Abstract
As creep of polymeric materials is potentially a safety concern for photovoltaic modules, the potential for module creep has become a significant topic of discussion in the development of IEC 61730 and IEC 61215. To investigate the possibility of creep, modules were constructed, using several thermoplastic encapsulant materials, into thin-film mock modules and deployed in Mesa, Arizona. The materials examined included poly(ethylene)-co-vinyl acetate (EVA, including formulations both cross-linked and with no curing agent), polyethylene/polyoctene copolymer (PO), poly(dimethylsiloxane) (PDMS), polyvinyl butyral (PVB), and thermoplastic polyurethane (TPU). The absence of creep in this experiment is attributable to several factors of which the most notable one was the unexpected cross-linking of an EVA formulation without a cross-linking agent. It was also found that some materials experienced both chain scission and cross-linking reactions, sometimes with a significant dependence on location within a module. The TPU and EVA samples were found to degrade with cross-linking reactions dominating over chain scission. In contrast, the PO materials degraded with chain scission dominating over cross-linking reactions. Although we found no significant indications that viscous creep is likely to occur in fielded modules capable of passing the qualification tests, we note that one should consider how a polymer degrades, chain scission or cross-linking, in assessing the suitability of a thermoplastic polymer in terrestrial photovoltaic applications.
Introduction
In the manufacturing of the photovoltaic modules there is a desire to use new thermoplastic encapsulant materials. This is motivated by the desire to reduce lamination time or temperature, reduce moisture permeation with new materials [1], use less corrosive materials [2, 3], improve electrical resistance [4, 5], or facilitate the reworking of a module after lamination. However, the use of any thermoplastic material in a high-temperature outdoor environment, where creep may occur, could raise safety and performance concerns. Therefore, there has been an increased concern in the photovoltaic (PV) community regarding the possibility of viscoelastic creep and has brought about concerns for the testing of modules according to IEC 61730 and IEC 61215 [6-8]. Small areas of a module may reach much higher temperatures (>150°C) during the “hot-spot” test or during partial shading of a module without bypass-diode protection [9, 10]; but the localized nature of this occurrence is different from the situation of prolonged operation in the hottest environments and mounting configurations. In very hot environments, modules are known to reach temperatures in excess of 100°C [11, 12]. One could envision an encapsulant with a melting point near 85°C with a highly thermally activated drop in viscosity, resulting in significant creep at 100°C. Creep is distinguished here from delamination, which may also occur when the degree of cure is less than intended, where a primer may not activate as intended [13].
Some early work with poly(ethylene)-co-vinyl acetate EVA encapsulation performed at the Jet Propulsion Laboratory (JPL) did consider the issue of creep during operation at high temperature [6, 14]. PV technology developers at that time were concerned that there was the possibility of displacement of the components within a heated module operating in the field, but did not formally investigate to verify creep using a variety of modules deployed in a hot location. To specifically prevent creep, EVA, that was cross-linked via a peroxide-initiated reaction, was advocated at that time. The use of 65% gel was found to facilitate passing the sales qualification tests (which included the “melt/freeze” test at that time) and was therefore suggested by JPL [6]. The use of EVA with at least 65% gel content was reaffirmed by Springborn Laboratories (later known as Specialized Technology Resources, Inc., or STR) [15], and currently, the use of EVA with 60–90% gel content after lamination is commonly used in the industry. However, if one wants to switch to a thermoplastic material, gel content is not a useful metric to evaluate creep. Therefore, one must find other ways to verify proper processing of a new encapsulant material.
Examination of EVA in fielded modules reveals gel contents above 90% [16, 17]. Often when EVA degrades, it leaves a clear border around the perimeter of the cell and a yellow/brown inner area. It has been observed that in the clear portion, the amount of oxygen incorporation and UV absorber (Cyasorb 531) was higher, but the gel content was slightly lower. Here, oxygen ingress is slow enough such that it is consumed on the cell perimeter and the oxygen serves to increase the rate of chain scission.
At elevated temperatures, EVA degrades by thermal deacetylation with the formation of acetic acid [16, 17]. However, at ambient or use conditions, this reaction pathway is unimportant and long-term fielded EVA samples have been shown to have <1% change in the amount of residual acetate groups [2, 18]. However, even this small amount of acid formation can lower the pH sufficiently to increase degradation rates of PV cells and interconnections.
Prior work studied the possible hazards associated with creep in modules, including how to screen for creep and how to test for the results of creep [19, 20]. Modules were fabricated with a variety of encapsulant materials, including polyethylene vinyl acetate (EVA, both cross-linked and with no curing agent), polyethylene/polyoctene copolymer (PO), poly(dimethylsiloxane) (PDMS), polyvinyl butyral (PVB), and thermoplastic polyurethane (TPU). These modules were subjected to high temperatures using outdoor aging (in Mesa, AZ or Golden, CO) or indoor aging (in a temperature step-stress test performed in a chamber), and the resulting creep was documented. The greatest creep was observed for EVA with no curing system present (3 mm in Mesa, AZ and 0.3 mm in Golden, CO), while the next greatest displacement was on the order of 30 μm. Curiously, the rate of creep for the fielded EVA specimens was seen to decrease, even though the enabling ambient temperature remained warm through the summer months of the exposure period. The observed creep was correlated with material-level tests to identify the best way to characterize phase transitions that could be predictive of creep in the field. The creep measured outdoors and indoors was found to correlate with each other, where the nonuniform temperature distribution within a module as well as the packaging components (frame or mounting clips) was found to greatly limit creep. Creep was also particularly correlated with the melting point, the phase transitions and rheological properties measured for the encapsulant materials. These data suggested that some noncuring thermoplastics were cross-linking, but did not present conclusive evidence to that effect.
The effort described here adds to the aforementioned study [19-21]. We demonstrate that, in some cases, most notably noncuring EVA (NC-EVA), a significant factor in the reduction of encapsulant creep is the formation of cross-links in the absence of cross-linking agents. The ability to cross-link polymers in the absence of specific chemistry to do so very significantly reduces the likelihood of damage resulting from creep in a field-deployed module. We explore the importance of proximity to the edge of the module to assess the role of diffusion of reactants and/or reaction products in the dominance of chain scission over cross-linking reactions.
Experimental Procedure
The specifics of the materials and construction are more thoroughly covered in M. D. Kempe et al. [21], but we will present a brief description of the sample preparation here.
Module construction
Encapsulant materials being used, or under investigation for use, in PV modules were obtained from industrial manufacturers. The nomenclature for as well as the phase characteristics of these materials are summarized in Table 1. A noncuring poly(ethylene-vinyl acetate) (NC-EVA) was formulated identically to a standard EVA formulation but without the inclusion of a peroxide to promote curing during lamination.
| Encapsulant material type and designation | Phase transitions | ||||||
|---|---|---|---|---|---|---|---|
| DSC | DMA at 0.1 rad/sec | ||||||
| Tg (°C) | Tm (°C) | Tf (°C) | Tg (°C) | Tm (°C) | Tc (°C) | ||
| |||||||
| Cured commercial PV EVA resin | EVA | −31 | 55 | 45 | −30 | 47 | |
| Commercial PV EVA resin with all components but the peroxide | NC-EVA | −31 | 65 | 45 | −28 | 69 | |
| Polyvinyl butyral | PVB | 15 | 17 | 121 | |||
| Aliphatic thermoplastic polyurethane | TPU | 2 | 3 | 84 | |||
| Pt catalyzed, addition cure polydimethyl siloxane gel (mock modules) | PDMS-M | −158 | −40 | −80 | |||
| Pt catalyzed, addition cure polydimethyl siloxane gel (Si modules) | PDMS-Si | −150 | −40 | −80 | |||
| Thermoplastic polyolefin #1 | TPO-1 | −43 | 93 | 81 | −35 | 105 | |
| Thermoplastic polyolefin #3 | TPO-3 | −44 | 61 | 55 | −41 | 79 | |
| Thermoplastic polyolefin #4 | TPO-4 | −34 | 106 | 99 | −21 | 115 | |
In previous experiments [19, 21], both crystalline silicon and thin-film mock samples were tested. Because we wanted to redeploy the silicon modules, only the thin-film mock modules were destructively evaluated in this work. The thin-film mock modules were constructed using two pieces of 3.18-mm thick, 61 × 122 cm glass obtained from a thin-film PV manufacturer. The rear surface of the back plate was painted black to simulate the optical absorption of a thin-film module. Thin-film mock modules were mounted by adhesively attaching fiberglass channel on the back, allowing the front piece of glass to move freely.
