that exclusively associated with immunological ART responders (Fig. S3). This demonstrates that the immune recovery associated to major metabolic changes in the gut ecosystem as compared to those caused by the HIV and ART itself: immune status is thus a stronger factor driving metabolite fluxes in the gut ecosystem.
3.3.2. Potential Biomarker Metabolites Related to Inflammatory and Immune Recovery
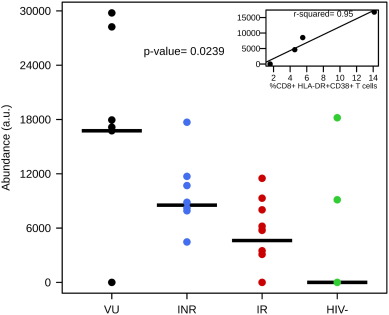
Fig. 1 a suggests that major changes in microbes' metabolic activity are associated to an optimal immunological response to ART. Although, it is plausible this is a side effect consequence, it is also possible that the microbial community and its molecular agents (i.e. metabolites) are drastically changed to help immune recovery during antiretroviral therapy. To test this, we select potential biomarkers related to inflammatory and immune recovery. Statistically significant differences for the innate immune activation markers CD4 + T-cells, nadir CD4 + T-cells, CD4/CD8 ratio and %CD8 + HLA-DR + CD38 + T cells (Table S1 ) between groups were tested by an independent t -test using them as continuous variables. To reduce noise in data analysis, pre-processing was performed to include only discriminating metabolites between groups (P < 0.05) whose empirical formulas and identities were assigned and confirmed (Fig. S3; Table S3 ). A total of 1190 metabolites were selected, from which 55 were potential candidates (Rho > 0.918; P < 0.05). Among them, we found a positive correlation between %CD8 + HLA-DR + CD38 + T cells and the level of a ceramide-related metabolite detected in this study, C16 ceramide (Rho = 0.952). Lower levels of %CD8 + HLA-DR + CD38 + T cells associated to healthy controls and also IR groups of individuals (P < 0.0239; Fig. 2 ). Note that intestinal tissues contain higher levels of ceramides than do other tissues (Bouhours and Bouhours, 1985 ) and that accumulation of ceramides by opportunistic pathogens is an indication of their ability to scavenge lipids from the host and destruction of tissues (Heung et al., 2006 ). Since, lower CD8 + T cell activation was associated to lower levels of inflammation it is plausible that the species responsible for the removal of lipids are drastically decreased in immune responders.
|
|
|
Fig. 2. Metabolite biomarkers associated to clinical variables. The relative abundance of C16 ceramide that is linked with innate immune activation marker %CD8 + HLA-DR + CD38 + T in each of the 4 group of individuals investigated is shown (P < 0.0239). Horizontal bars represent median. Inset represents the abundance of C16 ceramide versus %CD8 + HLA-DR + CD38 + T (Rho = 0.95). |
We also found 10 metabolite markers of epithelial barrier integrity, hepatic function, viral infectivity and inflammation that distinguish all groups, that included: i) the sialic acid derivative N -acetylneuraminic acid (Neu5Ac) and the dolichol derivatives dolichol phosphate (Dol-P) and dolichyl-β-d -glucosyl phosphate (Dol-β-G) that are markers of structural changes of the epithelial barrier ( Schenk et al., 2001 and Varki and Schauer, 2009 ); ii) arachidonic acid (AA) and its pro-inflammatory metabolic product leukotriene B4 (LTB4 ) and the cannabinoid N -acyl amide oleamide, that are markers of inflammation and immune recovery ( Bell et al., 2000 , Rao et al., 2011 and Archin and Archin, 2014 ); and iii) the haeme catabolic products biliverdin (BV), bilirubin (BR), and urobilogen (URO), that are markers of the hepatic function, HIV viral infectivity (Wegiel and Otterbein, 2012 ) and inflammation (Fowler, 2004 ).
As shown in Table S4, measurements of the levels of accumulated Dol-P and Dol-β-G indicated that they were significantly accumulated in the gut bacteria of all HIV-infected patients (VU, IR and INR) compared to controls (P < 0.044), with no statistically significant differences between VU, IR and INR subjects. Accumulation of Neu5Ac only occurred in bacteria of VU patients (P < 0.00076). This suggests the presence in HIV-infected patients of pathogens that are able to remove integral components of the host membranes, the abundance of some of which (i.e. those able to scavenge sialic acids) decreased in patients receiving ART.
Gut bacteria of all HIV-infected individuals accumulated AA, and no statistically significant differences were found between the three groups (VU, IR and INR); however, the level of AA in the gut microbiota was below the detection limit in healthy individuals (P < 0.000023; Table S4). This suggests the increased release of un-metabolized AA in the gut environment of HIV-infected individuals and its consequential accumulation by gut bacteria. The AA metabolic product LTB4 was found to be accumulated only in the gut bacteria of IR patients (P < 0.02291; Table S4). LTB4 is among the first soluble pro-inflammatory factors that are produced by the innate immune system from AA (Bertin et al., 2012 ). In vitro co-cultures of HIV-1-infected monocytes have been shown to secrete high levels of LTB4 , which substantially contribute to neuro-inflammation and neurotoxicity (Genis et al., 1992 ). Additionally, elevated levels of LTB4 have been found in the cerebrospinal fluids of patients with AIDS (Froldi et al., 1992 ). We hypothesized that gut bacteria accumulate this important pro-inflammatory mediator, which would otherwise be amassed in the gut environment, where it would induce inflammation ( Flamand et al., 2004 and Bertin et al., 2012 ). To test this hypothesis, we determined the amount of LTB4 in total faecal fluids. We found that LTB4 is extensively excreted into the gut environment at a high level in VU and INR individuals, while it was found at up to 32-fold lower levels in the faecal fluids of IR individuals and healthy controls (P < 0.05; Table S4). These measurements indicate that the biosynthesis and further excretion into the gut space of LTB4 increased as a consequence of HIV infection, which is in agreement with previous studies ( Froldi et al., 1992 and Genis et al., 1992 ). This process occurs whatever the patient is or is not treated with antiretroviral drugs and regardless of the level of immune recovery. Importantly, the accumulation of LTB4 in immunological responders occurs inside gut bacteria and not in faecal fluids, which prompted us to suggest that gut bacteria play a role in controlling the level of free LTB4 that is released into the gut environment. Consequently, bacteria might have evolved as yet unknown specific systems to transport pro-inflammatory signals and to regulate their concentration to assist in inflammation and, possibly, immune recovery. This was also supported by showing that the cannabinoid N -acyl amide oleamide, a molecule that regulates inflammation of the gastrointestinal tract ( Carroll et al., 1992 ), was also significantly accumulated by gut bacteria present only in the IR group of patients and was below the detection limit in the VU and INR patients and healthy controls (P < 0.00073; Table S4).
BV, the first reaction product of the breakdown of haeme catabolism, accumulated only in the gut bacteria of IR patients; BR, a degradation product of BV, accumulated only in the bacteria of INR individuals; and URO, the final degradation product of BR, was detectable only in the bacteria of VU patients (P < 0.00528; Table S4). None of these metabolites accumulated to an amount above the detection limit in healthy controls, suggesting that haeme catabolism represents a minor pathway in uninfected individuals. It has been shown that antiretroviral drugs are potent inhibitors of uridine glucuronyl transferases (UGT), which are hepatic enzymes responsible for BR metabolism that have also been identified in some bacteria (McPhee et al., 1996 ). This could be one explanation for the lack of accumulation of BR in HIV-untreated patients (VU), most likely because it is fully degraded by gut bacteria into URO, which is then accumulated intracellularly. However, accumulation of the haeme degradation products BV or BR inside the gut bacteria of patients undergoing ART could be linked to the inhibition of both hepatic and bacterial UGT enzymes. Indeed, one effect of some antiretroviral regimens is nearly constant hyperbilirubinemia (Wegiel and Otterbein, 2012 ). This is in agreement with our study: the accumulation of haeme degradation products (BV/BR) in patients (n = 2) treated with boosted atazanavir is slightly higher (P < 0.01) compared to that in patients (n = 2) treated with unboosted atazanavir (1.9-fold) and those (n = 16) treated with a different third antiretroviral drug (2.2-fold). Interestingly, while the gut bacteria of immune responders accumulated BV, those of non-responders accumulated BR (Table S4). This is of particular importance because it has been reported that BV, but not BR, reduces HIV viral infectivity (Wegiel and Otterbein, 2012 ) and is an important anti-inflammatory molecule (Fowler, 2004 ). Therefore, a link between immune recovery and the capacity of gut bacteria to accumulate BV is suggested. It is plausible that, under ART, gut bacteria play an indirect role in the rate of conversion of BV to BR by hepatic enzymes. Thus, because hepatic BV reductase is strongly induced by its substrate (BV) (Fowler, 2004 ), it is plausible that the accumulation of BV by gut bacteria may decrease its diffusion gradient from the colon to epithelial cells, thereby reducing the conversion of BV to BR. This would help to control local viral replication and reduce inflammation.
