Summary
Background
Diaphragm plication surgery is conducted to remove dyspnea, which results from mediastinal shift, atelectasia, and ventilation/perfusion dyssynchrony in lungs that occur because of an eventrated diaphragm. This study aims to determine whether diaphragm plication has any effect on respiration by analyzing the patients' changing values in the respiratory function test (RFT) after plication surgery.
Methods
Sixteen patients who underwent diaphragm plication surgery in our clinic because of plication eventration or paralysis were examined prospectively. Diaphragm eventration values were assessed using a calculation method that uses posteroanterior pulmonary radiographies taken during patient admission and control; then, these data were recorded. The amount of changes in the eventration levels and in restrictive respiratory failure parameters—forced expiratory volume in 1 second (FEV1), forced vital capacity (FVC) of RFTs—conducted in pre- and postoperative control periods were compared using statistical analysis methods. The compatibility between the amounts of RFT changes was examined through a satisfaction survey—using a questionnaire that consisted of multiple choice questions with answer options such as “better,” “the same,” and “worse”—to understand preoperative and postoperative symptom levels in the 12th month of postoperative control.
Results
According to postoperative levels, a decrease between 19% and 23% was observed in eventration amounts within the 1st postoperative month, 6th postoperative month, and 12th postoperative month. In addition, the highest average increase in FEV1 liter (lt) values was 0.2 lt and 0.25 in FVC (lt) values.
Conclusion
Researchers of this study believe that more distinctive decisions need to be taken while identifying patients for surgery in unilateral diaphragm eventrations, especially in the adult patient group; surgical option should be used for cases in which the eventrated diaphragm results in mediastinal shift and respiratory failure.
Keywords
Diaphragm;Eventration;Paralysis;Plication
1. Introduction
During calm respiration, approximately 75–80% of air is taken into the lungs because of diaphragm fibrillation. Each 1-cm vertical action of the diaphragm leads to shifting of 300–400 cm3 air.
Previous cardiac–thorax mediastinum surgery, trauma, neuromuscular diseases, infective cases, and malignity are believed to be causes of diaphragm eventration. In idiopathic cases, mostly viral factors are the main sources. Unilateral diaphragm eventration/paralysis does not necessarily exhibit any clear symptom in adults; however, bilateral diaphragm paralysis leads to the development of respiratory failure.
Although eventration manifests itself via respiratory distress, it is not symptomatic in every case. It is detected incidentally in a pulmonary radiography taken for another disease. So far, most of the studies on diaphragm eventration and surgical treatment offer subjective results. Some analytic studies have raised discussions about indications of surgical treatment. In this study, a total of 16 individuals who underwent an operation in our clinic because of diaphragm eventration and followed up for 1 year were examined prospectively. The eventration level of individuals was determined with the calculation method of the study group. The obtained results were compared with the values of restrictive respiratory failure parameters: FEV1 (forced expiratory volume in 1 second) and FVC (forced vital capacity). The data compiled from these results were evaluated within the scope of the existing literature.
2. Material and methods
A total of 16 patients who underwent diaphragm plication surgery in our clinic because of plication eventration or paralysis were examined prospectively between March 2009 and December 2012. Diaphragm eventration levels were calculated via a new calculation method through posteroanterior (PA) pulmonary radiographs taken during patient admission. Hemidiaphragm positions were detected by PA pulmonary radiography during patient control periods performed 1 months, 6 months, and 12 months after the operation. Through this method, the difference between preoperative and postoperative diaphragm levels was recorded. The amount of change in eventration levels and in restrictive respiratory failure parameters (FEV1, FVC) of respiratory function tests conducted during pre- and postoperative control periods were compared using statistical analysis methods. In this way, the effect levels of eventration changes on RFT were assessed. A survey using the symptomatic condition questionnaire—which featured answer options such as “better,” “the same,” and “worse” toward preoperative and postoperative symptom levels in the 12th month of postoperative control—was conducted. The compatibility between responses and the amounts of RFT changes was examined.