For the thin-film mock modules, the creep (displacement of the front glass relative to the back glass) was measured using a high-precision electronic depth gauge that was incremented ±1 μm. This gauge was mounted to a flat plate to ensure that it was positioned perpendicular to the side of the module, and in the plane of one of the glass plates. With this setup, the repeatability of creep measurement was better than ±20 μm.
Modules were deployed in Mesa, Arizona at the Arizona State University from May until September 2011 on a rack tilted at 33°latitude tilt and a 255 azimuth to more directly face the sun at the hottest part of the day. Additionally, a single NC-EVA thin-film mock module was exposed in Golden, Colorado at a 180 azimuth and 40°latitude tilt [8]. Insulation was placed on the backside to simulate a close-roof installation (resulting in maximum measured temperatures between 102°C and 104°C in Mesa, Arizona, AZ or Golden, CO)).
The modules were also examined indoors in environmental chambers in a step-stress experiment using a replicate set of modules. The temperatures applied during the test ranged from 65 to 110°C, with a 5°C increment, then from 110 to 140°C in a 10°C increment using a 200-h exposure time at each step.
Size exclusion chromatography and multiangle laser light scattering
Following field deployment, the formation of polymer chain cross-links on the mock modules was evaluated using size exclusion chromatography (SEC) in conjunction with multiangle laser light-scattering (MALLS, Waters Corporation GPCV 2000 instrument 34 Maple StreetMilford, MA 01757 USA) and viscometric detection (using a capillary viscometer detector CV from Waters). For the tetrahydrofuran (THF) solvent, four SEC styrene-divinyl benzene 8 × 300 mm columns from Shodex (4-1, Shiba Kohen 2-chome, Minato-ku, Tokyo, 105-8432, Japan) were used for separation. These were two linear GPC KF806M, one GPC KF802, and one GPC KF-801 columns. Columns were run at 40°C, with a flow rate of 1.0 mL/min, an injection volume of 0.219 mL, and a run time of 90 min.
Samples were cut out of the mock modules using a ceramic saw blade (Wale Apparatus Co., Inc.400 Front StreetHellertown, PA 18055) enabling samples to be taken at different distances from the edge. For the TPU and EVA, polymer was removed from the glass by extraction for 72 h at 40°C, utilizing mild agitation, in a solution of THF; however, this typically left behind some EVA as an insoluble fraction. For EVA, the residual glass-polymer specimens were then additionally soaked in trichlorobenzene (TCB) overnight at 140°C, with slight agitation, to solubilize some of the remaining polymer. The TCB soluble measurements of EVA represent material not soluble in THF, but soluble in TCB. For the thermoplastic polyolefins (TPO), the SEC measurements were done using only TCB solvent.
The TCB-based system utilized two Styragel HT 6E and one Styragel HT 2 styrene-divinyl benzene columns from Waters for SEC separation. The TCB columns used a temperature of 140°C, a flow rate of 1.01 mL/min, an injection volume of 0.2095 mL, and a run time of 80 min.
Samples of “virgin” (unaged) polymer film samples were dissolved in THF and TCB, as appropriate, at room temperature and 150°C, respectively, to serve as controls. All solutions were made with approximately 1 mg/mL polymer concentration.
Gel content measurement
Gel content measurement was accomplished using a Soxhlet extractor with toluene, THF, mixed isomeric xylenes, or methanol as the solvent. Test material was obtained in approximately 1 g samples by cutting out small samples using a ceramic wet saw, and subsequently separating the polymer from the glass manually using a blade. Polymer samples were taken at various distances from the edge of the glass.
Results and Discussion
NC-EVA
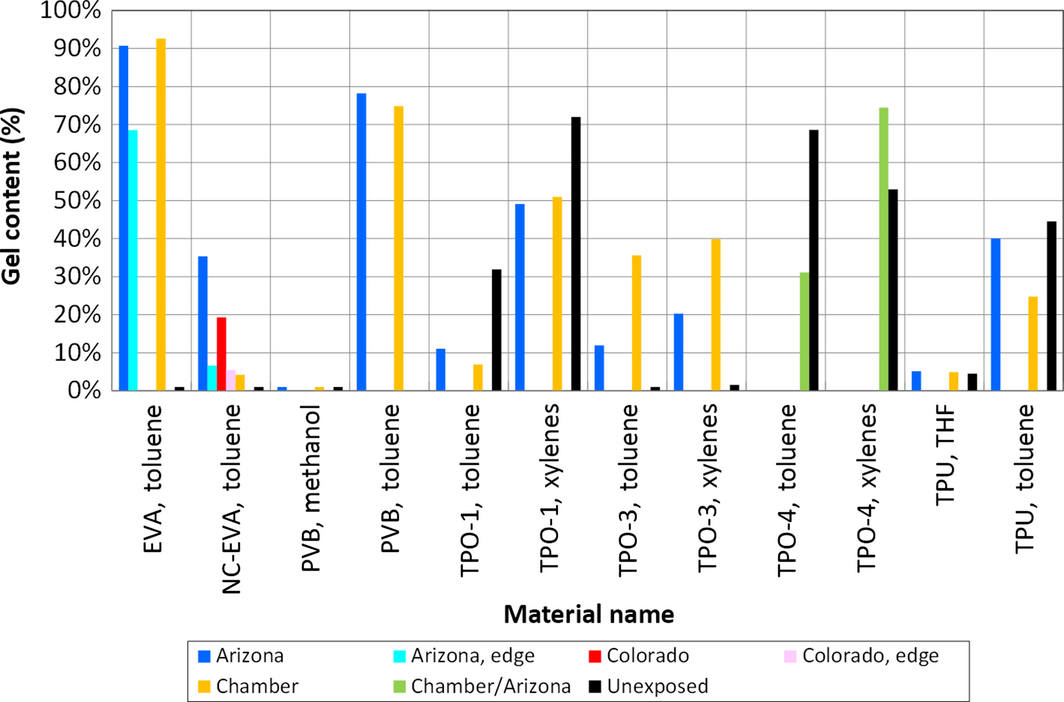
Three NC-EVA samples were removed from the mock modules deployed in Golden, Colorado and Mesa, Arizona. One cut from the edge of the module, one about 2 cm from the edge, and another about 4 cm. The unexposed sheets were completely soluble in both THF and TCB. For the exposed samples, a qualitative assessment during the preparation for SEC experimentation indicated that only ~40% of the NC-EVA was soluble in THF. Then, of this THF-insoluble fraction another ~40% (relative to initial amount) was soluble in TCB, leaving ~20%, which was not soluble in either THF or TCB. Qualitatively, this is in agreement with rigorous gel content measurements using toluene in a Soxhlet extractor (Fig. 1), where 5.5–35.4% gel was measured at various locations. This NC-EVA was formulated with EVA pellets containing trace amounts of butylated hydroxyl toluene (BHT) to which was added an hindered amine light stabilizer (HALS), methacryloxypropyl trimethoxysilane (Z6030), a phenylphosphonite, a benzophenone-based UV absorber, but no peroxide [5, 18].