Altogether, the results suggest that gut bacteria most likely assist inflammatory and immune recovery by accumulating pro-inflammatory molecules and controlling the production of molecules that might have an influence on viral replication.
3.4. Plasma Metabolomics
3.4.1. Metabolic Changes in the Bloodstream vs in the Gut Microbiota
The results of metabolite profiling of plasma samples were further evaluated to identify whether the metabolic changes in the gut ecosystem have a consequence in the bloodstream. The plasma metabolomes (Table S5 ) were less complex than those in the gut microbiota (Table S3 ). A scatter plot based on PCA scores obtained from the set of metabolites that fulfilled the statistical criteria for selection revealed no differences among the groups (Fig. 1 b; Fig. S1), indicating that alterations in the gut ecosystem caused by HIV and ART intervention had no major effect on the overall composition of metabolites in the bloodstream. It might seem contradictory that we observed numerous differences in the metabolic profile in the gastrointestinal tract but not in the plasma. The striking absence of abnormalities in the full set of plasma metabolites in VU patients can be explained by the fact that naïve patients were, by definition, free of the criteria required for ART initiation at the moment of the study and that they were therefore in good immunological condition. Whatever the case, the above results seem to be contradictory with a recent study (Cassol et al., 2013 ) that reported substantial changes in plasma metabolites in HIV subject compared to controls. However, note that this study accounts only for 35 metabolites that distinguish both groups, and that only those are accounted in the PCA plot, thus explaining the separation of HIV subjects and controls. In our study, the unsupervised scatter plot was generated using all statistically significant mass features detected (3602 in total); they include metabolites similar to those previously reported by Cassol et al. (2013) as distinguishing HIV-infected and uninfected individuals such as lipids and lipopolysaccharides (Table S5 ).
3.4.2. Plasma Signatures for Inflammation/Immune Recovery and Microbial Clearance
An independent t -test did not reveal any plasma metabolite as potential biomarker related to innate immune activation markers. However, 3 altered metabolites that are markers of viral infectivity and inflammation were found, whose biological relevance is discussed below.
One of the most significant alterations was found in the level of acetyl adenylate. This metabolite accumulated only in the bloodstreams of immunological responders (IR) and was below the detection limit in the VU and INR patients and healthy controls (P < 0.00201; Table S4). This led us to hypothesize that this molecule may be involved in a protective mechanism. Acetyl adenylate participates in the non-enzymatic acetylation of histones, the alteration of which has been associated with the purging of HIV reservoirs, an optimal immunological situation (Atarashi et al., 2013 ), cTreg expression (Chen et al., 2009 ), and the regulation of thymic function (Chen et al., 2009 ). Additionally, acetyl adenylate-mediated protein (including histone) acetylation has been associated with the reduced expression of factors involved in inflammatory processes (Kohli and Levy, 2009 ). To test this hypothesis, changes in histone acetylation were measured in the PBMC samples using western blot analysis for acetylated histone H3 (AcH3). Low levels of AcH3 were detectable by western blot analysis in VU, INR and healthy control individuals; however, an intense band indicating the hyper-acetylation of H3 was observed in PBMC samples of IR patients (Fig. S4). A correlation between the level of acetyl adenylate in plasma and the level of acetylated histone in PBMC was thus demonstrated. Therefore, the non-enzymatic acetylation of histones should be considered an important process to investigate in the field of HIV cures, as histone acetylation plays a role in reactivating latent HIV (Atarashi et al., 2013 ).
The deep analysis used to search for pro-inflammatory and anti-inflammatory mediators also revealed that neither HIV nor ART has an influence on the accumulation of leukotrienes such as LTB4 in the bloodstream, which was found to be secreted to the gastrointestinal tract (see above). However, lipoxin A5 (LXA5), an intermediate generated from AA, the appearance of which is linked to the resolution of inflammation (Serhan, 2014 ) and microbial clearance (Oh et al., 2011 ), was found to be accumulated in the plasma samples of all of the HIV-infected patients. However, it was below the detection limit in healthy controls (P < 0.01605; Table S4). Additionally, 5,6-epoxy,18-hydroxy-7E,9E,11Z,14Z,16E-eicosapentaenoic acid (5,6-epoxy-18-HEPE), an intermediate of the metabolism of eicopentaenoic acid (Gough et al., 2015 ), accumulated only in IR patients (P < 0.02775; Table S4). This metabolite is an intermediate in the biosynthesis of resolvins, which are potent anti-inflammatory and pro-solving lipid mediators that also actively clear inflammation and enhance microbial clearance ( Serhan, 2014 and Gough et al., 2015 ).
The consistent identification of anti-inflammatory molecules in plasma samples suggests that the host reacts to HIV infection and its induced chronic inflammation (Somsouk et al., 2014 ) by promoting the biosynthesis of pro-solving inflammatory and microbial clearance mediators, some of which were particularly abundant in immunological responders. These conclusions are in agreement with the findings that bacterial translocation occurs in individuals with chronic HIV infection but is minimal in healthy individuals and that over-production of these molecules may also help to control the clearance of microbial products that are released into the systemic circulation (Sandler and Douek, 2012 ).
3.5. Gut bacterial Proteomics
3.5.1. Associations between HIV, ART, CD4 + T Cell Recovery and Gut Microbes' Protein Synthesis
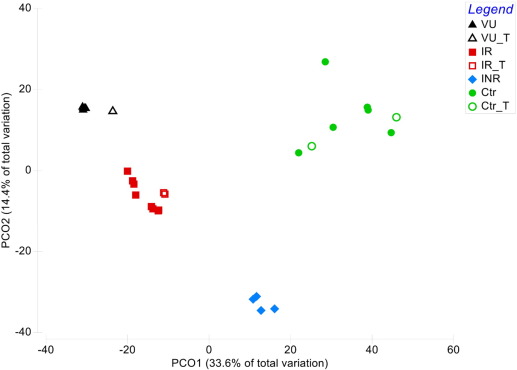
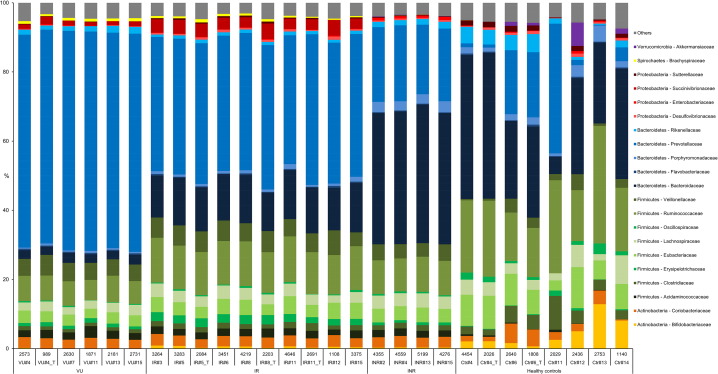
We selected a shotgun metaproteomic approach to measure the functional expression of the microbiomes in the VU, IR, INR and control groups. We captured the protein expression profiles and the differences in the composition and abundance of proteins being expressed and the active community members expressing them (Table S6 ) were evaluated in a subset of samples: 5 VU, 7 IR and 4 INR patients, and 6 HIV− individuals. Additionally, 6-weeks longitudinal samples for 6 of those individuals (VU: 1; IR: 3; HIV− : 2) were analysed. Samples were selected to cover the widest spectrum of sampling factors (Table S1 ). According to protein expression profiles, an unsupervised scatter plot based on PCA scores revealed a clear separation not only in the uninfected and infected groups of patients but also in the three HIV-infected sub-groups (Fig. 3 ). The taxonomic distribution of the identified expressed proteins at the family level (Table S5 ) also demonstrates the separation between the patient groups (Fig. 4 ) and that different bacteria are being active in each disease condition. No variations were observed in the level of expression (Fig. 3 ) and active taxa (Fig. 4 ) in the consecutive measurements. This trend was also found by examining bacterial metabolite profiles (Fig. 1 a). Taken together, changes in gut metabolism in HIV-infected individuals are a direct consequence of the differential expression of proteins by distinct sets of active bacteria and that the intra-individual variability in the bacterial proteomes and metabolomes was not significant in this study.
