Abstract
In this work, the performance of N-(3-trimethoxysilylpropyl)diethylenetriamine (DAEAPTS)-grafted mesoporous SBA-15 adsorbent for postcombustion carbon capture was studied. Wet grafting technique was adopted to functionalize SBA-15 surface. The adsorption of CO2 on the amine-grafted sorbent was measured by the thermogravimetric method over a CO2 partial pressure range of 0–90 kPa and a temperature range of 25–105°C under atmospheric pressure. The optimal amine loaded SBA-15 adsorbent containing 40 wt% DAEAPTS exhibited capture capacity up to 2.3 mmol/g under simulated gas conditions (88.2% CO2/N2) at 75°C. The CO2 adsorption–desorption kinetics of the grafted sorbents were found to be sufficiently fast in both dry and humid CO2 streams and it was observed that the grafted adsorbent achieved 75% of the total capacity in 5 min of adsorption time in dry 8.8% CO2/N2 and in less than 3.5 min in humid 8.8% CO2/N2. It was also observed that 95% of the total desorption occurred in less than 7 min at 150°C under pure N2. The grafted sorbents showed good reversibility and multicycle stability and the drop in capacity after 100 cycles in dry and humid 8.8% CO2/N2 streams was around 7.09% and 11.65%, respectively.
Introduction
Carbon dioxide (CO2) is one of the major greenhouse gases responsible for the global warming and climate change. According to the report of Intergovernmental Panel on Climate Change (IPCC), CO2 concentration in the atmosphere has increased considerably in the past few decades [1]. An increase in the use of fossil fuels, especially coal and petroleum has led to the increase in global annual CO2 emissions from ~21 Gt in 1970 to ~38 Gt in 2004 [1]. The world-wide concern over this issue has attracted considerable attention to cost-effective methods for CO2 capture from large point sources, such as coal-based power plants, natural and synthesis gas processing plants, and cement plants, and its sequestration carbon capture and storage (CCS) to mitigate the problem of global warming. However, the main focus of CCS at the moment is coal-based power plants.
Among the many process technology options used for CO2 capture from flue gas streams, there is a growing interest in using adsorption processes using temperature and/or pressure swing approaches as a promising alternative separation technique. Adsorption processes using solid sorbents capable of capturing CO2 from flue gas streams have many potential advantages, compared to the conventional aqueous amine-based process for CO2 capture, such as reduced energy for regeneration, greater capacity, selectivity, ease of handling, etc [2, 3]. However, most of the conventional physisorbents such as ACs (activated carbon) [3-5], zeolites [5, 6], carbon molecular sieve (CMSs) [7], and carbon nanotube (CNTs) [8] suffer from low CO2 adsorption capacities and lower selectivity toward CO2 at relatively low CO2 partial pressures. The presence of water vapor further limits the application of these sorbents by decreasing their capacity. To overcome these limitations, research work on functionalizing solid supports with amine functional groups to capture CO2 from flue gas has generated a lot of interest. It has been shown that, by incorporating certain basic functional groups into their porous structure, thus enhancing the CO2–adsorbent interactions, the adsorption capacity of these adsorbent materials could be significantly improved.
Recently, a variety of mesoporous silica materials loaded with basic nitrogen functionality, more specifically, organic amine functionality has been synthesized and characterized to chemisorb CO2 from flue gas streams (see Table S1) [9-19]. Table 1 shows different amine structures used in the literature for functionalization of mesoporous silica adsorbent [2]. All these adsorbent materials hold great potential for use in postcombustion CO2 capture from flue gas.
| Tetraethylenepentamine (TEPA) | |
| Polyethyleneimine (PEI) |
(R=H for linear, R=H or CHx for branched) |
| 3-aminopropyltrimethoxysilane (APTS) | |
| 3-aminopropyltriethoxysilane (APTES) | |
| N-[3-(trimethoxysilyl) propyl]- ethylenediamine (AEAPTS) | |
| N-(3-trimethoxysilylpropyl)- diethylenetriamine (DAEAPTS) | |
| Aziridine |
Amine-impregnated silica supports are promising and can effectively adsorb CO2 with relatively higher working capacity and their adsorption capacities are not impaired by the presence of moisture [9-18]. However, multicycle stability of the amine-impregnated solid sorbents has not been tested adequately under real flue gas conditions. Considerable leaching of the amines can be a major drawback for the use of impregnated amine-functionalized sorbents for CO2 capture. Zhao et al. [13] impregnated SBA-15 with tetraethylenepentamine (TEPA) and studied its multicycle stability for 10 cycles. The results showed CO2 adsorption capacities of 3.48 and 3.67 mmol/g after first cycle in dry and humid 10% CO2/90% N2 gas mixture at 75°C respectively. For both dry and humid streams, it was found that the capacities decreased by around 10% after 10 adsorption/desorption cycles using N2 as regeneration gas at 105°C. After 10 cycles, TEPA leaching was found ~8.2 wt% and 5.8 wt% of fresh adsorbent under dry and humid conditions respectively. Hicks et al. [20] loaded SBA-15 with TEPA. They observed that TEPA impregnated SBA-15 starts to drop capacity from 2nd cycle and capacity keeps on dropping further in subsequent cycles due to leaching of TEPA physisorbed onto SBA-15 surface.