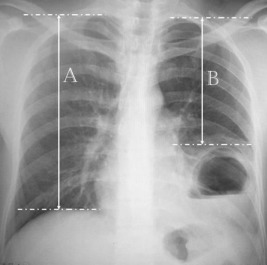
Using PA pulmonary radiography, the distance between the peak of the intactus hemidiaphragm and apex point of the ipsilateral hemithorax was measured with this new method. (1) Similarly, the distance between the peak of the eventrated hemidiaphragm and the apex point of ipsilateral hemithorax was measured. (2) The ratio of B/A and eventration level was calculated as a percentage ( Figure 1).
|
|
|
Figure 1. Preoperative chest X-ray. |
Example:
A = 25 cm, B = 20 cm
|
|
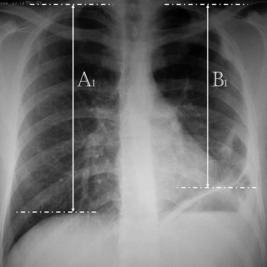
In the postoperative period, measurement was made using the same formula according to the last position of the plicated hemidiaphragm. The difference between postoperative and preoperative values was calculated, and the contribution of the plication operation on the diaphragm level was calculated (Figure 2).
|
|
|
Figure 2. Postoperative chest X-ray. |
Example:
A1 = 25 cm, B1 = 22 cm
|
|
Diaphragm eventration difference in preoperative and postoperative period: 8%.
2.1. Surgical technique
Diaphragmatic plication surgery was performed in all cases via posterolateral thoracotomy except in one case, for which a minimally invasive technique was used. During thoracotomy, the seventh or eighth intercostal space was chosen to enter the thoracic cavity and diaphragmatic eventration was observed. We started the procedure on the central tedious part of the diaphragm using 0 silk sutures; seven to nine lines of silk sutures were used. From the posteromedial to the anterolateral direction and by crossing the phrenical nerve fibers, six to seven consecutive stitches at each line were performed. These sutures were hanged, and parallel plication was performed. Finally, after knotting the sutures, diaphragmatic repositioning was added.
The study was approved by the Ataturk Training and Research Hospital for Chest Disease and Chest Surgery, Ankara, Ethical Committee.
SPSS data analysis (SPSS Inc., Chicago, IL, USA) was used to compare preoperative and postoperative spirometric data of the 16 cases, and the data were statistically evaluated using Wilcoxon test.
3. Results
The average age of the 16 patients was 51.5 (29–70) years. Overall, 12 males and four females participated in the study; 15 of the cases had left diaphragm eventration and one had right diaphragm eventration. All patients had respiratory distress, and two had coughing, two had chest pain, and one also had constipation. Findings related to etiology were not found in 10 cases. Three cases had in-car traffic accident, one had cardiac surgery, one had thorax surgery, and one had a penetrating injury from the thoracic region in the past. Thoracotomy + central plication was applied to 15 cases and video-assisted thoracoscopic surgery + single plication was applied to one case. In the postoperative period, atelectasia developed in three cases and pleural effusion developed in one case. No postoperative complication was observed in 12 cases. The average duration of the hospital stay was 5.8 (4–8) days (Table 1).
| Case | Sex | Age | Side | Symptoms | Complication | Operation | Etiology |
|---|---|---|---|---|---|---|---|
| 1 | M | 40 | Left | D–C | Left Tho–CP | Chest surgery | |
| 2 | M | 48 | Left | D | Left Tho–CP | Cardiac surgery | |
| 3 | M | 57 | Left | D | Left Tho–CP | ||
| 4 | M | 50 | Left | D | Left Tho–CPl | BCT | |
| 5 | M | 35 | Left | D–CP | Atelectasis | Left Tho–CPl | PCT |
| 6 | F | 50 | Left | D | Left Tho–CPl | ||
| 7 | F | 70 | Right | D | Atelectasis | Right Tho–CPl | |
| 8 | F | 54 | Left | D–CP | Atelectasis | Left Tho–CPl | |
| 9 | M | 56 | Left | D | Left VATS–SPl | ||
| 10 | M | 52 | Left | D–Co | Effusion | Left Tho–CPl | |
| 11 | M | 57 | Left | D–C | Left Tho–CPl | BCT | |
| 12 | M | 59 | Left | D | Left Tho–CPl | ||
| 13 | M | 62 | Left | D | Left Tho–CPl | ||
| 14 | M | 29 | Left | D | Left Tho–CPl | ||
| 15 | M | 45 | Left | D | Left Tho–CPl | ||
| 16 | F | 61 | Left | D | Left Tho–CPl | BCT |
BCT = blunt chest trauma; C = cough; Co = constipation; CP = chest pain; CPl = central plication; D = dyspnea; PCT = penetrating chest trauma; SPl = single plication, Tho = thoracotomy; VATS = video-assisted thoracoscopic surgery.
