Summary
Background
The traditional surgical approach to the excision of persistent urachal remnants is a lower midline laparotomy or semicircular infraumbilical incision. The aim of this study is to report our experience with laparoscopic urachus excision as a minimally invasive diagnostic and surgical technique.
Methods
This study was a prospective study involving patients who were diagnosed with persistent urachus and underwent laparoscopic excision. The morbidity, recovery, and outcomes of surgery were reviewed.
Results
Fourteen patients (8 men) with a mean age of 22.8 ± 6.42 years underwent laparoscopic excision. All patients presented with discharge from the umbilicus. Although four patients had no sonographic evidence of a patent urachus, a diagnostic laparoscopy detected a patent urachus that was excised laparoscopically. One patient required laparoscopic reoperation for persistent discharge, and one patient presented with bladder injury, which was repaired via a small Pfannenstiel incision without any morbidity. The mean operative time was 71.1 ± 0.28 minutes, and the mean duration of hospital stay was 1.3 ± 1.38 days. Pathological examination confirmed a benign urachal remnant in all cases.
Conclusion
Laparoscopy is a useful alternative for the management of persistent or infected urachus, especially when its presence is clinically suspected despite the lack of sonographic evidence. The procedure is associated with low morbidity, although a small risk of bladder injury exists, particularly in cases of severe active inflammation. Recurrence is uncommon and was caused by inadequate excision of inflammatory tissue in our series that was easily managed laparoscopically.
Keywords
bladder;laparoscopy;urachal cyst;urachus;urachal remnant;urachal sinus
1. Introduction
Urachal remnants represent a failure in the obliteration of the allantois at birth that connects the bladder to the umbilicus.1 It is a rare congenital anomaly, occurring in 1.6% of children under 15 years of age and in 0.063% of adults.2 The persistence of the urachal lumen after birth manifests in a variety of clinical presentations, of which recurrent periumbilical discharge is the most common.3 There are four variants of urachal anomalies: urachal cysts, sinus, diverticulum, and a patent urachus. Management of urachal remnants requires wide local excision of the urachus and inflamed adjacent extraperitoneal tissue, which is traditionally performed via a lower midline laparotomy incision. Although effective, it shares the same associated morbidities of any laparotomy incision with inherent postoperative pain, risk of wound infection, bleeding, and slow return to normal activities. Since its first description in 1993,4 laparoscopic surgery has been considered as an alternative to conventional open resection of urachal remnants. The laparoscopic approach has the benefits of being able to confirm the presence of urachal remnants and enable magnified dissection along the extraperitoneal plane until the dome of the bladder in the space of Retzius with minimal postoperative pain and rapid recovery to normal activities. The benefit of superior cosmesis compared to a lower midline incision is an added bonus. Studies reporting on the laparoscopic management of urachal remnants and outcomes are scarce. There are only a few isolated case reports or small case series owing to the rarity of this pathology. In this retrospective review, we present our centers experience with the laparoscopic management of symptomatic urachal remnants and our short-term outcomes.
2. Materials and methods
Between February 2010 and June 2013, 14 consecutive patients who were diagnosed with symptomatic urachal remnants underwent laparoscopic excision under the Department of Surgery, Sarawak General Hospital, Kuching, Malaysia. Patient demographics, ultrasonographic findings, operative procedure and findings, complications, postoperative outcomes, histopathological findings, and follow-up details were documented and stored in a prospectively maintained database. All patients presenting with recurrent umbilical discharge were enrolled and underwent sonographic assessment to confirm the diagnosis of persistent urachal remnants prior to being counseled for diagnostic laparoscopy with the intention of laparoscopic excision of urachal remnants if present. Patients who had overt infection and presented with umbilical abscesses were treated with drainage of abscesses via a small infraumbilical incision and prescribed with parenteral antibiotics until clinically there was regression of the surrounding cellulitis and periumbilical discharge. These patients were then discharged with oral antibiotics to complete a 10-day course of Amoxicillin and Sulbactam with an elective date for laparoscopic excision within 6 weeks. This study was approved by the hospitals ethics committee, and the permission to publish this paper was obtained from the Director General of Health, Malaysia.