In view of this drawback, there is a need to develop amine functionalized silica adsorbents with multicycle stability. Covalently tethered/grafted silica sorbents with high amine content capable of capturing CO2 reversibly show good promise for CO2 capture from flue gas with long term stability. Leal et al. [21] first used amine-grafted adsorbent for CO2 capture. They grafted 3-aminopropyltriethoxysilane (APTES) onto silica gel surface and observed that two amino groups were required to capture one CO2 molecule in the absence of H2O via ammonium carbamate formation whereas one amino group was sufficient to capture one CO2 molecule in the presence of H2O via ammonium bicarbonate formation. Delaney et al. [22] grafted several silane group amines like 3-aminopropyltrimethoxysilane (APTS), N-[3-(trimethoxysilyl) propyl]- ethylenediamine (AEAPTS), and N-(3-trimethoxysilylpropyl)diethylenetriamine (DAEAPTS) etc. on hexagonal mesoporous silica (HMS). This amine functionalized HMS material was reported to give better capacity than that found by Leal et al. [21] for functionalized silica gel. Harlick and Sayari [23] functionalized pore expanded MCM-41 (PE-MCM-41) with DAEAPTS and obtained CO2 adsorption capacity of 2.65 mmol/g at 25°C and 1 atm under dry 5% CO2 in N2 feed. Later, Sayari and Belmabkhout [24] continued their investigation on DAEAPTS-grafted PE-MCM-41 to study the stability of this adsorbent over multiple cycles. They found 15% loss in adsorption capacity after around 750 cycles under dry gas conditions whereas the adsorbent was found to be stable after 700 cycles in humid conditions. The loss in capacity under dry conditions was observed because of urea formation even at mild regeneration temperature of 70°C. Khatri et al. [25] and Zheng et al. [26] prepared several amine-grafted SBA-15 adsorbents and found that the grafted amine groups are thermally stable up to 250°C temperature. Jones research group [9, 27] proposed another amine-grafted adsorbent, hyperbranched aminosilica (HAS), to capture CO2 from flue gas mixture. The HAS adsorbent showed stability over 12 cycles (130°C desorption temperature) in humid conditions. The adsorption capacity reported in 10% CO2/90% Ar humid gas mixture at 25°C was 3.11 mmol/g [9]. Drese et al. [27] tuned the synthesis process of HAS adsorbent in order to improve the CO2 adsorption performance. After these modifications, the highest CO2 adsorption capacity and corresponding amine loading reported were 5.55 mmol/g and 9.78 mmol N/g respectively in 10% CO2/90% Ar humid gas mixture at 25°C. As each silane group amine used for grafting has different structure and nitrogen content, so type of amine used for grafting process also plays a key role in determining the performance of an adsorbent for CO2 capture. Hiyoshi et al. [28] grafted APTES, AEAPTS, DAEAPTS amines on SBA-15 surface and determined the order of CO2 adsorption capacity as DAEAPTS > AEAPTS > APTES. Chaffees group [29] prepared APTS, AEAPTS and DAEAPTS amines-grafted HMS adsorbents and examined their performance. It was seen that DAEAPTS-grafted adsorbent has generally higher capacity than the analogous APTS, AEAPTS-grafted adsorbents in dry conditions [29]. It was reported that all APTS, AEAPTS, DAEAPTS-grafted adsorbents were stable till 170°C in both pure N2 and mildly oxygenated (2% O2) N2 atmosphere and the adsorbents showed no affinity toward N2 or O2. Among the different amines used in the literature for grafting over silica support, DAEAPTS is very promising for synthesizing adsorbents using the grafting technique due to its higher nitrogen content (15.8 wt%) and higher CO2 adsorption capacity compared to other amines like APTS, AEAPTS, etc.
Although a lot of research work has already been carried out in the field of CO2 capture using the amine-grafted ordered mesoporous silica support but only few are concerned with grafted SBA-15. The systematic study of SBA-15 grafted with DAEAPTS dealing CO2 capture is still lacking. The CO2 adsorption capacities and their respective adsorption kinetics reported in literature so far for DAEAPTS-grafted SBA-15 adsorbent is inconsistent and quite low. There is no literature available on investigation of adsorption and desorption kinetics and multicycle performance of DAEAPTS-grafted SBA-15 adsorbent for CO2 capture. In view of the above, the main objective of this work is to perform systematic parametric study of DAEAPTS-grafted SBA-15 adsorbent for postcombustion CO2 capture applications in terms of adsorption/desorption performance, kinetics, and multicycle stability of DAEAPTS-grafted SBA-15 adsorbent.
Experimental
Materials
Triblock copolymer Pluronic P-123 surfactant (poly(ethylene glycol)-block-poly(propylene glycol)-block-poly(ethylene glycol), average MW ~5800), and tetraethyl orthosilicate (TEOS, 98%) obtained from Sigma-Aldrich (Oakville, Ontario, Canada) were used without further purification for the synthesis of SBA-15. HCl (36.5–38%) used as a pH-controlling agent and obtained from J.T. Baker (Avantor™ Performance Materials, Center Valley, PA). DAEAPTS (91.5%), hexane (99%) and anhydrous toluene (99.9%) used for grafting of amine on SBA-15 were also purchased from Sigma-Aldrich and used as received. Deionized water was obtained from a Milli-Q (Etobicoke, Ontario, Canada) integral pure and ultrapure water purification system. Pure carbon dioxide (99.99%) and ultra-high pure nitrogen (99.999%) gases were obtained from Praxair (Mississauga, Ontario, Canada) for the CO2 adsorption/desorption measurements.
Preparation of SBA-15 and amine-grafted SBA-15
The mesoporous SBA-15 material used as adsorbent in this work was synthesized according to the method reported elsewhere [30]. In a typical synthesis procedure, 4 g of Pluronic P123 was dissolved in 30 mL of deionized water and 120 mL of 2 mol/L HCl solution while stirring for 2 h at 25°C. The resultant solution was transferred into a teflon autoclave and solution temperature was raised to about 40°C. 8.5 g of TEOS was added dropwise to the homogenous solution under vigorous stirring for 30 min and a precipitated product appeared as a result. The resulting gel was stirred at 40°C for about 20 h at low stirring rate and then aged at 100°C for 48 h without stirring. The white precipitated solid obtained was then filtered, washed with deionized water, and dried in air at room temperature for 12 h and in an air oven at 100°C for another 12 h. The organic template was removed from the product by calcination in flowing air at 550°C for 5 h.
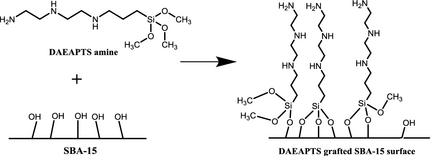
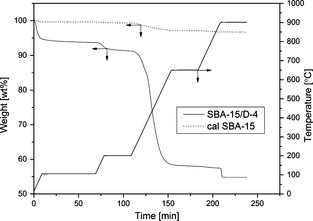
The DAEAPTS-grafted sorbents were prepared via wet grafting technique [23]. Figure 1 provides the schematic representation of the reaction between the DAEAPTS amine and hydroxyl groups on SBA-15 surface. As shown in Figure 1, one DAEAPTS molecule can form one, two or three linkages with the SBA-15 surface. In a typical preparation, about 0.5 g of calcined SBA-15 was dissolved and dispersed in about 150 mL of anhydrous toluene and allowed to mix for about 30 min at room temperature with low stirring. Then 0.2 mL of water was added to the above mixture and continuously stirred at room temperature for about 2 h. The temperature of the mixture was then increased and maintained at 100°C and desired quantity of DAEAPTS amine was added to it. The system was kept under vigorous stirring and reflux for about 24 h. The resultant slurry was washed first with toluene and then hexane and finally dried under vacuum at 70°C overnight. Amine-grafted SBA-15 sorbent thus formed is denoted as SBA-15/D-x where D represents the DAEAPTS amine and x represents the quantity of DAEAPTS amine used in mL per g of SBA-15 used during the synthesis process.
|
|
|
Figure 1. Schematic representation of N-(3-trimethoxysilylpropyl)diethylenetriamine (DAEAPTS) amine grafting over SBA-15 surface. |
Characterization
The N2 adsorption/desorption isotherms of the sorbents were measured using Autosorb iQ (Quantachrome, Boynton Beach, FL) in the relative pressure range of 0–0.997 at −196°C. The calcined SBA-15 sample was degassed at 250°C for 2 h under high vacuum before starting analysis whereas amine-grafted SBA-15 sample was degassed at 80°C for only 60 min under high vacuum in order to avoid amine loss. The total pore volume was calculated as the adsorbed volume of liquid nitrogen at the relative pressure of 0.997 and the micropore volume was calculated using the t-plot method. The multipoint Brunauer–Emmett–Teller (BET) method at a relative pressure range of 0.05–0.30 was used for the calculation of the surface area.
