Summary
Lipomatous ganglioneuroma (LG) is a rare variant of ganglioneuroma that is histologically characterized by a mature adipocytic component admixed with a conventional ganglioneuroma. We report the clinicopathological and immunohistochemical features of an LG in a 44-year-old Chinese male; additionally, we review the literature regarding this type of tumor. Magnetic resonance imaging revealed a left paravertebral soft-tissue mass at the T11–L3 levels. Grossly, the encapsulated neoplasm had a white to yellowish cut surface and rubbery consistency. Microscopic evaluation revealed an encapsulated lesion that consisted of areas of ganglioneuroma admixed with areas of mature fat. By immunohistochemistry, the ganglion cells were positive for chromogranin and synaptophysin, whereas the Schwann cells were positive for vimentin, S-100 protein, and glial fibrillary acidic protein (GFAP). This is the second known report of a retroperitoneal LG. The patient was well and without evidence of disease at 2 years' follow-up.
Keywords
ganglioneuroma;lipomatous ganglioneuroma;retroperitoneal tumors
1. Introduction
Ganglioneuroma is a rare, differentiated, benign, slow-growing tumor that commonly originates from primordial neural crest cells and is composed of mature Schwann cells, ganglion cells, and nerve fibers. Ganglioneuroma is most frequently diagnosed in patients between the ages of 10 years and 29 years and is most commonly located in the posterior mediastinum, followed by the retroperitoneum.1 We report a peculiar ganglioneuroma with the unusually prominent lipomatous component, namely, lipomatous ganglioneuroma (LG), occurring in the retroperitoneum. We include detailed clinicopathological and immunohistochemical features and present a review of the literature.
2. Case report
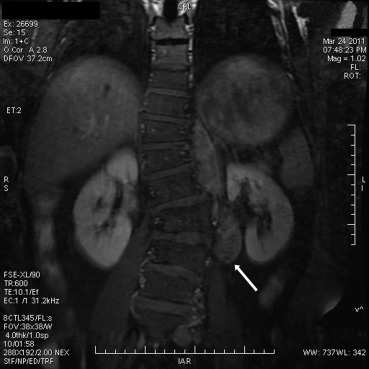
A 44-year-old male presented with intermittent pain in the left hypochondrium for twenty days. He had no significant past medical history. Magnetic resonance imaging revealed a left paravertebral soft-tissue mass of 9.0 cm in maximum diameter at the T11–L3 levels. The lesion was iso- and hypointense on T1-weighted images, heterogeneously iso- and hyperintense on T2-weighted images, where a higher cord-like signal intensity was present; in addition, it showed heterogeneously avid enhancement following the administration of the contrast agent (Fig. 1). He underwent a tumorectomy via the left posterolateral approach. Intraoperatively, a retroperitoneal mass was found at the left side of the vertebral column spanning from T11 to L3. The well-defined tumor was closely adjacent to the vertebrae and displayed no bone erosion. The patients postoperative course was uneventful. No additional treatment was administered. He was well and without evidence of disease at 2 years' follow-up.
|
|
|
Figure 1. Magnetic resonance imaging reveals a left paravertebral soft-tissue mass, which is enhanced with an injection of contrast medium at the T11–L3 levels (arrow). |
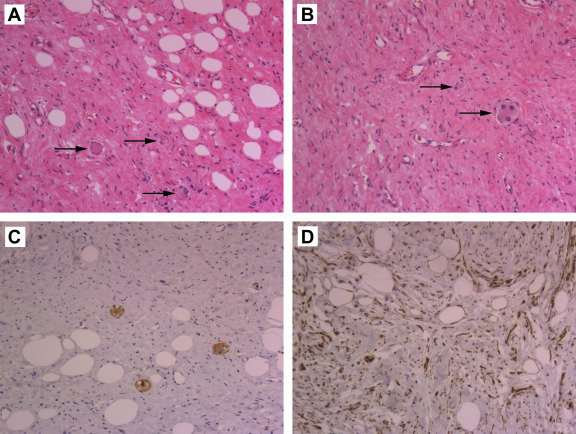
Grossly, the encapsulated neoplasm, measuring 9.5 cm × 9.0 cm × 2.5 cm, had a white to yellowish cut surface and rubbery consistency. Histological examination revealed an encapsulated lesion that consisted of areas of ganglioneuroma admixed with areas of mature fat (Fig. 2A). Ganglioneuroma regions showed fascicular growth patterns of mature Schwann cells within collagenous stroma. Obvious paliform structures were not observed. Scattered throughout the schwannian backdrop were mature ganglion cells. Characteristically, mature, nonatypical adipocytes were identified all over the lesion in varying proportions in various areas of the tumor, such as in lobules and the foci of fat cells. The spindled Schwann cells were ill defined and had scanty, weakly eosinophilic cytoplasm with serpentine nuclei. Nuclei atypia and mitoses were absent, and no cellular atypia, necrosis, or vascular invasion was observed. Mature ganglion cells were scattered or arranged in small clusters between the Schwann cells. The large polygonal ganglion cells had abundant granular cytoplasm. The nuclei were one to three in number and were large, round, vesicular and often eccentrically located; many of these had a prominent nucleolus (Fig. 2B). No neuroblasts were observed despite thorough sampling. Immunohistochemistry showed that the ganglion cells were positive for chromogranin and synaptophysin (Fig. 2C), whereas the schwannian cells were positive for vimentin, S-100 protein (Fig. 2D), and glial fibrillary acidic protein (GFAP).