The mean of preoperative FEV1 (lt) values among the 16 cases was calculated as 1.87 lt (1.05–2.95 lt), 2.03 lt (1.16–2.87 lt) in the 1st postoperative month, 2.08 lt (1.12–2.78 lt) in the 6th month, and 1.99 lt (1.09–2.63 lt) in the 12th month. Compared to preoperative values, a decrease was found in FEV1 (lt) values of three cases in the 1st month, of four cases in the 6th month, and of six cases in the 12th month of postoperative control. Compared to preoperative values, no change was found in FEV1 (lt) values of two cases in the 1st month, of two cases in the 6th month, and of one case in the 12th month of postoperative control (Table 2).
| Case | Preoperative | Postoperative 1 mo | Postoperative 6 mo | Postoperative 12 mo | Symp. | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| FEV1 | FVC | E | FEV1 | FVC | E | FEV1 | FVC | E | FEV1 | FVC | E | ||||||||||
| lt | % | lt | % | % | lt | % | lt | % | % | lt | % | lt | % | % | lt | % | lt | % | % | ||
| 1 | 1.52 | 50 | 2.15 | 56 | 23.8 | 1.64 | 54 | 2.36 | 62 | 15.3 | 1.59 | 53 | 2.54 | 66 | 13.3 | 1.49 | 50 | 2.05 | 53 | 16 | S |
| 2 | 1.82 | 77 | 2.22 | 79 | 37.6 | 1.66 | 70 | 1.80 | 64 | 5.7 | 2.34 | 96 | 2.45 | 85 | 20 | 1.91 | 82 | 2.20 | 80 | 21 | S |
| 3 | 2.95 | 77 | 4.17 | 90 | 26.9 | 2.48 | 65 | 3.20 | 69 | 15.7 | 2.32 | 60 | 3.19 | 69 | 15.1 | 2.55 | 68 | 3.46 | 76 | 12.5 | S |
| 4 | 1.65 | 54 | 2.18 | 57 | 36 | 1.96 | 65 | 2.68 | 70 | 8.7 | 2.30 | 76 | 3.07 | 81 | 11.1 | 1.95 | 68 | 2.69 | 75 | 13.2 | S |
| 5 | 1.59 | 66 | 2.15 | 77 | 34.4 | 1.44 | 58 | 1.68 | 58 | 14.9 | 1.36 | 57 | 1.74 | 62 | 15 | 1.11 | 45 | 1.48 | 51 | 23 | B |
| 6 | 2.46 | 62 | 3.15 | 67 | 33.5 | 2.74 | 68 | 3.15 | 67 | 12.5 | 2.67 | 67 | 3.42 | 73 | 7.8 | 2.43 | 63 | 3.07 | 66 | 14.3 | B |
| 7 | 1.67 | 51 | 2.03 | 50 | 20 | 1.68 | 52 | 2.20 | 54 | −10.7 | 2.45 | 76 | 3.21 | 79 | −8.3 | 2.63 | 83 | 3.17 | 79 | −4.3 | W |
| 8 | 2.13 | 59 | 2.67 | 60 | 31.4 | 2.58 | 70 | 3.07 | 68 | 14.7 | 2.04 | 55 | 2.46 | 54 | 18.8 | 2.01 | 55 | 2.28 | 50 | 18.1 | B |
| 9 | 1.91 | 59 | 2.79 | 68 | 45.6 | 2.21 | 70 | 3.15 | 80 | 0 | 2.40 | 94 | 3.50 | 117 | 4.5 | 2.21 | 74 | 3.04 | 80 | 13.4 | B |
| 10 | 1.05 | 64 | 1.42 | 71 | 50 | 1.16 | 73 | 1.42 | 73 | 36.9 | 1.16 | 71 | 1.43 | 72 | 44.9 | 1.09 | 80 | 1.15 | 68 | 33.5 | S |
| 11 | 2.13 | 66 | 2.66 | 67 | 32.6 | 2.23 | 69 | 2.70 | 68 | 13.7 | 2.17 | 67 | 2.56 | 64 | 15.1 | 2.42 | 79 | 3.12 | 83 | 13.1 | B |
| 12 | 2.13 | 60 | 2.52 | 57 | 26 | 2.87 | 82 | 3.20 | 70 | −11.1 | 2.78 | 79 | 3.16 | 73 | −16.7 | 2.45 | 72 | 2.77 | 65 | −5.7 | W |
