Abstract
Background
To evaluate in vivo the feasibility and safety of renal sympathetic denervation (RSD) with different catheters and various radiofrequency protocols.
Methods and results
Twenty-two pigs were included. First 2 pigs were enrolled in a feasibility protocol using one catheter and power from 5 W to 20 W. The next 10 pigs underwent RSD with three different catheters and four different RF-power settings of 5 W, 8 W, 10 W and 12 W in one minute per lesion (Protocol 1). The following 10 (Pigs 13 to 22) underwent RSD with five types of catheters (including the Symplicity® catheter), powers of 8 W and 10 W and two minutes RF-application (Protocol 2). Angiographic data were obtained at baseline, during and after RSD. At last, renal arteries were excised and analyzed macroscopically. The first pig developed severe renal stenoses with lesions of 15 to 20 W correlated with macroscopic alterations. The second feasibility pig did not develop renal stenosis with 5 and 8 W. In Protocol 1 from 60 RF-lesions, we observed 7 stenoses (≥ 30%). Three were severe (one of 80% with 10 W and two of 80% with 12 W). In Protocol 2 from 57 lesions we observed only 1 stenosis of 50% with 8 W with Symplicity® catheter. Severe stenosis was not observed.
Conclusion
In this study, renal sympathetic denervation showed safety using five types of catheters when applying RF-energy less than 10 W, within main stems of arteries larger than 3.0 mm diameter and a distance between lesions of at least 1 time catheter tip length.
Keywords
Radiofrequency renal sympathetic denervation;Various ablation catheters;Radiofrequency protocols;Safety of renal denervation
1. Introduction
Systemic hypertension represents a major risk factor for cardiovascular morbi-mortality, especially in occidental world [1] ; [2]. Approximately 90% of these patients do not have a secondary cause and, despite optimal clinical treatment, blood pressure (BP) levels remain uncontrolled in about one third [3]; [4]; [5]; [6] ; [7]. Patients taking 3 or more drugs, including a diuretic, and maintaining high BP levels are classified in a group of resistant hypertension. In this setting, sympathetic nervous system represents a major pathophysiological mechanism for development and maintenance of hypertension [8]. Surgical sympathectomy has been previously attempted however discontinued [9] ; [10]. Recently a new approach of endovascular renal sympathetic denervation (RSD) based on radiofrequency (RF) catheter ablation is gaining strength with convincing results [11] ; [12]. First studies used a fixed protocol with 4 to 6 focal lesions, in a spiral fashion, using a platinum–iridium 5 F 1.5 mm tip catheter, RF-power up to 8 Watts (W) and 120 s per lesion. Reasons for development of such protocol are not completely clear. Recently, new devices have been developed with particular protocols, based initially on those trials and have been applied in humans after few and short time animal testing. The present study sought to evaluate feasibility and safety of transcatheter RSD in animals testing different types of unipolar ablation catheters in various RF settings.
2. Methods
We conducted an observational and transversal evaluation of the feasibility and safety profile of RSD with radiofrequency (RF) catheter ablation in intact young pigs regarding vascular damage using various RF-generator settings and different types of ablation catheters. Secondary endpoints were the evaluation of macroscopic aspect of RF-lesions and general signs of traumatic manipulation. The Ethical Committee in Research from Medical School of Leipzig University approved the study.
2.1. Study sample
Twenty-two consecutive young healthy pigs underwent percutaneous RSD through radiofrequency catheter ablation. The first 2 pigs underwent feasibility protocols. Ten consecutive animals were assigned to Protocol 1 and a second group of ten animals were assigned to Protocol 2 (further described).
2.2. Definitions
Stenosis was defined by an angiographic narrowing of artery lumen equal or greater than 30% of its original diameter before any manipulation, measured by selective or unselective renal arteries angiography. Stenoses were classified according to the percentage of narrowing compared with the baseline lumen into light (30–50%), moderate (50–70%) and severe (≥ 70%). Irregularities were defined by an angiographic narrowing of artery lumen less than 30% of its original diameter. Traumatic manipulation included the presence of any other lesion rather than the ones placed by RF-catheters on the trajectory to the renal arteries or in their ostia, caused by catheter or sheath manipulation. Careful analyzes in this regard were conducted after renal arteries and aorta segment excision.
Macroscopic lesions were classified as present or not. In Pigs 1 and 2, macroscopic analyses were made with triphenyltetrazolium preparation, which is described further in this article.
2.3. Study protocols
The development of our protocols has been based on current RF-generator settings in previous trials [11] ; [12], which was fixed at 8 W, 60 °C and 2 min time RF-application per lesion. Following these criteria, programming of RF-generator included different power (W), temperatures (°C) and time in minutes (min). Different types of ablation catheters have been tested with these RF-settings as follows:
- Feasibility protocols:
In the first 2 pigs, two lesion protocols were defined using the palladium 6 F 4 mm non-irrigated tip catheter (Biosense Webster — Diamond Bar, CA, USA). Pig 1 received lesions with 15 and 20 W and 40 °C in the right renal artery and 10 to 15 W with 40 to 60 °C in the left one. Each lesion lasted 1 min. Lesions were placed in 3 segments and in opposite sites, meaning 6 lesions in each artery. After analyzing the results in Pig 1 (see in Results), we applied less lesions (3 lesions per artery) and less power in Pig 2 with 1 min RF-application.
