Abstract
The use and commercial applications of biosurfactants in the petroleum industries have been raised during the past decades. Marine bacteria and their efficiency in crude oil recovery has been less studied than terrestrial strain, hence this present study. A novel marine bacterium Bacillus simplex having promising biosurfactant production was isolated from a petroleum hydrocarbon-contaminated coastal sea sediment samples of Nagapattinam fishing harbor, Tamil Nadu, India. This strain showed most economical biosurfactant production with an agro-industrial waste substrate, sunflower oil cake at 54th h time incubation along with the cultural conditions of 20 ppt salinity, 35 °C temperature, and pH 7. The produced biosurfactant was purified, which was accounted at 908 ± 7 mg/L on dry weight basis. The biosurfactant was identified as lipopeptide with a molecular mass of 1111.1 Da which was deduced using TLC, biochemical estimation methods, FT-IR, NMR, and MALDI-TOF MS analysis. Furthermore, this purified lipopeptide surfactant showed consistent and enhanced crude oil recovering efficiency under different salinity conditions (0–30%). Based on the above facts, the isolated novel marine bacterium proved its cheaper production of novel biosurfactant and its promising oil recovering efficiency even at hypersaline conditions. Further, this is the first report of a biosurfactant from the bacterium Bacillus simplex.
Keywords
Bacillus simplex ; Crude oil recovery ; Lipopeptide surfactant ; Marine bacterium ; Sunflower oil cake
Introduction
Surfactants are amphipathic molecules with both hydrophilic and hydrophobic moieties that partition preferentially at the interface between fluid phases that have different degrees of polarity and hydrogen bonding, such as oil and water or air and water interfaces (Banat et al., 2000 ). Almost all surfactants that are currently being produced are derived from petroleum compounds. However, these synthetic surfactants are usually toxic themselves and hardly degraded by microorganisms. Therefore, they are potential sources of pollution and damage to the environment. These hazards associated with synthetic surfactants have, in recent years, drawn much attention to the microbial production of surfactants called biosurfactants (Urum and Pekdemir, 2004 ).
Biosurfactants have several advantages over synthetic surfactants with respect to high biodegradability, low toxicity, good biocompatibility, chemical diversity, greater stability, etc. These properties contribute its great demands in diverse industrial applications such as cosmetics, food, pharmaceutics, agriculture, and petrochemicals (Lotfabada et al., 2009 ). Petrochemical industries have attracted more attention because only 30% of oil in the reservoir can be recovered using primary and secondary recovery techniques (Banat, 1995 ) and tertiary oil recovery that could recover the residual oil using microorganisms or their products like biosurfactants. Biosurfactant molecules reduce the capillary forces that retain the oil in the reservoir, decreases oil viscosity and surface tension, increases emulsification etc., thus promoting its easy flow and resulting increased oil recovery. Nowadays, biosurfactants are mostly preferred in petroleum industries as well as bioremediation applications based on their excellent activities and from an ecological safety perspective (Desai and Banat, 1997 ).
Till date, many studies have been carried out with different microbes to explore the biosurfactant applications and its activity delivering different industrial and environmental significance of these compounds. Despite the fact that marine environment forms a vast majority of the Earths surface as well as comprising a wealthy basin of diverse microorganisms (Satpute et al., 2010 ), moreover, marine microbes have been reported for unique metabolic and physiological capabilities to thrive in extreme habitats and produce novel metabolites which are not often present in terrestrial isolates (Jensen and Fenical, 1994 ). However, very few literature has been solely reported about the marine bacterial isolates for biosurfactant production and its efficiency on enhanced oil recovery when compared to terrestrial isolates. Furthermore, only one commercialized industrial biosurfactant of marine origin which is in use for enhanced oil recovery that is “Emulsan” produced by marine Acinetobacter calcoaceticus RAG-1, isolated from the Mediterranean Sea, has been marketed by Petroferm Research Inc., Amelia Island, FL, USA ( Kosaric and Gray, 1987 ).
Based on the above facts, the present study undertook an investigation from the coastal region of Nagapattinam Fishing Harbor, Tamil Nadu, India for the effective isolation of a potential marine biosurfactant producer, its economic production using cheaper substrates, purification, and biochemical characterization and its crude oil recovering efficiency. To the best of our knowledge, the sampling site was unexplored earlier for the potent biosurfactant producer, and this site is highly contaminated by petroleum hydrocarbon compounds through the regular operating more than 300 mechanized boats for fishing where the probability of achieving most potential biosurfactant producers is expected to be high (Walter et al., 2010 ).
