Abstract
Objective
This was a prospective, single-center study evaluating the efficacy and cost-effectiveness of early ambulation (within 30 min) following femoral artery closure with the ProGlide® suture-mediated vascular closure device (PD) in patients undergoing diagnostic cardiac catheterization compared with manual compression.
Background
It is unclear whether early ambulation with ProGlide is safe or is associated with patient satisfaction and cost savings as compared with manual compression (MC).
Methods and results
Inclusion criteria were met in 170 patients (85 PD and 85 MC patients). Patients ambulated 20 ft. within 30 min (PD) or after the requisite 4 h recumbent time (MC) if feasible. Primary endpoint was time-to-ambulation (TTA) following device closure. We also directly compared the safety of closure, times-to-hemostasis (TTH), -ambulation (TTA) and -discharge (TTD) with MC and, using a fully allocated cost model, performed cost analysis for both strategies. Multivariate analysis was used to determine predictors of patient satisfaction. The primary endpoint of safe, early ambulation was achieved following closure (mean of 27.1 ± 14.9 min; 95% confidence interval [CI] 25.2–30.2). Predictors of patient satisfaction in the PD group were absence of pain during closure, decreased TTA, and drastic reductions in TTD; the latter contributed indirectly to significant cost savings in the PD group (1250.3 ± 146.4 vs. 2248.1 ± 910.2 dollars, respectively; P < 0.001) and incremental cost savings by strategy also favored closure over MC ($84,807).
Conclusions
ProGlide is safe and effective for femoral artery closure in patients who ambulate within 30 min after cardiac catheterization; translating into improved patient satisfaction and substantial cost savings.
Abbreviations
VCD, vascular closure devices;MC, manual compression;PD, Perclose ProGlide device;TTH, time-to-hemostasis;TTA, time-to-ambulation;TTD, time-to-discharge
Keywords
ProGlide;Vascular closure;Cost;Cardiac catheterization
1. Introduction
Femoral artery cannulation continues to be the predominant route for vascular access in coronary and structural heart interventions in the United States (US) despite the increasing use of radial artery access [1]; [2] ; [3]. However, femoral artery access site bleeding complications carry an increased morbidity and mortality as compared to other access routes [4]. Therefore, proficiency with arterial puncture and closure (either manual or device-related) is crucial and plays a vital role in the safety of diagnostic cardiac catheterization (cath). Minimizing femoral artery access site complications remains the rationale for the introduction and widespread use of vascular closure devices (VCD). However, with the wide variety of devices available each with unique mechanisms for vessel closure, the safety and efficiency of access site management using VCDs remains controversial. Large meta-analyses, registry and single-center studies provide conflicting data regarding VCD-related vascular complication event rates as compared with manual compression (MC) [5]; [6]; [7]; [8]; [9] ; [10]. Of the major advantages of VCDs, reduced time-to-ambulation (TTA), when associated with no increase in vascular complications [11], is among the most desirable in terms of patient satisfaction and cost-effectiveness [12] ; [13]. A wide variety of closure devices are currently available for use, each classified based on their method of closure; either collagen or procoagulant-based (e.g., Angioseal®, St. Jude Medical, St. Paul, MN), clip/staple-based (e.g., Starclose®, Abbott Scientific, Abbott Park, IL) or suture-mediated (e.g., Perclose ProGlide® and ProStar®, Abbott Scientific, Abbott Park, IL) [14].
Although data are available for the use of older versions of the suture-mediated VCDs in the setting of percutaneous coronary intervention, there are no data directly comparing femoral vessel closure using ProGlide (PD; the most recent generation in the Perclose device series) with manual compression, specifically following diagnostic cardiac cath. Moreover, whether patients can be safely ambulated and discharged early following femoral artery closure has only been demonstrated once with the StarClose VCD [12]. To date, this has not been prospectively evaluated with the Perclose devices. Here, we assessed prospectively, the safety and efficacy of early ambulation following use of ProGlide in patients undergoing diagnostic cardiac catheterization as compared to MC. We further assessed patient satisfaction and the cost-effectiveness of ProGlide-mediated vessel closure versus MC.
2. Methods
2.1. Patient population and study design
This study was a prospective study approved by the institutional review board at the University of Louisville School of Medicine (Louisville, Kentucky) and conducted at a single, high-volume quaternary-care center in Louisville Kentucky. Baseline patient characteristics are shown in Table 1. Two hundred and twelve consecutive patients presenting from April to June (2012) for elective diagnostic cardiac cath using either the right or left femoral arterial approach signed an informed consent. One hundred and seventy of these patients met entry criteria (Table 2) and were enrolled. Screen failures were excluded based on clinical and/or angiographic criteria. Catheterizations were performed under conscious sedation, using 5 or 6 French AvantiPlus® (Cordis, Bridgewater, NJ) or Pinnacle® (Terumo, Somerset, NJ) sheaths. We compared manual compression (85 patients) to vessel closure using the ProGlide suture-mediated VCD (PD) (85 patients). Of the six board-certified interventional cardiologists deploying the PD in the study, all had greater than three years (range 3.2–9 years) of experience using Perclose devices.