2.1. Surgical technique
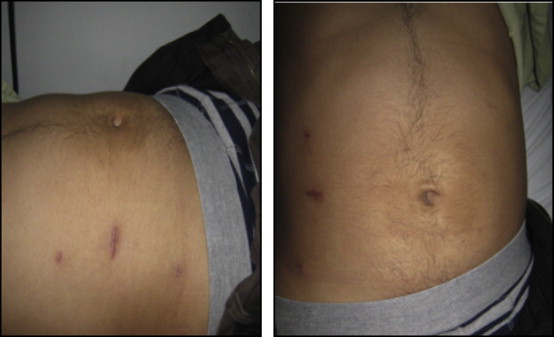
Laparoscopic excision was performed by one of three laparoscopically proficient surgeons at this tertiary-care hospital. All patients were administered a single dose of parenteral Amoxicillin/Sulbactam during induction of anesthesia. Foleys catheterization of the urinary bladder was routinely performed to initially decompress the bladder during the initial dissection of the urachal remnants and later enable retrograde distension of the bladder prior to ligating the insertion of the urachus at the dome of the bladder. The patient is placed in the supine position, with both arms secured at the sides of the body. The surgeon and the camera surgeon stand on the right side of the patient with the monitor on the opposite side of the patient. The first trocar (12 mm, Endopath Xcel; Ethicon Endo-Surgery, Ohio, USA) is inserted at the level of the umbilicus just medial to the anterior axillary line via direct visualization through the trocar. Once the port is placed inside the peritoneal cavity, a CO2 pneumoperitoneum is created with a pressure of 12 mmHg. A 10-mm, 30° angled lens camera is typically used. Under direct vision, 5-mm trocars are placed similarly medial to the anterior axillary line at the level of the right anterior iliac spine and two fingerbreadths inferior to the costal margin with care taken to ensure adequate spacing between all three ports (ideally, a hands breadth) to avoid crowding of instruments and enable triangulation (Fig. 1).
|
|
|
Figure 1. Postoperative port site scar 6 weeks after surgery. |
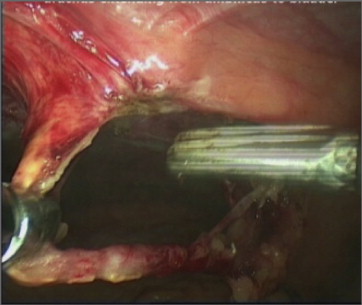
Wide local excision is performed using a traumatic ratcheted grasper to hold the urachal remnants, and dissection is accomplished using a combination of hook and scissors with electrocautery. The urachal remnants and adjacent tissues should be dissected off the transversalis fascia cranially until the umbilicus and caudally until the space of Retzius, where the urachal remnants insert into the dome of the bladder (Fig. 2). The urachus is usually easily identified midway between the umbilicus and the urinary bladder in the midline. The urachus is clipped with 5-mm hem-o-lock clips (Weck; Teleflex Medical, Co Westmeath, Ireland) and transected near the umbilicus. The bladder is distended to facilitate identification of the insertion of the urachal remnants into the bladder, which is ligated with a loop of chromic catgut and cut near the dome of the bladder.
|
|
|
Figure 2. Urachus extending from the umbilicus to the bladder. |
3. Results
The patient demographics are summarized in Table 1, whereas surgical outcomes are shown in Table 2. There were eight men (57.1%) and six women (42.9%) with a mean age of 22.8 ± 6.42 years. The most common presentation was umbilical discharge seen in 11 patients (78.6%), and the remaining three patients presented with an umbilical abscess (21.4%).
| Variable | Value |
|---|---|
| Number of patients | 14 |
| Age (y), mean (standard deviation) | 22.8 (6.42) |
| Sex (male/female) | 8:6 |
| Presence of sonographic evidence of patent urachus | 10 |
| Variable | Value |
|---|---|
| Operative time (min), mean (SD) | 71.1 (0.28) |
| Postoperative hospital stay (d), mean (SD) | 1.3 (1.38) |
| Operation-related complication | 1 |
| Conversion to laparotomy | 0 |
| Foley catheterization (d), mean (SD) | 6.2 (2.39) |
| Return to normal activities (d), mean (SD) | 4.5 (1.87) |
SD = standard deviation.