After the feasibility-step, the study was conducted with 2 protocols, each one applied in 10 pigs, respectively.
- Study protocols:
The first protocol was used from Pig 3 to Pig 12 (Protocol 1), and the second, from Pig 13 to Pig 22 (Protocol 2). Protocol 1 used 3 different catheters: Palladium 6 F 4 mm non-irrigated tip (Biosense Webster — Diamond Bar, CA, USA), the gold 6 F 4 mm non-irrigated tip (Biotronik — Berlin, Germany) and the gold 6 F 3.5 mm irrigated tip (Biotronik — Berlin, Germany), RF-powers of 5 W, 8 W, 10 W and 12 W and 1 min each RF-application. In Protocol 2, we used 5 different catheters: The previous 3 in Protocol 1 and added the platinum 5 F 4 mm non-irrigated tip — Marinr® (Medtronic Inc. — Minneapolis, MN, USA) and the platinum–iridium 5 F 2 mm tip Symplicity® Catheter System™ (Ardian/Medtronic Inc., California, USA) and RF-powers of 8 W and 10 W. Each lesion lasted 2 min in accordance to the fixed protocol of Symplicity®. Previously presented, the Symplicity® Catheter System™ provided only a fixed protocol of 8 W and 2 min each application [11] ; [12].
- Lesions placement and contact assurance:
In each pig, two to four lesions were applied per artery, depending on local anatomy, with an attempt of a 1 time-catheter-tip safe distance from each lesion in order to avoid RF-lesions in opposite sites within the same ring-segment, i.e. overlapping lesions. This could compromise the analysis of each lesion individually. Lesions were placed mainly at the inferior and superior aspects of the arteries to facilitate fluoroscopic detection and quantification of stenosis in antero-posterior 0°/0° incidence. This strategy was particularly useful to locate and mark lesions for macroscopic analysis. Contact assurance and force were estimated by angiographic analysis and local impedance changes from a non-contact position.
2.4. Procedure
Pigs were deep anesthetized, intubated and put on mechanical ventilatory support. An experienced veterinarian from the research laboratory of the Heart Center Leipzig — Leipzig University supervised the entire procedure. Standard monitoring with peripheral ECG leads, blood oxygen saturation, central venous pressure measurement (internal Jugular vein), central artery pressure (left femoral artery or right carotid artery) and controlled ventilator parameters. The right femoral artery was accessed by Seldinger technique with one 11 F in all pigs. Through this sheath, was introduced a long 8.5 F non-steerable sheath (for the first 5 pigs — SR0) or a 10.5 F steerable sheath (for the last 17 pigs — Agilis®) (both from St. Jude Medical, St. Paul, Minnesota, USA). The long sheath was advanced until the level of the renal arteries and their ostia were located by non-selective angiography. The ablation catheter was than advanced through the long sheath, which was placed nearby the ostia of the arteries, and gently placed inside the right and left renal arteries, respectively, to perform radiofrequency lesions according to the protocols (see Study protocols) (Fig. 1). The RF generators were the Stockert (Biosense Webster — Diamond Bar, CA, USA) and Symplicity®™ Generator (Ardian/Medtronic Inc., California, USA). All pigs received intravenous bolus of 5000 IU heparin after arterial assessment and reinfused to maintain the activated clotting time ≥ 250, measured each 30 min. We infused nitrate directly in arteries in some animals according to the presence of any stenosis after RSD.
|
|
|
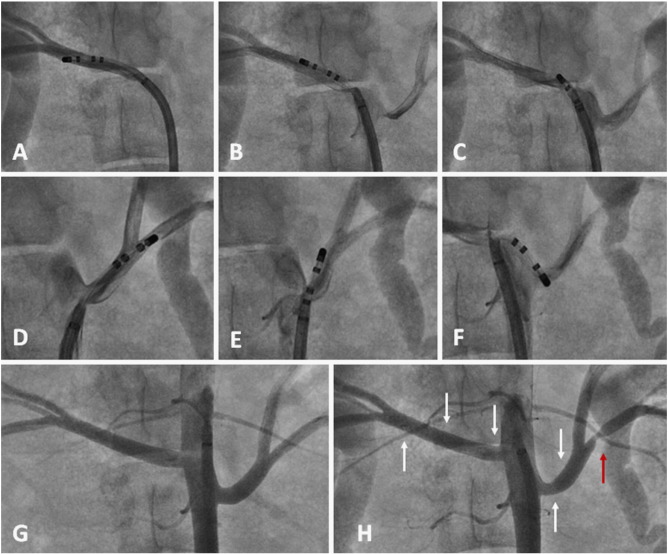
Fig. 1. Example of renal denervation in Pig 6 (Protocol 1) with the long sheath Agilis® and gold 4 mm non-irrigated tip catheter. Within the right artery (5.8 mm diameter): A — first (distal) lesion with 8 W; B — second (middle) lesion with 5 W; C — third (proximal) lesion with 8 W. Within the left artery (5.6 mm diameter): D — first (distal) lesion inside a small branch (2.1 mm diameter) with 8 W; E — second (middle) lesion with 5 W; F — third (proximal) lesion with 5 W. G — Non-selective angiography before ablation. H — Angiography after the entire set of lesions showing severe stenosis (80%) in the left inferior small branch after bifurcation (red arrow). |
2.5. Fluoroscopy analysis
The fluoroscopy intensifier was set to stay at the antero-posterior projection (AP) 0°/0° in all cases. The first contrast injection was in the Aorta (non-selective) or within each artery ostium as the baseline evaluation. After locating the arteries, the long sheath placed nearby the ostium of each artery permitted the introduction of ablation catheters. Angiography control assured the position of the catheter at baseline (before entering the renal artery), before and after each lesion. At last, the result was accessed with selective or non-selective angiography (Fig. 2). The measured baseline intraluminal diameter of the main arteries considered the catheter tip as reference in French scale (F) and converted into millimeters (mm). The degree of intraluminal stenosis was measured by comparison with the same segment before ablation and quantified as percent of stenosis (%).