Materials and Methods
Isolation and Screening of Potential Bacteria
Coastal sea sediment samples were collected using the Petersen grab sampler from three different contaminated locations of Nagapattinam fishing harbor, Tamil Nadu, India. During collection, aseptic methods were strictly applied and the samples were transferred to pre-sterilized 250 ml screw type bottle containers which were kept maintained at 4 °C in an ice box and were processed immediately in the lab. One gram of the central portion of the collected samples was serially diluted and spread plated on sea water prepared Bushnell Hass (SWBH) agar containing 0.1% crude oil. After week incubation, the morphological distinct colonies were pure cultured on sea water prepared nutrient agar. All the individual bacterial strains were freshly inoculated for SWBH broth containing 0.1% crude oil and tested for biosurfactant production using oil spreading test (Youssef et al., 2004 ). The strain showing maximum activity was used for further characterization of the biosurfactant.
Molecular Identification of the Potential Strain
Molecular identification of the potential strain was referred with 16S rRNA gene sequence analysis using the bacterial universal primer set of Eubac 27F (5′-AGAG TTTG ATCM TGGC TCAG-3′) and 1492R (5′-GGTT ACCT TGTT ACGA CTT-3′). The PCR was done on a thermal cycler (Eppendorf) with 50 μl reaction mix. The reaction mix contained 10 × amplification buffer (5 μl), 1.5 mM MgCl2 (5 μl), 1 μl of each forward and reverse primer, 1 μl dNTP, and 0.25 μl Taq polymerase. After an initial denaturation at 95 °C for 1 min, amplification was carried out with 35 cycles of 35 s at 94 °C, 40 s at 55 °C, 2 min at 72 °C followed by a final extension for 8 min at 72 °C. The PCR products were analyzed by electrophoresis using 1.2% agarose gel (Genei). The 16S rRNA gene sequence obtained from the potential bacterium was compared with other bacterial sequences by using NCBI megaBLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi ) for their pairwise identities. Phylogenetic analysis was done using the neighbor-joining tree based on the Maximum Composite Likelihood model with MEGA 6.0 (www.megasoftware.net ).
Fermentation Conditions
The potential strain was standardized for maximum biosurfactant production by adopting search technique, i.e., varying one parameter at a time. The fermentation process was carried out in a 3 L laboratory fermentor (Scigenics, India) with 2.1 L working volume using SWBH broth as the basal medium with the basal culture conditions of pH 8.0, 37 °C temperature, 34 ppt salinity, agitation at 150 rpm, and aeration at 1.0 VVM. In addition to that, accepta 2904 was used as an antifoaming agent on the required basis within the concentration of 10–50 ppm. The inoculum was prepared with the exponential phase culture of the potential strain using Zobell marine broth where the optical density (OD 620 nm) of the inoculum culture was adjusted to 0.1 as per the procedure of McFarland turbidity 0.5 standard which was equivalent to the bacterial concentration of 1 × 108 CFU/ml. The biosurfactant production was monitored using 24 h emulsification index (E24 ) (Sheppard and Mulligan, 1987 ).
Influence of Different Economic Substrates and Time Course on Biosurfactant Production
Different agro-industrial waste products were examined as the cheaper nutritional source for the enhanced biosurfactant production. Various cheaper substrates such as peanut oil cake, coconut oil cake, mustard oil cake, palm oil cake, and sunflower oil cake with 2% (v/w) were individually determined in the basal mineral medium for maximum biosurfactant production. For time course of biosurfactant production, the production medium was monitored for 120 h with an interval of 6 h starting from the lag phase to stationary phase under batch culture conditions during which the production peak time was determined. The time course on production was plotted with reference to biosurfactant production and bacterial growth.
Recapitulation of pH, Temperature, and Salinity Condition
Standardizing optimal parameters like salinity, temperature, and pH conditions play a vital role in fermentation progress for maximum production. The effect of various salinity parameters was standardized between 0 and 34 ppt (0, 10, 20, 30, 34 ppt), which was prepared by diluting the purified natural sea water, likewise, the influence of different temperature conditions from 25–45 °C were applied to the production medium. Further, different pH conditions between pH 6 and 9 were tested for the effective medium optimization.