Although abdominal ultrasonography was performed for all patients, only 10 patients (71.4%) had sonographic evidence of patent urachal remnants. Intraoperatively, 11 patients (78.6%) were found to have urachal sinus and three patients (21.4%) had urachal cysts. There were three patients who presented acutely with umbilical abscesses and overlying cellulitis. Two of these patients were initially managed with parenteral antibiotics and drainage of the abscesses via a small infraumbilical incision. Wide local excision was performed electively 6 weeks later after the inflammation had subsided. Laparoscopic wide local excision was performed in one patient in the acute setting but was complicated by iatrogenic bladder injury that was detected immediately and repaired via a small Pfannenstiel incision without any complication.
Laparoscopic wide local excision was successfully completed in all cases without conversion to a lower midline incision. The mean operative time was 71.1 ± 0.28 minutes. The mean postoperative duration of hospital stay was 1.3 ± 1.38 days. All patients were discharged with an indwelling Foley catheter, which remained in place for 3–10 days and was removed as an outpatient procedure. All patients had a successful removal of the catheter as an outpatient procedure.
There were no instances of intraoperative complications such as bleeding or bowel injury, although one patient required bladder repair owing to an inadvertent injury that recovered uneventfully as previously mentioned. No patients demonstrated leak on follow-up retrograde cystourethrography, and no patients had voiding difficulties.
There was one patient who complained of persistent umbilical discharge postoperatively. Ultrasonography showed no evidence of an umbilical collection or urachal remnants. Diagnostic laparoscopy revealed that the discharge originated from significant residual inflammatory tissue beneath the umbilicus. A laparoscopic excision of the inflamed tissue was performed, and his symptoms resolved. The histopathological examination revealed no urachal remnant in the resected tissue and the specimen was composed of inflammatory granulation tissue.
4. Discussion
The management of symptomatic urachal remnants in adults differs from that in children.2 ; 3 Conservative management is often advocated for children who are diagnosed within the 1st year of life. There are several reports showing that spontaneous involution of the urachus often occurs within 1 year of birth with complete resolution of symptoms.2; 5 ; 6 However, persistent urachal remnants in adults eventually become symptomatic and require surgical intervention.
The variable and nonspecific clinical presentation of the persistent urachal remnants represents a diagnostic challenge. Various imaging modalities have been used to confirm its presence including ultrasonography, computed tomography, voiding cystourethrography, and sinography. Ultrasonography is by far the most popular7; 8 ; 9 as it is easily performed, noninvasive, and has no radiation concerns. Furthermore, the location of urachal remnants on the posterior aspect of the anterior peritoneal wall is easily visualized by ultrasonography without interference from loops of bowels.8 ; 10 Computed tomography with its multiplanar images and high spatial resolution capability is a good diagnostic imaging modality, but it has been largely reserved for patients suspicious of urachal carcinoma in our hospital. Interestingly, in our series, four patients (28.6%) had no demonstrable urachal remnants on ultrasonography despite the typical clinical presentation. The diagnostic accuracy of ultrasonography for urachal remnants has been reported to vary from 61.1% to 91.3%.8; 11 ; 12 Widni et al8 demonstrated that ultrasonography had a positive predictive value of 83%, but a sensitivity of only 79%. Diagnostic laparoscopy is useful as a diagnostic modality in the event of equivocal sonographic findings and also enables definitive management of urachal remnants. This was the case where the four patients who had negative sonographic findings were confirmed to have urachal remnants and proceeded to undergo laparoscopic resection.