|
|
|
Figure 1. Results of gel content measurements using a Soxhlet extractor. “Edge” samples were taken <1 cm from the side of the module, all other samples were taken about 4 cm from the side. |
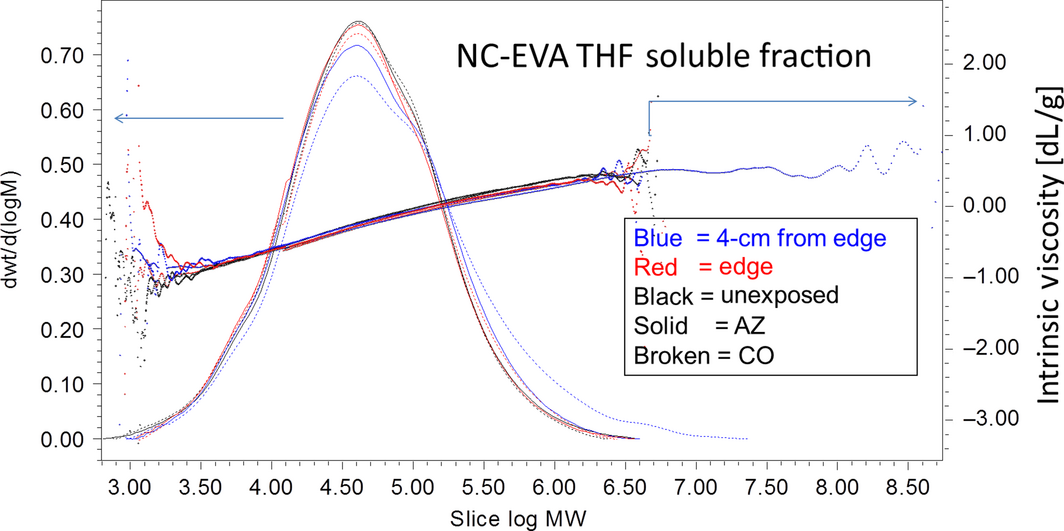
The cross-linking of the NC-EVA in the fielded module was further investigated using SEC in conjunction with MALLS and viscometry detection (Fig. 2). For the THF soluble fraction, very little change in the molecular weight distribution was seen for either location within the specimens. The sample taken 4 cm from the edge for the Colorado module has a high-molecular-weight fraction, but otherwise, the THF soluble fractions are similar to the unexposed material. The EVA specimens contain 33 wt% vinyl acetate. Qualitative experiments indicate that above about 30 wt% the solubility of EVA in THF is good, but below 30 wt% the solubility in THF is poor [22]. This indicates that even very slight modification/degradation of EVA molecules is sufficient to render them significantly less soluble in THF consistent with our data.
|
|
|
Figure 2. Weight fraction determined by SEC-MALLS (SEC, size exclusion chromatography) and viscometry for the NC-EVA (poly[ethylene]-co-vinyl acetate) thin-film mock module after exposure in Golden, Colorado (dotted lines) and Mesa, Arizona (solid lines). Samples dissolved in tetrahydrofuran (THF) by extraction at 40°C overnight. Average molecular weight (MW) of each slice of polymer distribution is in units of g/mol as determined by SEC-MALLS. Right axis, intrinsic viscosity (dL/g) of each slice. Lines are normalized to the area under the curve. |
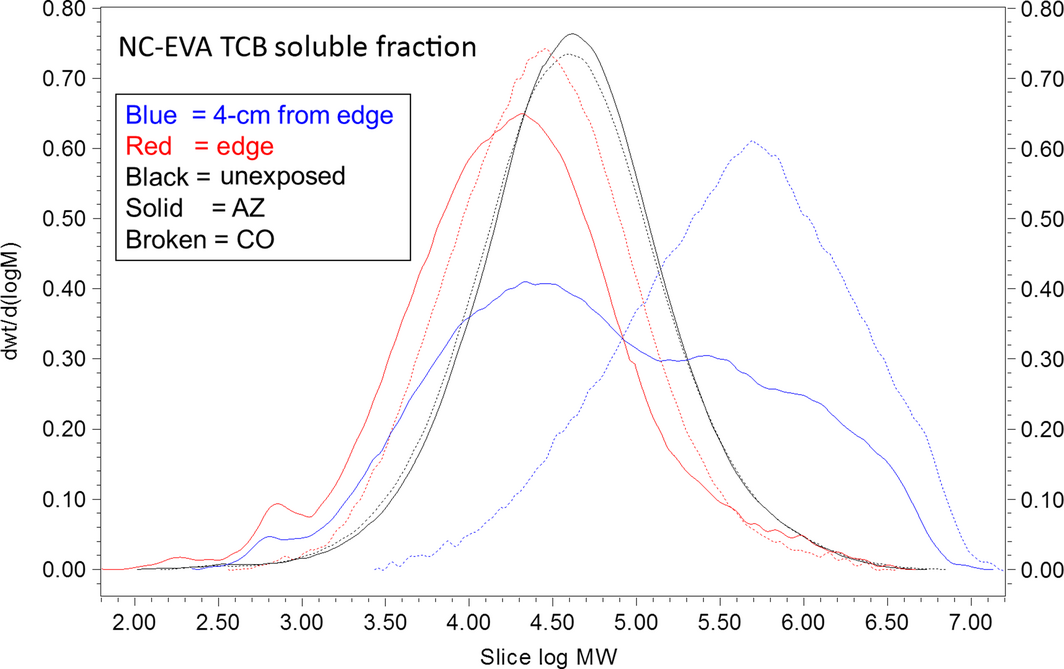
In contrast, the SEC profiles of the TCB-solubilized samples varied greatly depending on the exposure site, and on the distance from the module edge (Fig. 3). Samples taken at distances of 2 and 4 cm from the edge have a higher molecular weight fraction than the control indicating that significant cross-linking is occurring (Fig. 4).