Biosurfactant Purification
At the standardized cultural conditions, the cell-free supernatant was subjected to the acid precipitation method (Nitschke and Pastore, 2006 ) for recovering the crude biosurfactant and the acidified biosurfactant was neutralized using phosphate buffer (pH 7) solution. The crude extract was dissolved in 5 ml of acetonitrile, filtered through a 0.2 μm syringe filter and separated by Reverse Phase (RP)—C18 silica gel (230–400 mesh) column held at room temperature. The solvent system consisted of distilled water (solvent A) and acetonitrile (solvent B). The compounds were eluted at a flow rate of 0.5 ml/min with a stepwise gradient from the mixture A:B (0:100, vol/vol) to A:B (100:0, vol/vol) and the ratio were designed as 0:100, 2.5:97.5, 5: 95, 7.5:92.5 to 100:0 (vol/vol). Forty-one fractions were collected which were rotary vacuum evaporated and every fraction was screened for the emulsification index (E24 ). The purity of biosurfactant was checked by thin layer chromatography and the fraction showing maximum activity was lyophilized for further studies.
Biochemical Characterization and Molecular Weight Determination
For biochemical characterization, the purified biosurfactant was initially determined for biochemical characterization using silica gel TLC plate and the chromatograms were developed with 96% ethanol:water (7:3) for peptide, chloroform:acetic acid:water (60:30:10) for sugars, and chloroform:methanol:water (65:25:4) for lipids. Visualizing reagents used were ninhydrin reagent to detect peptides, anthrone reagent to examine sugars, and lipid portion was evidenced using rhodamine B reagent, followed by estimating its total content of protein, carbohydrate, and lipid using Lowrys method (Lowry et al., 1951 ), phenol sulphuric acid method (Dubois et al., 1956 ), and total free fatty acids (Folch et al., 1957 ), respectively. FT-IR and NMR was used for further analysis of this biosurfactant to elucidate its functional groups. One milligram of freeze-dried biosurfactant was grounded with 100 mg of KBr. Infrared absorption spectra were recorded on a Thermo Niocolet, AVATAR 330 FT-IR system with a spectral resolution and a wave number accuracy of 4 and 0.01 cm− 1 , respectively, and a KBr pellet was used as the background reference. 1 H NMR spectra of the biosurfactant were recorded on a Bruker AV600 NMR spectrometer (Germany) using deuterated chloroform (CDCl3 ) as the solvent. Chemical shifts were expressed in parts per million (ppm) down field from an internal standard of tetra methylsilane (TMS). MALDI-TOF MS was used to determine the molecular weight of this purified biosurfactant. MS was analyzed using a Voyager DE-Pro MALDI-TOF spectrometer (Applied Biosystems, Inc., CA, USA) in reflector mode with an accelerating voltage of 20 kV. Equal volume (2 μl) of the purified sample fraction was mixed with the equal volume of matrix solution, i.e. 0.1% a-cyano-4-hydroxycinnamic acid in acetonitrile–water–TFA (50:50:0.01, vol/vol/vol). After proper mixing, the sample was spotted on the target plate, dried, and placed inside the sample cabinet. Molecules were separated based on their mass and were detected by the ion detector set in reflector mode.
Application of Biosurfactant in Crude Oil Removal from Contaminated Sand
The application of the purified lipopeptide surfactant on enhanced oil recovery was estimated as per the earlier method described by Sobrinho et al. (2008) with some modification in which the acid washed beach sand impregnated with 5.0 ml of crude oil was used instead of waste motor oil per 100 g of soil in the recovery process as well as different salinity conditions were applied to the experimental setup to envisage biosurfactant efficiency on oil recovery. Experiments were carried out using 20 g crude oil impregnated sand with the addition of 60 ml of 100 μg/ml biosurfactant concentrations in 250 ml conical flasks under different salinity conditions ranging from 0 to 30% salinity. The different salinity conditions were applied using artificial sea salt (Himedia, India); in the abovementioned experiments, setup and controls were used for every parameter using the synthetic surfactant, sodium lauryl sulphate (SLS) as positive control and distilled water without added any surfactant as negative control. The experimental samples were incubated under rotary shaker conditions (150 rpm) and incubated for 24 h. After incubation, the samples were centrifuged at 5000 g for 20 min for the separation of the laundering solution and the sand. The remaining amount of oil residing in the sand after the oil recovery by the biosurfactant action was gravimetrically determined as the amount of material extracted from the sand by hexane.
Analytical Methods
The oil spreading test was done by adding 20 ml of distilled water to a Petri dish with a diameter of 8 cm in which 15 μl of crude oil was dropped to form a thin oil layer on the surface of the water and then 10 μl of a centrifuged supernatant was added onto the surface of the oil. The diameter of the clear zone was observed under light and measured. The E24 was evaluated with crude oil added to cell free broth in a ratio of 1:1 and vortexed vigorously for 2 min. After 24 h incubation, the height of the emulsified layer was measured and divided by the total height of the liquid layer and multiplied by 100. Both the tests were conducted at room temperature (26–28 °C). For the estimation of the dry weight of cell biomass on biosurfactant production, culture broth was centrifuged at 3000 rpm for 15 min and the obtained cell pellet was hot air oven dried and weighed. All the values were expressed as mean ± standard deviation of three replicate experiments.