Amine loading of the sample was measured using thermogravimetric analyzer (TGA/DSC 1 STARe System, Mettler Toledo, Mississauga, Ontario, Canada). The sample was kept under nitrogen atmosphere till 900°C inside a thermo-gravimetric analyzer. The amine loading results were calculated on the dry basis mass of amine-grafted adsorbent samples after water vaporization at 100°C in TGA. Elemental analysis was also performed on amine-grafted sorbents for carbon, nitrogen, hydrogen, and sulfur percentage using the Elementar analyzer (Model: Vario Micro, Elementar Analysensysteme GmbH, Mt. Laurel, NJ). From the elemental analysis results, the nitrogen (N) wt% and amine loading of the grafted and raw SBA-15 samples were calculated.
Fourier transform infrared spectroscopy (FTIR) spectra of the raw SBA-15 and grafted sorbent samples were obtained using the Nicolet 8700 FTIR spectrometer (Thermo Electron Corporation, Waltham, MA) equipped with Smart Environmental Chamber for tests under inert environment and Smart Collector accessory for analysis of highly light scattering solids. The results were analyzed using Omnic (Thermo Scientific, Waltham, MA) software. Each sample was scanned 100 times in the wave number range from 550 to 4000 cm−1 and at a resolution of 2 cm−1.
Solid-state 29Si NMR data were acquired on a Bruker Avance (Bruker, Billerica, MA) 500 spectrometer, operating at frequencies of 99.4 MHz for 29Si and 500.3 MHz for 1H. Spectra were obtained with magic angle spinning at a spinning frequency of 8 kHz and with ramped cross-polarization. The 1H 90° pulse of 4.5 μsec, contact time of 4 msec and the recycle delay of 5 sec was used. 1H decoupling was accomplished using a two-pulse phase modulation scheme (TPPM).
CO2 adsorption and desorption
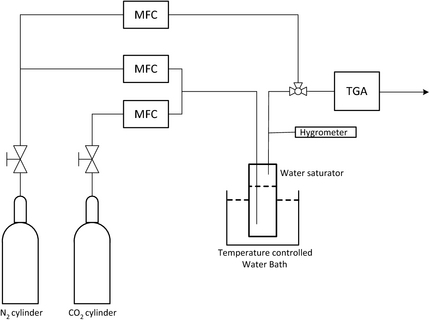
CO2 adsorption/desorption measurements of amine-grafted SBA-15 adsorbent were performed using a TGA/DSC 1 STARe System (Mettler Toledo). The TGA experimental set-up used for the tests is presented in Figure 2. The grafted adsorbents were ground and sieved to 150–250 μm size range before use. In a typical adsorption run, about 10–15 mg of powdered adsorbent material in platinum pan was used for the TGA measurements. The grafted adsorbent sample was activated before CO2 adsorption at 150°C under N2 stream with a flow rate of 150 cm3/min for about 60 min and the temperature was then decreased to the desired adsorption temperature. All the CO2 sorption capacities measured in this work are for 120 min of adsorption time at the specified adsorption temperature under CO2/N2 gas mixture at 1 atm. To study multicycle performance of the grafted SBA-15 sorbent, 100 consecutive cycles of adsorption/desorption were conducted thermogravimetrically using an 8.8% CO2/91.2% N2 gas mixture. Each cycle comprised 10 min of CO2 adsorption at 75°C and followed by 10 min of desorption at 120°C. The regeneration of the sorbent was accomplished via the temperature swing adsorption (TSA) method by purging pure N2. The moisture effect was studied by exposing the sorbent to a humid stream of CO2/N2 gas mixture after passing the dry gas mixture through a water saturator which was kept in a water bath maintained at 25°C. Relative humidity (RH) of the humid gas was measured using a hygrometer (Control Company, Friendswood, TX). RH of about 95% at 25°C (equivalent to about 2.98% moisture in the gas mixture) was maintained for the humid CO2/N2 gas mixture for the adsorption tests. The grafted sample was also tested for adsorption due to moisture alone using the humid N2 gas at RH of about 95% at 25°C.
|
|
|
Figure 2. Experimental set-up for performance evaluation of amine-grafted SBA-15 adsorbent. |
Results and Discussion
Characterization
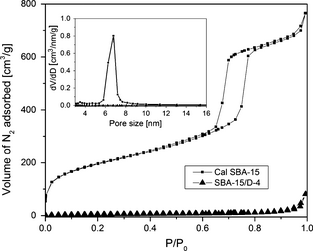
The textural properties such as surface area, pore volume, Barrett-Joyner-Halenda (BJH) pore size of calcined SBA-15, and amine-grafted samples are summarized in Table 2. The N2 adsorption/desorption isotherms of calcined SBA-15 shows typical type IV isotherm indicative of defined mesoporosity in the framework (Fig. 3). The pore size distribution of calcined SBA-15 as shown in Figure 3 (inset) exhibits a sharp peak centered around 6.8 nm, indicating the high uniformity of mesopore distribution. For calcined SBA-15, the BET surface area and total pore volume are about 676 m2/g and 1.184 cm3/g, respectively. On the other hand, amine-grafted sorbent SBA-15/D-4 has comparatively lower BET surface area and total pore volume of 15.9 m2/g and 0.122 cm3/g, respectively. BET surface area is further reduced to 0.4 m2/g (pore volume ~0 cm3/g) for SBA-15/D-8. This could be attributed to pore blocking/distorting in the mesoporous channels of the support due to the presence of DAEAPTS amine. It confirms that some DAEAPTS was not removed during the toluene and the hexane washing process and remained into the pores of the SBA-15 support blocking the connecting pore channels between the mesopores. Similar observations were also made by Mello et al. [31].
