Highlights
- Systematic searches identified 8 trials reporting days of cannabis use per month in delayed or information-only controls
- Sample-weighted mean days of use fell from 24.5 per month at Baseline to 19.9 at 2-4 months
- A meta-analysis showed an average reduction of 0.442 SD.
- While there was substantial continued use, this reduction sets a challenging benchmark for treatments.
Abstract
Introduction
Cannabis remains the most used illegal substance across the globe, and negative outcomes and disorders are common. A spotlight therefore falls on reductions in cannabis use in people with cannabis use disorder. Current estimates of unassisted cessation or reduction in cannabis use rely on community surveys, and few studies focus on individuals with disorder. A key interest of services and researchers is to estimate effect size of reductions in consumption among treatment seekers who do not obtain treatment. Effects within waiting list or information-only control conditions of randomised controlled trials offer an opportunity to study this question.
Method
This paper examines the extent of reductions in days of cannabis use in the control groups of randomised controlled trials on treatment of cannabis use disorders. A systematic literature search was performed to identify trials that reported days of cannabis use in the previous 30 (or equivalent).
Results
Since all but one of the eight identified studies had delayed treatment controls, results could only be summarised across 2–4 months. Average weighted days of use in the previous 30 days fell from 24.5 to 19.9, and a meta-analysis using a random effects model showed an average reduction of 0.442 SD. However, every study had at least one significant methodological issue.
Conclusions
While further high-quality data is needed to confirm the observed effects, these results provide a baseline from which researchers and practitioners can estimate the extent of change required to detect effects of cannabis treatments in services or treatment trials.
Keywords
Cannabis;Self-management;Natural recovery;Control conditions
1. Introduction
Cannabis remains the most used illegal drug across the world, and while rates of use are generally falling, the incidence of related harm is rising internationally (United Nations Office on Drugs and Crime, 2014). Australia has particularly high rates of use, with 35% of adults reporting lifetime consumption, and 10% using it in the previous 12 months (Australian Institute of Health and Welfare, 2014).
However, 70–80% of cannabis users stop using it by their mid-thirties (Chen & Kandel, 1998), and even over 5–6 years, substantial rates of cessation or reduced consumption in adolescents or young adults are seen (Kandel and Raveis, 1989; Pollard et al., 2014 ; Sussman and Dent, 2004). In common with other substances, most successful cessation occurs without treatment (Cunningham, 2000 ; Price et al., 2001). While these changes are typically greatest among infrequent or non-problematic users (Chen & Kandel, 1998), people with cannabis abuse or dependence also have substantial rates of recovery. For example, an analysis of data from Wave 1 of the National Epidemiologic Survey on Alcohol and Related Conditions (Agosti & Levin, 2007) found that 81% of people with lifetime cannabis dependence did not meet criteria over the previous year.
While community samples can provide good estimates of the degree and timing of recovery from cannabis use disorder, sample sizes need to be large to provide accurate estimates of these rates. So, a study of 1228 adolescents (Perkonnigg et al., 1999) found only 12 with lifetime cannabis dependence, and the resultant estimate of full remission (32%) therefore had a substantial standard error (26%). Furthermore, treatment trial researchers and services need estimates of remission in treatment seekers.
A study of control groups in treatment studies provides fertile ground for the estimation of changes in treatment seekers who do not receive substantial assistance. These studies have several advantages: high-quality trials typically have diagnostic interviews and other assessments that are able to characterise the samples well, the nature of treatments is standardised or tracked carefully, and substantial effort is put into ensuring that follow-up assessments maximise retention rates. While individual studies often have relatively small sample sizes in their control group, meta-analytic methods provide an opportunity to obtain estimations of effect sizes over multiple studies and substantial samples.
Accordingly, the aim of the current paper was to determine the degree of ‘natural recovery’ in the control groups from randomised controlled trials on substance use disorders, which reported changes in the frequency of cannabis use. ‘Natural recovery’ in this article refers to processes where consumption of cannabis is reduced or ceased without professional intervention. It was operationalised as the degree of change in cannabis use within groups receiving inactive or minimal interventions.
2. Methods
Electronic searches were performed in January 2015, to find studies that included a control group that had explored the topic of cannabis use treatment. The search used title, abstract and keywords of Medline, PsycINFO, Psychology Journals, and Psychology Subject Corner. The search terms were: (cannabis OR marijuana OR marihuana OR addiction OR abuse OR substance) AND (treatment OR randomi* control).