Laparoscopic excision of urachal remnants was first described by Trondsen in 1993,4 and since then there have been seven small case series, which are summarized in Table 3. Wide local resection of urachal remnants until the dome of the bladder along with adjacent inflammatory tissue is recommended. Inadequate resection risks recurrence of periumbilical discharge20 as well as possible malignant transformation of the urachal remnant.1
| Publication year and author | 2000, Cadeddu et al13 | 2005, Cutting et al14 | 2006, Okegawa et al15 | 2011, Li Destri et al16 | 2012, Araki et al17 | 2013, Patrzyk et al18 | 2013, Jeong et al19 | Present series |
|---|---|---|---|---|---|---|---|---|
| Country | USA | UK | Japan | Italy | Japan | Germany | Korea | Malaysia |
| No. of patients | 4 | 5 | 6 | 5 | 8 | 21 | 8 | 14 |
| Mean age (y) | 43.3 | 19 | 23.8 | 25.2 | 26.1 | 28.5 | 36.5 | 22.8 |
| Male/female | 3/1 | 5/0 | 6/0 | 1/4 | 4/4 | 8/13 | 7/1 | 8/6 |
| Type (n) | ||||||||
| Urachal cyst | 4 | 2 | 0 | 2 | 8 | 0 | 6 | 3 |
| Urachal sinus | 0 | 2 | 6 | 3 | 0 | 21 | 1 | 11 |
| Patent urachus | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 |
| Urachal adenocarcinoma | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| Mean operative time (min) | 180 | NR | 120.6 | 101 | 147.5 | 55.7 | 162 | 71.1 |
| Mean hospital stay (d) | 2.75 | ≤3 | 5.3 | 1.4 | NR | ≤3 | 14.6 | 1.3 |
| Mean duration of Foley catheterization (d) | 7 | NR | NR | NR | 1.9 | NR | 7.2 | 6.2 |
| Mean time to return to normal activities (d) | 11 | NR | 12.0 | 12.0 | 16 | NR | NR | 4.5 |
| Umbilical excision (Yes/No) | No | No | No | Yes | Yes | No | No | No |
| Bladder cuff resection (Yes/No) | Selective | Yes | NR | No | Selective | No | Selective | No |
| Complications | None | Umbilical hematoma (1) | None | None | None | None | None | Bladder injury (1) |
NR = not reported.
Various port placement techniques have been described. Most studies have adopted a three-trocar technique: one camera and two working ports.6 ; 18 The most common port positions are either epigastric or supraumbilical for the camera port, with the right and left midabdominal wall positions of the working ports forming the triangulation.13 ; 18 Another placement technique is based on the position of the three ports in the right or left lateral abdominal wall.15 ; 17 We adopted the latter technique, which affords excellent ergonomy during dissection of the median umbilical ligament from the umbilicus to the dome of the bladder and allows intracorporeal suturing if necessary. Care should be taken to position the trocars away from the umbilicus and potentially infected inflamed tissue lest there be port site infection. Identification of the urachus is the key to obtaining a good dissection during the procedure. Our approach is to start the dissection first at midway between the umbilicus and the bladder in the midline. This technique allows a good identification of urachus and a stepwise dissection either cranially toward the umbilicus or caudally toward the bladder. In addition, we favored the use of a laparoscopic hook over other instruments as it allows a steep angle dissection along the posterior abdominal wall.
The extent of resection remains a point of contention. En bloc resection of the umbilicus followed by umbilicoplasty has been described as the best method for complete resolution of symptoms and superior cosmesis in younger patients. 17 ; 21 We did not perform umbilectomy and did not encounter any recurrence except for one patient who had persistent discharge postoperatively from an inadequate resection of adjacent inflamed tissue. Diagnostic laparoscopy demonstrated the residual inflammatory tissue, and this was resected laparoscopically. The periumbilical discharge stopped immediately and the patient healed uneventfully. Thus, we feel that meticulous dissection of the urachal remnant along with its inflammatory tissue is more important than performing an umbilectomy as traditionally performed in open surgery. Furthermore, should there be any persistent symptoms, the laparoscopic procedure is easily repeatable with no increased morbidity and no poorer cosmesis.
With regard to the necessity for routine resection of the urachal remnant en bloc with a cuff of the bladder, there is also no consensus. Some argue that excision of a cuff of the bladder is necessary to avoid recurrence and avoid possible occurrence of carcinoma in residual urachal tisssue. 14 ; 22 However, most agree that excision of a bladder cuff is appropriate if there is an adherent urachal cyst attached to the dome of the bladder with communication between the cyst and bladder, especially if this is demonstrated by imaging. 13; 17 ; 19 Bladder cuff excision is also performed if there is suspicion of urachal carcinoma in patients who present with a midline abdominal mass with stippled calcification radiographically.23 We do not routinely resect a cuff of the bladder but instead transect the urachus where it inserts into the dome of the bladder, which is easily identifiable when the bladder is distended intraoperatively. We only use an absorbable catgut laparoscopic loop with a pusher to ligate the bladder end of the urachus for fear of stone formation should any nonabsorbable suture or clip(s) erode into the bladder and present itself as a focus for stone formation.