Results and Discussion
Screening and Identification for Potential Biosurfactant Bacteria
The procured sediment samples were serially diluted using sterile natural sea water and spread plated on SWBH agar containing 0.1% crude oil. After incubation, the plates were visualized for distinct morphological colonies which were isolated and pure cultured on sea water prepared nutrient agar. There were 23 morphological distinct bacterial strains were obtained which were named as SBN 1–23 and screened for biosurfactant production using oil spreading method. Among the isolates, the strain SBN 19 showed maximum activity of 54 ± 2 mm in oil spreading test, whereas the other strains showed lesser activity than it. The present study used oil spreading method for the effective screening, which is a reliable method to detect and screen potential producers from diverse microorganisms (Plaza et al., 2006 ), and the method is rapid, which can be applied when the activity and quantity of biosurfactant are low (Youssef et al., 2004 ). The phylogenetic position of the strain was examined by BLAST analysis and the results revealed that the strain belongs to the genus Bacillus of the family Bacillaceae. Based on the sequence homology, the strain SBN 19 showed the highest similarity of more than 99% with Bacillus simplex WN570. Following these comparisons, the strain was identified as Bacillus simplex SBN 19 and the 16S rRNA sequence was deposited in GenBank with the accession number KU170605. Phylogenetic tree of Bacillus simplex SBN 19 was plotted with their closest NCBI strains using the neighbor-joining tree based on the Maximum Composite Likelihood model ( Fig. 1 ). This strain was chosen for the further characterization based on its biosurfactant activity, and this strain has not been reported earlier for the biosurfactant characterization.
|
|
|
Fig. 1. Phylogenetic analysis was performed for the potential strain Bacillus simplex SBN 19 with their closest NCBI (BLASTn) strains based on the 16S rRNA gene sequences. This analysis was done using the neighbor-joining tree based on the Maximum Composite Likelihood model with MEGA 6.0. Numbers at nodes indicate levels of bootstrap values (expressed as percentages of 1000 replications) and the black dot indicates the potential strain of this study. |
Standardization of Economic Substrate and Time Course on Biosurfactant Production
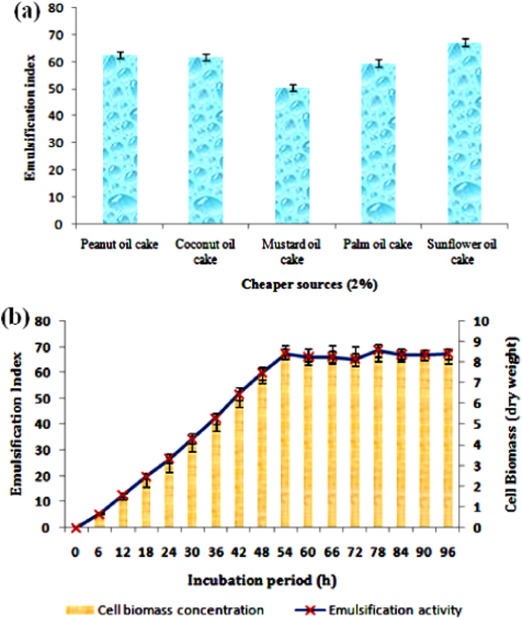
Different agro-industrial waste substrates were chosen for the examination of economic and enhanced biosurfactant production. All the examined cheaper source showed biosurfactant production above 50% E24 , but the maximum production was observed using sunflower oil cake with 67.2 ± 1.5 E24 (Fig. 2 a). The production of biosurfactant with cheaper substrates like oil cakes offers good alternative to traditional applications by their exploitation in the production of environmentally friendly green products. Fermentation processes utilizing oil cakes are attractive due to its relatively cheaper availability throughout the year, which makes it even more favorable when economics is considered (Ramachandran et al., 2007 ). Similar to this, Thavasi et al. (2011) used peanut oil cake for the production of biosurfactant from a marine Pseudomonas aeruginosa. The time course of biosurfactant production by Bacillus simplex as a function of time ( Fig. 2 b) depicted that the biosurfactant secretion was detected when the organism was in the early logarithmic phase of bacterial growth, which achieved its maximum during the end logarithmic phase or initiation of the stationary growth phase of the bacterium (54th h). Further, the maximum emulsification activity of 67.3 ± 3 was maintained till the predicted time course of the stationary phase with 8.49 ± 0.5 cell biomass. The production profile of biomass and biosurfactant as a function of time revealed that it was primary metabolite (Khopade et al., 2012 ). An earlier report also suggested that a similar growth-dependent pattern of biosurfactant production was observed in case of a marine Bacillus megaterium ( Thavasi et al., 2008 ).