|
|
|
Figure 3. Weight fraction determined by SEC-MALLS for the NC-EVA poly(ethylene-co-vinyl acetate) thin-film mock module after exposure in Golden, Colorado (dotted lines) and Mesa, Arizona (solid lines). Samples obtained from tetrahydrofuran (THF) insoluble fraction by extraction in TCB at 150°C overnight. Molecular weight (MW) is in units of g/mol determined by (SEC, size exclusion chromatography) SEC-MALLS. Lines are normalized to the area under the curve. |
|
|
|
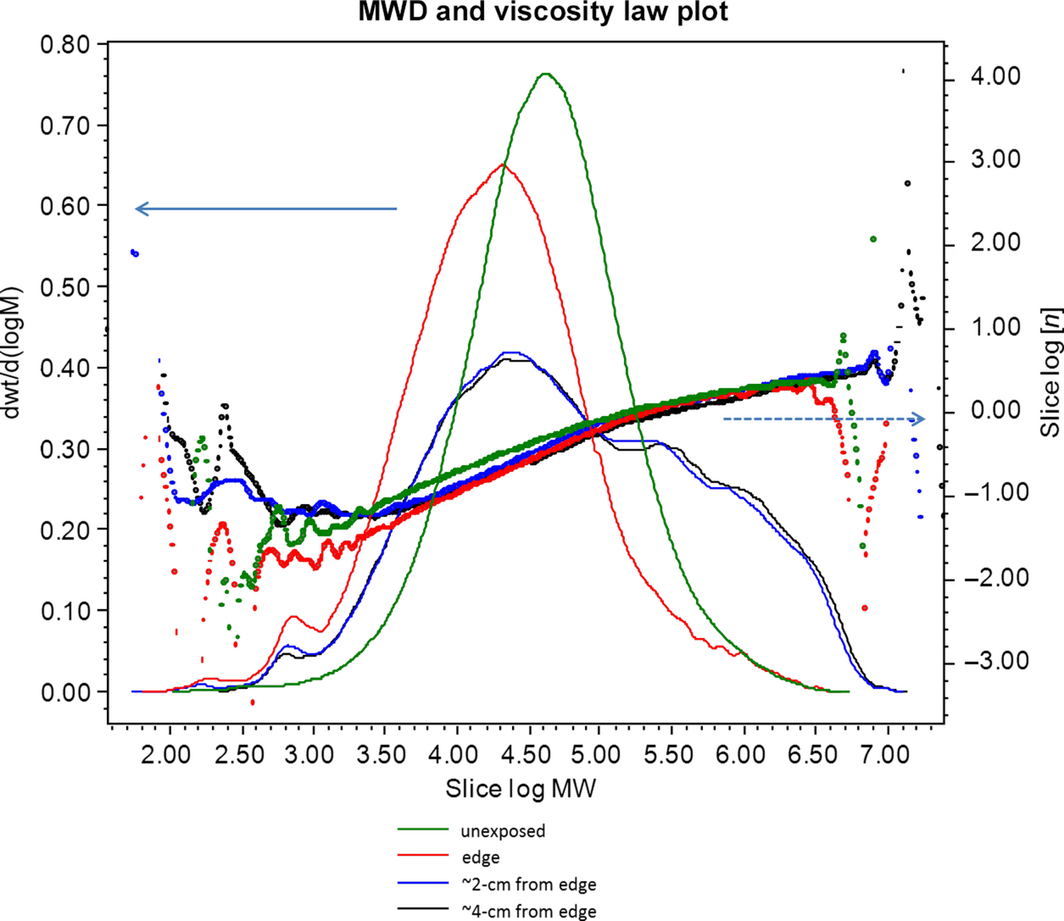
Figure 4. Weight fraction determined by (SEC, size exclusion chromatography) SEC-MALLS and viscometry for the NC-EVA thin-film mock module after exposure in Arizona. Samples dissolved in TCB by extraction at 150°C overnight. Molecular weight (MW) is in units of g/mol as determined by SEC-MALLS. Right axis, intrinsic viscosity (dL/g) of each slice. Lines are normalized to the area under the curve. |
The molecular weight distributions for Colorado and Arizona are very different. Of note in Table 2 are the apparently lower values for Mn in TCB versus THF. It is believed that this is an experimental artifact resulting from the silane component being isorefractive in THF but not in TCB. Because of the significantly lower temperature experienced in Colorado [19, 21], one would expect to see less degradation, that is cross-linking, than for the Arizona sample. Also of significance is the low-molecular-weight fractions, which if unmodified should have been previously extracted in THF. The absence of low-molecular-weight fractions at a distance of 4 cm from the edge in the Colorado sample indicates that the process of dissolution in TCB at 140°C is not significant in forming these fractions. Noting that low-molecular-weight fractions are more prevalent for the edge samples suggest that O2 may play an important role in the formation of low molecular weight THF-insoluble fractions.
| Sample | Mn (g/mol) | Mw (g/mol) | Mz (g/mol) | PDI |
|---|---|---|---|---|
| ||||
| Tetrahydrofuran (THF) | 20,000 | 98,000 | 450,000 | 4.8 |
| Unexposed | 20,000 | 98,000 | 450,000 | 4.8 |
| Trichlorobenzene (TCB) | 15,000 | 100,000 | 620,000 | 6.5 |
| Unexposed | 16,000 | 98,000 | 510,000 | 6.3 |
| Mesa, Arizona | ||||
| TCB soluble fraction | ||||
| Edge | 20,000 | 89,000 | 380,000 | 4.5 |
| ~2 cm from the edge | 20,000 | 85,000 | 330,000 | 4.2 |
| ~4 cm from the edge | 21,000 | 100,000 | 440,000 | 4.9 |
| THF soluble fraction | ||||
| Edge | 5,500 | 67,000 | 630,000 | 12.1 |
| ~2 cm from the edge | 8,700 | 390,000 | 2,200,000 | 45.2 |
| ~4 cm from the edge | 11,000 | 380,000 | 2,100,000 | 35.9 |
| Golden, Colorado | ||||
| TCB soluble fraction | ||||
| Edge | 20,000 | 88,000 | 340,000 | 4.4 |
| ~4 cm from the edge | 21,000 | 190,000 | 2,100,000 | 8.9 |
| THF soluble fraction | ||||
| Edge | 13,000 | 66,000 | 390,000 | 5.2 |
| ~4 cm from the edge | 100,000 | 980,000 | 3,100,000 | 8.7 |
Gel content measurements of both the Colorado and the Arizona test modules (Fig. 1) demonstrate a significantly lower gel content near the edge and a higher gel content away from the edge for NC-EVA. The same trend was also seen in the EVA with peroxide where the gel content of the edge sample was 68.5%, and 4 cm from the edge it was 90.7%, consistent with other observations [16, 17]. This is presumably because of an interaction with O2 at the periphery.
Numerous observations of fielded modules have observed discoloration of EVA in the center area of a cell; however, greater oxygen incorporation is observed around the cell perimeter [18]. Similarly, it has been observed that the gel content in the more discolored EVA at the center of the cell is higher than EVA at the perimeter in aged samples [16]. Thus, yellowing and cross-linking are correlated with lower oxygen incorporation, and lower cross-link densities are correlated with higher oxygen incorporation and oxidative bleaching. We hypothesize that oxygen can act to increase the rate of chain scission, but that its absence allows cross-linking chemistries to dominate more. Of particular note, is the observation that samples at a distance of 2 and 4 cm from the edge have nearly identical molecular weight profiles (Fig. 4). This implies that oxygen availability is the same at 2 and 4 cm, therefore the polymer environment is very likely anaerobic at distances >2 cm from the edge of the cells, consistent with the observed size of brown/yellow areas in some field deployed EVAs.
Marais and Hirata [23] measured the solubility of O2 in 33 wt% EVA at 25°C at 0.0023 cm3 (STC)/cm3/cm Hg (or 5.6 × 10−5 g/cm3) and a diffusivity (D) of about 4.0 × 10−7 cm2/sec. For the 140-day exposure, the characteristic distance for oxygen ingress () at a temperature of 25°C is 4.8 cm. For comparison, at 25°C the diffusivity of water in EVA is comparable at 4.8 × 10−7 cm2/sec, but its solubility at 100% RH is 100 times higher at 0.0021 g/cm3 [1, 24-26]. Considering that the perimeter of 2 cm is where all of this diffusant is consumed, a linear gradient at 25°C would result in 0.07 g/cm3 of water consumed in the perimeter of 2 cm over a 10-year period. Because EVA is not observed to be this highly degraded when fielded for long periods, water is not completely consumed so quickly upon diffusion into EVA. At the higher module temperatures in field deployment, oxygen would diffuse further than this estimate at 25°C. Thus, it is highly likely that oxygen ingress is enhancing chain scission in EVA, and is limited by its low solubility in EVA.
If the degree of branching is constant as a function of molecular weight, then the lines for intrinsic viscosity would be straight lines when plotted against Log(MW). The slight concave down curvature in Figures 2 and 4 indicates increased amounts of branching at higher molecular weights. For the THF soluble fractions, the difference in intrinsic viscosity between the aged samples is undetectable. But for the TCB soluble fraction there is a relative reduction in the intrinsic viscosity for low-molecular-weight fractions, indicating that they are becoming more compact and branched.
For the NC-EVA samples deployed in both Mesa, Arizona and Golden, Colorado, cross-linking was observed resulting from combined thermal/UV aging. Both SEC and gel content measurements indicate an increase in the frequency with which degradation results in chain scission for locations where, presumably, oxygen is able to diffuse. Very small amounts of degradation are able to significantly reduce the solubility of EVA in THF. Regardless of the initial degree of cure, or even the total absence of cure agents, typical PV EVA formulations are therefore expected to cure as they age.