| 13 | 2.12 | 55 | 2.28 | 50 | 34.6 | 2.58 | 68 | 2.86 | 63 | 11.2 | 2.46 | 64 | 2.71 | 59 | 12.3 | 2.30 | 62 | 2.65 | 57 | 15.2 | B |
| 14 | 1.61 | 47 | 2.29 | 55 | 18.7 | 1.89 | 55 | 2.52 | 61 | −7.4 | 1.92 | 58 | 2.71 | 64 | −5.3 | 1.85 | 56 | 2.63 | 63 | 0 | S |
| 15 | 1.93 | 62 | 2.60 | 67 | 23.5 | 1.98 | 63 | 2.90 | 74 | 5.6 | 2.24 | 74 | 3.58 | 96 | 4.9 | 2.38 | 77 | 3.72 | 94 | 5.2 | B |
| 16 | 1.31 | 58 | 1.44 | 55 | 23.5 | 1.42 | 63 | 1.57 | 59 | 2.9 | 1.12 | 46 | 1.14 | 40 | 1.4 | 1.21 | 51 | 1.35 | 43 | 1.9 | W |
| Mean | 1.87 | 60.4 | 2.42 | 64.1 | 31.1 | 2.03 | 65.3 | 2.51 | 66.2 | 8.03 | 2.08 | 68.3 | 2.67 | 72.1 | 9.6 | 1.99 | 66.5 | 2.55 | 67.6 | 11.9 | |
B = better; E = eventration; FEV1 = forced expiratory volume in 1 second; FVC = forced vital capacity; lt = liter; S = same; Symp. = symptom; W = worse.
The mean of preoperative FEV1 (%) values of the 16 cases was calculated as 60.4 (47–77), 65.3 (52–82) in the 1st postoperative month, 68.3 (46–96) in the 6th month, and 66.5 (45–83) in the 12th month. Compared to preoperative values, a decrease was found in FEV1 (%) values of three cases in the 1st month, of four cases in the 6th month, and of four cases in the 12th month of postoperative control. Compared to preoperative values, no change was found in FEV1 (%) values of one case in the 12th month of postoperative control (Table 2).
The mean of the preoperative FVC (lt) values of the 16 cases was calculated as 2.42 lt (1.42–4.17 lt), 2251 lt (1.42–3.20 lt) in the 1st postoperative month, 2.67 lt (1.14–3.58 lt) in the 6th month, and 2255 lt (1215–3272 lt) in the 12th month. Compared to preoperative values, a decrease was found in FVC (lt) values of three cases in the 1st month, of five cases in the 6th month, and of seven cases in the 12th month of postoperative control. Compared to preoperative values, no change was found in FVC (lt) values of two cases in the 1st month, of one case in 6th month, and of one case in the 12th month of postoperative control (Table 2).
The mean of preoperative FVC (%) values of the 16 cases was calculated as 64.1 (50–90), 66.2 (54–80) in the 1st postoperative month, 72.1 (40–117) in the 6th month and 67.6 (43–94) in the 12th month. Compared to preoperative values, a decrease was found in FVC (%) values of three cases in the 1st month, of five cases in the 6th month and of seven cases in the 12th month of postoperative control. Compared to preoperative values, no change was found in FVC (%) values of one case in the 1st month of postoperative control (Table 2).