|
|
|
Fig. 2. Influence of different cheaper substrates (a) and time course (b) on biosurfactant production from a marine Bacillus simplex SBN 19. |
Recapitulation of Salinity, Temperature, and pH Condition
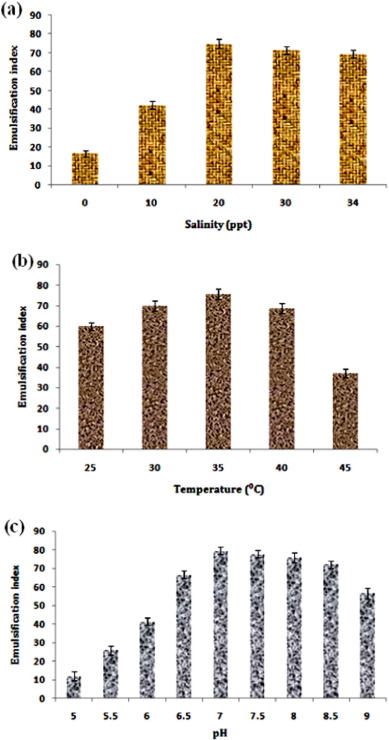
Since the bacterium is off from marine origin, so the optimization of salinity conditions makes an important estimation about the affinity of the organism to the salt concentration. The isolated strain showed maximum production of biosurfactant at 20 ppt with 74.7 ± 2.5% E24 (Fig. 3 a). Furthermore, the strain showed much decreased biosurfactant production as well as growth with decreasing salinity concentration which proved its strong adaptation in the marine environment. However, Kiran et al. (2009) observed that the 30 ppt salinity concentration showed maximum biosurfactant production with marine-derived fungus Aspergillus ustus MSF3. Regarding optimization for determining optimum temperature, the strain showed a strong dependence on biosurfactant production with varying temperature conditions and the peak production was achieved in 35 °C with 75.7 ± 2.2% E24 and lowest activity was observed in 45 °C with 37.2 ± 2.2% E24 (Fig. 3 b). Close to this, the optimal temperature for maximum rhamnolipid production by Pseudomonas aeruginosa J4 strain begins from 30 to 37 °C (Wei et al., 2005 ). The other important characteristic of most organisms is their strong dependence on the pH for cell growth and the production of metabolites. The isolated strain showed the highest biosurfactant production at pH 7.0 with 79.4 ± 2.2% E24 (Fig. 3 c); further, the isolated strain showed lowest biosurfactant production at acidic conditions below 6. Similar to the present investigation, the result observed by Mawgoud et al. (2008) revealed maximum biosurfactant production at pH 7 using B. subtilis .
|
|
|
Fig. 3. Effect of salinity (a), temperature (b), and pH (c) on biosurfactant production from a marine Bacillus simplex SBN 19. |
Purification and Biochemical Characterization of Biosurfactant
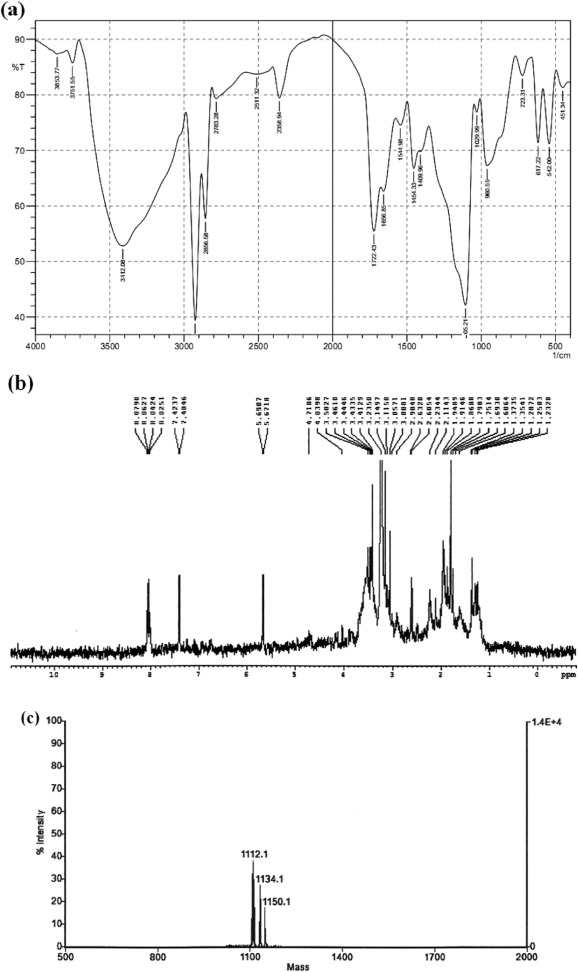
From the optimized conditions, the crude biosurfactant was extracted by acid precipitation technique, and further, it was purified using silica gel column chromatography. The maximum emulsification activity of 89 ± 2.3% E24 was observed at the 45:55 acetonitrile:water fraction which was accounted at 908 ± 7 mg/L on dry weight basis. The purified biosurfactant was analyzed for the presence of biochemical components using silica gel TLC plates, which showed peptide and lipid components at Rf values of 0.53 and 0.65, respectively. On biochemical estimating methods, the isolated biosurfactant showed the presence of 81 ± 0.8% protein and 19 ± 0.1% lipid and the carbohydrate was found to be absent. The biosurfactant was further confirmed to be lipopeptide nature based on the FT-IR and NMR spectral analysis (Fig. 4 a and b). Significant absorption peak at 3412 cm− 1 region exhibited the N–H stretching vibration of peptides. The characteristic absorbance at 1656 cm− 1 predicted the stretching mode of CO–N bond and the peak at 1544 cm− 1 evidenced the deformation mode of the N–H bond combined with C–N stretching. The important absorbance peaks at 723, 1409, 1454, 2856, and 2926 cm− 1 revealed the existence of aliphatic long hydrocarbon chain of the biosurfactants. Other important peak observed at 941 showed the presence of hydroxyl group (OH) present in the amino acid.