|
|
|
Fig. 3. Changes in protein expression profiles. The metaproteomic analysis PCO plot shows the similarity in the abundance of quantified proteins in each individual (Bray Curtis similarity; Bray and Curtis, 1957 ). An Analysis of Similarities (ANOSIM) revealed significant differences in protein abundance between groups with a global R of 0.668 at a significance level of 0.01%. Compared to the uninfected group, the Significant Percentages (SIMPER) analyses yielded an average dissimilarity of 82.22% in comparison to the VU group, 74.22% in comparison to the IR group and 72.31% in comparison to the INR group, with a cut off for low contributions at 90%. Symbol codes are included in the figure. The 6-weeks longitudinal samples, label as “_T” (for time), are incorporated in the PCA with empty symbols. |
|
|
|
Fig. 4. Taxonomic distribution of proteins that are expressed by gut bacteria at the family level. The taxonomy bar-chart was built with Unipept and is based on the peptides that we identified with the Mascot-Server. In the upper row, the number of totally identified peptides with the Mascot-Server are shown for each individual. The second row shows the id (#) of the individuals within groups, which are shown in the third row. The 6-weeks longitudinal samples, label as “_T” (for time), are incorporated right to the same initial sample. Note: we are aware that the classification based on family level allows a gross interpretation of the active members and their functions. Unfortunately, the peptide information commonly obtained by metaproteomics does not allow a deeper taxonomic characterization. Analyzing the phylogeny of all peptides (after quality check) already enabled the affiliation down to family level of only approximately 50% of the peptides in each sample; the others could be either assigned to levels of phyla or order. |
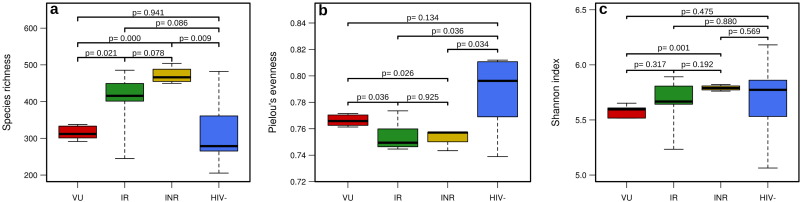
Proteome data can provide estimation of the taxonomic diversity of potentially active bacteria. We used Primer 6 (version 6.1.16) and PERMANOVA + (version 1.0.6) from the company Primer-E to determine the richness and evenness of the identified proteins. The alpha-diversity indexes (Fig. 5 ) showed that the healthy control subjects had highly variable active gut microbiota diversities. This may agree with the fact that in the absence of health pressures (i.e. the presence of a disease) the gut microbiota is an ecosystem with high inter-individual bacterial heterogeneity as compared to a scenario where a strong pressure (here, viral infection) exists. Also notice there is substantial inter-individual heterogeneity in the IR patients compared to that in VU and INR. This may agree with the fact that these patients are characterized by a healthier gastrointestinal tract and that the viral infection is not such a strong pressure factor as in VU and INR patients characterized by extensive structural, functional, and immunological changes in the intestines. No statistically significant differences were found in species richness (Fig. 5 a), evenness (Fig. 5 b) and Shannon index (Fig. 5 c) when comparing controls and VU groups (P > 0.134), indicating that the viral infection per se does not influence significantly the active fraction of the microbiota. This may explain the similarity between the microbes' metabolic activity in HIV-negative and VU groups of individuals (Fig. 1 a). However, we found that ART-treated patients had significantly higher richness (Fig. 5 a) of active species and a lower evenness index (Fig. 5 b) compared to VU (P < 0.026) and healthy control (P < 0.036) groups; no significant differences between INR and IR groups were found (P > 0.078). This indicates that in treated patients the relative abundances of taxa in the active subset were less equally distributed among widespread bacterial groups.
|
|
|
Fig. 5. Diversity parameters of potentially active microbiota based on the analysis of proteins identified and quantified using MaxQuant. (a) Species richness. (b) Pielous evenness. (c) Shannon index. P -values are given per each of the comparisons. |
3.5.2. Active Bacterial Biomarkers of HIV-Infection
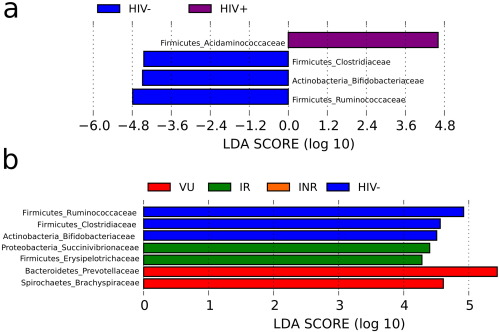
Bacteria belonging to the Prevotellaceae, Acidaminococcaceae Bacteroidaceae, Desulfovibrionaceae, Enterobacteriaceae, Erysipelotrichaceae, Eubacteriaceae, Lachnospiraceae and Veillonellaceae families have been previously identified as active biomarkers in HIV-infected patients. This was revealed by high-resolution total bacterial community profiling and direct DNA sequencing (Mutlu et al., 2014 and Vázquez-Castellanos et al., 2015 ). By examining the taxonomic assignments of the proteins that are expressed (Table S6 ; Fig. 4 ), only the enrichment of proteins from the Acidaminococcaceae family was identified as an active biomarker of HIV infection (Fig. 6 a). This suggests that bacteria of this taxon are among the most active biomarkers that distinguish infected from uninfected individuals and that, from a functional point of view, the metabolic potential of the other bacterial groups to contribute to HIV-induced structural, functional and immunological deficits may be lower than was previously thought. It should be noted that an increase in the relative abundance of bacteria of the Acidaminococcaceae family has been associated with future linear growth deficits (Isanaka et al., 2009 ) and that growth failure is common among infected patients with HIV (Rush et al., 2008 ). We further observed three additional biomarkers for the HIV group, the Clostridiaceae, Bifidobacteriaceae and Ruminococcaceae families, which, opposite to the previous group, were significantly decreased in HIV-infected patients (Fig. 6 a). Such groups have not been found to be differentially abundant when examining 16S rRNA datasets in HIV-infected patients (Mutlu et al., 2014 and Vázquez-Castellanos et al., 2015 ), possibly because of the low abundance of these bacterial groups in the total gut community.
|
|
|
Fig. 6. Linear discriminative analysis (LDA) effect size (LEfSe) analyses of statistically significant active families. (a) Negative LDA scores (red) are enriched in HIV-infected patients, while positive LDA scores (blue) are enriched in healthy controls. (b) LDA scores for active families are enriched in each of the examined groups. |
We used the linear discriminative analysis (LDA) effect size (LEfSe) biomarker discovery tool to elucidate which active families were driving divergences between the groups (P < 0.01) and to identify active biomarkers in immunological responders. We found 3 families for healthy controls, 2 for VU and 2 for IR with LDA scores higher than 4 (Fig. 6 b). None was found for the INR group. Active biomarkers for healthy controls are bacteria that belong to the Ruminococcaceae, Clostridiaceae and Bifidobacteriaceae families; bacteria of these families were significantly enriched in healthy controls. In the VU group, Prevotellaceae and Brachyspiraceae were the active biomarkers, as they were significantly enriched in this group. Finally, bacteria of the families Succinivibrionaceae and Erysipelotrichaceae were significantly enriched in the IR group, and thus they were biomarkers of immune recovery in HIV-infected patients.
3.5.3. Bacteria Controlling the Accumulation of Mediators Implicated in Immune Recovery
Members of the Succinivibrionaceae family which were found to be significantly activated in IR patients (see Fig. 6 b), have also been found to be enriched at the level of total community composition in ART-treated HIV individuals with good CD4 T cell reconstitution compared to healthy subjects (Vázquez-Castellanos et al., 2015 ). Bacteria of the family Succinivibrionaceae are quite undiscovered bacteria whose protective role against gut inflammation has been suggested in the porcine colon during nematode infection (Li et al., 2012 ). It is therefore plausible that these bacteria might play an active metabolic role in immune recovery. One possibility is that they participate in the active transport and accumulation of anti-inflammatory, viral inhibitors and pro-solving lipid mediators, such as LTB4 , BV and cannabinoid N -acyl amides, which were found to be accumulated inside the gut bacterial cells of immune responders (Table S4). This hypothesis is supported by the fact that a solute-binding domain of the SLC5/6 protein transporter (gi | 671,503,691) binned to Succinivibrionaceae was exclusively expressed in this group of patients (Table S6 ). No other transporter protein was found to be specifically expressed in the IR group of patients. Experimental evidence has shown that SLC5/6 transporters do participate in the transport of complex molecules, such as neurotransmitters (Wright et al., 2004 ).
To prove whether bacteria of the family Succinivibrionaceae are able to actively transport molecules that help resolving inflammation and immune recovery, an experimental assay was performed. Briefly, cultures of Succinivibrio dextrinosolvens (type strain DSM-3072 ; DSMZ, Braunchweig, Germany), a gut bacterium of the Succinivibrionaceae family, were established in medium 330 (anaerobic, 37 °C, 2 weeks) in the presence of BV and oleamide (10 mg/L; Sigma Aldrich, Madrid, Spain). As control strains, we used Pseudomonas aeruginosa PAO, Escherichia coli K12, Staphylococcus aureus and Enterococcus faecalis , which were grown as previously reported ( Kubacka et al., 2007 ) in the presence of BV and oleamide (10 mg/L). After cultivation, bacterial cells were separated, and the concentrations of both metabolites in the supernatant and bacterial cells were measured (see Materials and Methods). We found a residual percentage of both metabolites at the end of the assay in supernatants of S. dextrinosolvens cultures, and concomitant accumulation inside bacterial cells (Fig. S5). Although, this strain may not be the same as the one detected in the gut ecosystem of IR patients, it demonstrates that bacteria of the Succinivibrionaceae family are able to efficiently transport molecules implicated in inflammatory and immune recovery. Note that no accumulation (or a level below the detection limit) of both metabolites was detected inside the control strains (Fig. S5).