| Sample | BET surface area (m2/g) | Total pore volume (cm3/g) | Micropore volumea (cm3/g) | BJH diameter (nm) |
|---|---|---|---|---|
| ||||
| Calcined SBA-15 | 676 | 1.184 | 0.065 | 6.8 |
| SBA-15/D-1 | 20.4 | 0.204 | ~0 | 3.5 |
| SBA-15/D-1.5 | 18.1 | 0.176 | ~0 | 3.5 |
| SBA-15/D-4 | 15.9 | 0.122 | ~0 | 3.3 |
| SBA-15/D-8 | 0.4 | ~0 | ~0 | ~0 |
|
|
|
Figure 3. N2 adsorption/desorption isotherm and pore size distribution of calcined SBA-15 and amine-grafted sample SBA-15/D-4. |
FTIR analysis was carried out to find out the types of functional groups and chemical bonds present in calcined SBA-15 and amine-grafted SBA-15. In Figure 4, the spectrum for calcined SBA-15 showed peak at 1080 cm−1 with a broad shoulder at 1200 cm−1, which is typical of (SiO)n siloxane stretching [32]. The peak at 807 cm−1 for calcined SBA-15 is indicative of Si–O–Si vibration, which represents the silica framework of calcined SBA-15 [33]. For all the amine-grafted SBA-15 spectra, the N–H bond in the amine groups is verified by peaks at 3288, 1648, and 1570 cm−1, whereas, alkane C–H bond is testified by peaks at 2935, 2874, 2820, 1470, and 1298 cm−1, respectively, and CH2–N bond is supported by peak at 1407 cm−1, which shows the grafting of DAEAPTS amine over SBA-15 surface [25, 32-34]. The broad band in the spectrum of calcined SBA-15 in this figure at 3458 cm−1 is due to the presence of silanol groups and the sharp peak at 3746 cm−1 corresponds to unassociated silanol groups. The presence of adsorbed water by calcined SBA-15 sample is confirmed by peak at 1625 cm−1 [26]. The peaks at 3746 cm−1 and band at 3458 cm−1 due to silanol groups in calcined SBA-15 spectrum is not observed in the spectra for amine-grafted SBA-15 samples. This is in accordance to the reaction of silanol groups in calcined SBA-15 with DAEAPTS amine molecules to form amine-grafted SBA-15 sample. All the amine-grafted SBA-15/D-x samples show the same characteristic IR bands but the peak intensities are much lower for SBA-15/D-8 sample. The reduction in peak intensities for SBA-15/D-8 sample indicates structural degradation of this sample due to much higher amine loading used for this sample.
|
|
|
Figure 4. FTIR analysis of calcined SBA-15 and amine-grafted SBA-15 adsorbent samples. |
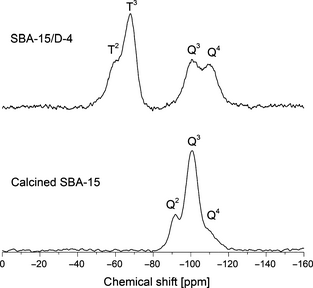
Figure 5 shows the typical 29Si NMR spectra of raw calcined SBA-15 and amine functionalized sample SBA-15/D-4. The spectrum for calcined SBA-15 displays clear and distinct peaks at −91 and −100 ppm for Q2 (geminal silanol) and Q3 (isolated silanol) sites, respectively. The shoulder at −109 ppm in calcined SBA-15 spectrum corresponds to the Q4 (internal siloxane) sites (see Table S2). In general, Qn sites correspond to the chemical species Si(OSi)n(OH)4−n. The overlapping nature of these peaks is because of the large variety of bond angles in SBA-15 silica support structure. All these Q2, Q3, and Q4 sites are characteristic of the SBA-15 silica framework. The SBA-15/D-4 spectrum shows peaks for Q3 sites at −100 ppm and Q4 sites at −109 ppm. The Q3 and Q4 peaks in this spectrum indicate the presence of SBA-15 support material. The SBA-15/D-4 spectrum also shows peaks at −60 ppm for T2 sites and at −68 ppm for T3 sites (see Table S2), where Tm sites are associated with the chemical species RSi(OSi)m(OR')3−m in the sample structure. The presence of these T2 and T3 peaks confirm the formation of covalent bonding via Si-O-Si linkages between DAEAPTS amine molecules and SBA-15 silica support. Similar observations were also made by other research groups in the literature [26, 31, 35, 36].
|
|
|
Figure 5. 29Si MAS NMR spectra of N-(3-trimethoxysilylpropyl)diethylenetriamine (DAEAPTS) amine functionalized sample SBA-15/D-4 and calcined SBA-15. |
Amine content of a grafted adsorbent indicates the measure of CO2 capture and it can be determined from the thermal analysis of the adsorbent. Figure 6 depicts the typical thermal behavior of amine-grafted SBA-15/D-4 adsorbent as measured by TGA under dry N2 environment. Here, temperature was first maintained at 105°C for about 60 min and then held at 200°C and 650°C for 30 min each. After reaching about 900°C, the sample was exposed to air for another 30 min. It can be seen from Figure 6 that there is a weight loss of about 6.3% upto about 105°C. This is attributed to the vaporization of water and adsorbed CO2 from atmosphere. It is also found that there is no significant drop in weight (only about 2.4 wt%) for the amine-grafted sorbent with increase in temperature till 200°C. This is attributed to the loss of methoxy ligands [37]. It confirms that there is very negligible amine leaching till 200°C. So, it can be concluded that the grafted amine sorbent is stable up to 200°C.
|
|
|
Figure 6. A typical thermal behavior curve of calcined SBA-15 and DAEAPTS-grafted adsorbent SBA-15/D-4 using TGA. DAEAPTS, N-(3-trimethoxysilylpropyl)diethylenetriamine; TGA, thermogravimetric analyzer. |
Table 3 presents the amine loading of amine-grafted SBA-15 samples with different amounts of amine used during synthesis as obtained using TGA and elemental analysis. Amine loading of each grafted sorbent was calculated using TGA as the weight drop from 100°C to 900°C and then converting it into dry basis mass at 100°C. Amine loading results were also obtained from elemental analysis. There is a good agreement between the amine loading values obtained from TGA as well as elemental analysis with a maximum error of 12.57%.