|
|
|
Fig. 4. FT-IR (a), NMR spectrum (b), and MALDI-TOF mass spectrometry (c) of a lipopeptide surfactant from a marine Bacillus simplex SBN 19. |
The 1 H NMR results also showed that the presence of a long aliphatic chain (CH2 )n was observed between 1.2328–2.6320 ppm, simultaneously, the CH2 –OH group of the amino acid was predicted at the 4.0398–4.7186 ppm. The important absorption peak of hydrogen atom in the amide group (C
O–NH) was observed between 5.6907–8.0790 ppm and hydrogen atom of the group CH2 C
O was predicted at 2.9048–3.2358 ppm which revealed the conjugation between the amino acids and the fatty acid. Further, the chemical complex studied using FT-IR and NMR spectral analysis showed the evidence of peptide and aliphatic long hydrocarbon fatty acid chain. The functional group analysis carried out in the present investigation corroborates with the similar pattern of wave numbers and functional groups observed for lipopeptide surfactants from different research (Pereira et al ., 2013 ; de-Faria et al ., 2011 ; Pornsunthorntawee et al ., 2008 ). The molecular weight of this lipopeptide surfactant was studied using MALDI-TOF spectral analysis, which revealed a cluster of three molecules at m /z 1112.1, 1134.1, and 1150.1. These molecules were attributed to the protonated ion and to the adducts of sodium and potassium ions ( Fig. 4 c). The molecular mass of this lipopeptide is 1111.1 Da, and this molecular weight was different from the earlier reports which represent it as new lipopeptide surfactants.
Application of Biosurfactant in Crude Oil Removal from Contaminated Sand
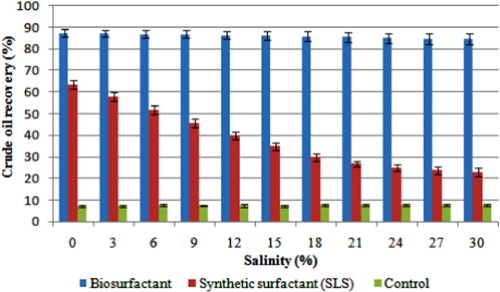
In the present study, the purified lipopeptide surfactant was used for enhanced crude oil recovery from the contaminated sand under different salinity conditions. The biosurfactant revealed a consistent and promising oil recovery rate on all the tested different parameters (Fig. 5 ). The maximum tested salinity concentration of 30% showed 84.7 ± 2.4% crude oil recovery, which was very close result to the peak recovery rate examined at 0% salinity (87.4 ± 1.7%). But the positive control using SLS removed maximum concentration of crude oil at 0% salinity (63.4%) whereas removed only 22.9% at the highest tested concentration of 30% salinity. This comparative observation clearly evidenced the consistent crude oil recovering efficiency of this new lipopeptide surfactant even at extreme salinity conditions. The negative control using distilled water without any added surfactant removed only 7.1–7.9% of crude oil from the sand. Further, these results hold as the significant data for the promising application of this isolated biosurfactant in petroleum industries for enhanced oil recovery and cleaning of oil contaminants, reducing oil viscosity during crude oil transportation as well as for bioremediation, in particular, this biosurfactant can act excellently even at severe salinity condition.