Potential studies were evaluated for inclusion in this study by the first author, based on whether they: (a) provided data on cannabis use, which allowed the calculation of pre–post effect sizes in a group of participants randomised to receive inactive (e.g. waitlist) or minimal interventions (e.g. drug-related information only); (b) were in English; (c) did not comprise case studies or personal accounts; (d) did not include participants with severe mental disorders (i.e., schizophrenia, bipolar disorder, posttraumatic stress disorder, major depressive disorder). In order to report results on a single measure, we restricted the studies to those allowing a calculation of cannabis use in the previous 30 days.
The formal examination of effect sizes used Comprehensive Meta-Analysis (Borenstein, Hedges, Higgins, & Rothstein, 2005), and the primary analysis applied a random effects model. This is the appropriate approach to use when samples or treatments are potentially different, regardless of whether significant heterogeneity is evidenced (Borenstein, Hedges, Higgins, & Rothstein, 2009). We report effects as standardised mean differences (Cohens d). Analyses of degree of change require estimates of test–retest correlations of the measures, or reported analyses of changes within groups. While Timeline Followback assessments of cannabis use can have a 7–14 day test–retest reliability of 0.92 (Robinson, Sobell, Sobell, & Leo, 2014), we do not know the reliability of the 3–12 month assessments of cannabis use in the current trials. We use an estimate of 0.70 for the primary analyses below, but also undertake sensitivity analyses with test–retest correlations of .60 and .80. Where means and standard deviations were reported on different sample sizes at baseline and follow-up, we used the follow-up sample size for the analysis, estimating baseline scores for retained participants from reported data using the full sample. We also present sample-weighted mean days of use at baseline, post and follow-up assessments.
3. Results
The search of cannabis treatment in general population samples elicited 2554 articles. Reviewing article titles to confirm that they met the search criteria left 374, and this number was reduced to 55 after reading abstracts. Further searching using reference lists and cited reference search yielded 12 potential articles, and 3 others were suggested by reviewers. Review papers were examined (Carballo et al., 2007; Dutra et al., 2008; Sobell et al., 2000 ; Tanner-Smith et al., 2013) to identify any additional papers, but none were added from that procedure. A final decision on inclusion was determined after reading the full paper, and any that raised potential questions on inclusion were reviewed by all authors, until consensus was reached. Studies by Copeland et al. (2001), Lozano et al. (2006), Kadden et al. (2007), Kay-Lambkin et al. (2009), Fernandes et al. (2010), Peters et al. (2011), Stein et al. (2011), Walker et al. (2011), Litt et al. (2013) and Hoch et al. (2014) were excluded due to an inability to calculate a within-group effect size on cannabis use per month from the data provided. The control groups of Stephens et al. (1994), Hendriks et al. (2011) and Budney et al. (2000) provided too much support for them to meet inclusion criteria as a control treatment condition.
Details of the eight included studies are displayed in Table 1, their results are provided in Table 2 and their methodological quality is summarized in Table 3. The studies had a total of 600 control participants. Average weighted mean days of use in the previous 30 days fell from 24.5 to 19.9 at 2–4 months across the eight studies. Only one of the included studies (Fischer et al., 2012) provided data over a longer follow-up, preventing an assessment of the degree of sustained change across the studies. That study saw little change in use at 12 months (M = 22.1, SD = 9.2).