3.5.4. Correlations between Markers of Innate and Adaptive Immunity and Active Gut Microbiota
Finally, we used the Generalized Linear Model (GLM) to explore the interactions between the abundance level of active taxonomic groups expressing proteins (Table S6 ) and immunological and genetic markers involved in HIV immune-pathogenesis. In particular, we focused on finding statistically significant associations using active bacterial biomarkers (Erysipelotrichaceae and Succinivibrionaceae) of the IR group. There was a correlation between the degree of the CD4+ T cell activation marker %HLA-DR+ CD38+ and the percentage of active Erysipelotrichaceae (Rho Spearman correlation index = − 0.821; P = 0.034) and between the CD4/CD8 ratio and the percentage of active Succinivibrionaceae (Rho = 0.857; P = 0.023). No other active bacterial group, from those identified by proteomic analysis, were systemic markers for those variables. Note that, %HLA-DR+ CD38+ and CD4/CD8 ratio are immunological markers of immune activation and are predictors of inflammation and mortality during HIV infection ( Hunt et al., 2011 and Serrano-Villar et al., 2014 ). Indeed, IR patients are characterized by lower inflammation compared to VU patients due to the absence of viral replication and a partially recovered immune system. The over-abundance of active Erysipelotrichaceae and Succinivibrionaceae in IR individuals may thus correlate with better levels of immuno-activation and immune function, and the level of abundance may help reduce excess mortality.
We further found that the abundance of active Bifidobacteriaceae was associated with a higher level of HDL cholesterol (Rho = 0.954; P = 0.00080). This bacterial group was significantly depleted in HIV-infected patients compared to healthy controls at the level of active microbiota (Fig. 6 a); in HIV-infected patients, depletion was most associated with the VU and INR groups, while it was detected at a higher level in the IR group (Fig. 6 ). Bifidobacteria are powerful bacteria that can protect the gut, boost the immune system and control inflammatory responses (Fujimura et al., 2010 ). Therefore, it is plausible that the occurrence, albeit at low levels (Fig. 6 ), of active Bifidobacteria in the gut of IR patients may be an additional element that contributes to the improvement of health status in these patients, e.g., favouring the HDL cholesterol levels that are known to be associated with lower cardiovascular risk.
Finally, Acidaminococcaceae, which is an active (or functional) biomarker of HIV-infection as identified here (Fig. 6 a), was associated with the level of CD4 (Rho = 0.821; P = 0.034), and no other active bacterial group was associated with this marker. As Acidaminococcaceae was also a biomarker at the level of the total microbiota as determined by 16S rRNA analysis (Vázquez-Castellanos et al., 2015 ), a correlation between abundance and function (in this study) of this bacterial group during HIV infection and CD4 recovery is demonstrated. No such agreement between abundance and function was found for Erysipelotrichaceae, Succinivibrionaceae and Bifidobacteria, as they were biomarkers when considering the active components of the microbiota but not the total microbiota (Vázquez-Castellanos et al., 2015 ).
4. Discussion
As for other diseases and disorders (Marchesi et al., 2015 ), there is a strong interest in identifying next-generation information to describe how HIV infection impacts the gut environment and the microbiota inhabiting it (Vyboh et al., 2015 ). The majority of previous studies investigated the gut microbiota during viral infection at the level of total species (DNA). These studies observed different shifts as a consequence of HIV infection and ART. Our results provided an additional vision about the impact of HIV infection, antiretroviral therapy and immune recovery in the gut microbiota at the level of protein synthesis and metabolism, which are indicators of the active processes occurring in the gastrointestinal tract. The study included 8 healthy HIV-uninfected controls and 29 HIV-infected patients, 6 of whom were examined longitudinally.
The data herein presented showed that each of the four groups investigated possesses a unique active microbiota. Remarkably, during HIV infection and in the absence of antiretroviral therapy, the numerous structural, functional, and immunological changes in the intestines (Vujkovic-Cvijin et al., 2013 ) seems to provoke the activation of a specialized subset of bacterial phylotypes that are different to those in uninfected individuals (Fig. 4 ), but that are equally diverse, rich and active. Therefore, HIV infection per se did not alter significantly gut ecosystem function, but rather affect to fundamental properties. The metabolic deficits observed in the comparisons of infected and uninfected patients could be explained by HIV-induced changes in, for example, epithelial disruption (Sandler and Douek, 2012 ). Indeed, it is plausible that during HIV infection, pathogenic species scavenging mucosa components such as sialic and dolichol-moieties (Crick and Rip, 1989 ) and ceramide lipids (Bouhours and Bouhours, 1985 ) are significantly increased. These molecules can be accumulated inside bacterial cells, where they can encourage pathological microbe growth and induce loss of epithelial cell integrity and translocation of pathogenic bacterial products into the bloodstream, commonly observed in HIV infected patients (Severi et al., 2007 and Cassol et al., 2013 ).
Comparisons of patients receiving ART or not, demonstrated that patients receiving ART, whatever the level of immune recovery, had the highest and most similar active bacterial richness and lowest evenness. This contrasts with the previous evidences showing a lower bacterial richness and diversity in patients receiving ART, particularly in the INR group, using 16S RNA amplicons (Noguera-Julian et al., 2016 ). This is an important result, as the active microbiota, namely that with protein synthesis, potentially is more essential for the host than the inactive (total) microbiota because of factors such as metabolic turnover. Albeit being similarly diverse and even, both groups of individuals contain a distinctive set of active taxa (Fig. 4 ) that behave functionally distinct (Fig. 1 a). While the increase in richness and active taxa in INR individuals is not translated to changes in the overall ecosystem function compared to that in the VU patients and healthy control, in IR patients it does. We suggest therefore, that the degree of immune recovery is not associated to a change in the richness of active bacteria, but rather to an ecosystem similarly diverse but with different set of metabolically distinct species.
We found that the changes associated to IR group of individuals were not evident in the blood, particularly among ART treated patients. Therefore, we found that gut bacteria metabolism is a best marker of adequate immune recovery. This was found both at the level of microbes´ protein synthesis and microbes´ metabolic activity regardless sampling factors, thus reinforcing the significance of the findings. This was not suggested by previous studies when evaluating the total microbial composition (Vujkovic-Cvijin et al., 2013 , Mutlu et al., 2014 and Vázquez-Castellanos et al., 2015 ). This prompted us to suggest that inferring metabolic consequences during HIV infection based solely on taxonomic classification (total microbiota at the DNA level) is unrealistic and that studies at the active (functional) level are needed (Moya and Ferrer, 2016 ).
Although HIV cannot target bacteria, it is worth considering what effects that antiretroviral drugs used to treat the viral infection might have on the gut microbiota. One possibility is that immune recovery might induce alterations in microbiome profiles in the gut environment. Indeed, recent studies linked bacterial richness with immune function (Nowak et al., 2015 ). However, the results of this study reinforce also the hypothesis that favourable changes in the gut ecosystem may also be counted as additional contributors to the recovery of mucosal and systemic immunity. In this context, we found that the antiretroviral therapy most likely drastically decreased the abundance of pathogenic species involved in glycan and lipid (ceramide) modification, thereby most likely favouring the integrity of the colonic surface and the maintenance of its functions (Deeks et al., 2013 and Masoodi et al., 2013 ). Other concrete examples that demonstrate the role of gut bacteria as being fundamental for aiding in inflammation and immune recovery involve the metabolism of pro- and anti-inflammatory mediators. Indeed, our data demonstrate that gut bacteria, most likely bacteria of the family Succinivibrionaceae, may play a role in controlling the accumulation of pro-inflammatory molecules that would otherwise promote inflammation in the gut environment (Serhan, 2014 ). This is the case for the leukotriene LTB4 , which might be less likely to accumulate in the gastrointestinal tract when it is accumulated in the gut bacteria. Leukotrienes are potent pro-inflammatory lipid mediators (Bertin et al., 2012 ). Therefore, reducing their concentration in the large intestine may exert a positive effect that mitigates inflammation and the loss of mucosal epithelium. This may play a direct role in promoting immune recovery because the capacity of gut bacteria to accumulate LTB4 was only observed in ART responders. Similarly, gut bacterial members (e.g., bacteria of the family Succinivibrionaceae and possibly Erysipelotrichaceae as suggested by taxonomic assignments of transport proteins being expressed) may also contribute to the accumulation of additional molecules that have the ability to reduce viral infection (i.e., BV) and signalling molecules (i.e., oleamide) that help to reduce inflammation (Froldi et al., 1992 , Genis et al., 1992 and Bertin et al., 2012 ). Other bacteria such as the Bifidobacteria may also be implicated in additional health benefits.