| Sample | Amine loading from TGA (wt%) | Amine loading from elemental analysis (wt%) | % error |
|---|---|---|---|
| |||
| SBA-15 | 0 | 0.00 | 0.00 |
| SBA-15/D-1 | 28.21 | 32.27 | 12.57 |
| SBA-15/D-1.5 | 33.49 | 37.54 | 10.77 |
| SBA-15/D-4 | 39.64 | 42.44 | 6.59 |
| SBA-15/D-8 | 46.22 | 51.01 | 9.38 |
Adsorption performance
Effect of amine loading on CO2 adsorption performance
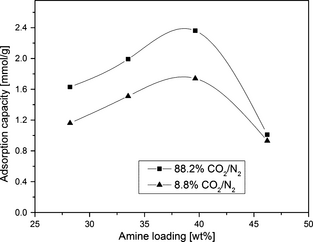
A series of DAEAPTS-grafted SBA-15 sorbents with varying amine content were prepared to investigate the effect of amine loading on CO2 uptake performance. Figure 7 presents the variation of CO2 adsorption capacity with amine loading at two different CO2 compositions of 8.8% CO2/N2 and 88.2% CO2/N2. For both the CO2 compositions in this figure, the CO2 adsorption capacity attained the highest value for SBA-15/D-4 sample. It was observed that CO2 adsorption capacity kept on increasing as the amine loading increased from 28.21 wt% to 39.64 wt%. Whereas beyond 39.64 wt%, CO2 adsorption capacity decreased sharply indicating that SBA-15/D-4 sample has the optimum amine loading of 39.64 wt% with CO2 adsorption capacity of 2.36 mmol/g in 88.2% CO2/11.8% N2 gas mixture at 75°C. Possible explanation of capacity reduction over 40 wt% amine loading may be due to the bulk deposition of amine over the SBA-15 surface and consequently due to pore blocking/distorting in the mesoporous channels of the support as observed from textural data shown in Table 2. For example, the BJH pore size for SBA-15/D-4 sample is 3.3 nm, whereas BJH pore size for SBA-15/D-8 is almost negligible. The bulk deposition of amine leads to reduced accessibility of amino group by the CO2 molecules due to more proximity of amino groups and entanglements provided by large DAEAPTS molecules, which creates diffusional resistance and hence reduced capacity.
|
|
|
Figure 7. CO2 adsorption capacity variation with amine loading for different SBA-15/D-x adsorbent samples in 88.2% CO2/11.8% N2 and 8.8% CO2/91.2% N2 gas mixtures at 75°C and 1 atm. |
Table 4 compares the CO2 adsorption capacity and amine adsorption efficiency of amine-grafted SBA-15 samples with different amine loadings in 8.8% CO2/91.2% N2 gas mixture at 75°C. As per Table 4, the highest amine efficiency is shown by SBA-15/D-4 sample (0.23 mmol CO2/mmol N) whereas the amine efficiency for SBA-15/D-8 is 0.10. The reduced amine adsorption efficiency and CO2 adsorption capacity of SBA-15/D-8 sample supports that amine loading after 40 wt% increase the diffusional resistances significantly which limit the amount of CO2 adsorbed by the sample.
| Sample | Amine loading (wt%) | Adsorption capacity (mmol/g) | N content (mmol N/g) | Amine adsorption efficiency (mmol CO2/mmol N) |
|---|---|---|---|---|
| ||||
| SBA-15 | 0.00 | 0.04 | 0.00 | 0.00 |
| SBA-15/D-1 | 28.21 | 1.16 | 5.82 | 0.20 |
| SBA-15/D-1.5 | 33.49 | 1.51 | 6.77 | 0.22 |
| SBA-15/D-4 | 39.64 | 1.74 | 7.65 | 0.23 |
| SBA-15/D-8 | 46.22 | 0.93 | 9.20 | 0.10 |
Effect of adsorption temperature and CO2 partial pressure
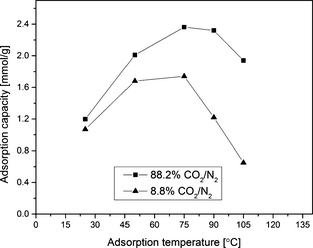
Figure 8 presents the CO2 adsorption performance of SBA-15/D-4 sample at different adsorption temperatures for 88.2% CO2 and 8.8% CO2 in N2. In 88.2% CO2/11.8% N2 gas mixture, the adsorption capacity for this sample increased from 1.20 mmol/g at 25°C to 2.36 mmol/g at 75°C and then decreased to 1.94 mmol/g at 105°C. Similarly for 8.8% CO2/91.2% N2 gas mixture, the adsorption capacity achieved maximum value of 1.74 mmol/g at 75°C. Therefore, CO2 adsorption capacity increases with an increase in adsorption temperature till 75°C and then starts decreasing beyond this temperature. The CO2 adsorption capacity of DAEAPTS-grafted sample in the lower temperature range (about 25°C) is quite low in both 88.2% CO2/N2 and 8.8% CO2/N2 gas mixtures. This can be attributed to the slow diffusion of CO2 to the active amino sites inside amine-grafted SBA-15 pores due to pore blocking by amine molecules, and relatively slow reaction of CO2 and amine. These lead to kinetic limitations, even though the lower temperatures are thermodynamically more suitable for CO2 capture [13]. The kinetic limitation and diffusional resistance diminishes as the temperature increases. But beyond 75°C, the CO2 adsorption capacity decreased in both gas mixtures. This indicates that the thermodynamic equilibrium between CO2 and DAEAPTS-grafted SBA-15 governed CO2 adsorption instead of chemical kinetics [38]. Hence, an increase in adsorption temperature beyond 75°C that would enhance the kinetic reaction rate was not favorable in achieving highest CO2 uptake.
|
|
|
Figure 8. Amine-grafted sorbent SBA-15/D-4 variation of CO2 adsorption capacity with adsorption temperature for 88.2% CO2 and 8.8% CO2 in N2 at 1 atm. |
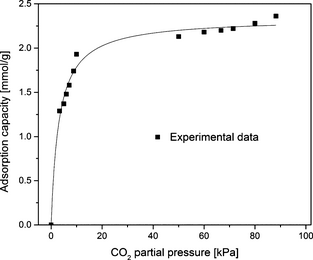
Figure 9 presents the effect of partial pressure of CO2 on adsorption capacity of SBA-15/D-4 sample measured using the thermogravimetric method under different CO2 partial pressures and at 75°C which is optimum temperature for this sorbent. The adsorption capacity increases sharply with CO2 partial pressure initially at lower partial pressures, while further increasing in CO2 partial pressure results in a plateau of CO2 adsorption capacity near a value about 2.3 mmol/g. It is worth mentioning here that the adsorption capacities reported in this work are working adsorption capacities, not equilibrium adsorption capacities. This is because of incomplete degassing to remove CO2 and moisture via the TGA method and incomplete adsorption even after long exposure time. It was observed in this work that even after exposing the adsorbent under CO2 for a longer time up to 20 h, adsorption was still occurring but extremely slowly.