|
|
|
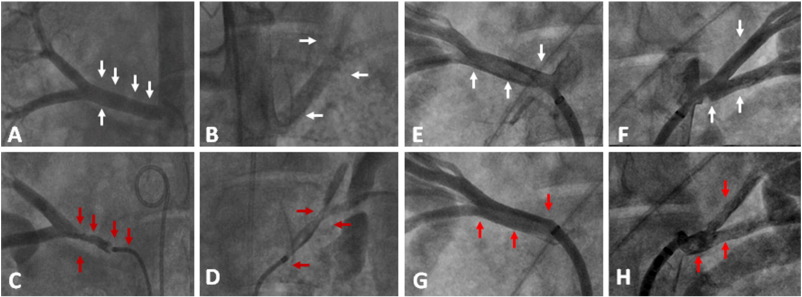
Fig. 2. Renal denervation in feasibility protocol: Pig 1 (A to D — left half of figure) and Pig 2 (E to H — right half of figure). Pig 1: A and B show angiographies before denervation from the right and left arteries, respectively. White arrows are showing the locations where lesions were placed (power of 10 W to 20 W — see text). C and D show selective angiography of right and left renal arteries, respectively, after denervation. Red arrows show where lesions were placed. Notice the proximity of each lesion placed at the upper aspect of the right artery, as well as the greater stenosis degree where the lesions were at opposite sides within the same ring segment. At the left artery is of note that distal lesions were placed very close to the bifurcation and within the same ring segment. Intravenous local nitrate could not dilate the arteries. Pig 2: E and F show selective angiography before denervation from the right and left arteries, respectively. White arrows show the locations where the lesions were placed (Power of 5 W and 8 W — see text). Red arrows at G and H show lesions locations after the procedure. On G there were a slightly irregularity at the distal lesion in the ostium of the first inferior branch of right artery (5 W lesion) and on H, just few irregularities within the upper branch of left artery (8 W lesion). |
2.6. Anatomy/macroscopy analysis
After ablation and euthanasia, animals had the kidneys with the arteries and Aorta segment excised. Kidneys were than separated with resections after the first main bifurcation of the renal arteries. The main renal arteries stems with the Aorta segment received markers (thin stitches) where each lesion was placed based on fluoroscopic information. In the first 2 pigs from the feasibility protocol, the triphenyltetrazolium preparation was used to observe macroscopic lesions (Fig. 2 ; Fig. 3).
|
|
|
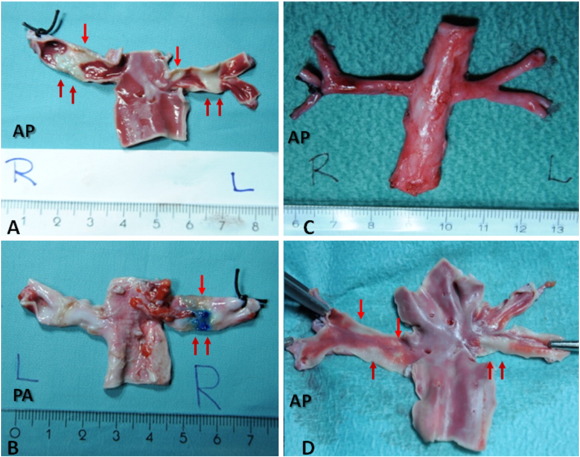
Fig. 3. Macroscopic evaluation of Pig 1 (A and B) and Pig 2 (C and D) after denervation with triphenyltetrazolium preparation. On A the injured tissue is colored in white which are showed by red arrows. Notice the severe damage and extension of lesions, especially at the right artery. On B is noticed a blue coloration at the adventitia (red arrows) meaning transmural necrosis. Figure C shows the Aorta segment with the renal arteries of Pig 2 before triphenyltetrazolium preparation. Figure D shows the lesions marks in white (red arrows). At this last, even the distally 5 W lesion could produce tissue damage. Notice no macroscopic damage within the aorta in A or D. |
2.7. Statistical analysis
Baseline variables were expressed as mean ± SD. Fishers exact test was used to compare differences of lesion formation with the different catheters in Protocol 2. P value < 0.05 was considered significative.