|
|
|
Fig. 5. Comparison of crude oil recovering efficiency of a lipopeptide surfactant from a native marine Bacillus simplex SBN 19 with the positive control using the synthetic surfactant, SLS, and the negative control using distilled water at different salinity conditions. |
Conclusion
The isolated novel marine bacterium Bacillus simplex in the present study showed a significant response to all the cheaper substrates utilized; in particular, the maximum production was observed with the sunflower oil cake which evidenced its most economical cheaper production. The purified lipopeptide surfactant exhibited a different molecular mass than the earlier reports which constituted it as a novel compound. Furthermore, an interesting observation on enhanced and consistent crude oil recovering efficiency at wide range of salinity parameters proved that it could be an ideal product for the petroleum industries as well as environmental applications even at hypersaline environmental conditions.
Conflict of Interest
The authors declare that they have no competing interests.
Acknowledgment
The authors very gratefully acknowledge Shri R. J. Antony Raj, Chairman and Mrs. A. Agnes Antony Raj, Secretary, SIMPRA, SIRO Institution, Thanjavur, Tamil Nadu, India, for providing lab facilities and supporting our research.
References
- Banat, 1995 I.M. Banat; Biosurfactants production and possible uses in microbial enhanced oil recovery and oil pollution remediation: a review; Bioresour. Technol., 51 (1995), pp. 1–12
- Banat et al., 2000 I.M. Banat, R.S. Makkar, S.S. Cameotra; Potential commercial applications of microbial surfactants; Appl. Microbiol. Biotechnol., 53 (2000), pp. 495–508
- de-Faria et al., 2011 A.F. de-Faria, D.S. Teodoro-Martinez, G.N. de-Oliveira Barbosa, B.G. Vaz, I.S. Silva, J.S. Garcia, M.R. Totola, M.N. Eberlin, M. Grossman, O.L. Alves, L.R. Durrant; Production and structural characterization of surfactin (C14/Leu7) produced by Bacillus subtilis isolate LSFM-05 grown on raw glycerol from the biodiesel industry ; Process Biochem., 46 (2011), pp. 1951–1957
- Desai and Banat, 1997 J.D. Desai, I.M. Banat; Microbial production of surfactants and their commercial potential; Microbiol. Mol. Biol. Rev., 61 (1997), pp. 47–64
- Dubois et al., 1956 M. Dubois, K.A. Gilles, J.K. Hamilton, P.A. Rebers, F. Smith; Colorimetric method for determination of sugars and related substances; Anal. Chem., 28 (1956), pp. 350–356
- Folch et al., 1957 J. Folch, M. Lees, G.H. Sloane-Stanley; A simple method for the isolation and purification of total lipides from animal tissues; J. Biol. Chem., 226 (1957), pp. 497–509
- Jensen and Fenical, 1994 P.R. Jensen, W. Fenical; Strategies for the discovery of secondary metabolites from marine bacteria: ecological perspectives; Annu. Rev. Microbiol., 48 (1994), pp. 559–584
- Khopade et al., 2012 A. Khopade, B. Ren, X. Liu, K. Mahadik, L. Zhang, C. Kokare; Production and characterization of biosurfactant from marine Streptomyces species B3 ; J. Colloid Interface Sci., 367 (2012), pp. 311–318
- Kiran et al., 2009 G.S. Kiran, T.A. Hema, R. Gandhimathi, J. Selvin, T.A. Thomas, T.R. Ravji, K. Natarajaseenivasan; Optimization and production of a biosurfactant from the sponge associated marine fungus Aspergillus ustus MSF3. ; Colloids Surf. B: Biointerfaces, 73 (2009), pp. 250–256
- Kosaric and Gray, 1987 N. Kosaric, N.C.C. Gray; Cairns WL. Introduction: Biotechnology and the surfactant industry; ,in: N. Kosaric, W.L. Cairns, N.C.C. Gray (Eds.), Biosurfactants and Biotechnology, Surfactant Science Series, vol. 25, , Marcel Dekker, Inc., New York (1987), pp. 1–19
- Lotfabada et al., 2009 T.B. Lotfabada, M. Shourianc, R. Reza; An efficient biosurfactant-producing bacterium Pseudomonas aeruginosa MR01, isolated from oil excavation areas in south of Iran ; Colloids Surf. B: Biointerfaces, 11 (2009), pp. 445–3455