| Author (date) | Sample type | Basis of participation | Disorder | Country | Control group | Measure |
|---|---|---|---|---|---|---|
| Stephens et al. (2000) | COM | Wanting help quitting | 98% current CUD | US | Delayed treatment | # days used cannabis per month |
| Litt et al. (2005) | COM | Treatment | 100% current CUD | US | Delayed treatment | % days used cannabis in the past 90 |
| Walker et al. (2006) | SCH | Information re their CU | 68% current CUD (86% lifetime CUD) | US | Delayed treatment | # days used cannabis in the past 60 |
| Stephens et al. (2007) | COM | Feedback on CU (not treatment) | 93% current CUD | US | Delayed feedback | # days used cannabis per week |
| Martin and Copeland (2008) | COM + OP | Information, discussion | 85% CUD | AU | Delayed treatment | # days used cannabis in the past 90 |
| Fischer et al. (2012) | UNI | …1 | CU | CAN | General health information | # days used cannabis in the past 30 |
| Gates et al. (2012) | COM | Information or counselling on CU concerns | 98% probable CUD on SDS | AU | Delayed treatment | # days used cannabis in the past 28 |
| Rooke et al. (2013) | COM | Wanting to reduce or cease CU | CU | AU | Cannabis information | # days used cannabis in past month |
AU: Australia; CAN: Canada; US: United States of America;
OP: Outpatients; COM: Community; HM: Homeless/unstably housed; SCH: School; UNI: University;
CU: Cannabis use; CUD: Cannabis use disorder (DSM-IIR or DSM-IV Cannabis Dependence or Abuse);
SDS: Severity of Dependence Scale (Gossop et al., 1992).
1. Mass advertising described the intervention study. Specific details on the basis of participation are not provided.
| Study | Baseline | 2–4 months | ||||
|---|---|---|---|---|---|---|
| N | M | SD | N | M | SD | |
| Stephens et al. (2000) | 86 | 24.9 | 6.1 | 79 | 17.1 | 10.7 |
| Litt et al. (2005)1 | 148 | 30.0 | 4.7 | 148 | 25.2 | 10.2 |
| Walker et al. (2006)2 | 50 | 18.4 | 8.5 | 50 | 16.4 | 10.3 |
| Stephens et al. (2007)3 | 64 | 26.0 | 8.2 | 64 | 24.6 | 8.2 |
| Martin and Copeland (2008)4 | 20 | 18.5 | 10.5 | 20 | 18.2 | 10.5 |
| Fischer et al. (2012) | 32 | 23.9 | 6.1 | 32 | 23.1 | 6.9 |
| Gates et al. (2012)5 | 81 | 23.9 | 6.3 | 61 | 13.4 | 12.2 |
| Rooke et al. (2013) | 119 | 20.8 | 8.7 | 58 | 14.1 | 8.8 |
| Total N, weighted mean | 600 | 24.5 | 512 | 19.9 | ||
Conversion formulae from reported means (M) to give days of use in the past 30 days:.
1. % days used in past 90: M × 30.
2. Days used in past 60: M/2.
3. Days per week: (M/7) × 30.
4. Days used in past 90: M/3.
5. Days used in past 28: (M/28) × 30.
| Study | Symptom/diagnostic measure | Treatment received by controls | Follow-up retention | Intention to treat (and management of missing data) | Single-blind follow-up |
|---|---|---|---|---|---|
| Stephens et al. (2000) | CUD: Self-report CU: Self & collateral report | BL: No current formal treatment 4 mths: 6% had treatment 18% in self-help groups | 92% to 4 mths | No | No—Self-report (phone interview if no response) |
| Litt et al. (2005) | CUD: SCID CU: TLFB, Self & collateral report, urinalysis. | BL: No current Mj therapy, self-help group 4 mths: NR | 93% to 4 mths | No (Secondary analyses: BL substitution) | No |
| Walker et al. (2006) | CUD: GAIN CU: Self-report | NR | 98% to 3 mths | No | NR—Self-report; different staff at follow-up |
| Stephens et al. (2007) | CUD: SCID CU: TLFB, self-report, urinalysis. | BL: No current Mj therapy, self-help group At 7-wks, 6 & 12 mths: 1–4% of whole sample in treatment 2–7% in self-help groups | 97% at 7 wks | Yes (BL substitution. Checked with imputation, omission) | No |
| Martin and Copeland (2008) | CUD: Structured interview (GAIN) & self-report (SDS) CU: TLFB, self-report, urinalysis. | BL: No treatment in previous 90 days 3 mths: NR | 80% at 3 mths | Yes (BL substitution) | NR (Independent researcher) |
| Fischer et al. (2012) | CU: Interviewer-administered questionnaire | NR | 52% at 12 mths | No Analysed completers of all assessments | NR |
| Gates et al. (2012) | Probable CUD: SDS CU: TLFB, self-report. | BL: No current Mj therapy 3 mths: 46% sought treatment, 39% used medication | 75% at 3 mths | Yes (Multiple imputation) | No |
| Rooke et al. (2013) | CUD: GAIN CU: TLFB, Self-report | BL: No formal Mj treatment in last 3 mths 3 mths: Excluded 4% who had treatment | 66% at 6 wks 52% at 3 mths | No (Complier average causal effect analyses. Checked with LOCF, omission) | Automated self-report |
CU: Cannabis use; CUD: Cannabis Use Disorder;
SCID: Structured Interview for DSM-IV; GAIN: Global Appraisal of Individual Needs (Initial or final) (Dennis, 1998 ; Dennis, 1999);
TLFB: Timeline Follow-Back; SDS: Severity of Dependence Scale (Gossop et al., 1992);
LOCF: Last observation carried forward; BL: Baseline; Mj: Marijuana; NR: Not Reported.