3. Results
3.1. Baseline characteristics
From August to November 2011, twenty two consecutive young healthy pigs underwent percutaneous RSD through radiofrequency catheter ablation. The first 2 pigs (Pig 1 and Pig 2) were included in the observations and were basically used for procedure feasibility. The next 10 pigs (Pig 3 to Pig 12) were included in the Protocol 1 and the last 10 pigs (Pig 13 to Pig 22) underwent Protocol 2. The second protocol was adjusted after analyzing the former, as previously described (see Study protocols). The pigs weighted between 54 and 75 kg. Comparison between groups demonstrated significantly that animals from Protocol 2 were less weighted (59 ± 6.3 vs. 69 ± 2.8 kg — P < 0.05) and renal arteries had smaller diameter (right: 4.75 ± 0.7 vs. 5.8 ± 0.4 mm/left: 5.4 ± 0.6 vs. 6.0 ± 0.6 mm — P < 0.05). In addition, in Protocol 2, the right arteries had smaller length compared with Protocol 1 (26.4 ± 9.2 vs. 38.8 ± 7.2 mm — P < 0.05). Baseline characteristics are described in Table 1.
| Characteristics | Feasibility Pig 1 | Feasibility Pig 2 | Protocol 1 (mean ± SD) | Protocol 2 (mean ± SD) | Total Value (mean ± SD) | P value |
|---|---|---|---|---|---|---|
| Length (cm) | 125 | 122 | 125 ± 1.9 | 125 ± 2.2 | 124.4 ± 2.1 | 0.04 |
| Weight (kg) | 68 | 67 | 69 ± 2.8 | 59 ± 6.3 | 65.7 ± 6.1 | < 0.05 |
| SBP (mm Hg) | 100 | 98 | 91.5 ± 7.7 | 85.5 ± 9.4 | 89.1 ± 9.5 | NS |
| DBP (mm Hg) | 61 | 55 | 51 ± 5.1 | 48.5 ± 2.8 | 50.7 ± 4.8 | NS |
| Heart rate (bpm) | 83 | 72 | 84 ± 6.8 | 86 ± 6.2 | 82.7 ± 6.6 | NS |
| CVP (mm Hg) | 12 | 11 | 12 ± 2.4 | 13 ± 1.6 | 12.3 ± 2.0 | NS |
| SO2 (%) | 100 | 99 | 99.5 ± 1.2 | 99 ± 3.2 | 98.5 ± 2.3 | NS |
| Anal Temp (°C) | 39.4 | 39.7 | 39.8 ± 0.4 | 39.8 ± 0.5 | 39.7 ± 0.4 | NS |
| Aorta diameter (mm) | 7.9 | 12 | 10.9 ± 1.5 | 10.5 ± 0.6 | 10.6 ± 1.2 | 0.30 |
| LA main diameter (mm) | 3.97 | 7.77 | 6 ± 0.6 | 5.4 ± 0.6 | 5.5 ± 0.8 | < 0.05 |
| RA main diameter (mm) | 5.25 | 7 | 5.8 ± 0.4 | 4.75 ± 0.7 | 5.6 ± 0.7 | < 0.05 |
| LA length (mm) | 20.2 | 12.8 | 27.2 ± 6.2 | 25.05 ± 7.0 | 24.3 ± 6.8 | 0.19 |
| RA length (mm) | 49.7 | 31.9 | 38.8 ± 7.2 | 26.4 ± 9.2 | 32.4 ± 10.4 | < 0.05 |
3.2. Radiofrequency lesions and end points
A total of 131 lesions were achieved in the 22 pigs: feasibility protocol — 15 lesions; Protocol 1 — 60 lesions; Protocol 2 — 56 lesions. These data is summarized in Table 2.
| Type of catheter | Number of lesions with 5 W | Number of stenoses (%) | Number of lesions with 8 W | Number of stenoses (%) | Number of lesions with 10 W | Number of stenoses (%) | Number of lesions with 12 W | Number of stenoses (%) | Total of RF-applications per catheter | Total of stenoses per catheter (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Gold 4 mm | 14 | 0 (0%) | 11 | 3 (27.3%) | 2 | 0 (0%) | 1 | 0 (0%) | 28 | 3 (10.7%) |
| Gold irrigated | 2 | 0 (0%) | 4 | 0 (0%) | 5 | 0 (0%) | 3 | 1 (33.3%) | 14 | 1 (7.1%) |
| Palladium | 6 | 0 (0%) | 5 | 0 (0%) | 2 | 1 (50%) | 5 | 2 (40%) | 18 | 3 (16.7%) |
| Total RF-applications | 22 | 0 (0%) | 20 | 3 (15%) | 9 | 1 (11.1%) | 9 | 3 (33.3%) | 60 | 7 (11.7%) |
3.3. Feasibility protocol
The catheter used was the palladium tip for 1 min each RF-lesions. In Pig 1 we delivered 5 lesions in the right artery with 15 W to 20 W and 4 lesions in the left artery with 10 W to 15 W alternately. In the right artery, lesions were placed very close to one another, thus some lesions appeared to be overlapped. Severe stenoses have been observed almost immediately after placement of each lesion independently on power (15 W or 20 W). In the left artery, the first distal lesion was placed with 10 W in a small branch after the first bifurcation with 2.50 mm diameter. The next 2 lesions (with 15 W and 10 W) were slightly overlapped at the distal segment of the main left stem, which measured 3.97 mm diameter. These 3 places showed severe stenoses. The forth lesion (15 W) was placed at the proximal segment of the main stem (3.97 mm) and did not show important stenosis. Angiographic stenoses and respective macroscopic analyzes are shown in Fig. 2 ; Fig. 3. At last, local infusion of nitrate did not show artery dilatation.