|
|
|
Figure 9. Amine-grafted sorbent SBA-15/D-4 variation of CO2 adsorption capacity with CO2 partial pressure at 75°C and 1 atm. |
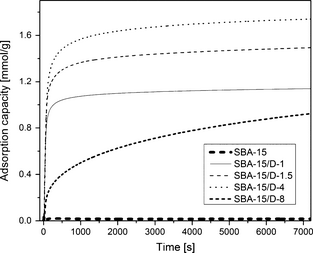
Kinetics
The kinetics play a vital role in determining the performance of a sorbent. In order for the sorbent to have any meaningful use, its kinetics needs to be sufficiently fast. Figure 10 presents the CO2 adsorption capacity as a function of time for amine-grafted adsorbents with different amine loading at 75°C and 1 atm. For SBA-15/D-1, SBA-15/D-1.5, and SBA-15/D-4, there is a sharp increase in CO2 adsorption capacity in the first few minutes of adsorption time indicating fast kinetics whereas for SBA-15/D-8 sample, the kinetics are slow. The slow kinetics of SBA-15/D-8 can be attributed to the SBA-15 support pore blocking/distorting due to bulk amine deposition over the SBA-15 support.
|
|
|
Figure 10. CO2 adsorption capacities of different SBA-15/D-x adsorbents as a function of time in 8.8% CO2/91.2% N2 gas mixture at 75°C and 1 atm. |
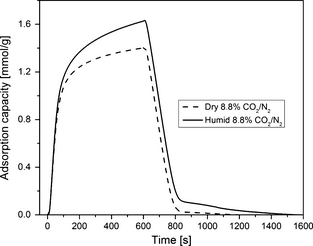
Adsorption and desorption kinetics of amine-grafted adsorbent SBA-15/D-4 sample is presented in Figure 11 in dry and humid 8.8% CO2/91.2% N2 gas at 75°C. In this test, CO2 was adsorbed on the sample for 10 min at 75°C and then desorbed at 150°C in pure N2 for 10 min. In dry gas, the sample reached 75% of the maximum CO2 sorption capacity of 1.74 mmol/g in only 5 min of adsorption time whereas in humid gas, it reached 75% capacity in only 3.5 min of adsorption time. Although the adsorption kinetics are fast in both dry and humid gas but humid gas adsorption kinetics are relatively better. For both dry and humid gas, more than 95% of the total desorption happened in less than 7 min of desorption time.
|
|
|
Figure 11. Comparison of adsorption–desorption kinetics of SBA-15/D-4 sample in dry and humid 8.8% CO2/91.2% N2 gas mixture at 75°C and 1 atm. |
Effect of moisture
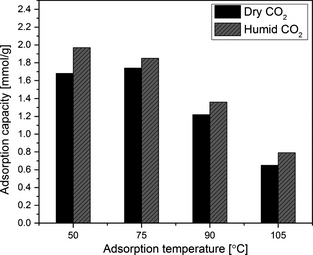
Amine-grafted sample SBA-15/D-4 was tested for CO2 adsorption using humid CO2/N2 gas mixture to determine the effect of moisture in CO2 adsorption performance. Figure 12 compares the CO2 adsorption capacity of amine-grafted SBA-15/D-4 sample in humid and dry 8.8% CO2/91.2% N2 gas mixture at different adsorption temperatures. The increase in adsorption capacity in humid gas at different adsorption temperatures may be partially attributed to the formation of bicarbonate species in presence of moisture [39] and partially to moisture adsorption. Therefore, CO2 adsorption capacity is not much affected by the presence of moisture as the overall capacity has increased in the presence of moisture in humid CO2 stream.
|
|
|
Figure 12. Comparison of CO2 adsorption capacity of SBA-15/D-4 sample in dry and humid CO2 streams at different adsorption temperatures. 8.8% CO2/91.2% N2 gas mixture was used for these tests. |
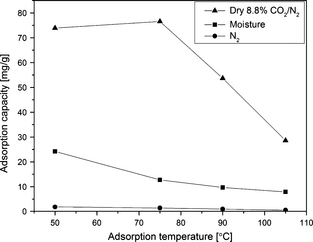
Figure 13 presents the adsorption capacities of dry CO2, moisture and N2 at different adsorption temperatures. N2 adsorption is negligible for this adsorbent, which means the adsorbent is highly selective toward CO2. Adsorption capacity due to moisture alone is very low as compared to CO2 adsorption capacity.
|
|
|
Figure 13. Comparison of dry CO2, moisture and N2 adsorption capacities at different adsorption temperatures for SBA-15/D-4 sample. |
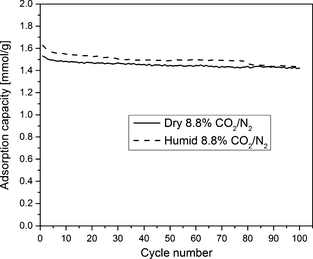
Multicycle stability
One of the most important criteria in evaluating adsorbent for postcombustion CO2 capture applications is multicycle stability of the adsorbent. Figure 14 presents 100 short adsorption/desorption cycles in dry and humid 8.8% CO2 in N2 at 1 atm. For this test, each cycle comprised 10 min of adsorption at 75°C in CO2/N2 gas mixture and 10 min of desorption at 120°C in pure N2 gas. The capacity observed after 1st and 100th cycle under dry gas was 1.53 and 1.42 mmol/g, respectively. For the humid gas, the 1st and 100th cycle capacity was 1.63 and 1.44 mmol/g, respectively. The drop in capacity after 100 cycles in humid gas was 11.65% and in dry gas was 7.09%. Amine leaching, calculated from the drop in weight of the sample (from TGA data), after 100 cycles in humid gas was 0.45 wt% and in dry gas was 4.25 wt%. So, with short cycles in 8.8% CO2/N2 gas mixture, multicycle stability in dry gas is slightly better than the cyclic stability in the humid gas. Better multicycle stability can be achieved by using more adsorption and desorption time. The TGA availability for this work allowed us to use a maximum of 10 min of adsorption and desorption time as performing 100 cycles require considerably a large amount of time. The stabilizing effect of moisture on the multicycle performance of amine-grafted samples is also believed to be more pronounced for a longer cyclic time period. Overall, the sorbent exhibits decent stability over 100 adsorption/desorption cycles in both dry and humid gas mixtures.