| Demographic characteristics | |||
|---|---|---|---|
| ProGlide | Manual compression | P | |
| n = 85 | n = 85 | ||
| Age — year | |||
| Mean | 60 ± 12 | 59 ± 10 | NS |
| Male sex — no. (%) | 73 (86) | 76 (89) | NS |
| Race — no. (%) | |||
| White | 64 (75) | 66 (78) | NS |
| Black | 20 (24) | 17 (20) | NS |
| Asian | 0 (0) | 2 (2) | NS |
| Other | 1 (1) | 0 (0) | NS |
| Body mass index (BMI)a | |||
| Mean | 27 ± 3.0 | 25 ± 4.0 | NS |
| Hypertension — no. (%) | 70 (82) | 64 (75) | NS |
| Diabetes — no. (%) | 36 (42) | 37 (44) | NS |
| Dyslipidemia — no. (%) | 54 (63) | 61 (72) | NS |
| Smoking — no. (%) | 66 (78) | 65 (77) | NS |
| Current | 34 (52) | 39 (60) | NS |
| Previous | 32 (48) | 26 (40) | NS |
| Family history CAD — no. (%) | 30 (35) | 21 (25) | < 0.05 |
| Procedure characteristics | |||
| Pre-procedural angiography — no. (%) | 85 (100) | 85 (100) | NS |
| Retrograde puncture — no. (%) | 85 (100) | 85 (100) | NS |
| Right groin approach — no. (%) | 78 (92) | 83 (98) | NS |
| Vessel diameter ≥ 5.0 mm — no. (%) | 85 (100) | 85 (100) | NS |
Continuous data are presented as means ± SD; categorical data are presented as counts (percentages).
a. Body mass index is the weight in kilograms divided by the square of the height in meters.
| Major inclusion criteria |
|---|
| Candidates for elective cardiac diagnostic catheterization performed percutaneously via the right or left femoral arterial approach |
| Vessel size > 5 mm by visual estimate |
| Access using 5F or 6F introducer sheath |
| Confirmed angiographic absence of current or previously treated significant (≥ 50% stenosis) femoral artery atherosclerosis |
| Lack of obvious fluoroscopic evidence of significant femoral vessel calcification |
| Confirmed post-procedural puncture site between the common femoral artery bifurcation and inferior border of the inferior epigastric artery |
| Major exclusion criteria |
| Vessel size < 5 mm by visual estimate (or plaque burden resulting in lumen < 5 mm) |
| Inability to control post-procedural hypertension in the cath lab (systolic ≥ 180 mm Hg, diastolic ≥ 100 mm Hg) |
| Inability to ambulate 20 ft due to co-morbidity or functional limitation |
| Access site complications prior to ProGlide deployment |
2.2. Study device description
The ProGlide is the fifth generation of the single-use 6F disposable suture-mediated Perclose devices that delivers two needles and a single suture through the arterial wall (adventitia to lumen) for closure of the femoral arteriotomy following percutaneous diagnostic or interventional cath or peripheral procedures that utilize 5–21F sheaths. The ProGlide single device delivery system is for use with any 0.035 in. guidewire and contains a “knot-pusher”. Further details of its use have been previously reported [14].
2.3. Procedural definitions and study variables
All patients underwent pre-procedural fluoroscopy-guided identification of the mid-femoral head to determine the puncture site. Post-cardiac cath femoral angiography was performed on all patients. In patients who met inclusion criteria (Table 2), hemostasis was achieved either by (i) placement of the PD with a check for hemostasis made and time recorded, or (ii) following transfer to holding for manual compression. If immediate hemostasis was not achieved manual compression was performed for 3 min and additional adjunctive compression followed as necessary; not to exceed 5 min (min) or device success was not achieved. If additional time for manual compression was required, this time was recorded. In the MC group, manual compression was performed by experienced cath lab nurses with the application of a Neptune® hemostasis Pad(s) (TZ Medical Inc., Portland, OR) and the holding time was determined by the French size of the sheath used; with 3 min of compression per French size being the standard. If after 15 (5F) or 18 (6F) min, hemostasis was not achieved, manual pressure was held for additional 5 min increments until hemostasis was achieved. The time in min from removal of sheath to when no compression was required to control bleeding at the access site was defined as time-to-hemostasis (TTH). Mechanical compression devices were not used as an adjunct to achieve hemostasis in any patient.
Once hemostasis was achieved, patients were ambulated. Prior to ambulation, the access site was assessed for bleeding and/or complications. The primary endpoint for the study was mean TTA following PD artery closure. Early ambulation was defined as TTA ≤ 30 min in the PD patients and the requisite recumbent time for MC patients was 4 h from TTH. The 4 h limit for the MC group is an institutional policy and consistent with previous reports [15] ; [16] and was followed to ensure congruence between groups when assessing complication rates. Patients in the device group underwent a ‘challenge test’ consisting of simultaneous head and leg lifts to assess the adequacy of hemostasis. Those in the PD group who did not achieve hemostasis within 5 min of device deployment or who converted to manual compression were ambulated at the discretion of the interventionalist to ensure patient safety. Continued hemostasis was assessed while patients were ambulating. Time-to-ambulation (TTA) was defined as the time in min from hemostasis to the time that the patient ambulated 20 ft without assistance or re-bleeding. Patients with persistent access site issues were managed and those methods were recorded.