According to angiographic results in Pig 1, we defined a different set of lesions in Pig 2. We applied 4 lesions in the right artery with 5 W and 8 W alternately and 2 lesions in the left artery with 8 W. We applied RF-energy in less places compared to Pig 1 in order to avoid overlapping lesions. We observed only irregularities and no severe macroscopic lesions (Fig. 2 ; Fig. 3). Lesions in the left artery of Pig 2 were placed after artery bifurcation, within the branches, due to the anatomy of a short main stem of 7.7 mm diameter. The upper and lower branches measured 3.50 mm and 3.75 mm diameter, respectively, and we did not observe any stenosis.
3.4. Protocol 1
In this protocol, three catheters were evaluated: the Palladium tip, gold non-irrigated tip and gold irrigated tip. We evaluated RF-powers of 5 W, 8 W, 10 W and 12 W with all three catheters. A total of 60 spots of RF applications were performed, each spot for 1 min. This data is summarized in Table 2.
3.4.1. Analysis of RF-power and stenosis
We observed that RF applications performed with 5 W did not produce any kind of injury, regardless of the type of catheter used. From the 20 RF applications performed using 8 W of power, we observed 3 (15%) stenoses. Using the power of 10 W, one stenosis was observed from 10 RF-applications (11.1%). From 9 RF-applications with 12 W we observed 3 stenoses (33.3%). Details from these lesions are indicated in Fig. 4.
|
|
|
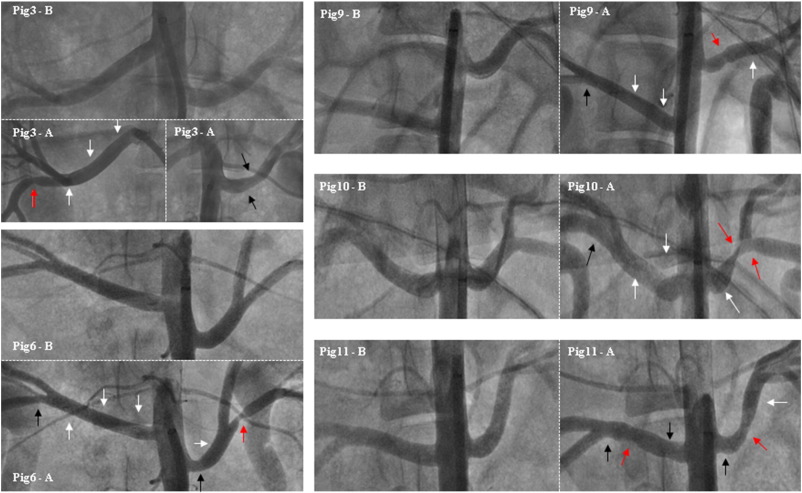
Fig. 4. Samples in Protocol 1 (one minute RF-time) which presented some degree of angiographic damage (Pigs 3, 6, 9, 10 and 11). The legend is represented by: “Pig number — B (before) and A (after)” the procedure. White arrows show where lesions where applied without apparent angiographic damage. Black arrows indicate irregularities. Red arrows indicate lesions considered to be stenoses (intraluminal reduction ≥ 30%). Stenosis in Pig 3 in a small branch from right artery with 8 W and gold non-irrigated tip catheter. Stenosis in Pig 6 in a small branch with 8 W and gold non-irrigated tip catheter. Stenosis in Pig 9 with gold non-irrigated tip catheter and 8 W. Stenosis in Pig 10 at the left artery with palladium tip catheter after 2 overlapped lesions using 10 W (distal) and 12 W (middle). Two stenoses in Pig 11 using palladium tip catheter in the right artery with 12 W and gold tip catheter with 12 W in the left artery. |
3.4.2. Analysis of catheter type and stenoses
We observed 3 stenoses out of 28 RF-applications with the gold non-irrigated tip catheter. All three with 8 W power. With gold irrigated tip catheter we observed 1 stenosis with 12 W from a total of 14 RF-applications. At last, with the Palladium tip catheter we observed 3 stenoses from 18 RF-applications: One with 10 W and 2 with 12 W. (Table 2 and Fig. 4 — red arrows).