The mean of preoperative eventration values of the 16 cases was calculated as 31.1% (18.7–50%), 8.03% (−11.1–36.9%) in the 1st postoperative month, 9.6% (−16.7–44.9%) in the 6th month, and 11.9% (−5.7–33.5%) in the 12th month (Table 3).
| Min. | Max. | Mean | SD | p | ||
|---|---|---|---|---|---|---|
| FEV1 (lt) | Preoperative | 1.05 | 2.95 | 1.8738 | 0.45681 | |
| Postoperative 1 mo | 1.16 | 2.87 | 2.0325 | 0.51673 | 0.04 | |
| Postoperative 6 mo | 1.12 | 2.78 | 2.0825 | 0.51528 | 0.03 | |
| Postoperative 12 mo | 1.09 | 2.63 | 1.9994 | 0.52149 | 0.17 | |
| FEV1 (%) | Preoperative | 47.00 | 77.00 | 60.4375 | 8.51641 | |
| Postoperative 1 mo | 52.00 | 82.00 | 65.3125 | 7.77791 | 0.04 | |
| Postoperative 6 mo | 46.00 | 96.00 | 68.3125 | 13.98437 | 0.04 | |
| Postoperative 12 mo | 45.00 | 83.00 | 66.5625 | 12.38800 | 0.06 | |
| FVC (lt) | Preoperative | 1.42 | 4.17 | 2.4200 | 0.64742 | |
| Postoperative 1 mo | 1.42 | 3.20 | 2.5175 | 0.61052 | 0.22 | |
| Postoperative 6 mo | 1.14 | 3.58 | 2.6794 | 0.72610 | 0.07 | |
| Postoperative 12 mo | 1.15 | 3.72 | 2.5519 | 0.75239 | 0.46 | |
| FVC (%) | Preoperative | 50.00 | 90.00 | 64.1250 | 11.23610 | |
| Postoperative 1 mo | 54.00 | 80.00 | 66.2500 | 6.67832 | 0.30 | |
| Postoperative 6 mo | 40.00 | 117.00 | 72.1250 | 17.68568 | 0.10 | |
| Postoperative 12 mo | 43.00 | 94.00 | 67.6875 | 14.24415 | 0.37 | |
| Eventration | Preoperative | 18.70 | 50.00 | 31.1313 | 8.77259 | |
| Postoperative 1 mo | −11.10 | 36.90 | 8.0375 | 11.99016 | ||
| Postoperative 6 mo | −16.70 | 44.90 | 9.6188 | 13.91513 | ||
| Postoperative 12 mo | −5.70 | 33.50 | 11.9000 | 10.35670 |
FEV1 = forced expiratory volume in 1 second; FVC = forced vital capacity; SD = standard deviation.
According to postoperative levels, a decrease between 19% and 23% was observed in eventration amounts within the 1st postoperative month, 6th postoperative month, and 12th postoperative month. In addition, the highest average increase in FEV1 (lt) values was 0.2 lt, and 0.25 in FVC (lt) values. According to the analysis on values of FEV1 and FVC (%), the highest average increase in FEV1 (%) values was 7.87%, and that in FVC (%) values was 8% (Table 4).
| FEV1 | FVC | Eventration | |||
|---|---|---|---|---|---|
| lt | % | lt | % | % | |
| Postoperative 1 mo | 0.15 | 4.87 | 0.09 | 2.12 | 23.09 |
| Postoperative 6 mo | 0.2 | 7.87 | 0.25 | 8 | 21.51 |
| Postoperative 12 mo | 0.09 | 6.12 | 0.13 | 3.56 | 19.23 |
FEV1 = forced expiratory volume in 1 second; FVC = forced vital capacity; lt = liter.
A survey using a three-option satisfaction questionnaire was conducted in all cases in order to compare the respiratory distress level perceived during the 12th month of postoperative control compared to the preoperative level. Of all cases, seven reported that the respiratory distress was removed following the operation; six reported that no change occurred in their symptoms, and three reported that there was an increase in respiratory distress levels compared to the preoperative period (Table 2).
4. Discussion
In this study, diaphragm eventration of all cases was unilateral and diffused, and 93.7% had left hemidiaphragm pathology. Furthermore, 62.5% were not diagnosed with any etiologic factor. One patient had a heart surgery, one had a pulmonary surgery, and four cases had thoracic trauma history.