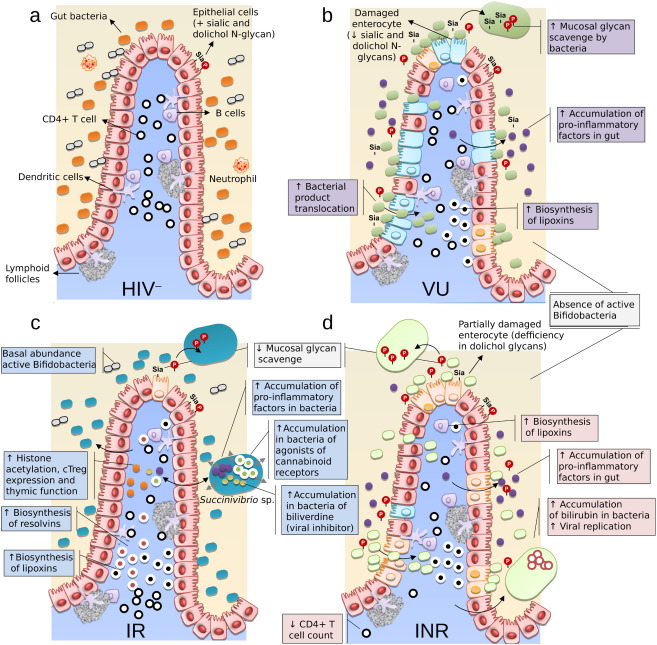
While gut bacteria help to control the accumulation of important molecules that are associated with inflammation, immune recovery and viral infectivity, we also demonstrated that human cells respond by increasing the biosynthesis of anti-inflammatory and pro-solving lipid mediators that circulate in the plasma and help diminishing inflammation (Isanaka et al., 2009 ), and controlling HIV replication (e.g. by non-enzymatic acetylation of histones). An orchestrated solution to inflammation is thus plausible between the host and the gut ecosystem that is aimed at helping to reduce inflammation and boost immune recovery (for a summary see Fig. 7 ). It is therefore plausible that the metabolic connection between the host and the gut ecosystem is stronger than previously thought and that they can work together to improve immune recovery. It will be important to identify the mechanisms by which such metabolic connections are established and to clearly identify whether an HIV-infected individual responds better to ART as a result of the additional contributions of the microbiota or whether the gut bacteria metabolism is altered as a consequence of the improvement in health of the host after ART. This last hypothesis may agree with the fact that human genetics and human health shape the gut microbiome (Goodrich et al., 2014 ). Although, we cannot rule out either of these two possibilities, this study supports the first hypothesis: a specialized subset of active microorganisms structured in a more even configuration than the total gut community (Vujkovic-Cvijin et al., 2013 and Vázquez-Castellanos et al., 2015 ) is implicated in key inflammation and immune traits. Thus, we reasoned that the accumulation of pro-inflammatory molecules during HIV infection may be one of the most easily controlled factors that can modulate the composition of the active microbiota, which is shifted to reduce their concentration in the gut environment and to help immune and inflammation recovery.
|
|
|
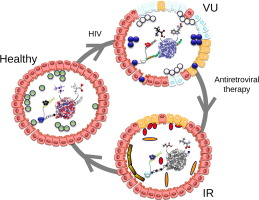
Fig. 7. Proposed mechanisms for the contribution of gut bacteria and human cell products to the recovery of mucosal and systemic immunity during HIV infection. (a) The intestinal epithelium in a healthy individual. A continuous enterocyte lining with intact tight junctions promotes barrier integrity and prevents the translocation of commensal bacteria from the intestinal lumen into the intestinal wall. The microbiota comprises active microbiota species that yield a defined repertoire of expressed proteins and metabolites that interact with the gut mucosa. Human cells promote immune-tolerance mechanisms. No inflammation occurs in this healthy state. (b) The intestinal epithelium in an HIV-infected individual not receiving ART. These individuals are characterized by progressive immunodeficiency, immune hyper-activation and excess mortality. The microbiota comprises a distinct set of active microbiota species that yield a different repertoire of expressed proteins and metabolites than those documented in healthy individuals. The cleavage of sialic and dolichol components from epithelial cells by gut bacteria causes abnormal glycan composition in enterocytes; this leads to the destruction of tight junctions and increased microbial translocation. Human cells increase the production of lipotoxins, which accumulate in the bloodstream. The biosynthesis of leukotrienes also increases, and they accumulate in the gut environment (faecal fluid), promoting inflammation and toxicity. (c) The intestinal epithelium in an HIV-infected individual who is an immunological ART responder. The individual is characterized by the variable recovery of the mucosal architecture and immune functions, low-level inflammation and subtle health vulnerability. Distinct active species (including Bifidobacteria), proteins and metabolites characterize this individual. The cleavage of epithelial cell components by gut bacteria is limited to the cleavage of dolichol-glycans, which lowers the destruction of enterocytes and limits microbial translocation. Gut bacteria (e.g. Succinivibrionaceae family) accumulate AA and its inflammatory mediator LTB4 in addition to the cannabinoid oleamide and biliverdin (a viral inhibitor), which may contribute to health recovery by inhibiting viral replication, stimulating the immune system, and ultimately reducing inflammation. Human cells reciprocally increase the biosynthesis of anti-inflammatory and pro-solving lipid mediators (the resolving intermediate 5,6-epoxy-18-HEPE) and lipotoxins, which circulate in the bloodstream. (d) The intestinal epithelium in an HIV-infected ART non-responder. This individual is characterized by chronic mucosal impairment, systemic immune impairment, chronic inflammation and excess mortality. The set of microbial products in this individual are most similar to those in the ART-untreated patients. N -glycan metabolism occurs as in (c). Lipotoxins and leukotrienes are also produced as in (b). Gut bacteria accumulate bilirubin. |
We would like to stress that one of the limitations of this study is that, while we have the ability to show associations, we can only demonstrate causality to a limited extent. In addition to that, although the set of active bacteria and molecular agents identified herein may not be the only ones contributing to immune recovery in HIV-infected patients, they certainly may play a key role in HIV progression and chronic immune response, and thus life expectancy, in HIV patients during effective ART. Also, the observed associations do not necessarily indicate an effect of gut microbiota on immune-reconstitution and inflammation. Other possible explanations could be that immune-reconstitution and inflammation influence gut microbiota or that other unexplored factors linked to HIV infection, antiretroviral therapy, immune-reconstitution and inflammation could impact on gut bacteria environment.
We are aware also that the only way to definitively evaluate the impact of ART on the gut microbiome is to study the microbiota in patients prior to ART initiation followed by the analyses of changes during ART. Similarly, the only way to evaluate definitively the effect of HIV is to study HIV-uninfected patients and to analyze the effects following HIV infection. While these studies will likely come and provide definitive data, the complexity inherent to these study designs has hampered so far its development in humans, and we feel that cross-sectional studies still provide valuable data to approach the problem. Increasing the size of all of the cohorts would improve the significance of the findings. Following on from this, while the present manuscript is focused on the associations between the gut microbiota metabolism and systemic markers, the existence of a group of patients with repeated measurements after six-weeks offers the opportunity to account for intra-individual variability in a subgroup of patients. We overall observed no changes in the analysed data in this subgroup, thus reinforcing the significance of the findings, which were herein confirmed also by two distinct but complementary omics approaches.
While our study is focused on HIV-infected individuals, our findings should be of general interest because our data suggest the need to assess the metabolic contribution of gut bacteria to recovery or to the maintenance or persistence of defects in innate and adaptive immunity across different diseases.
5. Conclusions
We herein report the study of HIV-infected individuals analyzing the effects of HIV infection, antiretroviral therapy and immune recovery on gut microbiota, particularly on microbes´ protein synthesis and microbes´ metabolic activity. We found that, compared to a healthy situation, the HIV infection per se is a pressure factor to which gut microbiota react without an overall effect in diversity, richness and activity. We further found that the antiretroviral intervention is safe and well tolerated from a metabolic point of view, albeit it provokes that an additional set of bacterial species react, the nature of which is different depending on the immune recovery level of the individuals. Although we cannot rule out the possibility that some of these changes are partially a random consequence, our data demonstrate also that others are a cause by which a subset of bacterial species become active to help controlling the accumulation of molecular agents and promote immune and inflammation recovery in HIV-infected patients undergoing antiretroviral therapy. We also found relationships between gut bacteria and host which most likely influence the immune recovery.
The following are the supplementary data related to this article.
Table S1.
Individual characteristics, clinical variables and HIV immune pathogenesis markers. The Table has been provided as a separate Excel table due to its extensive size (https://dl.dropboxusercontent.com/u/62046725/Copia%20de%20Table%20S1%20%28new%20format%29.xls).
Table S3.