3.5. Protocol 2
During Protocol 2 we evaluated five catheters: the Palladium tip, gold non-irrigated tip, gold irrigated tip, platinum 5 F tip and the Symplicity® catheter using powers of 8 W and 10 W (except the Symplicity® system), each for 2 min. Fifty-six ablation spots were performed as shown in Table 3.
| Type of catheter tip according to power | 8 W | Number of lesions (%) | 10 W | Number of lesions (%) | Total of lesions according to catheter | Total of lesions caused according to catheter |
|---|---|---|---|---|---|---|
| Gold 4 mm | 5 | 0 (0%) | 5 | 0 (0%) | 10 | 0 (0%) |
| Gold irrigated | 6 | 0 (0%) | 5 | 0 (0%) | 11 | 0 (0%) |
| Palladium | 6 | 0 (0%) | 6 | 0 (0%) | 12 | 0 (0%) |
| Symplicity®/Ardian | 12 | 1 (%) | X | X | 12 | 1 (%) |
| Platinum | 5 | 0 (0%) | 6 | 0 (0%) | 11 | 0 (0%) |
| Total of lesions according to power | 34 | 1 (%) | 22 | 0 (0%) | 56 | 1 (%) |
3.5.1. Analysis of RF-power, catheters and stenoses
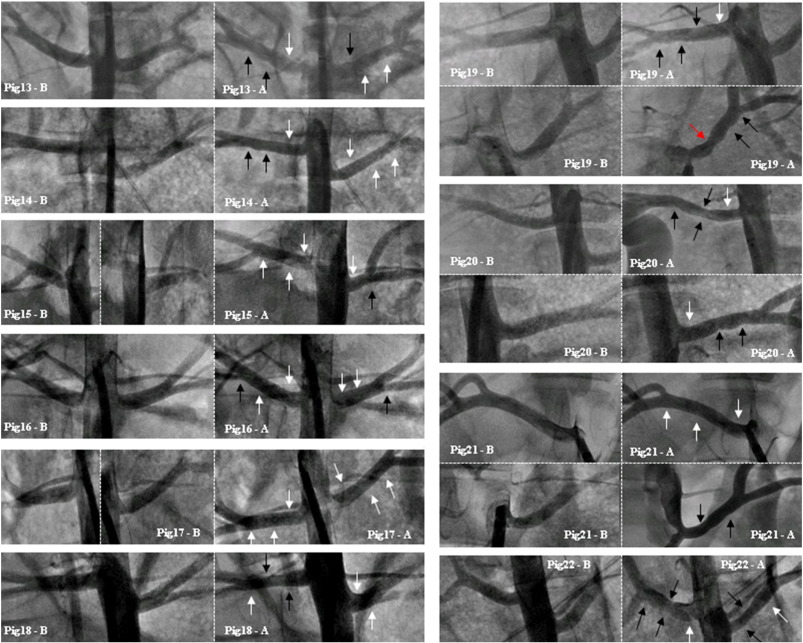
From 56 RF applications we could observe only one stenosis with the Symplicity® catheter, which was considered to have 50% lumen reduction (Fig. 5 — red arrow at Pig 19-A). The power of 10 W has not been tested using this catheter, since the maximum 8 W is the valid algorithm from the respective RF generator (Table 3).
|
|
|
Fig. 5. All pigs in Protocol 2 (two-minute RF-time). The legend is represented by: “Pig number — B (before) and A (after)” the procedure. White arrows show the placement of lesions without angiographic damages. Black arrows indicate lesions with irregularities after ablation. The red arrow in Pig 19 shows stenotic lesion considered 50% with the Symplicity® catheter. |
3.5.2. Analysis of mechanical trauma or dissection
During and after all procedures, we did not find mechanical trauma and any artery dissection during and after RSD by angiographic and macroscopic analyses.
4. Discussion
Based on pathophysiological aspects of resistant essential hypertension, targeting sympathetic innervation became an attractive aim of treatment [6]; [7]; [8] ; [13]. In the early 70s, patients with resistant hypertension were treated surgically with sympathectomy [9] ; [10]. However, this procedure was discontinued owing to complexity, invasiveness and high incidence side effects. Intravascular RF-ablation intends to disrupt renal sympathetic innervation less invasively. The majority of sympathetic nerves are located within 0.5 to 1.0 depth from the intima layer of renal arteries [14]. This fact corresponds to the anatomical basis for RSD. First studies in humans, also using conventional ablation catheters, have proved safety and positive reproducible clinical results [11]; [12]; [15]; [16]; [17] ; [18]. Moreover some studies suggest additional clinical benefits [15] ; [16] and a presumable positive cost-effectiveness [17] with a safe profile. However, no experimental data clearly supports the apparent choice of ablation protocols applied in clinical trials. To our knowledge, this is the first study in vivo testing different RF-protocols and various catheters on feasibility and safety of RSD procedure.
In the present study, we have found very important stenoses with great RF-power delivered (15–20 W) in Pig 1 using a palladium tip conventional ablation catheter. The first distal lesion placed within left artery in Pig 1 was in a small branch after the first bifurcation (2.50 mm diameter), and showed stenotic lesion with less power (10 W). The 2° and 3° stenotic lesions, with 15 W and 10 W respectively, overlapped each other at the distal segment of the main left stem, which measured 3.97 mm diameter. The 4° lesion (15 W) at the proximal segment of the main stem (3.97 mm) did not show important stenosis. Therefore we observed that was unsafe to apply lesions with RF-power equal or higher than 15 W, within small artery branches (≤ 2.50 mm) and in a close distance between spots (overlapping lesions).