One of the major symptoms of diaphragm paralysis or eventration is respiratory distress. In patients with eventration, the diaphragm function is either decreased or does not exist. In adults, tachycardia and dyspnea, which develop during rest or exertion, are the prominent complaints. The main feature of these complaints is that they often intensify with posture changes.1 Piehler et al2 examined 142 patients with diaphragm paralysis and found that the main symptoms of eventration are primarily respiratory distress, coughing, and chest pain. In parallel with this result, the present study found that all of our 16 cases had dyspnea, and they also had other symptoms such as coughing, chest pain, and constipation. Respiratory distress results from multifactorial changes in respiratory physiology involving atelectasia and ventilation/perfusion dyssynchrony arising from pulmonary blood flow toward the contralateral lung as a result of the paradoxical movement of the hemidiaphragm and mediastinal shift.3
In a standard PA and lateral chest radiography, 1–2 cm eventration of the right hemidiaphragm compared to the left is regarded as normal. The position of the intactus hemidiaphragm was considered a basal measurement point in the eventration measuring method using PA pulmonary radiography. It is inaccurate to say that a higher positioned right hemidiaphragm will cause errors in calculations. As the position of the left hemidiaphragm is lower than the right one in 95% of the general population, the position difference between hemidiaphragms can be statistically ignored. Similarly, the left or right hemithorax location of the eventration does not lead to any difference. We believe that the new method used in this study is more reliable because of the lack of difference between the postoperative and preoperative positions of the intact hemidiaphragm, and the proportional value of eventration that is calculated with this method.
The paradoxical movement of diaphragm plication and eventration and mediastinal shift can be prevented, and the capacity of tidal volume and maximal respiration is improved.4 The important point in plication is to obtain suture tension, which will maintain diaphragm tension. After the plication, the diaphragm detected in the lower position leads to the expansion of the hemithorax and improves the functional residual capacity. Frequently, a decrease is observed in FVC and FEV1, which are among the restrictive-type respiratory failure parameters in the diaphragm function disorder.5 In a study conducted on 17 patients by Graham et al,6 it was found that open transthoracic plication results in significant recovery in respiratory distress and RFT. Similarly, Higgs et al7 followed 15 patients for 10 years and reported that the open transthoracic approach is a suitable choice to improve RFT values, dyspnea scores, and patient satisfaction. Freeman et al8 reported a recovery in RFT values in a 25-case series. However, the follow-up period was restricted to 6 months. Ribets9 study found a 15% increase in FEV1 and a 20% increase in vital capacity following plication. In the present study, the cases were followed for 1 year, and their spirometric changes were examined. At the end of the 1-year follow-up period, it was found that although a 19% recovery was observed in eventration levels, 0.09 lt (6.12%) increase was found in FEV1 and 0.13 lt (3.56%) increase was found in FVC. The statistical analysis indicated that there was a significant increase in the 1st month and 6th month of postoperative FEV1 (lt) and (%) values compared to preoperative values. However, neither a statistically nor a clinically significant increase was observed in long-term results. In the analysis of FVC values, no statistical significance was found (Table 3).
In the analysis of the symptoms of cases in this study, it was found that symptoms did not change in six cases in parallel with spirometric values, and an increase was found in the symptoms of three cases compared to the preoperative period. The responses of eight cases were compatible with spirometric measurement values in symptom examination. In three patients who reported that their symptoms ceased, an increase was found in RFT values. In two patients who reported that their symptoms intensified, RFT values increased. It is clear that further studies are required to investigate whether the reason for the recovery from symptoms may have resulted from the decrease in paradoxical movements of the diaphragm contrary to the RFT values.