Thermoplastic polyolefins
In this work three different thermoplastic polyolefins were evaluated. Thermoplastic polyolefin #1 (TPO-1) was designed to be a material that might be susceptible to creep, but still pass the qualification test because its melting transition was above 85°C. However, when fielded, the movement of the glass was barely detectable (~30 μm) [19, 20]. Thermoplastic polyolefin #4 (TPO-4) is similar to TPO-1, but with a higher melting point and viscosity, which resulted in no detectable movement. Finally, thermoplastic polyolefin #3 (TPO-3) was made by a different manufacturer and has been used in the construction of commercial modules. TPO-3 has a melting point between 61°C and 79°C (depending on the measurement method) which is just a bit higher than EVA. Because it had a much higher viscosity than NC-EVA [20], the 0.090 mm of creep was not sufficient to see the effects of chain scission/cross-linking in the creep response curve for the outdoor exposed modules. However, a significant increase in viscosity was seen in the indoor stressed samples at temperatures >90°C [21]. All three of these materials were analyzed in SEC to evaluate cross-linking and chain scission reactions.
All three TPO samples were completely soluble in TCB at 150°C and were analyzed in this solvent using the multidetector SEC system (Fig. 5). Similar to NC-EVA, for all three materials the sample taken from the edge showed the greatest reduction in molecular weight indicating that a similar O2-induced chain scission mechanism may be active at the periphery. By far, the greatest shift was for TPO-4, followed by TPO-1, then TPO-3. Similarly, the samples taken at a distance of 4 cm from the edge showed a small drop in molecular weight. This is in stark contrast to the NC-EVA samples which experienced significant cross-linking in the interior anaerobic regions. This difference in cross-linking behavior between EVA and these TPOs could be a result of the stabilization chemistry used, or more likely there could be a greater ability of EVA to form cross-links through a mechanism involving the acetate monomers as seen elsewhere [27]. Tertiary carbons in the backbone are prone to experiencing chain scission. Therefore, it is likely that the chain scission reactions dominate throughout the polyolefin copolymers. In combination, these effects can explain the difference in the cross-linking/chain scission behavior of EVA as compared to TPOs.
|
|
|
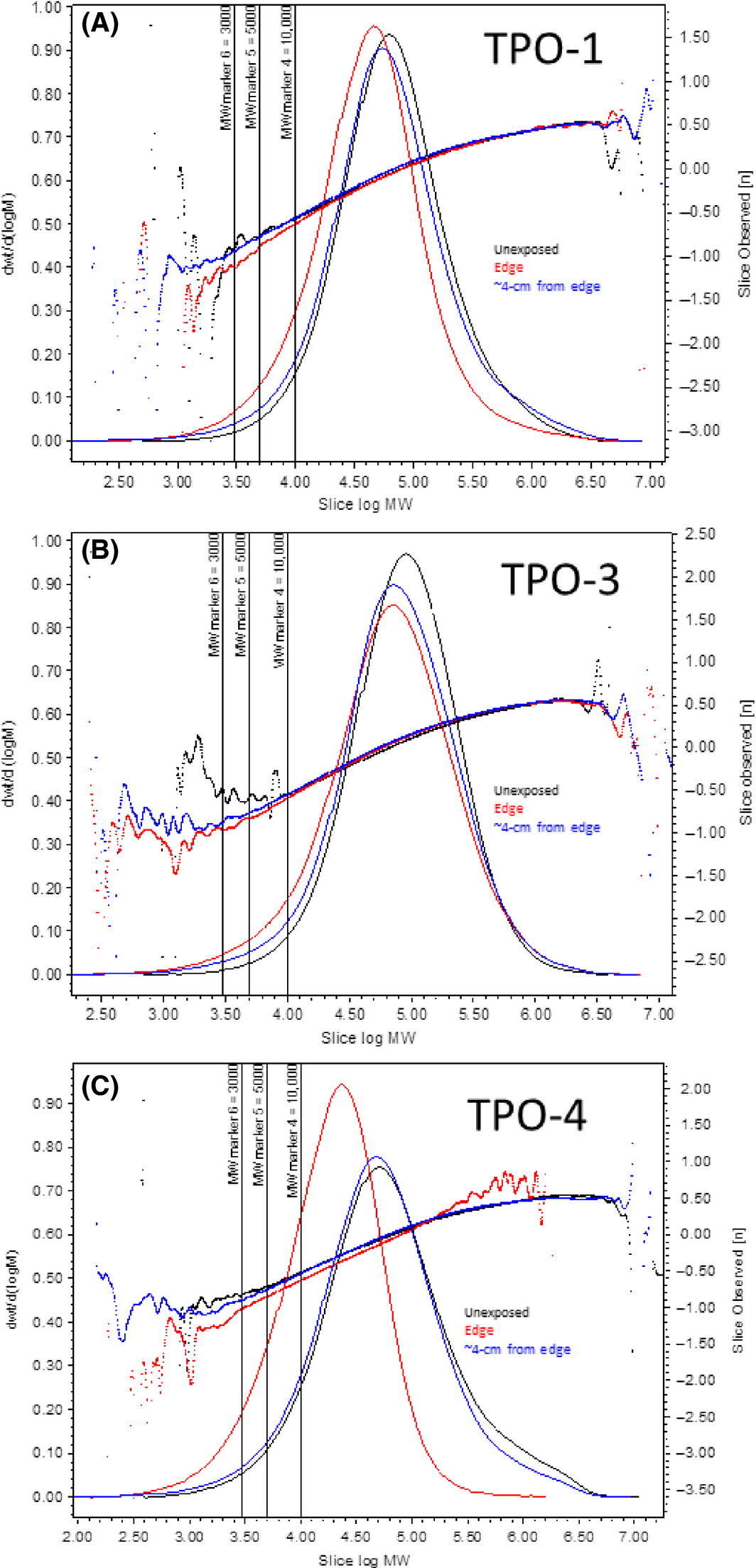
Figure 5. Weight fraction determined by SEC-MALLS (SEC, size exclusion chromatography) and viscometry for the TPO-1 thin-film mock module after exposure in Arizona. Samples dissolved in TCB by extraction at 150°C overnight. Molecular weight (MW) is in units of g/mol. Right axis, intrinsic viscosity (dL/g) in slice, horizontal lines. Blue: 4 cm from the edge; red: edge sample; black: unexposed. (A) TPO-1, (B) TPO-3, and (C) TPO-4. Lines are normalized to the area under the curve. |
Similar to EVA, the slight concave down curvature in Figure 5 indicates the presence of increased amounts of branching at higher molecular weights. For TPO-1 and TPO-4, and to a lesser extent TPO-3, the aged samples showed a relative reduction in intrinsic viscosity near the edge but not at 4 cm distance from the perimeter. Thus, O2 is degrading the polymers, making them more compact and branched.