In Protocol 1 we observed 7 stenoses from 60 RF-applications. Interestingly, three of them occurred with 8 W and with the gold non-irrigated tip catheter. Two of them occurred in an artery branch smaller than 3.0 mm diameter and one within a main stem graded as 30% (Fig. 4). The three severe stenoses occurred with 8 W (small branch of 2.64 mm from left artery — Fig. 4), 10 W and 12 W (overlapped lesions in Pig 10 — Fig. 4). In Protocol 2 we did not observe severe stenosis applying 8 W and 10 W, except one stenosis of 50% with the Symplicity® catheter within the artery main stem of 5.4 mm diameter (Fig. 5). In this protocol, we applied lesions only within main stems.
Interestingly, comparing animals from Protocols 1 and 2, we observed that the second group was composed from smaller samples and with narrower arteries. This feature, however, did not interfere in overall result.
Other studies in animal models showed that local changes in endothelial monolayer led to thrombus formation even after 6 months follow-up [19] ; [20]. As shown previously by optical coherence tomography from renal arteries, most patients who underwent RSD with non-irrigated technology presented acute vascular injury after the procedure. These alterations were mainly edema, thrombus and vasospasms [21]. This first study observed a trend towards the intraluminal thrombus formation, i.e. in 89% of the cases using the EnligHTN™ system (catheter-basket ablation system) and in 53% with the Simplicity (P = 0.07). A significantly greater thrombus load per renal artery was observed after RSD with the EnligHTN™ system compared with the Simplicity system (4.6 ± 3.1 vs. 1.5 ± 1.8, P = 0.006) [21]. In our study we did not observed any thrombus formation during angiographic or macroscopic analyses.
Some groups used conventional RF-ablation catheters for RSD with varied results, however showing safety with particular protocols [18]; [22]; [23] ; [24]. This experimental study showed safety with either conventional RF-catheters or the Symplicity® catheter, in a stepwise testing approach of the different catheters and RF-settings. The safety was addressed when ablation was attempted with a RF-power less than 10 W, in artery segments above 3.0 mm diameter and a distance between lesions at least 1 time the catheter tip length.
In addition, using a 8.5 F non-steerable and a 10.5 F steerable sheath, we did not observe any mechanical trauma or dissection. This experience was also recently published in humans [25].
4.1. Limitations
Our study evaluated acute effects of RSD in animal samples with similar anatomy than human and tested different catheter types and RF-settings. One limitation includes the small sample number to observe different variables. We did not conduct a study with chronic observations, regarding ethical issues in animal studies. In this study we did not conduct histological evaluation, thus it was not possible to define the accomplishment of transmural lesion. Nevertheless the main endpoint was safety from occurrence of stenosis or macroscopic lesions after necropsy.
Protocol 1 included more different power settings to test in a small sample of animals, which could have led to a non-uniform distribution of RF-lesion placement.
5. Conclusion
Radiofrequency ablation for renal sympathetic denervation is safe when applied with power less than 10 W, in arteries with a diameter above 3.0 mm and a distance between lesions at least 1 time the catheter tip length to avoid overlapping of lesions. Renal arteries denervation with all tested catheters showed to be safe respecting the conditions mentioned before.
Conflict of interests
The authors do not declare conflict of interests for this study.
Acknowledgments
We would like to thank the great contribution from the team of veterinarians and biologists of the animals' experimental laboratory of the Heart Center of Leipzig, University of Leipzig.
References
- [1] P.M. Kearney, M. Whelton, K. Reynolds, et al.; Global burden of hypertension: analysis of worldwide data; Lancet, 365 (2005), pp. 217–223
- [2] K. Wolf-Maier, R.S. Cooper, H. Kramer, et al.; Hypertension treatment and control in five European countries, Canada, and the United States; Hypertension, 43 (2004), pp. 10–17
- [3] W.C. Cushman, C.E. Ford, J.A. Cutler, et al.; Success and predictors of blood pressure control in diverse North American settings: the antihypertensive and lipid-lowering treatment to prevent heart attack trial (ALLHAT); J. Clin. Hypertens., 4 (2002), pp. 393–404
- [4] S. Erdine, et al.; ESH-ESC guidelines for the management of hypertension; Herz, 3 (2006), pp. 331–338
- [5] C. Rosendorff, H.R. Black, Cannon CP, et al.; Treatment of hypertension in the prevention and management of ischemic heart disease: a scientific statement from the American Heart Association Council for High Blood Pressure Research and the Councils on Clinical Cardiology and Epidemiology and Prevention; Circulation, 115 (2007), pp. 2761–2788