Patients were discharged once satisfactory ambulation was achieved, access sites were appropriately managed, and access site pain was tolerable. Post-deployment time-to-discharge (TTD) was defined as the elapsed time from achievement of sheath removal to the time when the patient was medically able to be discharged.
Procedural success and closure method complications were recorded (Table 3). Surgical complications specific to the closure device were as follows: rupture or perforation of femoral artery, need for surgical removal of device needle(s) and capture of vessel posterior wall with resultant vessel occlusion.
| Variable | |||
|---|---|---|---|
| n = 85 | n = 85 | P | |
| ProGlide | MC | ||
| Device success (%)a | 85 (100) | (NA) | NA |
| Procedural success (%)b | 85 (100) | 82 (96) | 0.12 |
| Major vascular site complications | |||
| Need for surgical vascular repair | 0 (0) | 0 (0) | NA |
| Ultrasound-guided compression, stent graft deployment or catheter-based embolization | 0 (0) | 0 (0) | NA |
| Ipsilateral limb ischemia (decreased or absent distal Doppler signal, or poor distal run-off by lower extremity angiogram) | 0 (0) | 0 (0) | NA |
| Transfusion of any blood products required for access site-related bleeding (Hb drop ≥ 3 g/dL) | 0 (0) | 3 (3.5) | 0.25 |
| Overall (major) | |||
| Minor vascular site complications | |||
| Non-surgical access site complications | 0 (0) | 0 (0) | NA |
| Access site hematoma < 6 cm | 2 (2.4) | 1 (1.2) | 0.56 |
| Access site hematoma > 6 cm | 0 (0) | 4 (4.7) | < 0.05 |
| Access site persistent oozing | 1 (1.2) | 2 (2.4) | 0.56 |
| Femoral bruit | 0 (0) | 0 (0) | NA |
| Pulsatile mass | 0 (0) | 0 (0) | NA |
| Hemoglobin drop (above 3 g/dL) without need for transfusion | 0 (0) | 0 (0) | NA |
| US guided compression for pseudoaneurysm | 0 (0) | 0 (0) | NA |
| Overall (minor) | 3 (3.5) | 7 (8.2) | 0.33 |
| Overall complications | 3 (3.5) | 10 (11.8) | 0.08 |
Data are presented as counts (percentages). MC = manual compression.
a. Defined as freedom from major vascular complications following achievement of complete hemostasis using the Perclose alone or with adjunctive compression of ≤ 5 min.
b. Defined as freedom from major vascular complications following achievement of complete hemostasis using any method.
Patients overall satisfaction was evaluated by questionnaire using a previously validated scoring method that included the Visual Analog Scale (VAS-11) point system [12]; [17] ; [18] and addressed the following: (i) intensity of access site pain during closure and prior to discharge; (ii) TTH; (iii) TTA; (iv) TTD; and (v) overall general satisfaction. Pain intensity was specifically assessed using a Visual Analog Scale (VAS-11) point system that ranked the level of patient discomfort at access site was based on a pain scale of 0–11 (0 for no pain, 11 for pain being as bad as imaginable for this procedure) taken during two distinct time points: during closure and prior to discharge. Classification of pain was as follows: no pain (0), mild pain (1–3), mild to moderate pain (4–7) moderate to severe pain (8–10) and severe pain (11). To determine overall patient satisfaction, patients were asked to rank their experience based on a satisfaction scale of 0–11 (0 for the worst experience imaginable and 11 for the best experience imaginable for this procedure). Classification of overall satisfaction with their procedural experience was as follows: worst (0), very bad (1–2), bad (3–4), indifferent (5–6), good (7–8), very good (9–10), and best imaginable (11). Those who had previous cardiac caths (from any access site) compared the present to previous procedures.
Cost analysis was performed using a fully allocated cost model [19]. Total direct costs (costs of the device, infrastructure, and personnel) were compared between both groups for the year 2012 (reported in US dollars). The analysis was based on the actual institutional cost of equipment used (e.g., closure device and/or hemostatic pad) and the institutional costs for post-procedural management (which included individual nursing costs per patient treated and cost for hospitalization). Hospitalization costs were determined prospectively. In-hospital unit costs and length of stay costs were adjusted to reflect ancillary imaging and blood bank needs (e.g., ultrasound and red blood cell transfusion) and also included fixed costs obtained from our hospital cost model for a 24–72 h period. For a national comparison of per patient costs, refer to http://www.ahaonlinestore.com/ProductDisplay.asp?ProductID=637. The effect of recumbent time prior to ambulation was further examined using sensitivity analysis to determine whether a cost advantage existed between protocols (PD vs. manual compression).
2.4. Study endpoints
The primary endpoint of this study was mean TTA following PD closure; with ‘early ambulation’ in the PD arm defined as TTA ≤ 30 min without re-bleeding. Our secondary endpoints were TTH and TTD along with analysis of patient safety (determined by procedural success and access site complications), satisfaction (determined by patient questionnaire after hemostasis and prior to discharge), and the cost-effectiveness of the strategy used. Safety of closure method used was measured by procedural (device) success, and procedure-related major and minor complications. Variables used to measure efficacy of closure method were the TTH, TTA, and TTD.