Putative metabolites identified and quantified in bacterial metabolomes of HIV-infected and non-infected individuals using LC-MS (-), LC-MS (+) and CE. For differential quantitative metabolomics, we compared the metabolomes (in triplicate) of cells by evaluating the peak area from the chromatographic peaks. Total numbers of significant mass features per group of samples are as follows: LC (+) 244,548, LC (−) 89,230 and CE 2,724. A list of masses that were identified is presented for all individuals. Because the samples interact during the separation technique and MS, it is crucial to use quality controls (QCs) during LC-MS to ensure analytical reproducibility. Indeed, QC samples are required throughout the analytical runs at periodic intervals during analysis to monitor variations in the signals across time and at the beginning of the sequence to stabilize the system. QC samples were prepared for LC-MS by pooling and mixing equal volumes of each sample. After gently vortexing the samples, the mixture was filtered and subsequently transferred to an analytical vial and analysed. In all cases, the mass (in Dalton [Da]), retention time (RT; as Da@RT), and abundance in each sample is shown. To correct for multiple comparisons the Mann–Whitney U test was performed. This test compares the groups by pairs and reports for each of them an independent p -value (p MWU). Subsequently it was corrected with Benjamini–Hochberg false discovery rate, which also provides the corresponding p -value (p BH). Based on that, a final table with statistically truly significant (P < 0.05) metabolites identified and quantified was prepared which is herein presented. The Table has been provided as a separate Excel table due to its extensive size (https://dl.dropboxusercontent.com/u/62046725/Copia%20de%20Table%20S3%20%28new%20format%29.xls). For raw data (including p-values for the comparison by groups), please contact the authors directly.
Table S5.
Putative metabolites identified and quantified in plasma metabolomes of HIV-infected and non-infected individuals using LC-MS (-) and LC-MS (+). Total numbers of significant mass features per group of samples are as follows: LC (+) 73227 and LC (−) 4141. For details, see the Table S3 legend. The Table has been provided as a separate Excel table due to its extensive size (https://dl.dropboxusercontent.com/u/62046725/Copia%20de%20Table%20S5%20%28new%20format%29.xlsx).
Table S6.
Protein expression datasets. (A) List of proteins identified and quantified with label-free quantification (LFQ) values. (B) Abundance levels of proteins assigned to taxonomic groups (family level). The Table has been provided as a separate Excel table due to its extensive size (https://dl.dropboxusercontent.com/u/62046725/Copia%20de%20Table%20S6%20%28new%20format%29.xls). For raw data, please contact the authors directly.
Supplementary material
Author Contributions
M.F., S.S-V., and A.M. contributed to the conceptualization of the study; S.S-V., D.R., M.M.M., M.F., C.B., S.D., R.B., J.S., J.F.V-C., and M.J.G. contributed to the methodology; D.R., C.B and M.F contributed metabolomics data design, data collection, data analysis and data interpretation; S.D., J.S and M.F., contributed proteomics data design, data collection, data analysis and data interpretation; S.S.V., T.S., S.M., V.E., M.V., J.S., A.L., A.M., C.B., and M.F. contributed resources (data collection, data analysis, and data interpretation); D.R, R.B., S.D., and M.F. contributed to figures preparation; M.F., and S.S-V., wrote the original draft, which was reviewed by D.R., A.M., J.S., and C.B., and approved by all authors; and M.F., S.S-V., A.L., A.M., and C.B. acquired funding.
Conflict of Interest
The authors declare that no competing interests exist.
Acknowledgements
This work was supported by the Health Research Institute of University Hospital Clínico San Carlos, “Generalitat Valenciana ” (PROMETEOII/2014/065 ), by the Spanish Ministry of Economy and Competitiveness (SAF2012-31187 , SAF2013-49788-EXP , SAF2015-65878 from MINECO ), Instituto de Salud Carlos III (PIE14-00045 and Redes Temáticas de Investigación en SIDA RETICRD12/0017/0029 and RD12/0017/0037 ), and the Junta de Andalucía (Proyecto de Excelencia CTS-6313 ). The present investigation was also funded by the Spanish Ministry of Economy and Competitiveness and the Federal Ministry of Education and Research (BMBF) within the ERA NET PathoGenoMics2 program, grant number 0315441A . This work was further funded by grants BIO2011-25012 and BIO2014-54494-R from the Spanish Ministry of Economy and Competitiveness . The authors gratefully acknowledge the financial support provided by the European Regional Development Fund (ERDF) . C.B and D.R would like to acknowledge funding from the Spanish Ministry of Economy and Competitiveness (CTQ2014-55279-R ). J.F.V.C. was supported by a fellowship “Ayudas Predoctorales de Formación en Investigación en Salud” from the Instituto de Salud Carlos III (Spain) and the CONACYT-SECITI (México) . S.S.V. is supported by a grant from the Spanish Ministry of Science and Innovation (Contratos Juan Rodés, ECC/1051/2013 ), and T.S. is supported by a grant from the European Society of Pediatric Infectious Diseases (ESPID) . Funding Agencies did not have any role neither in the writing of the manuscript or the decision to submit it nor have paid to write this article. The authors wish to acknowledge the participation of all of the study participants who contributed to this work as well as the clinical research staff of the participating institutions who made this research possible.
References
- Archin and Archin, 2014 N.M. Archin, D.M. Archin; Emerging strategies to deplete the HIV reservoir; Curr. Opin. Infect. Dis., 27 (2014), pp. 29–35
- Atarashi et al., 2013 K. Atarashi, T. Tanoue, K. Oshima, W. Suda, Y. Nagano, H. Nishikawa, S. Fukuda, T. Saito, S. Narushima, K. Hase, S. Kim, J.V. Fritz, P. Wilmes, S. Ueha, K. Matsushima, H. Ohno, B. Olle, S. Sakaguchi, T. Taniguchi, H. Morita, M. Hattori, K. Honda, et al.; Treg induction by a rationally selected mixture of Clostridia strains from the human microbiota; Nature, 500 (2013), pp. 232–236
- Bargiela et al., 2015 R. Bargiela, F. Mapelli, D. Rojo, B. Chouaia, J. Tornés, S. Borin, M. Richter, M.V. Del Pozo, S. Cappello, C. Gertler, M. Genovese, R. Denaro, M. Martínez-Martínez, S. Fodelianakis, R.A. Amer, D. Bigazzi, X. Han, J. Chen, T.N. Chernikova, O.V. Golyshina, M. Mahjoubi, A. Jaouanil, F. Benzha, M. Magagnini, E. Hussein, F. Al-Horani, A. Cherif, M. Blaghen, Y.R. Abdel-Fattah, N. Kalogerakis, C. Barbas, H.I. Malkawi, P.N. Golyshin, M.M. Yakimov, D. Daffonchio, M. Ferrer; Bacterial population and biodegradation potential in chronically crude oil-contaminated marine sediments are strongly linked to temperature; Sci. Report., 5 (2015), p. 11651
- Bell et al., 2000 C.J. Bell, E.J. Elliott, J.L. Wallace, D.M. Redmond, J. Payne, Z. Li, E.V. O'Loughlin; Do eicosanoids cause colonic dysfunction in experimental E. coli O157:H7 (EHEC) infection? ; Gut, 46 (2000), pp. 806–812
- Bertin et al., 2012 J. Bertin, C. Barat, D. Bélanger, M.J. Tremblay; Leukotrienes inhibit early stages of HIV-1 infection in monocyte-derived microglia-like cells; J. Neuroinflammation, 9 (2012), p. 55
- Bouhours and Bouhours, 1985 D. Bouhours, J.F. Bouhours; Developmental changes if monohexosylceramide and free ceramide in the large intestine of the rat; J. Biochem., 98 (1985), pp. 1359–1366
- Bray and Curtis, 1957 J.R. Bray, J.T. Curtis; An ordination of the upland forest communities of southern Wisconsin; Ecol. Monogr., 27 (1957), pp. 325–349