- [6] D.A. Calhoun, et al.; Resistant hypertension: diagnosis, evaluation, and treatment: a scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research; Circulation, 24 (2008), pp. 117–125
- [7] H. Lowel, C. Meisinger, M. Heier, H. Hymer, D. Alte, H. Volzke; Epidemiology of hypertension in Germany. Selected results of population representative cross-sectional studies; Dtsch. Med. Wochenschr., 131 (2006), pp. 2586–2591
- [8] G. Grassi, S. Bertoli, G. Seravalle; Sympathetic nervous system: role in hypertension and in chronic kidney disease; Curr. Opin. Nephrol. Hypertens., 21 (2012), pp. 46–51
- [9] D.M. Morrissey, V.S. Brookes, W.T. Cooke; Sympathectomy in the treatment of hypertension; review of 122 cases; Lancet, 28 (1953), pp. 403–408
- [10] R.H. Smithwick; Sympathectomy, splanchniecoctomy and vagotomy; Rev. Surg., 30 (1973), pp. 153–173
- [11] H. Krum, M. Schlaich, R. Whitbourn, et al.; Catheter-based renal sympathetic denervation for resistant hypertension: a multicentre safety and proof-of-principle cohort study; Lancet, 373 (2009), pp. 1275–1281
- [12] M.D. Esler, H. Krum, P.A. Sobotka, M.P. Schlaich, R.E. Schmieder, M. Böhm; Renal sympathetic denervation in patients with treatment-resistant hypertension (the Symplicity HTN-2 Trial): a randomized controlled trial; Lancet, 376 (2010), pp. 1903–1909
- [13] P.A. Smith, L.N. Graham, A.F. Mackintosh, et al.; Relationship between central sympathetic activity and stages of human hypertension; Am. J. Hypertens., 17 (2004), pp. 217–222
- [14] D.S. Atherton, N.L. Deep, F.O. Mendelsohn; Micro-anatomy of the renal sympathetic nervous system: a human postmortem histologic study; Clin. Anat., 25 (2012), pp. 628–633
- [15] E. Pokushalov, A. Romanov, G. Corbucci, S. Artyomenko, V. Baranova, A. Turov, N. Shirokova, A. Karaskov, S. Mittal, J.S. Steinberg; A randomized comparison of pulmonary vein isolation with versus without concomitant renal artery denervation in patients with refractory symptomatic atrial fibrillation and resistant hypertension; J. Am. Coll. Cardiol., 60 (2012), pp. 1163–1170
- [16] F. Mahfoud, M. Schlaich, I. Kindermann, C. Ukena, B. Cremers, M.C. Brandt, U.C. Hoppe, O. Vonend, L.C. Rump, P.A. Sobotka, H. Krum, M. Esler, M. Böhm; Effect of renal sympathetic denervation on glucose metabolism in patients with resistant hypertension: a pilot study; Circulation, 123 (2011), pp. 1940–1946
- [17] B.P. Geisler, B.M. Egan, J.T. Cohen, A.M. Garner, R.L. Akehurst, M.D. Esler, J.B. Pietzsch; Cost-effectiveness and clinical effectiveness of catheter-based renal denervation for resistant hypertension; J. Am. Coll. Cardiol., 60 (2012), pp. 1271–1277
- [18] M.G. Kiuchi, G.L. Maia, M.A. de Queiroz Carreira, T. Kiuchi, S. Chen, B.R. Andrea, M.L. Graciano, J.R. Lugon; Effects of renal denervation with a standard irrigated cardiac ablation catheter on blood pressure and renal function in patients with chronic kidney disease and resistant hypertension; Eur. Heart J., 34 (2013), pp. 2114–2121
- [19] M.K. Rippy, D. Zarins, N.C. Barman, A. Wu, K.L. Duncan, C.K. Zarins; Catheter-based renal sympathetic denervation: chronic preclinical evidence for renal artery safety; Clin. Res. Cardiol., 100 (2011), pp. 1095–1101
- [20] K. Steigerwald, A. Titova, C. Malle, E. Kennerknecht, C. Jilek, J. Hausleiter, J.M. Nahrig, K.L. Laugwitz, M. Joner; Morphological assessment of renal arteries after radiofrequency catheter-based sympathetic denervation in a porcine model; J. Hypertens, 30 (2012), pp. 2230–2239
- [21] C. Templin, M. Jaguszewski, J.R. Ghadri, I. Sudano, R. Gaehwiler, J.P. Hellermann, R. Schoenenberger-Berzins, U. Landmesser, P. Erne, G. Noll, T.F. Lüscher; Vascular lesions induced by renal nerve ablation as assessed by optical coherence tomography: pre- and post-procedural comparison with the Simplicity catheter system and the EnligHTN multi-electrode renal denervation catheter; Eur. Heart J., 34 (2013), pp. 2141–2148
- [22] Gajendra Manakshe, R. Chakravarthi, Shaista Hussaini, et al.; Renal sympathetic denervation for treatment of resistant hypertension — indigenous technique; Indian Heart J., 65 (2013), pp. 239–242
- [23] L. Armaganijan, et al.; Renal denervation using an irrigated catheter in patients with resistant hypertension: a promising strategy?; Arq. Bras. Cardiol., 102 (2014), pp. 355–363
- [24] M.G. Kiuchi, et al.; Acute effect of renal sympathetic denervation on blood pressure in refractory hypertensive patients with chronic kidney disease; Int. J. Cardiol., 190 (2015), pp. 29–31
- [25] B.R. Andrea, et al.; Renal sympathetic denervation with long steerable sheath and mapping system — first-in-man experience in three consecutive patients; Int. J. Cardiol., 174 (2014), pp. 408–410
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?