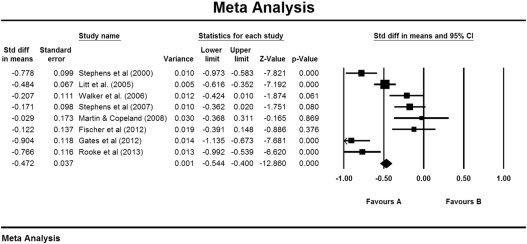
Results of the meta-analysis using a test–retest correlation of .70 are displayed in Fig. 1. The figure displays the average effect using a fixed-effects model. With a random effects model, there was an average change in cannabis use of −.442 SD (CI: −.657 to −.228), which was highly significant (p < .001). A test of heterogeneity gave Q (7) = 57.71, p < .001, providing support for the selection of the random effects model. Examination of the classic fail-safe N found that 293 missing studies would be required to give p > .05. Sensitivity analyses using random-effects models and test–retest correlations of .60 (−.460, CI: −.685 to −.235) and .80 (−.415, CI: −.613 to −.217) made little difference to the result.
|
|
|
Fig. 1. Control group changes over 2–4 months in non-psychotic groups. |
Four studies specifically targeted people who wanted to quit or reduce cannabis use, or recruited them for an intervention trial (Table 1). The remainder recruited people for information or discussion about their cannabis use. Inspection of Fig. 1 shows that the latter group included three of the four studies with weaker effects (Martin and Copeland, 2008; Stephens et al., 2007 ; Walker et al., 2006), but it also included the trial with the strongest effects (Gates et al., 2012).
An evaluation of the methodological quality of the control group data is in Table 2. A strength of the studies was their follow-up rates over the control period, with six having rates of 75% or above and four having rates above 90%. None clearly had single-blind follow-up, but two studies had an independent assessor conducting the follow-up, and three used only self-report. Four studies checked participant reports of cannabis use during follow-up against collateral data or urinalysis. All but two studies verified that most participants had a cannabis use disorder, although only two used a gold-standard structured clinical interview. A significant potential threat to the interpretation of results as being reflective of unassisted recovery was the lack of reports on other concurrent treatment in four trials, and a high level of reported treatment in one (Gates et al., 2012). Every study had at least one significant issue that should induce caution in the interpretation of its results.
4. Discussion
Control groups from the eight randomised controlled trials showed a significant mean reduction in days of cannabis use. At 2–4 months' follow up, participants used cannabis on 4.6 fewer days a month than at baseline, reflecting over one additional day of abstinence each week, and giving a total of more than a week of total abstinence each month. The average effect size of −.415 to −.442 SD offers a challenging base from which treatment effects are to be obtained. Our results will assist in minimum sample size calculations for randomised controlled trials, and provide a yardstick for the evaluation of changes from services for cannabis misuse.
While we regard these self-initiated changes as substantial, they fall short of major changes in sustained cessation, which supports the contention that self-initiated cannabis cessation is difficult. This observation is consistent with a comparison of reviews on placebo interventions for different substances by Moore and Aubin (2012), which found that nicotine provided the lowest abstinence rate (8%), followed by cannabis (15%) and alcohol (18%), with opioids (44%) and cocaine (47%) providing the strongest responses. While the meta-review potentially resulted in multiple counting of trials and there were few cannabis trials in a single included review, the study highlighted the limited nature of cannabis cessation rates, even in placebo conditions.