We advocate the routine catheterization of the bladder preoperatively to enable intraoperative decompression of the bladder to facilitate dissection. Once dissection is complete, it enables distension of the bladder to help identify where the urachus inserts into the dome of the bladder and also ensure that there is no communicating distensible urachal cyst. Postoperatively, the Foleys catheter is left in situ for 3–10 days and an ascending cystourethrogram is performed prior to its removal to ensure there is no bladder leak. We initially advocated catheterization for 10 days as routine, but later we tended to remove the catheters by Day 3 after the surgery as many patients were eager to return to normal activities and were hindered by their urinary catheters.
The timing of surgery is important. Treatment of infected urachal anomalies in an acute setting with traditional open excision should be avoided, as open incisions are associated with a significant risk of surgical site infection and prolonged convalescence. The appropriate treatment should be appropriate antibiotics, adequate analgesia and incision, and drainage of the abscesses if seen via ultrasonography followed by resection of the urachal remnant later.12 ; 13 We similarly do not recommend immediate resection of urachal remnants at presentation and prefer ultrasonography, drainage of any abscesses detected via ultrasonography using a small infraumbilical incision with adequate analgesia and antibiotic therapy. Surgical resection should be performed after the inflammation has subsided, which is heralded by resolving overlying cellulitis and reduced discharge. Attempts at performing resection in the presence of active inflammation is made difficult by the amount of inflammatory tissue that needs to be removed and the difficulty in identifying where the urachus inserts into the bladder despite intraoperative distension. We encountered one such event where surgical resection was performed before the inflammation had completely resolved, leading to the accidental opening of the dome of the bladder, which was immediately identified and repaired via a small Pfannenstiel incision after the urachal remnant and inflammatory tissue had been completely removed laparoscopically. The patient recovered uneventfully and the Foleys catheter was removed uneventfully postoperatively on Day 7 without any leak seen radiographically.
5. Conclusion
Urachal remnants should be managed in a systematic manner. Ultrasonography should be performed to confirm the diagnosis and identify abscesses. If a urachal abscess is found, drainage of the abscess should be performed via a small infraumbilical incision with appropriate antibiotic therapy and adequate analgesia. Excision should be performed electively once the infection and inflammation has abated and it should be performed laparoscopically. Laparoscopy is also useful to confirm the diagnosis in instances where ultrasonography fails to identify the presence of urachal remnants but the recurrent umbilical discharge is highly suggestive. The advantages of laparoscopic resection include superior magnified dissection of the preperitoneal plane, superior cosmesis compared to a lower midline laparotomy scar, low morbidity, less postoperative pain, and earlier return to normal activities. En bloc umbilical resection and routine resection of a bladder cuff are unnecessary.
Acknowledgments
The authors thank the Director General of Health, Malaysia, for permission to publish this paper. We also thank Prof. Clarence Lei for his invaluable advice on surgical techniques.