- Buzón et al., 2010 M.J. Buzón, M. Massanella, J.M. Llibre, A. Esteve, V. Dahl, M.C. Puertas, J.M. Gatell, P. Domingo, R. Paredes, M. Sharkey; HIV-1 replication and immune dynamics are affected by raltegravir intensification of HAART-suppressed subjects; Nat. Med., 16 (2010), pp. 460–465
- Carroll et al., 1992 K.K. Carroll, N. Guthrie, K. Ravi; Dolichol: function, metabolism, and accumulation in human tissues; Biochem. Cell Biol., 70 (1992), pp. 382–384
- Cassol et al., 2013 E. Cassol, V. Misra, A. Holman, A. Kamat, S. Morgello, D. Gabuzda; Plasma metabolomics identifies lipid abnormalities linked to markers of inflammation, microbial translocation, and hepatic function in HIV patients receiving protease inhibitors; BMC Infect. Dis., 13 (2013), p. 203
- Chen et al., 2009 S.S. Chen, A.J. Jenkins, H. Majewski; Elevated plasma prostaglandins and acetylated histone in monocytes in type 1 diabetes patients; Diabet. Med., 26 (2009), pp. 182–186
- Crick and Rip, 1989 D.C. Crick, J.W. Rip; Age-associated changes in dolichol and dolichyl phosphate metabolism in the kidneys and liver of mice; Biochim. Biophys. Acta, 1004 (1989), pp. 180–186
- Deeks et al., 2013 S.G. Deeks, S.R. Lewin, D.V. Havlir; The end of AIDS: HIV infection as a chronic disease; Lancet, 382 (2013), pp. 1525–1533
- Dillon et al., 2014 S.M. Dillon, E.J. Lee, C.V. Kotter, G.L. Austin, Z. Dong, D.K. Hecht, S. Gianella, B. Siewe, D.M. Smith, A.L. Landay, C.E. Robertson, D.N. Frank, C.C. Wilson, et al.; An altered intestinal mucosal microbiome in HIV-1 infection is associated with mucosal and systemic immune activation and endotoxemia; Mucosal Immunol., 7 (2014), pp. 983–994
- Elliott et al., 2014 J.H. Elliott, F. Wightman, A. Solomon, K. Ghneim, J. Ahlers, M.J. Cameron, M.Z. Smith, T. Spelman, J. McMahon, P. Velayudham, G. Brown, J. Roney, J. Watson, M.H. Prince, J.F. Hoy, N. Chomont, R. Fromentin, F.A. Procopio, J. Zeidan, S. Palmer, L. Odevall, R.W. Johnstone, B.P. Martin, E. Sinclair, S.G. Deeks, D.J. Hazuda, P.U. Cameron, R.P. Sékaly, S.R. Lewin; Activation of HIV transcription with short-course vorinostat in HIV-infected patients on suppressive antiretroviral therapy; PLoS Pathog., 10 (2014), Article e1004473
- Flamand et al., 2004 L. Flamand, P. Borgeat, R. Lalonde, J. Gosselin; Release of anti-HIV mediators after administration of leukotriene B4 to humans; J. Infect. Dis., 189 (2004), pp. 2001–2009
- Fowler, 2004 C.J. Fowler; Oleamide: a member of the Endocannabinoid family?; Brit. J. Pharmacol., 141 (2004), pp. 195–196
- Friedman et al., 2010 J. Friedman, T. Hastie, R. Tibshirani; Regularization paths for generalized linear models via coordinate descent; J. Stat. Softw., 33 (2010), pp. 1–22
- Froldi et al., 1992 M. Froldi, A. Castagna, M. Parma, A. Piona, A. Tedeschi, A. Miadonna, M. Lorini, E.N. Orazio, A. Lazzarin; Mediator release in cerebrospinal fluid of human immunodeficiency virus-positive patients with central nervous system involvement; J. Neuroimmunol., 38 (1992), pp. 155–161
- Fujimura et al., 2010 K.E. Fujimura, N.A. Slusher, M.D. Cabana, S.V. Lynch; Role of the gut microbiota in defining human health; Expert Rev. Anti-Infect. Ther., 8 (2010), pp. 435–454
- Genis et al., 1992 P. Genis, M. Jett, E.W. Bernton, T. Boyle, H.A. Gelbard, K. Dzenko, R.W. Keane, L. Resnick, Y. Mizrachi, D.J. Volsky, et al.; Cytokines and arachidonic metabolites produced during human immunodeficiency virus (HIV)-infected macrophage-astroglia interactions: implications for the neuropathogenesis of HIV disease; J. Exp. Med., 176 (1992), pp. 1703–1718
- Girardi et al., 2007 E. Girardi, C.A. Sabin, A.D. Monforte; Late diagnosis of HIV infection: epidemiological features, consequences and strategies to encourage earlier testing; J. Acquir. Immune Defic. Syndr., 46 (Suppl. 1) (2007), pp. S3–S8
- Godzien et al., 2015 J. Godzien, V. Alonso-Herranz, C. Barbas, E. Grace Armitage; Controlling the quality of metabolomics data: new strategies to get the best out the QC sample; Metabolomics, 11 (2015), pp. 518–528
- Goodrich et al., 2014 J.K. Goodrich, J.L. Waters, A.C. Poole, J.L. Sutter, O. Koren, R. Blekhman, M. Beaumont, W. Van Treuren, R. Knight, J.T. Bell; Human genetics shape the gut microbiome; Cell, 159 (2014), pp. 789–799
- Gough et al., 2015 E.K. Gough, D.A. Stephens, E.E. Moodie, A.J. Prendergast, R.J. Stoltzfus, J.H. Humphrey, A.R. Manges; Linear growth faltering in infants is associated with Acidaminococcus sp. and community-level changes in the gut microbiota ; Microbiome, 3 (2015), p. 24
- Hansen et al., 2014 S.H. Hansen, A. Stensballe, P.H. Nielsen, F.A. Herbst; Metaproteomics: evaluation of protein extraction from activated sludge; Proteomics, 14 (2014), pp. 2535–2539
- Hernández et al., 2013 E. Hernández, R. Bargiela, M.S. Diez, A. Friedrichs, A.E. Pérez-Cobas, M.J. Gosalbes, H. Knecht, M. Martínez-Martínez, J. Seifert, M. von Bergen, A. Artacho, A. Ruiz, C. Campoy, A. Latorre, S.J. Ott, A. Moya, A. Suárez, V.A. Martins dos Santos, M. Ferrer; Functional consequences of microbial shifts in the human gastrointestinal tract linked to antibiotic treatment and obesity; Gut Microbes, 4 (2013), pp. 306–315
- Heung et al., 2006 L.J. Heung, C. Luberto, M. Del Poeta; Role of sphingolipids in microbial pathogenesis; Infect. Immun., 74 (2006), pp. 28–39
- Hunt et al., 2011 P.W. Hunt, H.L. Cao, C. Muzoora, I. Sewanyana, J. Bennett, N. Emenyonu, A. Kembabazi, T.B. Neilands, D.R. Bangsberg, S.G. Deeks, J.N. Martin, et al.; Impact of CD8 + T-cell activation on CD4 + T-cell recovery and mortality in HIV-infected Ugandans initiating antiretroviral therapy; AIDS, 25 (2011), pp. 2123–2131
- Isanaka et al., 2009 S. Isanaka, C. Duggan, W.W. Fawzi; Patterns of postnatal growth in HIV-infected and HIV-exposed children; Nutr. Rev., 67 (2009), pp. 343–359
- Jagtap et al., 2013 P. Jagtap, J. Goslinga, J.A. Kooren, T. McGowan, M.S. Wroblewski, S.L. Seymour, T.J. Griffin; A two-step database search method improves sensitivity in peptide sequence matches for metaproteomics and proteogenomics studies; Proteomics, 13 (2013), pp. 1352–1357
- Kohli and Levy, 2009 P. Kohli, B.D. Levy; Resolvins and protectins: mediating solutions to inflammation; Br. J. Pharmacol., 158 (2009), pp. 960–971
- Kubacka et al., 2007 A. Kubacka, C. Serrano, M. Ferrer, H. Lünsdorf, P. Bielecki, M.L. Cerrada, M. Fernández-García, M. Fernández-García; High-performance dual-action polymer-TiO2 nanocomposite films via melting processing ; Nano Lett., 7 (2007), pp. 2529–2534
- Li et al., 2012 R.W. Li, S. Wu, W. Li, K. Navarro, R.D. Couch, D. Hill, J.F. Urban Jr.; Alterations in the porcine colon microbiota induced by the gastrointestinal nematode Trichuris suis; Infect. Immun., 80 (2012), pp. 2150–2157
- Marchesi et al., 2015 J.R. Marchesi, D.H. Adams, F. Fava, G.D. Hermes, G.M. Hirschfield, G. Hold, M.N. Quraishi, J. Kinross, H. Smidt, K.M. Tuohy, L.V. Thomas, E.G. Zoetendal, A. Hart, et al.; The gut microbiota and host health: a new clinical frontier; Gut, 65 (2015), pp. 330–339
- Masoodi et al., 2013 M. Masoodi, D.S. Pearl, M. Eiden, J.K. Shute, J.F. Brown, P.C. Calder, T.M. Trebble; Altered colonic mucosal polyunsaturated polyunsaturated fatty acid (PUFA) derived lipid mediators in ulcerative colitis: new insight into relationship with disease activity and pathophysiology; PLoS ONE, 8 (2013), p. e76532
- McGrath et al., 2015 N. McGrath, J.W. Eaton, M.L. Newell, V. Hosegood; Migration, sexual behaviour, and HIV risk: a general population cohort in rural South Africa; Lancet HIV, 2 (2015), pp. e252–e259