- Lowry et al., 1951 O.H. Lowry, N.J. Rosenbrough, A.L. Farr, R.J. Randall; Protein measurement with the folin phenol reagent; J. Biol. Chem., 193 (1951), pp. 265–275
- Mawgoud et al., 2008 A.M.A. Mawgoud, M.M. Aboulwafa, N.A. Hassouna; Characterization of surfactin produced by Bacillus subtilis isolate BS5 ; Appl. Biochem. Biotechnol., 150 (2008), pp. 289–303
- Nitschke and Pastore, 2006 M. Nitschke, G.M. Pastore; Production and properties of a surfactant obtained from Bacillus subtilis grown on cassava wastewater ; Bioresour. Technol., 97 (2006), pp. 336–341
- Pereira et al., 2013 J.F.B. Pereira, E.J. Gudina, R. Costa, R. Vitorino, J.A. Teixeira, J.A.P. Coutinho, L.R. Rodrigues; Optimization and characterization of biosurfactant production by Bacillus subtilis isolates towards microbial enhanced oil recovery applications ; Fuel, 111 (2013), pp. 259–268
- Plaza et al., 2006 G. Plaza, I. Zjawiony, I. Banat; Use of different methods for detection of thermophilic biosurfactant-producing bacteria from hydrocarbon contaminated bioremediated soils; J. Pet. Sci. Eng., 50 (2006), pp. 71–77
- Pornsunthorntawee et al., 2008 O. Pornsunthorntawee, N. Arttaweeporn, S. Paisanjit, P. Somboonthanate, M. Abe, R. Rujiravanit, S. Chavadej; Isolation and comparison of biosurfactants produced by Bacillus subtilis PT2 and Pseudomonas aeruginosa SP4 for microbial surfactant-enhanced oil recovery ; Biochem. Eng. J., 42 (2008), pp. 172–179
- Ramachandran et al., 2007 S. Ramachandran, S.K. Singh, C. Larroche, C.R. Soccol, A. Pandey; Oil cakes and their biotechnological applications – a review; Bioresour. Technol., 98 (2007), pp. 2000–2009
- Satpute et al., 2010 S.K. Satpute, I.M. Banat, P.K. Dhakephalkar, A.G. Banpurkar, B.A. Chopade; Biosurfactants, bioemulsifiers and exopolysaccharides from marine microorganisms; Biotechnol. Adv., 28 (2010), pp. 436–450
- Sheppard and Mulligan, 1987 J.D. Sheppard, C.N. Mulligan; The production of surfactin by Bacillus subtilis grown on peat hydrolysate ; Appl. Microbiol. Biotechnol., 27 (1987), pp. 110–116
- Sobrinho et al., 2008 H.B.S. Sobrinho, R.D. Rufino, J.M. Luna, A.A. Salgueiro, G.M.C. Takaki, L.F.C. Leite, L.A. Sarubbo; Utilization of two agro industrial by-products for the production of a surfactant by Candida sphaerica UCP0995 ; Process Biochem., 43 (2008), pp. 912–917
- Thavasi et al., 2008 R. Thavasi, S. Jayalakshmi, T. Balasubramanian, I.M. Banat; Production and characterization of a glycolipid biosurfactant from Bacillus megaterium using economically cheaper sources ; World J. Microbiol. Biotechnol., 24 (2008), pp. 917–925
- Thavasi et al., 2011 R. Thavasi, V.R.M. Subramanyam Nambaru, S. Jayalakshmi, T. Balasubramanian, I.M. Banat; Biosurfactant production by Pseudomonas aeruginosa from renewable resources ; Indian J. Microbiol., 51 (2011), pp. 30–36
- Urum and Pekdemir, 2004 K. Urum, T. Pekdemir; Evaluation of biosurfactants for crude oil contaminated soil washing; Chemosphere, 57 (2004), pp. 1139–1150
- Walter et al., 2010 V. Walter, C. Syldatk, R. Hausmann; Screening concepts for the isolation of biosurfactant producing microorganisms; Adv. Exp. Med. Biol., 672 (2010), pp. 1–13
- Wei et al., 2005 Y.H. Wei, C.L. Chou, J.S. Chang; Rhamnolipid production by indigenous Pseudomonas aeruginosa J4 originating from petrochemical wastewater ; Biochem. Eng. J., 27 (2005), pp. 146–154
- Youssef et al., 2004 N. Youssef, K. Duncan, D. Nagle, K.N. Savage, R.M. Knapp, M.J. McInerney; Comparison of methods to detect biosurfactant production by diverse microorganisms; J. Microbiol. Methods, 56 (2004), pp. 339–347
Document information
Published on 27/03/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?