Interpretation of our results must be moderated by the issues raised in our methodological review of the studies, which identified at least one significant limitation in every study. Perhaps most important was the potential for other treatment to have been responsible for at least some of the observed reductions in cannabis use. The results highlight areas for future improvement of randomised controlled trials on cannabis use disorder that will not only provide increased confidence in the estimates of change in control groups, but also in the reported outcomes of the whole trial.
While there has been research into unassisted cessation of substance misuse for more than 40 years (Carballo et al., 2007 ; Sobell et al., 2000), it is only in the last 15 that this work has focused specifically on cannabis. To our knowledge, the current review is the first to examine ‘natural recovery’ in the control groups of randomised controlled trials. Regression to the mean may account for some of the observed change, but our results are consistent with population studies (Agosti and Levin, 2007 ; Perkonigg et al., 1999), which have similarly observed the potential for recovery from both cannabis use and cannabis dependence, suggesting that at least some individuals can reduce their cannabis use without significant help.
A limitation of this review was the fact that the initial literature search relied on one author, although the resolution of any identified issues and final decisions on inclusion were by consensus of all authors, and no additional papers were identified from reviews. Other limitations included the small number of identified trials with control groups that had no or minimal treatment, and the fact that minimal treatment controls can typically be conducted for periods of only 2–4 months at most. We excluded eight studies because of an absence of data on cannabis consumption over a specific period, in order to preserve comparability of the results across studies: if those studies had provided consumption data, we could potentially have doubled the number of studies in our review. We recommend that future studies routinely include both abstinence rates and average consumption data as part of their results (Peters et al., 2011). However, despite the restricted number of studies, the total sample size of 600 provided a substantial group for estimation of consumption changes.
5. Conclusions
This is the first meta-analysis to explore changes in cannabis use in control conditions of treatment studies. Results of the current study demonstrate that modest average reductions in the frequency of average cannabis use can be seen, although there was substantial variability in effect size between studies, and some uncertainty over the potential role of outside treatment in the effects. The study gives weight to further exploration of the concept of natural recovery in people with cannabis use disorders and provides researchers and practitioners a baseline from which to estimate likely changes or needed effects sizes in intervention studies.
References
- Agosti and Levin, 2007 V. Agosti, F.R. Levin; Predictors of cannabis dependence recovery among epidemiological survey respondents in the United States; The American Journal of Drug and Alcohol Abuse, 33 (2007), pp. 81–88
- Australian Institute of Health and Welfare, 2014 Australian Institute of Health and Welfare; National drug strategy household survey detailed report 2013; AIHW, Canberra (2014)
- Borenstein et al., 2005 M. Borenstein, L. Hedges, J. Higgins, H. Rothstein; Comprehensive meta-analysis (version 2); Biostat, Englewood, NJ (2005)
- Borenstein et al., 2009 M. Borenstein, L. Hedges, J. Higgins, H. Rothstein; Introduction to meta-analysis; Wiley, Chichester (2009)
- Budney et al., 2000 A.J. Budney, S.T. Higgins, K.J. Radonovich, P.L. Novy; Adding voucher-based incentives to coping skills and motivational enhancement improves outcomes during treatment for marijuana dependence; Journal of Consulting and Clinical Psychology, 68 (2000), pp. 1051–1061