The light-scattering measurements at an angle of 90° also provide insight into the cross-linking behavior of the materials (Fig. 6). Here, the strength of the signal is related to the product of concentration and molecular weight. Thus, when two elution peaks are seen in light-scattering detectors, the first peak consists of a small amount of very large molecular weight material, and the second peak is of a much lower molecular weight material where the signal strength is attributable to significantly larger quantities of polymer. Looking at the changes in the relative ratios of these peaks gives a sensitive indication of changes in the large molecular weight fraction.
|
|
|
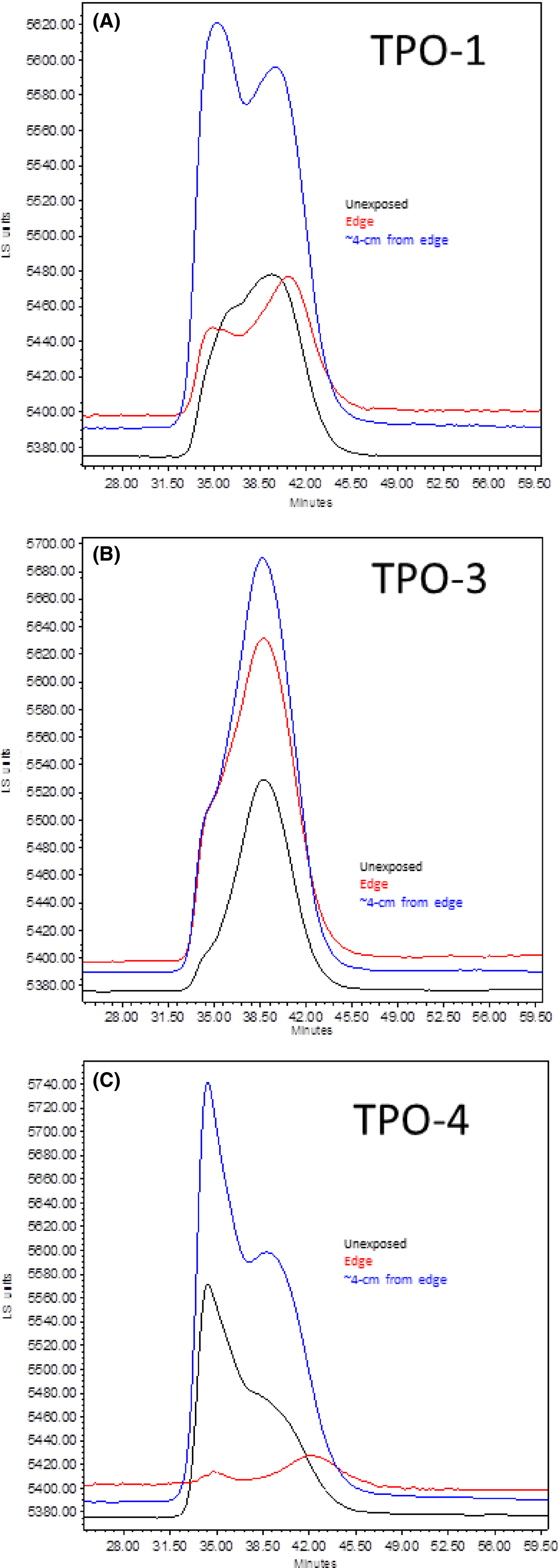
Figure 6. Light-scattering chromatographs of TPOs measured at an angle of 90°. Blue: 4 cm from the edge; red: edge sample; black: unexposed. (A) TPO-1, (B) TPO-3, and (C) TPO-4. |
For TPO-1, the unaged material has a small amount of large molecular weight material. Upon exposure, the position of this large molecular weight fraction moves to larger molecular weights and becomes more pronounced (Fig. 6). Comparison with Figure 5 indicates that this larger molecular weight fraction is only a very small part of the total polymer. Also of note is the fact that the sample taken from a distance of 4 cm from the edge demonstrates more cross-linking than the sample on the edge of the module. Although chain scission dominates the degradation for TPO-1, there is still some evidence cross-linking reactions have occurred. Overall, these changes produced decreases in the mass average molecular weight (Mw) near the edge, and slight increases further in the module, while increasing the polydispersity of the sample as it ages (Table 3).
| Sample | Mn (g/mol) | Mw (g/mol) | Mz (g/mol) | PDI |
|---|---|---|---|---|
| ||||
| TPO-1 | ||||
| Unexposed | 36,200 | 124,000 | 472,000 | 3.4 |
| Edge | 20,200 | 81,200 | 454,000 | 4.0 |
| ~4 cm from the edge | 26,100 | 134,000 | 738,000 | 5.1 |
| TPO-3 | ||||
| Unexposed | 50,400 | 135,000 | 337,000 | 2.7 |
| Edge | 27,300 | 130,000 | 461,000 | 4.8 |
| ~4 cm from the edge | 35,000 | 137,000 | 439,000 | 3.9 |
| TPO-4 | ||||
| Unexposed | 24,400 | 172,000 | 1,040,000 | 7.0 |
| Edge | 8,980 | 30,100 | 96,200 | 3.4 |
| ~4 cm from the edge | 18,300 | 135,000 | 868,000 | 7.4 |
For TPO-4, there is initially a significant amount of large molecular weight material (Fig. 5) such that the light-scattering chromatograph is dominated by the large molecular weight fraction (Fig. 6). Upon exposure, the small molecular weight fraction becomes more distinct. In the edge sample, Mw is reduced much more than the number average molecular weight (Mn), indicating a higher probability that large chains are reduced, resulting in a reduction in the PDI (Table 3). Neither the light-scattering data nor the molecular weight distribution can clearly demonstrate the formation of cross-links. However, considering that TPO-4 and TPO-1 were made by the same manufacturer and intended to differ primarily in their melting temperatures, it is likely that cross-linking is occurring in TPO-4 but that it is masked by a much stronger chain scission reaction.
For TPO-3, there is initially a weak signal for a large molecular weight fraction in the light-scattering data. After exposure, the large molecular weight fraction becomes more pronounced. Here, Mw remains relatively unchanged after exposure, but Mn is reduced (Table 3). For Mw to be constant, chain scission and cross-linking reactions must be occur at a similar rate. In Figure 5B, both the edge and 4-cm distance from the edge samples demonstrate the formation of a large molecular weight tail which is more pronounced for the edge sample. Thus, oxygen may be enhancing both cross-linking and chain scission reactions slightly in this material.
While TCB at 150°C was a good solvent for all the TPOs, the xylenes and toluene solvent did not produce complete solubility in the Soxhlet extractor (Fig. 1). For TPO-1, toluene was a better solvent than xylenes; but for TPO-4, xylenes were a marginally better solvent than toluene. Similarly, toluene was a poor solvent for PVB and TPU producing gel contents between 28.8% and 78.2%. But when PVB and TPU were tested using methanol and THF in the Soxhlet extractor, gel contents of 0% and ~4.9% were obtained, respectively. This illustrates how solvent quality, if it is marginal or poor, can give misleading results. Additionally, one must be cautious with TCB at 150°C because the thermal history of the extraction could alter the results, though we did not see evidence for this in our experiments. In the Soxhlet extractor, even with xylenes boiling around 140°C, the liquid is condensed and significantly cooled when in contact with the polymer. One would therefore not expect to see significant degradation of the polymers, even when xylenes are used.
For TPO-1 and TPO-4, a higher gel content was seen for the unexposed sample (Fig. 1). A reduction in the molecular weight is consistent with the SEC experiments, but the poor quality of the solvent leaves open the question of the amount of gel in the materials both before and after the experiment.
Toluene was a better solvent than xylenes for TPO-3 (Fig. 2), but both solvents indicate nearly zero gel content initially. Even though the aged samples indicate significant increases in gel content, the SEC data indicated there was only a small change in the molecular weight distribution (Table 3). Toluene is probably just barely able to dissolve this material such that the small amount of cross-linking occurring is able to significantly affect the measurement of the gel content without really changing the molecular weight distribution significantly.
For all three TPOs, chain scission seems to dominate over cross-linking reactions and is enhanced by the presence of oxygen in the perimeter regions. For TPO-1 and TPO-3 (and possibly TPO-4) there are also some cross-linking chemistry occurring but with a less noticeable dependence on the presence of oxygen. Dominance of chain scission over cross-linking was unexpected because indoor experiments [19, 21] with TPO-1 and TPO-3 mock modules clearly demonstrate a reduction in creep rates above 110°C and 90°C, respectively. This could be because UV light is largely responsible for chain scission, or that the small amount of cross-linked fractions here has a large effect on the rheology of the polymers.