As regards plication operation in general, the first investigation on the indication of the operation was made by Svanberg10 in 1956. Since then, it has been argued whether or not the repair of diaphragm at the lower levels with plication is functional.10 Similarly, although in the research of Tiryaki et al11 paradoxical movements in the diaphragm and recurrence in postoperative follow-up were not observed, symptom recurrence was observed in the cases. Mulvey et al12 followed 12 cases for 2–4 years and found no clear change in the spirometric measurements of pulmonary volumes. According to a research investigating 17 diaphragm plication cases with a wide and lengthy follow-up period, which was reported at the 2006 European Association for Cardio-Thoracic Surgery (EACTS)/European Society of Thoracic Surgeons (ESTS) congress, the postoperative mean FEV1 value was 71%, an increase of 64% from the preoperative level.13 In the present study, an increase was found in RFT values in nine (56.2%) cases compared to preoperative values. However, considering that the mean increase was 0.09 lt for FEV1 and 0.13 lt FVC, it is evident that the discussions on the indication of diaphragm plication operation are justified.
Literature reviews show that most of the studies on diaphragm eventration and surgical treatment offer subjective results based on patient satisfaction. Some analytic studies have brought forward discussions regarding indications of surgical treatment to the agenda. However, the common point of all these studies is that the eventration levels of operated cases are unknown. We hope that the present results, which were obtained through a new calculation formula toward the detection of the eventration level with digital data, will bring a new dimension to the existing discussions about surgical indications.
We believe that more selective decisions should be taken while determining whether patients are appropriate for surgery in unilateral diaphragm eventrations, especially in the adult patient group; plication operations should not be used for low-level diaphragm eventrations that do not result in mediastinal shift.
References
- 1 T.V. Thomas; Congenital eventration of the diaphragm; Ann Thorac Surg, 10 (1970), pp. 180–192
- 2 J.M. Piehler, P.C. Pairolero, D.R. Gracey, P.E. Bernatz; Unexplained diaphragmatic paralysis: a harbinger of malignant disease?; J Thorac Cardiovasc Surg, 84 (1982), pp. 861–864
- 3 M.Z. Schwartz, R.M. Filler; Plication of the diaphragm for symptomatic phrenic nerve paralysis; J Pediatr Surg, 13 (1978), pp. 259–263
- 4 K.S. Stone, J.W. Brown, D.F. Canal, H. King; Long-term fate of the diaphragm surgically plicated during infancy and early childhood; Ann Thorac Surg, 44 (1987), pp. 62–65
- 5 J.J. McNamara, D.L. Paulson, H.C. Urschel Jr., M.A. Razzuk; Eventration of the diaphragm; Surgery, 64 (1968), pp. 1013–1021
- 6 D.R. Graham, D. Kaplan, C.C. Evans, C.R. Hind, R.J. Donnelly; Diaphragmatic plication for unilateral diaphragmatic paralysis: a 10-year experience; Ann Thorac Surg, 49 (1990), pp. 248–252
- 7 S.M. Higgs, A. Hussain, M. Jackson, R.J. Donnelly, R.G. Berrisford; Long term results of diaphragmatic plication for unilateral diaphragm paralysis; Eur J Cardiothorac Surg, 21 (2002), pp. 294–297
- 8 R.K. Freeman, T.C. Wozniak, E.B. Fitzgerald; Functional and physiologic results of video-assisted thoracoscopic diaphragm plication in adult patients with unilateral diaphragm paralysis; Ann Thorac Surg, 81 (2006), pp. 1853–1857
- 9 M. Ribet; Diaphragmatic plication; Ann Thorac Surg, 50 (1990), pp. 855–856
- 10 L. Svanberg; Clinical value of analysis lung function in some intrathoracic diseases: spirometric, bronchospirometric and angiopneumographic investigation; Acta Chir Scand, 111 (1956), p. 1196
- 11 T. Tiryaki, Z. Livanelioğlu, H. Atayurt; Eventration of the diaphragm; Asian J Surg, 29 (2006), pp. 8–10
- 12 A.D. Mulvey, R.J. Aquilina, M.W. Elliott, J. Moxham, M. Green; Diaphragmatic dysfunction in neuralgic amyotrophy: an electrophysiologic evaluation of 16 patients presenting with dyspnea; Am Rev Respir Dis, 147 (1993), pp. 66–71
- 13 M.I. Versteegh, J. Braun, P.G. Voigt, et al.; Diaphragm plication in adult patients with diaphragm paralysis leads to long term improvement of pulmonary function and level of dyspnea; Eur J Cardiothorac Surg, 32 (2007), pp. 449–456
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?