2.5. Statistical analysis
Categorical data are expressed as frequency counts and percentages and continuous data are expressed as mean ± SD or as median and interquartile ranges (IQR) when variables lacked a normal distribution. Categorical data were compared using χ2 or Fishers exact tests. Continuous data were compared between groups using two-tailed Students t-tests or Wilcoxon rank-sum test as appropriate. Univariate and multivariate analyses were performed using stepwise logistic regression to identify factors associated with an overall satisfaction rating of ≥ 9. Covariates with P < 0.1 in the univariate analysis were entered into the multivariate model. Odds ratios (ORs) and 95% confidence intervals (CIs) were reported for continuous and categorical data. P < 0.05 was considered statistically significant. All statistical analyses were performed using the SPSS (version 19) statistical software (SPSS, Inc., Chicago, Illinois).
3. Results
3.1. Subject enrollment
A total of 212 consecutive patients undergoing diagnostic cardiac cath were screened and 170 were not excluded by angiographic or clinical criteria and were enrolled at a single center (42) excluded based on criteria outline in Table 2: hypertension (8), ambulation (2), calcification (12), stenosis (15), and vessel size (5). Eighty-five patients received standard MC and 85 received percutaneous closure with the ProGlide suture-mediated VCD. Baseline characteristics are listed in Table 1. Overall, each group was similar demographically and procedurally.
3.2. Device safety
The overall safety and performance of the procedure was measured by procedural success and device success (Table 3). The PD was successfully deployed in 100% of patients attempted; one patient required rewiring and an additional device due to suture breakage of the first device. Manual compression was successful in all but three patients (Table 3). In the PD group one patient experienced persistent oozing from the access site and two had small hematomas (< 6 cm). In the MC group, two patients experienced persistent oozing and four had large hematomas (> 6 cm). Taken together, these minor complications were not significantly different between the 2 groups (Table 3, PD = 3.5% and MC = 8.2%; P = 0.33). There were no major complications in the PD group and no deaths or retroperitoneal bleeds in either group. Three patients in the MC group required blood transfusions due to significant access site bleeding (Hb drop ≥ 3 g/dL) which was not seen in the PD group; yet, this difference was merely a trend (Table 3, PD = 0.0% and MC = 3.5%; P = 0.25).
3.3. Closure method efficacy
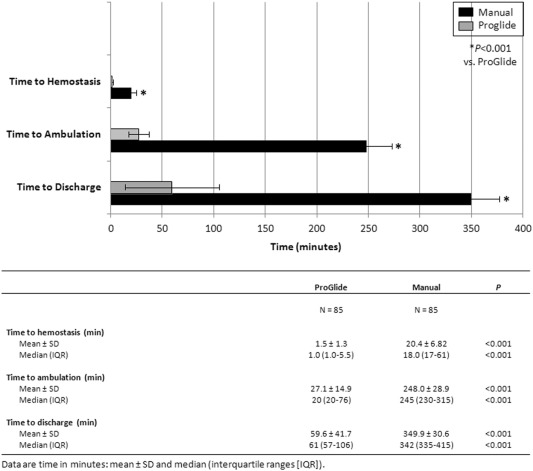
Patients with major complications requiring hospitalization were excluded from this analysis (3 patients in the MC group). As depicted in Table 4, our primary endpoint of TTA was met in the PD group with the majority of patients ambulating early following PD closure (mean TTA 27.1 ± 14.9 min; 95% confidence interval [CI] 25.2–30.2). As expected, TTA was reduced substantially in the PD group as compared to MC (mean TTA of 27.1 ± 14.9 min and 4.13 h ± 28.9 min, respectively; Fig. 1, P < 0.001). Time-to-hemostasis was compared between the two groups; PD patients had a significantly lower TTH (Fig. 1, P < 0.001). Post-deployment TTD was drastically reduced in the PD group with a mean TTD of 59.6 (± 41.7) min compared to MC (mean of 5.8 h ± 30.6 min; Fig. 1, P < 0.001).
| Variable | ProGlide Group | |
|---|---|---|
| n = 85 | 95% CI | |
| Time-to-ambulation (min) | ||
| Mean ± SD | 27.1 ± 14.9 | 25.2–30.2 |
| Median (IQR) | 20 (20–76) | 19.5–24.6 |
Data are time in minutes: mean ± SD and median (interquartile ranges [IQR]). CI = confidence interval.