Thermoplastic polyurethane
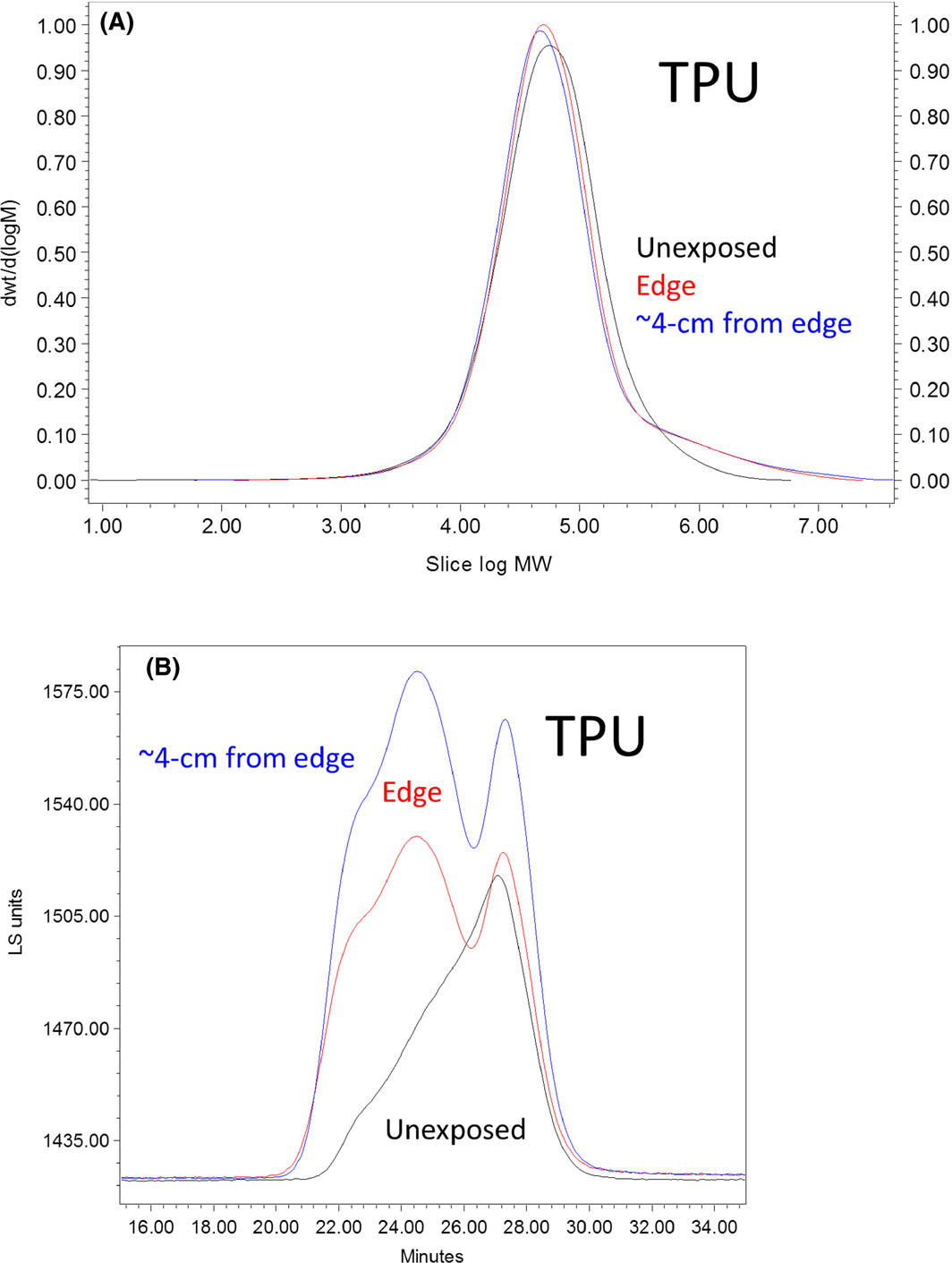
Even after exposure in Arizona region, the thermoplastic polyurethane (TPU) was highly soluble in THF and could be run in the SEC system using THF. Gel content measurements in THF (Fig. 1) indicate that 5.1, 4.9, and 4.5% of the polymer were not soluble for samples at a distance of 4 cm from the edge in the Arizona region, environmental chamber, and unexposed conditions, respectively. Upon exposure, a large molecular weight tail was seen on the GPC curve due to cross-linking of polymer chains, and there is significant indication of cross-linking in the light-scattering chromatographs (Fig. 7B). However, no signs of chain scission were seen in the mass chromatograms (Fig. 7A). The absence of small scale for cross-linking is further supported by the fact that Mn actually increased upon exposure (Table 4). Thus, a significant amount of polymer is becoming highly cross-linked.
|
|
|
Figure 7. Weight fraction determined by SEC-MALLS (SEC, size exclusion chromatography) and viscometry for the thermoplastic polyurethane thin-film mock module after exposure in Arizona. Samples dissolved in tetrahydrofuran (THF) by extraction at 40°C overnight. Molecular weight (MW) is in units of g/mol. Blue: 4 cm from the edge; red: edge sample; black: unexposed. Lines are normalized to the area under the curve. |
| Sample | Mn (g/mol) | Mw (g/mol) | Mz (g/mol) | PDI |
|---|---|---|---|---|
| TPU | ||||
| Unexposed | 20,700 | 89,900 | 370,000 | 4.3 |
| Edge | 24,700 | 176,000 | 2,540,000 | 7.1 |
| ~4 cm from the edge | 23,900 | 229,000 | 5,160,000 | 9.6 |
Initially, the TPU seems to have three bumps in the light-scattering plot (Fig. 7). After exposure, the two larger molecular weight fractions become more pronounced and shifted to larger molecular weights. The shift is slightly larger for the sample near the edge indicating that oxygen has a small additional effect, increasing the rate of cross-link formation. This could be indicative of the use of a block copolymer or a polymer blend where each different polymer types have differing ability to form cross-links.
Conclusions
Modules were deployed in a very hot environment with insulation on their backside in an effort to reproduce one of the worst case situations where creep of the encapsulant might occur. In previous experiments [19, 21], even when noncuring polymer encapsulant formulations were used, only insignificant creeping of the cells or other module components was observed. This was attributed to several factors such as some nonuniformity in the temperature distribution creating cold spots limiting creep. Measurable creep, occurring asymptotically with time, was observed for the more homogeneously constructed thin-film mock modules, particularly for NC-EVA.
For the NC-EVA and the TPOs, the rate of cross-linking and chain scission reactions vary with position in the sample indicating that a chemical species must be entering or leaving the module package affecting the kinetics. It is plausible that oxygen ingress is increasing the rate of chain scission relative to cross-linking and increasing the overall degradation rate.
The EVAs and the TPU tested here increased in their polymer molecular weight attributable to the formation of cross-links. For TPU, no evidence for chain scission was observed, but for EVA chain scission was observed and was enhanced presumably by the presence of oxygen in the perimeter areas. In TPU, these changes did not result in an increase in gel content, but in EVA an increase in gel content was seen even in the chain scission-prone perimeter regions with the NC-EVA sample.
In contrast, the TPOs experienced an overall decrease in molecular weight with aging. SEC data for TPO-1 and TPO-3 indicate some cross-linking reactions are occurring, but this is much less prominent than chain scission reactions. The overall reduction in molecular weight is a small effect and not likely to result in module creep as demonstrated elsewhere [19-21]. When using any thermoplastic material in a PV module, one should determine if cross-linking or chain scission dominates degradation reactions sufficiently to cause a concern for a reduction in molecular weight and subsequent creep.