|
|
|
Figure 2. Histological findings (hematoxylin and eosin staining, A and B) and immunohistochemical features of LG (avidin-biotinylated peroxidase complex method with hematoxylin counterstaining, C and D). (A) Microscopic findings of the specimen reveal an interlacing pattern of adipose tissue, Schwann cells, and mature ganglion cells (arrows; ×100). (B) The ganglion cells are scattered in a background of spindle cells (arrows), the nuclei of which are one to three in number and are large, round, vesicular, and often eccentrically located (×100). (C) Synaptophysin stain shows cytoplasmic positivity in ganglion cells (×100). (D) Both adipic and Schwann cells are positive for S-100 (×100). LG = lipomatous ganglioneuroma. |
3. Discussion
LG, previously called “ganglioneuroma with fatty replacement”, is an extremely rare variant of ganglioneuroma first reported in 1999 by Hara et al.2 It is characterized by a mature adipocytic component admixed with a conventional ganglioneuroma. To date, only five cases of LG have been reported in English literature, including our patient (Table 1).2; 3; 4 ; 5
| Reference | Sex/age (y) | Location | Size (cm) | Follow-up |
|---|---|---|---|---|
| Hara et al2 | F/54 | Mediastinum | 11.0 | NA |
| Duffy et al3 | F/27 | Mediastinum | NA | NA |
| Ko et al4 | F/53 | Mediastinum | 9.0 | NA |
| Adachi et al5 | M/73 | Retroperitoneum | 2.0 | NED/18 months |
| Present case | M/44 | Retroperitoneum | 9.5 | NED/24 months |
Most lesions presented clinically as asymptomatic tumors and were discovered fortuitously. As documented in Table 1, LG seems to occur in middle-aged adult patients (range: 27–73 years; mean: 50.2 years). The sex distribution for the LG cases was 3 females and 2 males. The size of the lesions ranged from 2.0 cm to 11.0 cm (mean: 8.0 cm). The posterior mediastinum and retroperitoneum were the only LG-affected sites reported to date.
LG is grossly sharply marginated with or without encapsulation. Cut sections show alternating areas of whitish and yellowish tumor tissue.2; 3; 4 ; 5 The hallmark microscopic feature of LG is a varying combination of ganglioneuroma-like and lipomatous elements. The ganglioneuroma-like vasculature is composed of relatively mature ganglion cells, Schwann cells, and nerve fibers. The ganglion cells can be identified by their abundant eosinophilic cytoplasm, large nuclei, and prominent nucleoli.3 The lipomatous areas consist of mature adipocytes without atypia. Immunohistochemistry indicates that Schwann cells diffusely and strongly express S-100 protein (Fig. 2C) and GFAP; the ganglion cells are positive for chromogranin and synaptophysin (Fig. 2D).
The histogenesis of the fat element in LG is controversial. Hara et al2 reported that the presence of adipose tissue in the tumor is because of the fat replacement of degenerated tumor. They supposed that the patients neoplasm had arisen years earlier and subsequently experienced spontaneous degeneration with fatty replacement. However, Adachi et al5 reported that it is likely that the adipocytes in the tumor derive from the tumor cells themselves, adipocytic differentiation or lipometaplasia, a perspective with which we agree. As all the reported tumors were well circumscribed, infiltration or entrapment of the surrounding adipose tissue could not have generated this. Therefore, this possibility seems to be unlikely. Moreover, the neural crest is considered as ectomesenchyme, which is capable of differentiating into various cell types. Notably, tumor cells originating in the neural crest may have the capacity for lipomatous differentiation.5
The differential diagnosis of fat-containing retroperitoneal masses includes, but is not limited to, lipoma, liposarcoma, spindle cell lipoma (SCL), and angiomyolipoma. The adipocytic tumors are the most common soft-tissue neoplasms in the retroperitoneum. In fact, in our study, the initial clinical diagnosis was lipoma. Briefly, the simultaneous presence of fatty cells, Schwann cells, and ganglion cells, which is a distinct feature of LG, could not be observed in any of the above mentioned lesions. The absence of atypical lipoblasts and the presence of a characteristic schwannian background can exclude well-differentiated liposarcoma. SCL usually develops in the subcutaneous tissue of the neck and upper back of male patients; the retroperitoneum is an unlikely anatomical site. Differentiating an angiomyolipoma, especially one with a prominent lipomatous component, from an LG is based on the formers distinctive arrangement of the spindle cells around thick-walled vessels and the spindle cells' immunoreactivity for smooth-muscle markers (SMA) and melanocytic markers (HMB45 and Melan-A).
In conclusion, we report the fifth case of LG and the second case of such tumor in the retroperitoneum. More cases of LG must be studied to draw definitive conclusions about its distinct behavior and management.
Acknowledgments
The authors thank Kyle M. and Rachel D. (American Journal Experts) for editing the present article.
References
- 1 C. Gary, H. Robertson, B. Ruiz, et al.; Retropharyngeal ganglioneuroma presenting with neck stiffness: report of a case and review of literature; Skull Base, 20 (2010), pp. 371–374
- 2 M. Hara, S. Ohba, K. Andoh, et al.; A case of ganglioneuroma with fatty replacement: CT and MRI findings; Radiat Med, 17 (1999), pp. 431–434
- 3 S. Duffy, M. Jhaveri, J. Scudierre, et al.; MR Imaging of a posterior mediastinal ganglioneuroma: fat as a useful diagnostic sign; AJNR Am J Neuroradiol, 26 (2005), pp. 2658–2662
- 4 S.M. Ko, D.Y. Keum, Y.N. Kang; Posterior mediastinal dumbbell ganglioneuroma with fatty replacement; Br J Radiol, 80 (2007), pp. e238–e240
- 5 S. Adachi, N. Kawamura, K. Hatano, et al.; Lipomatous ganglioneuroma of the retroperitoneum; Pathol Int, 58 (2008), pp. 183–186
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?