|
|
|
Fig. 1. Study endpoints. Upper panel: data are mean ± SD. |
3.4. Patient satisfaction
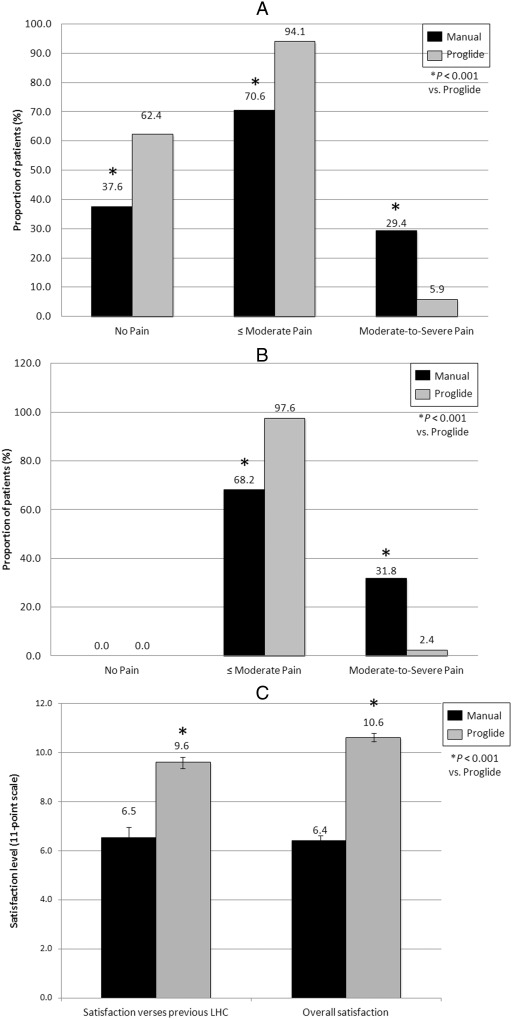
Overall patient satisfaction including procedure-related pain intensity was assessed peri-procedurally. Mean pain scores on a scale of 0–11 during closure/compression for PD and MC groups were 1.4 ± 2.11 and 4.3 ± 2.51, respectively (P < 0.05). During closure/manual compression, patients reporting a pain scale of “0” (no pain) was 62.4% (53/85) in the PD group, and 37.6% (32/85) in the MC group (P < 0.001), with 94.1% (80/85) of PD and 70.6% (60/85) of the MC patients reporting pain scales ≤ 7 (≤ moderate pain, P < 0.001); only 5.9% (5/85) of PD patients reported moderate-to-severe (8–11) compared to 29.4% (25/85) of MC patients (Fig. 2A, P < 0.001). Prior to discharge, 97.6% (83/85) of PD and 68.2% (58/85) of MC patients reported pain levels ≤ moderate (P < 0.001); only 2.4% (2/85) of PD patients reported moderate-to-severe pain compare to 31.8% (27/85) of MC patients (Fig. 2B, P < 0.001). No patients in either group were completely free of pain or discomfort at discharge (mean pain scores at discharge were 0.53 ± 0.93 and 4.1 ± 2.63 for PD and MC groups, respectively) (Fig. 2B, P < 0.05).
|
|
|
Fig. 2. Procedural patient satisfaction. Procedural discomfort was assessed following hemostasis (A) and prior to discharge (B) achieved using either suture-mediated vessel closure (ProGlide) or manual compression (Manual). Data are frequency counts by questionnaire expressed as percentages. (C) Overall patient satisfaction was assessed prior to discharge on a scale of 0–11. Data are mean ± SD. |
Forty-five percent of PD patients and 55% of MC patients underwent previous cardiac cath (P = 0.22). Patients in the PD group reported a significantly higher satisfaction with their procedure compared previous caths (9.6 ± 0.5 versus 6.5 ± 0.9, respectively; P < 0.001, Fig. 2C). Likewise, patients undergoing closure reported a significantly higher overall satisfaction with their procedure compared to patients undergoing MC (10.6 ± 0.3 versus 6.4 ± 0.4, respectively; P < 0.001, Fig. 2C).
Multivariate analysis demonstrated that patients in the PD group who reported a ‘very good’ or better procedural experience (≥ 9) were ~ 5-fold more likely to report no pain during closure (Table 5; odds ratio [OR] 5.11; 95% CI 3.12–8.35; P < 0.01). Both decreased TTA (OR 3.18; 95% CI 1.34–5.36; P < 0.01) and TTD (OR 4.61; 95% CI 2.54–6.21; P < 0.01) also predicted patients overall satisfaction in favor of PD over MC (Table 5). In both MC (OR 1.01; 95% CI 0.98–1.03; P = 0.04) and PD groups (OR 2.01; 95% CI 1.10–3.67; P = 0.02), ≤ moderate pain was more often associated with increased overall patient satisfaction (Table 5).
| Variable | Odds ratio | 95% CI | P |
|---|---|---|---|
| Pain intensity (during closure/compression) | |||
| MC | |||
| No pain | 1.06 | 0.96–1.89 | 0.58 |
| ≤ moderate pain | 1.01 | 0.98–1.03 | 0.04 |
| PD | |||
| No pain | 5.11 | 3.12–8.35 | < 0.01 |
| ≤ moderate pain | 2.01 | 1.10–3.67 | 0.02 |
| Time savings | |||
| MC | |||
| Time-to-ambulation (per minute decrease) | 1.13 | 0.51–2.42 | 0.74 |
| Time-to-discharge (per minute decrease) | 1.07 | 0.83–1.62 | 0.62 |
| PD | |||
| Time-to-ambulation (per minute decrease) | 3.18 | 1.34–5.36 | < 0.01 |
| Time-to-discharge (per minute decrease) | 4.61 | 2.54–6.21 | < 0.01 |
Manual compression (MC), ProGlide device (PD) and confidence interval (CI). Other covariates studied with P > 0.1 in the univariate analysis were: presence of hematoma (≤ 6 or ≥ 6 cm); no pain, ≤ moderate pain, and ≤ moderate-to-severe pain prior to discharge; and time-to-hemostasis.