- McHardy et al., 2013 I.H. McHardy, X. Li, M. Tong, P. Ruegger, J. Jacobs, J. Borneman, P. Anton, J. Braun; HIV infection is associated with compositional and functional shifts in the rectal mucosal microbiota; Microbiome, 1 (2013), p. 26
- McPhee et al., 1996 F. McPhee, P.S. Caldera, G.W. Bemis, A.F. McDonagh, I.D. Kuntz, C.S. Craik; Bile pigments as HIV-1 protease inhibitors and their effects on HIV-1 viral maturation and infectivity in vitro; Biochem. J., 320 (1996), pp. 681–686
- Mesuere et al., 2015 B. Mesuere, G. Debyser, M. Aerts, B. Devreese, P. Vandamme, P. Dawyndt; The Unipept metaproteomics analysis pipeline; Proteomics, 15 (2015), pp. 1437–1442
- Mesuere et al., 2012 B. Mesuere, B. Devreese, G. Debyser, M. Aerts, P. Vandamme, P. Dawyndt; Unipept: tryptic peptide-based biodiversity analysis of metaproteome samples; J. Proteome Res., 11 (2012), pp. 5773–5780
- Moya and Ferrer, 2016 A. Moya, M. Ferrer; Functional redundancy-induced stability of gut microbiota subjected to disturbance; Trends Microbiol., 24 (2016), pp. 402–413
- Mutlu et al., 2014 E.A. Mutlu, A. Keshavarzian, J. Losurdo, G. Swanson, B. Siewe, C. Forsyth, A. French, P. Demarais, Y. Sun, L. Koenig, S. Cox, P. Engen, P. Chakradeo, R. Abbasi, A. Gorenz, C. Burns, A. Landay; A compositional look at the human gastrointestinal microbiome and immune activation parameters in HIV infected subjects; PLoS Pathog., 10 (2014), p. e1003829
- Noguera-Julian et al., 2016 M. Noguera-Julian, M. Rocafort, Y. Guillén, J. Rivera, M. Casadellà, P. Nowak, F. Hildebrand, G. Zeller, M. Parera, R. Bellido, C. Rodríguez, J. Carrillo, B. Mothe, J. Coll, I. Bravo, C. Estany, C. Herrero, J. Saz, G. Sirera, A. Torrela, J. Navarro, M. Crespo, C. Brander, E. Negredo, J. Blanco, F. Guarner, M.L. Calle, P. Bork, A. Sönnerborg, B. Clotet, R. Paredes; Gut microbiota linked to sexual preference and HIV infection; EBioMedicine, 5 (2016), pp. 135–146
- Nowak et al., 2015 P. Nowak, M. Troseid, E. Avershina, B. Barqasho, U. Neogi, K. Holm, J.R. Hov, K. Noyan, J. Vesterbacka, J. Svärd, K. Rudi, A. Sönnerborg, et al.; Gut microbiota diversity predicts immune status in HIV-1 infection; AIDS, 29 (2015), pp. 2409–2418
- Oh et al., 2011 S.F. Oh, P.S. Pillai, A. Recchiuti, R. Yang, C.N. Serhan; Pro-resolving actions and stereoselective biosynthesis of 18S E-series resolvins in human leukocytes and murine inflammation; J. Clin. Invest., 121 (2011), pp. 569–581
- Rao et al., 2011 J.S. Rao, H.W. Kim, M. Kellom, D. Greenstein, M. Chen, A.D. Kraft, G.J. Harry, S.I. Rapoport, M. Basselin; Increased neuroinflammatory and arachidonic acid cascade markers, and reduced synaptic proteins, in brain of HIV-1 transgenic rats; J. Neuroinflammation, 8 (2011), p. 101
- Rappsilber et al., 2003 J. Rappsilber, Y. Ishihama, M. Mann; Stop and go extraction tips for matrix-assisted laser desorption/ionization, nanoelectrospray, and LC/MS sample pretreatment in proteomics; Anal. Chem., 75 (2003), pp. 663–670
- Rappsilber et al., 2007 J. Rappsilber, M. Mann, Y. Ishihama; Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips; Nat. Protoc., 2 (2007), pp. 1896–1906
- Rojo et al., 2015a D. Rojo, M.J. Gosalbes, R. Ferrari, A.E. Pérez-Cobas, E. Hernández, R. Oltra, J. Buesa, A. Latorre, C. Barbas, M. Ferrer, A. Moya, et al.; Clostridium difficile heterogeneously impacts intestinal community architecture but drives stable metabolome responses ; ISME J., 9 (2015), pp. 2206–2220
- Rojo et al., 2015b D. Rojo, A. Hevia, R. Bargiela, P. López, A. Cuervo, S. González, A. Suárez, B. Sánchez, M. Martínez-Martínez, C. Milani, M. Ventura, C. Barbas, A. Moya, A. Suárez, A. Margolles, M. Ferrer, et al.; Ranking the impact of human health disorders on gut metabolism: systemic lupus erythematosus and obesity as study cases; Sci. Report., 5 (2015), p. 8310
- Rush et al., 2008 J.S. Rush, N. Gao, M.A. Lehrman, C.J. Waechter; Recycling of dolichyl monophosphate to the cytoplasmic leaflet of the endoplasmic reticulum after the cleavage of dolichyl pyrophosphate on the lumenal monolayer; J. Biol. Chem., 283 (2008), pp. 4087–4093
- Sandler and Douek, 2012 N.G. Sandler, D.C. Douek; Microbial translocation in HIV infection: causes, consequences and treatment opportunities; Nat. Rev. Microbiol., 10 (2012), pp. 655–666
- Schenk et al., 2001 B. Schenk, F. Fernandez, C.J. Waechter; The ins(ide) and out(side) of dolichyl phosphate biosynthesis and recycling in the endoplasmic reticulum; Glycobiology, 11 (2001), pp. 61R–70R
- Segata et al., 2011 N. Segata, J. Izard, L. Waldron, D. Gevers, L. Miropolsky, W.S. Garrett, C. Huttenhower; Metagenomic biomarker discovery and explanation; Genome Biol., 12 (2011), p. R60
- Serhan, 2014 C.N. Serhan; Pro-resolving lipid mediators are leads for resolution physiology; Nature, 510 (2014), pp. 92–101
- Serrano-Villar et al., 2014 S. Serrano-Villar, T. Sainz, S.A. Lee, P.W. Hunt, E. Sinclair, B.L. Shacklett, A.L. Ferre, T.L. Hayes, M. Somsouk, P.Y. Hsue, M.L. Van Natta, C.L. Meinert, M.M. Lederman, H. Hatano, V. Jain, Y. Huang, F.M. Hecht, J.N. Martin, J.M. McCune, S. Moreno, S.G. Deeks, et al.; HIV-infected individuals with low CD4/CD8 ratio despite effective antiretroviral therapy exhibit altered T cell subsets, heightened CD8 + T cell activation, and increased risk of non-AIDS morbidity and mortality; PLoS Pathog., 10 (2014), p. e1004078
- Severi et al., 2007 E. Severi, D.W. Hood, G.H. Thomas; Sialic acid utilization by bacterial pathogens; Microbiology, 153 (2007), pp. 2817–2822
- Shevchenko et al., 2006 A. Shevchenko, H. Tomas, J. Havlis, J.V. Olsen, M. Mann; In-gel digestion for mass spectrometric characterization of proteins and proteomes; Nat. Protoc., 1 (2006), pp. 2856–2860
- Somsouk et al., 2014 M. Somsouk, J.D. Estes, C. Deleage, R.M. Dunham, R. Albright, J.M. Inadomi, J.N. Martin, S.G. Deeks, J.M. McCune, P.W. Hunt; Gut epithelial barrier and systemic inflammation during chronic HIV infection; AIDS, 29 (2014), pp. 43–51
- Varki and Schauer, 2009 A. Varki, R. Schauer; Sialic Acids; A. Varki, R.D. Cummings, J.D. Esko, H.H. Freeze, P. Stanley, C.R. Bertozzi, G.W. Hart, M.E. Etzler (Eds.), Essentials of Glycobiology (second ed.), Cold Spring Harbor Laboratory Press, Cold Spring Harbor (NY) (2009) (Chapter 14)
- Vázquez-Castellanos et al., 2015 J.F. Vázquez-Castellanos, S. Serrano-Villar, A. Latorre, A. Artacho, M.L. Ferrús, N. Madrid, A. Vallejo, T. Sainz, J. Martínez-Botas, S. Ferrando-Martínez, M. Vera, F. Dronda, M. Leal, J. Del Romero, S. Moreno, V. Estrada, M.J. Gosalbes, A. Moya, et al.; Altered metabolism of gut microbiota contributes to chronic immune activation in HIV-infected individuals; Mucosal Immunol. , 8 (2015), pp. 760–772
- Vujkovic-Cvijin et al., 2013 I. Vujkovic-Cvijin, R.M. Dunham, S. Iwai, M.C. Maher, R.G. Albright, M.J. Broadhurst, R.D. Hernandez, M.M. Lederman, Y. Huang, M. Somsouk, S.G. Deeks, P.W. Hunt, S.V. Lynch, J.M. McCune, et al.; Dysbiosis of the gut microbiota is associated with HIV disease progression and tryptophan catabolism; Sci. Transl. Med., 5 (2013), p. 193ra91
- Vyboh et al., 2015 K. Vyboh, M.A. Jenabian, V. Mehraj, J.P. Routy; HIV and the gut microbiota, partners in crime: breaking the vicious cycle to unearth new therapeutic targets; J. Immunol. Res., 2015 (2015), p. 614127
- Wegiel and Otterbein, 2012 B. Wegiel, L.E. Otterbein; Go green: the anti-inflammatory effects of biliverdin reductase; Front. Pharmacol., 3 (2012), p. 47
- Wright et al., 2004 E.M. Wright, D.D. Loo, B.A. Hirayama, E. Turk; Surprising versatility of Na+ -glucose cotransporters: SLC5 ; Physiology (Bethesda), 19 (2004), pp. 370–376
Document information
Published on 06/04/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?