The materials and “module” designs examined here help to identify the requirements for a module that could be subject to creep during a 25-year field lifetime. It would be difficult to construct an encapsulant that would experience creep when deployed. The encapsulation would have to have a low melting transition (≤70°C as in NC-EVA) providing a low viscosity at operating conditions, the module would have to use a mounting scheme that did not fix the relative positions of the front and back sheets, the module would have to be installed in a very hot climate in a hot mounting configuration, and the encapsulation would have to age predominantly by chain scission rather than cross-linking. Such a module construction might still pass 1000 h of 85°C exposure without creep but fail in the field. Examination at 90°C or 105°C for 200 h (MST 56) has been recently added to IEC 61730–2 to more rigorously screen for modules that might be prone to creep over prolonged time.
Acknowledgments
This work was part of a large collaborative effort of a number of people working on standards developed at many institutions. The authors gratefully acknowledge the support of the following individuals: Adam Stokes, Alain Blosse, Ann Norris, Bernd Koll, Bret Adams, Casimir Kotarba (Chad), David Trudell, Ed Gelak, Greg Perrin, Hirofumi Zenkoh, James Galica, Jayesh Bokria, John Pern, Jose Cano, Kartheek Koka, Keith Emery, Kent Terwilliger, Kolapo Olakonu, Masaaki Yamamichi, Mowafak Al-Jassim, Nick Powell, Niki Nickel, Pedro Gonzales, Peter Hacke, Ryan Smith, Ryan Tucker, Steve Glick, Steve Rummel, and Tsuyoshi Shioda. This work was supported by the U.S. Department of Energy under Contract No. DE-AC36-08-GO28308 with the National Renewable Energy Laboratory.
Conflict of Interest
None declared.
References
- Kapur, J., K. Proost, and C. A. Smith. 2009. Determination of moisture ingress through various encapsulants in glass/glass laminates. Pp. 001210–001214in Photovoltaic Specialists Conference (PVSC), 2009 34th IEEE.
- Kempe, M. D., G. J. Jorgensen, K. M. Terwilliger, T. J. McMahon, C. E. Kennedy, and T. T. Borek. 2007. Acetic acid production and glass transition concerns with ethylene-vinyl acetate used in photovoltaic devices. Sol. Energy Mater. Sol. Cells91:315–329.
- Meyer, S., S. Timmel, S. Richter, M. Werner, M. Gläser, S. Swatek, et al. 2014. Silver nanoparticles cause snail trails in photovoltaic modules. Sol. Energy Mater. Sol. Cells121:171–175.
- Reid, C. G., S. ferrigan, I. Fidalgo, and J. T. Woods. 2013. Contribution of PV encapsulant composition to reduction of potential induced degradation (PID) of crystalline silicon PV cells. 28th European Photovoltaic Solar Energy Conference and Exhibition.
- Reid, C. G., J. G. Bokria, and J. T. Woods. 2013. UV Aging and Outdoor exposure correlation for EVA PV encapsulants. Pp. 882508-882508-11in 2013 SPIE, San Diego, California.
- Cuddihy, E. F., A. Gupta, C. D. Coulbert, R. H. Liang, A. Gupta, and P. Willis, et al. 1983. Applications of ethylene vinyl acetate as an encapsulation material for terrestrial photovoltaic modules. DOE/JPL/1012-87 (DE83013509).
- Dietrich, S., M. Pander, M. Ebert, and J. Bagdahn. 2008. Mechanical assessment of large photovoltaic modules by test and finite element analysis.
- Wohlgemuth, J., M. Kempe, D. Miller, and S. Kurtz. 2011. Developing standards for PV packaging materials. SPIE. Proceedings of SPIE Conference
- Wohlgemuth, J., and W. Herrmann. 2005. Hot spot tests for crystalline silicon modules. Pp. 1062–106331st IEEE Photovoltaic Specialists Conference, 3-7 Jan. 2005.
- Wohlgemuth, J.. 2013. Hot Spot Testing of PV Modules. SPIE, San Diego, California.
- Kurtz, S., K. Whitfield, D. Miller, J. Joyce, J. H. Wohlgemuth, and M. D. Kempe, et al. 2009. Evaluation of high-temperature exposure of rack-mounted pohotovoltaic modules. 34th IEEE PVSC.
- D. C. Miller, M. D. Kempe, S. H. Glick, and S. R. Kurtz. 2010. Creep in photovoltaic modules: Examining the stability of polymeric materials and components. 35thIEEE Photovoltaic Specialists Conference (PVSC), 20–25 June 2010.
- Miller, D. C.2013. Examination of a standardized test for evaluating the degree of cure of EVA encapsulation (#1066/0731). National Renewable Energy Laboratory, Golden, Colorado.
- Cuddihy, E. F., and P. B. Willis. 1984. Antisoiling technology: theories of surface soiling and performance of antisoiling surface coatings. DOE/JPL/1012-102 (DE85006658).
- Holley, W. W., and S. C. Agro. 1998. Advanced EVA-based encapsulants. Final Report January 1993-June 1997 NREL/SR-520-25296.
- Pern, F. J., and A. W. Czanderna. 1992. Characterization of ethylene vinyl acetate (EVA) encapsulant: effects of thermal processing and weathering degradation on its discoloration. Sol. Energy Mater. Sol. Cells25:3–23.
- Patel, M., S. Pitts, P. Beavis, M. Robinson, P. Morrell, N. Khan, et al. 2013. Thermal stability of poly(ethylene-co-vinyl acetate) based materials. Polym. Testing32:785–793.
- Klemchuk, P., M. Ezrin, G. Lavigne, W. Holley, J. Galica, and S. Agro. 1997. Investigation of the degradation and stabilization of EVA-based encapsulant in field-aged solar energy modules. Polym. Degrad. Stab.55:347–365.
- Kempe, M. D., D. C. Miller, J. H. Wohlgemuth, S. R. Kurtz, J. M. Moseley, and Q.-U.-A. S. J. Shah, et al. 2012. A field evaluation of the potential for creep in thermoplastic encapsulant materials. Proceedings of the 38thIEEE Photovoltaic Specialists Conferences.
- Moseley, J. M., D. C. Miller, Q.-U.-A. S. J. Shah, K. Sakurai, M. D. Kempe, and G. TamizhMani, et al.,2011. The melt flow rate test in a reliability study of thermoplastic encapsulation materials in photovoltaic modules. Pp. 1–20NREL/TP-5200-52586.
- Kempe, M. D., D. C. Miller, J. H. Wohlgemuth, S. R. Kurtz, J. M. Moseley, and Q.-U.-A. S. J. Shah, et al., 2015. Field Testing of Thermoplastic Encapsulants in High-Temperature Installations. Submitted to Energy Science & Engineering.
- Lux, C., U. Blieske, E. Malguth, and N. Bogdanski. 2013. “Variations in Cross-Link Properties of EVA of Un-Aged and Aged PV-Modules. 29th European Photovoltaic Solar Energy Conference and Exhibition.
- Marais, S., Y. Hirata, D. Langevin, C. Chappey, T. Nguyen, and M. Metayer. 2002. Permeation and Sorption of Water and Gases Through EVA Copolymers Films. Mater. Res. Innovations6:79–88.
- Kempe, M. D.2006. Modeling of rates of moisture ingress into photovoltaic modules. Sol. Energy Mater. Sol. Cells90:2720–2738.
- Hulsmann, P., D. Philipp, and M. Kohl. 2009. Measuring temperature-dependent water vapor and gas permeation through high barrier films. Rev. Sci. Instrum.80:113901.
- McIntosh, K. R., N. E. Powell, A. W. Norris, J. N. Cotsell, and B. M. Ketola. 2011. The effect of damp-heat and UV aging tests on the optical properties of silicone and EVA encapsulants. Prog. Photovoltaics Res. Appl.19:294–300.
- Rätzsch, M., and U. Hofmann. 1991. The Crosslinking of Ethylene-Vinyl Acetate Copolymers with Sodium Alcoholates. J. Macromolecular Sci.: Part A - Chemistry28:145–157.
Document information
Published on 01/06/17
Submitted on 01/06/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?