|
|
|
Figure 14. Multicycle stability of SBA-15/D-4 sample in dry and humid 8.8% CO2/91.2% N2 gas mixture at 75°C and 1 atm. |
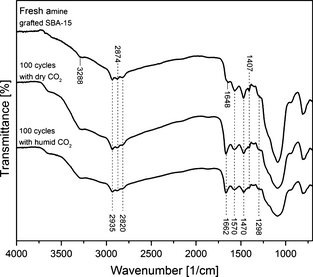
Figure 15 shows the FTIR analysis of amine-grafted SBA-15 samples: fresh, after 100 cycles with dry CO2 and humid CO2. As discussed above in the FTIR characterization section, the peaks at 2935, 2820, 1570, 1470, 1407, and 1298 cm−1 are typical of amine grafting as shown in spectrum for fresh amine-grafted SBA-15 sample. All these peaks are also retained by the grafted adsorbent sample after 100 cycles in dry and humid CO2, which imply that the amine grafting is preserved even after 100 adsorption/desorption cycles. Although 100 cycles in dry and humid gas has led to some amine leaching and capacity reduction as mentioned above. The peak at 1662 cm−1 in the spectrums of amine-grafted samples after 100 cycles in dry and humid gas is due to the presence of C=N bond generated upon amine oxidation after 100 cycles in dry and humid CO2 [40]. Overall, the amine-grafted samples after 100 cycles in dry and humid CO2 has maintained amine grafting but some of the grafted amine is degraded and resulted in capacity reduction.
|
|
|
Figure 15. FTIR analysis of fresh amine-grafted SBA-15 sample and amine-grafted samples after 100 cycles with dry and humid CO2/N2 gas mixtures. |
Conclusion
In this work, postcombustion carbon capture performance of DAEAPTS-grafted SBA-15 adsorbent was evaluated. DAEAPTS-grafted SBA-15 adsorbents with varying amine loading were prepared and characterized. The effect of adsorption temperature, CO2 partial pressure, and amount of amine loading on the CO2 adsorption capacity of SBA-15-DAEAPTS adsorbents were investigated in order to determine the optimum adsorption conditions. Based on CO2 adsorption capacity, amine adsorption efficiency and adsorption kinetics, it was found that SBA-15/D-4 sample has the optimal amine loading of 40 wt%. The synthesized SBA-15 adsorbents containing 40 wt% DAEAPTS exhibited maximum capture capacity of 2.3 mmol/g under simulated gas conditions (88.2% CO2/N2) at 75°C. The adsorption capacity increases sharply with CO2 partial pressure initially at lower partial pressures (less than 10 kPa), while further increasing in CO2 partial pressure results in a plateau of CO2 adsorption capacity near a value about 2.3 mmol/g. As in the postcombustion carbon capture, CO2 is available at around 10 kPa partial pressure, so this adsorbent has good potential under these conditions. The grafted adsorbent was also tested for N2 adsorption and the results showed negligible N2 adsorption; hence, the selectivity of adsorbent toward CO2 is sufficiently high.
The effect of moisture on CO2 capture performance of DAEAPTS-grafted SBA-15 adsorbent was also investigated. It was observed that the adsorption performance of grafted adsorbent is not much affected by the presence of moisture in the humid CO2 stream. The grafted sorbent was also tested for 100 short adsorption–desorption cycles in both dry and humid CO2 streams to investigate the multicycle stability and adsorption/desorption kinetics. The cycle time was kept low to allow fast adsorption and regeneration of the adsorbent. In both dry and humid streams, the sample showed good cyclic stability. It was found that the drop in capacity after 100 cycles in dry and humid CO2 streams was around 7.09% and 11.65%, respectively. The grafted sample displayed considerably fast adsorption–desorption kinetics in both dry and humid streams, reaching 75% of the capacity in 5 min in dry 8.8% CO2/N2 and in only 3.5 min in humid 8.8% CO2/N2. During temperature swing desorption using N2 as the desorbing gas, 95% of the total regeneration occurred in less than 7 min of desorption time at 150°C.
Acknowledgment
The financial support of Carbon Management Canada and Canadian Centre for Clean Coal/Carbon and Mineral Processing Technology is acknowledged. We also express our deep gratitude to Guy Bernard and Roderick Wasylishen, Department of Chemistry, University of Alberta, for their support and help in recording the solid state 29Si NMR of amine functionalized samples.
Conflict of Interest
None declared.
References
- Core Writing Team, Pachauri, R. K., and A. Reisinger, eds. Climate change 2007: synthesis report. IPCC, Geneva, Switzerland.
- Samanta, A., A. Zhao, G. K. H. Shimizu, P. Sarkar, and R. Gupta. 2012. Post-combustion CO2 capture using solid sorbents–a review. Ind. Eng. Chem. Res.51:1438–1463.
- Sjostrom, S., and H. Krutka. 2010. Evaluation of solid sorbents as a retrofit technology for CO2 capture. Fuel89:1298–1306.
- Na, B. K., K. K. Koo, H. M. Eum, H. Lee, and H. K. Song. 2001. CO2 recovery from flue gas by PSA process using activated carbon. Korean J. Chem. Eng.18:220–227.
- Zhang, Z., W. Zhang, X. Chen, Q. Xia, and Z. Li. 2010. Adsorption of CO2 on zeolite 13X and activated carbon with high surface area. Sep. Sci. Technol.45:710–719.
- Chue, K. T., J. N. Kim, Y. J. Yoo, S. H. Cho, and R. T. Yang. 1995. Comparison of activated carbon and zeolite 13X for CO2 recovery from flue gas by pressure swing adsorption. Ind. Eng. Chem. Res.34:591–598.
- Rutherford, S. W., and D. D. Do. 2000. Adsorption dynamics of carbon dioxide on a carbon molecular sieve 5A. Carbon38:1339–1350.
- Razavi, S. S., S. M. Hashemianzadeh, and H. Karimi. 2011. Modeling the adsorptive selectivity of carbon nanotubes for effective separation of CO2/N2 mixtures. J. Mol. Model.17:1163–1172.
- Li, W., S. Choi, J. H. Drese, M. Hornbostel, G. Krishnan, P. M. Eisenberger, et al. 2010. Steam-stripping for regeneration of supported amine-based CO2 adsorbents. ChemSusChem3:899–903.
- Xu, X., C. Song, B. G. Miller, and A. W. Scaroni. 2005. Influence of moisture on CO2 separation from gas mixture by a nanoporous adsorbent based on polyethylenimine-modified molecular sieve MCM-41. Ind. Eng. Chem. Res.44:8113–8119.
- Ma, X., X. Wang, and C. Song. 2009. Molecular basket sorbents for separation of CO2 and H2S from various gas streams. J. Am. Chem. Soc.131:5777–5783.
- Son, W. J., J. S. Choi, and W. S. Ahn. 2008. Adsorptive removal of carbon dioxide using polyethyleneimine-loaded mesoporous silica materials. Microporous Mesoporous Mater.113:31–40.
- Zhao, A., A. Samanta, P. Sarkar, and R. Gupta. 2013. Carbon dioxide adsorption on amine impregnated mesoporous SBA-15 sorbents: experimental and kinetics study. Ind. Eng. Chem. Res.52:6480–6491.