3.5. Cost analysis
Table 6 compares the institutional cost per closure strategy. With the exception of the mean additional cost of ProGlide (278.2 ± 77.4 dollars) use per-patient versus the mean cost of Neptune Pads used per-patient (41.6 ± 22.4 dollars), per-patient institutional costs were significantly reduced when the ProGlide closure system was used (P < 0.001). When considered in the absence of hospitalizations (100% of which occurred in the MC group; P < 0.001), savings in catheterization lab holding and nursing expense accounted for the majority of the average total cost savings (117.7% and 118.9% reductions in average cost, respectively; P < 0.001). When compared to MC, a cost savings of almost $1000 per-patient was observed in PD group (Table 6; 1250.3 ± 146.4 versus 2248.1 ± 910.2 dollars, respectively; P < 0.001). The incremental cost savings by closure strategy also favored the use of ProGlide over MC in this study (Table 6; $106,278.70 versus $191,086.50). Sensitivity analysis was performed (comparing the cost of bed-time [TTA] between groups) to better understand how varying the mandatory recumbent times in the MC group, driven by our institutional protocol, might impact the demonstrated cost savings. We found the PD strategy to be cost-effective so long as TTD in the MC group was ≥ 64 min, as it was in all MC cases. As such, even when shorter ambulation times are considered (e.g., 2 h, the case in many US labs), our cost analysis effectively estimates the cost benefit of early ambulation when the PD strategy is utilized.
| Hospital costs | ProGlide | Manual | P |
|---|---|---|---|
| n = 85 | n = 85 | ||
| Total procedural cost/patient, without hemostasis product — $ | |||
| Mean ± SD | 564.5 ± 132.3 | 553.7 ± 121.0 | 0.37 |
| Hemostasis cost/patient — $a | |||
| Mean ± SD | 278.2 ± 77.4 | 41.6 ± 22.4 | < 0.001 |
| Post-procedural cath lab holding/patient — $b | |||
| Mean ± SD | 308.5 ± 78.8 | 1190.8 ± 333.6 | < 0.001 |
| Total adjusted nursing costs/patient — $c | |||
| Mean ± SD | 99.1 ± 41.1 | 389.5 ± 98.2 | < 0.001 |
| Hospital adjusted in-patient expenses/patient — intermediate level– $d | |||
| Mean ± SD | 0.0 | 2052.6 ± 250.2 | < 0.001 |
| Total cost/patient — $ | |||
| Mean ± SD | 1250.3 ± 146.4 | 2248.1 ± 910.2 | < 0.001 |
| (Incremental savings — $) | (983.6) | ||
| Total cost/strategy — $ | 106,278.70 | 191,086.50 | |
| (Incremental savings — $) | (84,807.80) |
All data are U.S. dollars ($): mean ± SD.
a. All patients in the device group (ProGlide) received a ProGlide device ($197.9/device) ± Neptune® Pads ($35.2/pad) while all in the manual compression group (Manual) received only Neptune® Pad(s).
b. Based on an institutional post-procedural cath lab holding cost per hour of $205 per patient.
c. Based on an adjusted nursing cost per hour of $67 per patient (extrapolated from adjusted annualized nursing salary per nurse per year).
d. Based on hospital adjusted expenses per in-patient day of $1551.
4. Discussion
The primary objective of this single-center prospective study was to determine whether early ambulation following femoral artery closure using the newest version of the single-use 6F Perclose suture-mediated ProGlide VCD was safe. To date, no study has compared the efficacy of early ambulation (within 30 min of closure) using Perclose with manual compression in patients undergoing diagnostic cardiac catheterization. Nor has any study assessed the cost-effectiveness of this strategy. The major findings of our study were the following:
- To our knowledge, ours is the only prospective study using the Perclose system to report safe early ambulation within 30 min of closure. We also describe the earliest reported discharge time of any femoral VCD following diagnostic cardiac cath (~ 60 min).
- Predictors for greater patient satisfaction associated with the use of the ProGlide were (i) a greater number of patients undergoing painless vessel closure, (ii) very early TTA, and (iii) timely TTD.
- Despite the additional up-front cost of the ProGlide device compared to MC, significant reductions in cath lab and nursing time spent per-patient drastically reduced the total direct costs of the procedure, primarily due to very early discharge; driven further by lack of need for hospitalization in the PD group.
Since the early days of cardiac catheterization, the femoral artery has been the most widely used site for percutaneous access. Increased access site bleeding and vascular complications were the driving forces for the eventual elimination or miniaturization of the requisite bulky equipment used during these procedures. Unfortunately, since the subsequent reduction in sheath sizes (4, 5 and 6 French) did not translate into reductions in bleeding complications or patient discomfort [16], alternative methods for access site management emerged. Accordingly, the current era of cardiac catheterization has witnessed a resurgence of transradial artery access which has since been shown to reduce vascular complications and major access site bleeding [1] ; [20]. As mechanical compression devices demonstrated no clinically appreciable advantages in terms of femoral access site management, the inevitable corollary was the development of VCDs in an effort to eliminate or limit the risk, patient discomfort and prolonged costly immobilization associated with manual compression [12] ; [14].