- Carballo et al., 2007 J.L. Carballo, J.R. Fernandez-Hermida, R. Secades-Villa, L.C. Sobell, M. Dum, O. Gracia-Rodriguez; Natural recovery from alcohol and drug problems: A methodological review of the literature from 1999 through 2005; H.K.H. Klingemann, L.C. Sobell (Eds.), Promoting self-change from addictive behaviors: Practical implications for policy, prevention, and treatment, Springer, New York (2007), pp. 87–101
- Chen and Kandel, 1998 K. Chen, D.B. Kandel; Predictors of cessation of marijuana use: An event history analysis; Drug and Alcohol Dependence, 50 (1998), pp. 109–121
- Copeland et al., 2001 J. Copeland, W. Swift, R. Roffman, R. Stephens; A randomized controlled trial of brief cognitive–behavioral interventions for cannabis use disorder; Journal of Substance Abuse Treatment, 21 (2001), pp. 55–64
- Cunningham, 2000 J.A. Cunningham; Remissions from drug dependence: Is treatment a prerequisite?; Drug and Alcohol Dependence, 59 (2000), pp. 65–76
- Dennis, 1998 M.L. Dennis; Global appraisal of individual needs (GAIN) manual: Administration, scoring and interpretation; Lighthouse, Bloomington, IL (1998)
- Dennis, 1999 M.L. Dennis; Global appraisal of individual needs (GAIN) manual; Chestnut Health Systems, Bloomington, IL (1999)
- Dutra et al., 2008 L. Dutra, G. Stathopoulou, S.L. Basden, T.M. Leyro, M.B. Powers, M.W. Otto; A meta-analytic review of psychosocial interventions for substance use disorders; The American Journal of Psychiatry, 165 (2008), pp. 179–187
- Fernandes et al., 2010 S. Fernandes, M. Ferigolo, M.C. Benchaya, T. Moreira, P.S. Pierozan, C.G. Mazoni, H.M.T. Barros; Brief motivational intervention and telemedicine: A new perspective of treatment to marijuana users; Addictive Behaviors, 35 (2010), pp. 750–755
- Fischer et al., 2012 B. Fischer, W. Jones, P. Shuper, J. Rehm; 12-month follow-up of an exploratory ‘brief intervention’ for high-frequency cannabis users among Canadian University students; Substance Abuse Treatment, Prevention, and Policy, 7 (2012), pp. 15–20
- Gates et al., 2012 P.J. Gates, M.M. Norberg, J. Copeland, E. Digiusto; Randomized controlled trial of a novel cannabis use intervention delivered by telephone; Addiction, 107 (2012), pp. 2149–2158
- Gossop et al., 1992 M. Gossop, P. Griffiths, B. Powis, J. Strang; Severity of dependence and route of administration in heroin, cocaine and amphetamines; British Journal of Addiction, 87 (1992), pp. 1527–1536
- Hendriks et al., 2011 V. Hendriks, E. van der Schee, P. Blanken; Treatment of adolescents with a cannabis use disorder: Main findings of a randomized controlled trial comparing multidimensional family therapy and cognitive behavioral therapy in The Netherlands; Drug and Alcohol Dependence, 119 (2011), pp. 64–71
- Hoch et al., 2014 E. Hoch, G. Buhringer, A. Pixa, K. Dittmer, J. Henker, A. Seifert, H.U. Wittchen; CANDIS treatment program for cannabis use disorders: Findings from a randomized multi-site translational trial; Drug and Alcohol Dependence, 134 (2014), pp. 185–193
- Kadden et al., 2007 R.M. Kadden, M.D. Litt, E. Kabela-Cormier, N.M. Petry; Abstinence rates following behavioral treatments for marijuana dependence; Addictive Behaviors, 32 (2007), pp. 1220–1236
- Kandel and Raveis, 1989 D.B. Kandel, V.H. Raveis; Cessation of illicit drug use in young adulthood; Archives of General Psychiatry, 46 (1989), pp. 109–116
- Kay-Lambkin et al., 2009 F.J. Kay-Lambkin, A.L. Baker, T.J. Lewin, J.A.R. Carr; Computer-based psychological treatment for comorbid depression and problematic alcohol and/or cannabis use: A randomized controlled trial of clinical efficacy; Addiction, 104 (2009), pp. 378–388
- Litt et al., 2005 M.D. Litt, R.M. Kadden, R. Stephens; Coping and self-efficacy in marijuana treatment: Results from the marijuana treatment project; Journal of Consulting and Clinical Psychology, 73 (2005), pp. 1015–1025
- Litt et al., 2013 M.D. Litt, R.M. Kadden, N.M. Petry; Behavioral treatment for marijuana dependence: Randomized trial of contingency management and self-efficacy enhancement; Addictive Behaviors, 38 (2013), pp. 1764–1775
- Lozano et al., 2006 B.E. Lozano, R.S. Stephens, R.A. Roffman; Abstinence and moderate use goals in the treatment of marijuana dependence; Addiction, 101 (2006), pp. 1589–1597