- Drage, T. C., A. Arenillas, K. M. Smith, and C. E. Snape. 2008. Thermal stability of polyethyleneimine based carbon dioxide adsorbents and its influence on selection of regeneration strategies. Microporous Mesoporous Mater.116:504–512.
- Yue, M. B., L. B. Sun, Y. Cao, Y. Wang, Z. J. Wang, and J. H. Zhu. 2008. Efficient CO2 capturer derived from as-synthesized MCM-41 modified with amine. Chem. Eur. J.14:3442–3451.
- Qi, G., Y. Wang, L. Estevez, X. Duan, N. Anako, A. H. A. Park, et al. 2011. High efficiency nanocomposite sorbents for CO2 capture based on amine-functonalized mesoporous capsules. Energy Environ. Sci.4:444–452.
- Goeppert, A., S. Meth, G. K. S. Prakash, and G. A. Olah. 2010. Nanostructured silica as a support for regenerable high-capacity organoamine-based CO2 sorbents. Energy Environ. Sci.3:1949–1960.
- Lee, S., T. Filburn, M. Gray, J. W. Park, and H. J. Song. 2008. Screening test of solid amine sorbents for CO2 capture. Ind. Eng. Chem. Res.47:7419–7423.
- Khatri, R., S. S. C. Chuang, Y. Soong, and M. Gray. 2005. Carbon dioxide capture by diamine-grafted SBA-15: a combined Fourier transform infrared and mass spectrometry study. Ind. Eng. Chem. Res.44:3702–3708.
- Hicks, J. C., J. H. Drese, D. J. Fauth, M. L. Gray, G. Qi, and C. W. Jones. 2008. Designing adsorbents for CO2 capture from flue gas-hyperbranched aminosilicas capable of capturing CO2 reversibly. J. Am. Chem. Soc.130:2902–2903.
- Leal, O., C. Bolivar, C. Ovalles, J. J. Garcia, and Y. Espidel. 1995. Reversible adsorption of carbon dioxide on amine surface-bonded silica gel. Inorg. Chim. Acta240:183–189.
- Delaney, S. W., G. P. Knowles, and A. L. Chaffee. 2002. Hybrid mesoporous materials for carbon dioxide separation. Prepr. Symp. Am. Chem. Soc. Div. Fuel Chem.47:65.
- Harlick, P. J. E., and A. Sayari. 2007. Applications of pore-expanded mesoporous silica. 5. Triamine grafted material with exceptional CO2 dynamic and equilibrium adsorption performance. Ind. Eng. Chem. Res.46:446–458.
- Sayari, A., and Y. Belmabkhout. 2010. Stabilization of amine-containing CO2 adsorbents: dramatic effect of water vapor. J. Am. Chem. Soc.132:6312–6314.
- Khatri, R. A., S. S. C. Chuang, Y. Soong, and M. Gray. 2006. Thermal and chemical stability of regenerable solid amine sorbent for CO2 capture. Energy Fuels20:1514–1520.
- Zheng, F., D. N. Tran, B. Busche, G. E. Fryxell, R. S. Addleman, T. S. Zemanian, et al. 2005. Ethylenediamine-modified SBA-15 as regenerable CO2 sorbent. Ind. Eng. Chem. Res.44:3099–3105.
- Drese, J. H., S. Choi, R. P. Lively, W. J. Koros, D. J. Fauth, M. L. Gray, et al. 2009. Synthesis-structure-property relationships for hyperbranched aminosilica CO2 adsorbents. Adv. Funct. Mater.19:3821–3832.
- Hiyoshi, N., K. Yogo, and T. Yashima. 2005. Adsorption characteristics of carbon dioxide on organically functionalized SBA-15. Microporous Mesoporous Mater.84:357–365.
- Knowles, G. P., S. W. Delaney, and A. L. Chaffee. 2006. Diethylenetriamine[propyl(silyl)]-functionalized (DT) mesoporous silicas as CO2 adsorbents. Ind. Eng. Chem. Res.45:2626–2633.
- Zhao, D., J. Feng, Q. Huo, N. Melosh, G. H. Fredrickson, B. F. Chmelka, et al. 1998. Triblock copolymer syntheses of mesoporous silica with periodic 50 to 300 angstrom pores. Science279:548–552.
- Mello, M. R., D. Phanon, G. Q. Silveira, P. L. Llewellyn, and C. M. Ronconi. 2011. Amine-modified MCM-41 mesoporous silica for carbon dioxide capture. Microporous Mesoporous Mater.143:174–179.
- Knöfel, C., C. Martin, V. Hornebecq, and P. L. Llewellyn. 2009. Study of carbon dioxide adsorption on mesoporous aminopropylsilane-functionalized silica and titania combining microcalorimetry and in situ infrared spectroscopy. J. Phys. Chem. C113:21726–21734.
- Yang, Y., F. Li, C. Yang, W. Zhang, and J. Wu. 2012. Grafting morphologies of TEPA on SBA-15(P) and its effect on CO2 adsorption performance. Acta Phys. Chim. Sin.28:195–200.
- Danon, A., P. C. Stair, and E. Weitz. 2011. FTIR study of CO2 adsorption on amine-grafted SBA-15: elucidation of adsorbed species. J. Phys. Chem. C115:11540–11549.
- Luechinger, M., R. Prins, and G. D. Pirngruber. 2005. Functionalization of silica surfaces with mixtures of 3-aminopropyl and methyl groups. Microporous Mesoporous Mater.85:111–118.
- Chong, A. S. M., X. S. Zhao, A. T. Kustedjo, and S. Z. Qiao. 2004. Functionalization of large-pore mesoporous silicas with organosilanes by direct synthesis. Microporous Mesoporous Mater.72:33–42.
- Harlick, P. J. E., and A. Sayari. 2006. Applications of pore-expanded mesoporous silicas. 3. Triamine silane grafting for enhanced CO2 adsorption. Ind. Eng. Chem. Res.45:3248–3255.
- Heydari-Gorji, A., Y. Yang, and A. Sayari. 2011. Effect of the pore length on CO2 adsorption over amine-modified mesoporous silicas. Energy Fuels25:4206–4210.
- Serna-Guerrero, R., Y. Belmabkhout, and A. Sayari. 2010. Triamine-grafted pore-expanded mesoporous silica for CO2 capture: effect of moisture and adsorbent regeneration strategies. Adsorption16:567–575.
- Calleja, G., R. Sanz, A. Arencibia, and E. S. Sanz-Pérez. 2011. Influence of drying conditions on amine-functionalized SBA-15 as adsorbent of CO2. Top. Catal.54:135–145.
Document information
Published on 01/06/17
Submitted on 01/06/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?