4.1. Safety and efficacy
The most recent comprehensive meta-analysis collectively comparing all three commercially available VCDs (Angioseal, Perclose, and Starclose) showed no difference in complications rates when compared to MC alone [8]. However, analysis of the suture-mediated VCD Perclose relative to other VCDs confirmed its safety particularly when used in diagnostic cases, suggesting that safety of VCDs is not a class effect [8] ; [10]. Our study was not designed to simply verify the safety of the ProGlide suture-mediated VCD relative to MC in patients who underwent diagnostic cardiac catheterization. Instead we sought to thoroughly evaluate the efficiency of ProGlide-mediated femoral artery closure in terms of safe, early TTA and TTD and further assess patient experience and the economic value of each hemostasis strategy. Previous prospective trials evaluating the safety of VCDs versus manual compression during invasive cardiac procedures reported similar procedural success and complication rates [15]; [17]; [21]; [22]; [23] ; [24]. However, to our knowledge this is the first prospective study using a Perclose VCD to clearly demonstrate the safety of early ambulation (within 30 min) following any type of cardiac catheterization. With the exception of a small subset of patients (2.2%) in a single study who were ambulated in the cath lab following Perclose use [21], only one other study has reported early ambulation times (within 30 min) following the use of a VCD after cardiac cath; in this case, following use of the Starclose device [12]. In the former study, no specific effort was made to ambulate patients early. Instead, closure success determined ambulation times and TTA was left to the discretion of the nursing team with the overwhelming majority of patients (~ 80%) ambulating between 2 and 6 h [21].
4.2. Satisfaction
Given the discomfort related the mechanical compression of the femoral artery following cardiac catheterization and the requisite recumbent time required for effective manual compression, previous investigators have attempted to qualify and quantify patient satisfaction associated with various methods of groin hemostasis [12]; [13]; [15] ; [17]. Whereas Baim et al. reported less discomfort in patients treated with the older Techstar® and ProstarPlus suture-mediated systems in the STAND trials [15], others found no difference in patient satisfaction when either the older or newer ProGlide suture-mediated systems were compared to MC [13] ; [17]. In one of these studies evaluation of patient satisfaction was assessed only after 30 days, by telephone, and in an undisclosed number of patients [17]; neither study was restricted to patients undergoing diagnostic catheterization. An important distinction, considering that a major criterion reportedly driving patient satisfaction was TTA: (i) which ranged from 2 to 4 h following diagnostic cath; (ii) was greater than 7 h following intervention; and (iii) with no stratification of patient satisfaction based on procedure type attempted. Greater patient satisfaction and reduced discomfort in our study following the use of ProGlide were primarily driven by the absence of pain during closure in the vast majority of patients studied and by the substantial reduction in TTA and TTD as compared patients who underwent MC.
4.3. Cost savings
In addition to impacting patient satisfaction, the economic consequences of delaying ambulation were clearly illustrated in this study translated into a delay in discharge. We report the earliest TTD to date which proved to be the most economically relevant endpoint studied. Here, we assessed cost using a fully allocated cost model without consideration of accounting costs or “charges”. Our cost analysis uncovered significant cost savings on a per-patient basis driven primarily by the reduction in time spent at our institution. This translated into tens of thousands of dollars saved when ProGlide was utilized as a hemostasis strategy versus manual compression. Sensitivity analysis further demonstrated that the PD strategy remains cost-effective relative to MC within most clinically relevant TTA or TTD timeframes.
4.4. Limitations
Given that this is a single-center non-randomized trial, there are inherent limitations. First, the lack of randomization introduces the possibility of selection bias particularly since only patients who were clinically and anatomically suitable for closure underwent PD; though randomization is difficult in cohorts where post-procedural TTA is controversial. Second, our single-center experience allows only for analysis based on our routine clinical practice. Third, our study reported only in-hospital outcomes without out-patient follow-up; thus, conclusions about long-term consequences of closure strategy cannot be drawn. Finally, given that access fees and charges always tend to be higher in insured patients (albeit a large portion of our population) it is possible that we grossly underestimated to cost savings reported in our study [19].
4.5. Conclusions
The results of this prospective trial comparing ProGlide VCD to MC indicate that a strategy of femoral artery closure using ProGlide is not only safe but effective when patients ambulate within 30 min following diagnostic cardiac catheterization. These data clearly demonstrate that procedural satisfaction associated with ProGlide suture-mediated vessel closure is driven by absence of pain during closure, early TTA and very early TTD; the latter being the primary influence behind the economic benefit to using the ProGlide strategy.
Disclosures
None of the authors have any conflicts of interest.