References
- 1 M. Blichert-Toft, O.V. Nielsen; Congenital patent urachus and acquired variants. Diagnosis and treatment. Review of the literature and report of five cases; Acta Chir Scand, 137 (1971), pp. 807–814
- 2 T. Ueno, H. Hashimoto, H. Yokoyama, M. Ito, K. Kouda, H. Kanamaru; Urachal anomalies: ultrasonography and management; J Pediatr Surg, 38 (2003), pp. 1203–1207
- 3 V. Galati, B. Donovan, F. Ramji, J. Campbell, B.P. Kropp, D. Frimberger; Management of urachal remnants in early childhood; J Urol, 180 (suppl 4) (2008), pp. 1824–1827
- 4 E. Trondsen, O. Reiertsen, A.R. Rosseland; Laparoscopic excision of urachal sinus; Eur J Surg, 159 (1993), pp. 127–128
- 5 B. Zieger, B. Sokol, W.K. Rohrschneider, K. Darge, J. Tröger; Sonomorphology and involution of the normal urachus in asymptomatic newborns; Pediatr Radiol, 28 (1998), pp. 156–161
- 6 A.M. Lipskar, R.D. Glick, N.G. Rosen, et al.; Nonoperative management of symptomatic urachal anomalies; J Pediatr Surg, 45 (2010), pp. 1016–1019
- 7 J.H. Yiee, N. Garcia, L.A. Baker, R. Barber, W.T. Snodgrass, D.T. Wilcox; A diagnostic algorithm for urachal anomalies; J Pediatr Urol, 3 (2007), pp. 500–504
- 8 E.E. Widni, M.E. Hollwarth, E.Q. Haxhija; The impact of preoperative ultrasound on correct diagnosis of urachal remnants in children; J Pediatr Surg, 45 (2010), pp. 1433–1437
- 9 B.R. Yapo, B. Gerges, A.J.A. Holland; Investigation and management of suspected urachal anomalies in children; Pediatr Surg Int, 24 (2008), pp. 589–592
- 10 J.S. Yu, K.W. Kim, H.J. Lee, Y.J. Lee, C.S. Yoon, M.J. Kim; Urachal remnant diseases: spectrum of CT and US findings; Radiographics, 21 (2001), pp. 451–461
- 11 Y.J. Choi, J.M. Kim, S.Y. Ahn, J.T. Oh, S.W. Han, J.S. Lee; Urachal anomalies in children: a single center experience; Yonsei Med J, 47 (2006), pp. 782–786
- 12 M.O. McCollum, A.E. MacNeily, G.K. Blair; Surgical implications of urachal remnants: presentation and management; J Pediatr Surg, 38 (2003), pp. 798–803
- 13 J.A. Cadeddu, K.E. Boyle, M.D. Fabrizio, P.G. Schulam, L.R. Kavoussi; Laparoscopic management of urachal cysts in adulthood; J Urol, 164 (2000), pp. 1526–1528
- 14 C.W. Cutting, R.G. Hindley, J. Poulsen; Laparoscopic management of complicated urachal remnants; BJU Int, 96 (2005), pp. 1417–1421
- 15 T. Okegawa, A. Odagane, K. Nutahara, E. Higashihara; Laparoscopic management of urachal remnants in adulthood; Int J Urol, 13 (2006), pp. 1466–1469
- 16 G. Li Destri, D. Scillaci, R. Latino, M. Castaing, B. Scilletta, A.D. Cataldo; The urachal pathology with umbilical manifestation: overview of laparoscopic technique; J Laparoendosc Adv Surg Tech A, 21 (2011), pp. 809–814
- 17 M. Araki, T. Saika, D. Araki, et al.; Laparoscopic management of complicated urachal remnants in adults; World J Urol, 30 (2012), pp. 647–650
- 18 M. Patrzyk, L. Wilhelm, K. Ludwig, C.D. Heidecke, W. von Bernstorff; Improved laparoscopic treatment of symptomatic urachal anomalies; World J Urol, 31 (2013), pp. 1475–1481
- 19 H.J. Jeong, D.Y. Han, W.A. Kwon; Laparoscopic management of complicated urachal remnants; Chonnam Med J, 49 (2013), pp. 43–47
- 20 I.L. Goldman, A.A. Caldamone, M. Gauderer, N. Hampel, C.W. Wesselhoeft, J.S. Elder; Infected urachal cyst: a review of 10 cases; J Urol, 140 (1998), pp. 375–378
- 21 R. Uyama, Y. Yoshizawa, S. Sasaya, H. Nemoto, T. Ikeda, Y. Sanada; Laparoscopic resection of urachal remnant with plasty of umbilicus: report of five cases; J Jpn Surg Assoc, 65 (2004), pp. 1371–1375
- 22 B.G. Cilento Jr., S.B. Bauer, A.B. Retik, C.A. Peters, A. Atala; Urachal anomalies: defining the best diagnostic modality; Urology, 52 (1998), pp. 120–122
- 23 C.A. Sheldon, R.V. Clayman, R. Gonzalez, R.D. Williams, E.E. Fraley; Malignant urachal lesions; J Urol, 131 (1984), pp. 1–8
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?