- Martin and Copeland, 2008 G. Martin, J. Copeland; The adolescent cannabis check-up: Randomized trial of brief intervention for young cannabis users; Journal of Substance Abuse Treatment, 34 (2008), pp. 407–414
- Moore and Aubin, 2012 R.A. Moore, H. Aubin; Do placebo response rates from cessation trials inform on strength of addictions?; International Journal of Environmental Research and Public Health, 9 (2012), pp. 192–211
- Perkonigg et al., 1999 A. Perkonigg, R. Lieb, M. Höfler, P. Schuster, H. Sonntag, H.-U. Wittchen; Patterns of cannabis use, abuse and dependence over time: Incidence, progression and stability in a sample of 1228 adolescents; Addiction, 94 (1999), pp. 1663–1678
- Peters et al., 2011 E.N. Peters, C. Nich, K.M. Carroll; Primary outcomes in two randomized controlled trials of treatments for cannabis use disorders; Drug and Alcohol Dependence, 118 (2011), pp. 408–416
- Pollard et al., 2014 M.S. Pollard, J.S. Tucker, K. de la Haye, H.D. Green, D.P. Kennedy; A prospective study of marijuana use change and cessation amng adolescents; Drug and Alcohol Dependence, 144 (2014), pp. 134–140
- Price et al., 2001 R.K. Price, N.K. Risk, E.L. Spitznagel; Remission from drug abuse over a 25-year period: Patterns of remission and treatment use; American Journal of Public Health, 91 (2001), pp. 1107–1113
- Robinson et al., 2014 S.M. Robinson, L.C. Sobell, M.B. Sobell, G.I. Leo; Reliability of the timeline followback for cocaine, cannabis and cigarette use; Psychology of Addictive Behaviors, 28 (2014), pp. 154–162
- Rooke et al., 2013 S. Rooke, J. Copeland, M. Norberg, D. Hine, J. McCambridge; Effectiveness of a self-guided web-based cannabis treatment program: Randomized controlled trial; Journal of Medical Internet Research, 15 (2013) e26
- Sobell et al., 2000 L.C. Sobell, T.P. Ellingstad, M.B. Sobell; Natural recovery from alcohol and drug problems: Methodological review of the research with suggestions for future directions; Addiction, 95 (2000), pp. 749–764
- Stein et al., 2011 M.D. Stein, C.E. Hagerty, D.S. Herman, M.G. Phipps, B.J. Anderson; A brief marijuana intervention for non-treatment-seeking young adult women; Journal of Substance Abuse Treatment, 40 (2011), pp. 189–198
- Stephens et al., 1994 R.S. Stephens, R.A. Roffman, E.E. Simpson; Treating adult marijuana dependence. A test of the relapse prevention model; Journal of Consulting and Clinical Psychology, 62 (1994), pp. 92–99
- Stephens et al., 2000 R.S. Stephens, R.A. Roffman, L. Curtin; Comparison of extended versus brief treatments for marijuana use; Journal of Consulting and Clinical Psychology, 68 (2000), pp. 898–908
- Stephens et al., 2007 R.S. Stephens, R.A. Roffman, S.A. Fearer, C. Williams, R.S. Burke; The marijuana check-up: Promoting change in ambivalent marijuana users; Addiction, 102 (2007), pp. 947–957
- Sussman and Dent, 2004 S. Sussman, C.W. Dent; Five-year prospective prediction of marijuana use cesation of youth at continuation high schools; Addictive Behaviors, 29 (2004), pp. 1237–1243
- Tanner-Smith et al., 2013 E.E. Tanner-Smith, S.J. Wilson, M.W. Lipsey; The comparative effectiveness of outpatient treatment for adolescent substance abuse: A meta-analysis; Journal of Substance Abuse Treatment, 44 (2013), pp. 145–158
- United Nations Office on Drugs and Crime, 2014 United Nations Office on Drugs and Crime; World drug report; United Nations, New York (2014)
- Walker et al., 2006 D.D. Walker, R.A. Roffman, R.S. Stephens, J. Berghuis, W. Kim; Motivational enhancement therapy for adolescent marijuana users: A preliminary randomized controlled trail; Journal of Consulting and Clinical Psychology, 74 (2006), pp. 628–632
- Walker et al., 2011 D.D. Walker, R. Stephens, R. Roffman, J. Demarce, B. Lozano, S. Towe, B. Berg; Randomized controlled trial of motivational enhancement therapy with nontreatment-seeking adolescent cannabis users: A further test of the teen marijuana check-up; Psychology of Addictive Behaviors, 25 (2011), pp. 474–484
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?