References
- [1] M. Brueck, et al.; A randomized comparison of transradial versus transfemoral approach for coronary angiography and angioplasty; JACC Cardiovasc. Interv., 2 (2009), pp. 1047–1054
- [2] G.J. Dehmer, et al.; A contemporary view of diagnostic cardiac catheterization and percutaneous coronary intervention in the United States: a report from the CathPCI Registry of the National Cardiovascular Data Registry, 2010 through June 2011; J. Am. Coll. Cardiol., 60 (2004), pp. 2017–2031
- [3] M.S. Lee, et al.; Minimizing femoral artery access complications during percutaneous coronary intervention: a comprehensive review; Catheter. Cardiovasc. Interv., 84 (2009), pp. 62–69
- [4] S.V. Rao, et al.; Relationship of blood transfusion and clinical outcomes in patients with acute coronary syndromes; JAMA, 292 (2004), pp. 1555–1562
- [5] B. Ahmed, et al.; Significantly improved vascular complications among women undergoing percutaneous coronary intervention: a report from the Northern New England Percutaneous Coronary Intervention Registry; Circ. Cardiovasc. Interv., 2 (2009), pp. 423–429
- [6] N. Arora, et al.; A propensity analysis of the risk of vascular complications after cardiac catheterization procedures with the use of vascular closure devices; Am. Heart J., 153 (2007), pp. 606–611
- [7] B.J. Doyle, et al.; Major femoral bleeding complications after percutaneous coronary intervention: incidence, predictors, and impact on long-term survival among 17,901 patients treated at the Mayo Clinic from 1994 to 2005; JACC Cardiovasc. Interv., 1 (2008), pp. 202–209
- [8] E. Nikolsky, et al.; Vascular complications associated with arteriotomy closure devices in patients undergoing percutaneous coronary procedures: a meta-analysis; J. Am. Coll. Cardiol., 44 (2004), pp. 1200–1209
- [9] S. Trimarchi, et al.; Retroperitoneal hematoma after percutaneous coronary intervention: prevalence, risk factors, management, outcomes, and predictors of mortality: a report from the BMC2 (Blue Cross Blue Shield of Michigan Cardiovascular Consortium) registry; JACC Cardiovasc Interv., 3 (2010), pp. 845–850
- [10] P.T. Vaitkus, et al.; A meta-analysis of percutaneous vascular closure devices after diagnostic catheterization and percutaneous coronary intervention; J. Invasive Cardiol., 16 (2004), pp. 243–246
- [11] T.A. Sanborn, et al.; Impact of femoral vascular closure devices and antithrombotic therapy on access site bleeding in acute coronary syndromes: The Acute Catheterization and Urgent Intervention Triage Strategy (ACUITY) trial.; Circ Cardiovasc Interv., 3 (2008), pp. 57–62
- [12] M.N. Burke, J. Hermiller, M.R. Jaff; StarClose vascular closure system (VCS) is safe and effective in patients who ambulate early following successful femoral artery access closure—results from the RISE clinical trial; Catheter Cardiovasc Interv., 80 (2007), pp. 45–52
- [13] J.L. Martin, et al.; A randomized trial comparing compression, Perclose Proglide and Angio-seal VIP for arterial closure following percutaneous coronary intervention: the CAP trial; Catheter. Cardiovasc. Interv., 71 (2008), pp. 1–5
- [14] H.L. Dauerman, R.J. Applegate, D.J. Cohen; Vascular closure devices: the second decade; J. Am. Coll. Cardiol., 50 (2007), pp. 1617–1626
- [15] D.S. Baim, et al.; Suture-mediated closure of the femoral access site after cardiac catheterization: results of the suture to ambulate aNd discharge (STAND I and STAND II) trials; Am. J. Cardiol., 85 (2000), pp. 864–869
- [16] R.J. Krone, L. Johnson, T. Noto; Five year trends in cardiac catheterization: a report from the registry of the Society for Cardiac Angiography and Interventions; Catheter. Cardiovasc. Diagn., 39 (1996), pp. 31–35
- [17] D.C. Duffin, et al.; Femoral arterial puncture management after percutaneous coronary procedures: a comparison of clinical outcomes and patient satisfaction between manual compression and two different vascular closure devices; J. Invasive Cardiol., 13 (2001), pp. 354–362
- [18] G.A. Hawker, et al.; Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale (SF-36 BPS), and Measure of Intermittent and Constant Osteoarthritis Pain (ICOAP); Arthritis Care Res (Hoboken), 63 (Suppl. 11) (1982), pp. S240–S252
- [19] S.A. Finkler; The distinction between cost and charges; Ann. Intern. Med., 96 (1982), pp. 102–109
- [20] L. Campeau; Percutaneous radial artery approach for coronary angiography; Catheter. Cardiovasc. Diagn., 16 (1989), pp. 3–7
- [21] D.B. Fram, et al.; Suture closure of the femoral arteriotomy following invasive cardiac procedures: a detailed analysis of efficacy, complications, and the impact of early ambulation in 1200 consecutive, unselected cases; Catheter. Cardiovasc. Interv., 53 (2001), pp. 163–173
- [22] U. Gerckens, et al.; Management of arterial puncture site after catheterization procedures: evaluating a suture-mediated closure device; Am. J. Cardiol., 83 (1999), pp. 1658–1663
- [23] H. Rickli, et al.; Comparison of costs and safety of a suture-mediated closure device with conventional manual compression after coronary artery interventions; Catheter. Cardiovasc. Interv., 57 (2002), pp. 297–302
- [24] B.W. Starnes, et al.; Percutaneous arterial closure in peripheral vascular disease: a prospective randomized evaluation of the Perclose device; J. Vasc. Surg., 38 (2003), pp. 263–271
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?