Phytoremediation is a promising method for mitigating nitrogen pollution due to its low cost and generation of biomass that can be repurposed. However, given the risk of plant invasion and the low tolerance to extreme conditions of plants fcurrently used for phytoremediation, broad implementation requires the discovery of new species. This study assessed the phytoremediation suitability and invasive potential of Phyllanthus fluitans, a promising free-floating macrophyte for which no peer-reviewed physiological data exist. P. fluitans is morphologically similar to Lemna minor, the most widely used phytoremediation species, but has the added benefit of larger fronds that reduce wind-driven displacement risk. Plants were placed in 0.002M solutions of ammonium-to-nitrate ratios ranging from 0:1 to 1:0 across two independent trials. Plants receiving solely nitrate exhibited drastically higher fresh weight (p<0.05), dry weight, leaf count (p<0.05), root length, and survival rate compared to all other groups. Ammonium dose-dependently increased mortality, causing toxicity symptoms like chlorosis, necrosis, and stunted growth, resulting in complete die-off in the sole ammonium group. Additionally, the reduction of a mineral supplement between trials revealed P. fluitans to be highly sensitive to elevated water hardness. Collectively, nitrate preference and poor resilience render P. fluitans a poor phytoremediation candidate. However, under optimal conditions, the species can more than double its biomass in 24 days. This indicates that the invasive potential of P. fluitans varies drastically depending on environmental conditions, a finding that environmentalists can use to make condition-specific risk assessments and manage invasions accordingly.
1.0 INTRODUCTION
Nitrogen is an element essential for all forms of life. However, excess inorganic nitrogen can pollute water, degrading its quality and making it unsafe for consumption, and can additionally cause ecological damage by triggering harmful algal blooms and dead zones, disrupting nutrient cycles, and diminishing biodiversity [1,2]. The primary source of anthropogenic nitrogen pollution is agricultural fertilizer [3], of which only 10–15% enters the human food chain; the remainder runs off into the environment, with most ultimately reaching surface and ground waters [4].
Nitrogen pollution harms aquatic ecosystems through a eutrophication cascade: excessive nitrogen triggers explosive, and often toxic, algal growth that blocks sunlight, and once the nitrogen supply is exhausted, microbial decomposition of the algal biomass depletes the dissolved oxygen required by aerobic aquatic organisms. Over the past 50 years, the incidence of this cascade has increased more than tenfold [5]. High-profile examples include the 2014 Lake Erie microcystin crisis, which deprived over 500,000 residents of safe drinking water [6], and the Gulf of Mexico hypoxic zone, which now spans approximately 6,705 square miles [7]. In the United States alone, the economic costs of freshwater eutrophication are estimated at approximately $2.2 billion annually [8]. To address these severe environmental and economic impacts, researching effective methods to remediate eutrophic waters is critical.
The ability of aquatic macrophytes to remediate wastewater has been receiving increased attention, as these plants can take up and store large amounts of inorganic nitrogen [2]. Phytoremediation, the use of plants to remove contaminants, is among the most environmentally friendly and cost-effective approaches available. However, the efficiency of phytoremediation systems depends almost entirely on the physiology of the species used. Researchers in this field therefore agree that studying the morphology, growth characteristics, and nitrogen preference of macrophytes, especially underresearched species, is essential for designing systems that are both optimized and well-suited to specific forms of pollution [9,10]. The results of the present study contribute to this effort by evaluating the phytoremediation suitability of Phyllanthus fluitans, a species for which no peer-reviewed data on nitrogen uptake currently exist.
1.1 Phytoremediation
In response to nitrogen pollution and increasingly stringent regulations, researchers have developed several remediation methods, including bacterial biofilm filters, stormwater retention ponds, constructed wetlands, and physicochemical removal [11,12,13]. Although effective, each has notable limitations. Physicochemical removal and bacterial biofilm filtration, the most widely used approaches, are energy-intensive and expensive, and thus carry their own environmental concerns [14].
In recent years, phytoremediation has gained attention as a viable alternative, owing to its low cost, minimal maintenance requirements, low ecological risk, and high efficiency. Phytoremediation systems are also highly versatile as they can synergize with other remediation treatments and are effective against a broad range of pollutants beyond nitrogen [12,13]. This is especially relevant because nitrogen pollution frequently co-occurs with other contaminants [15], giving phytoremediation an advantage over methods targeting nitrogen alone. Documented pollutants that phytoremediation can mitigate include phosphorus, lithium, microplastics, cadmium, manganese, copper, zinc, and silver [15,16,17,18,19]. Phytoremediation also shows promise in aquaculture, one of the fastest-growing sectors of the global food economy. Although aquaculture effectively supplies seafood, unsustainable practices can directly contribute to nitrogen pollution, and multiple studies demonstrate the beneficial effects of integrating phytoremediation into aquaculture operations [11,13,20].
However, phytoremediation also has unique limitations, the most salient of which include relatively slow remediation rates, climate and seasonal dependence, intolerance to poor water conditions, and the risk of introducing invasive species. Hence, identifying new species that maximize the strengths of phytoremediation while minimizing these drawbacks is a prerequisite for its broader adoption as a primary method of addressing nitrogen pollution.
1.2 Key Markers of Nitrogen Phytoremediation Suitability: Nitrogen Preference and Growth Rate
Growth rate is conventionally the most important criterion for phytoremediation suitability and serves as a primary proxy for estimating nitrogen uptake (N uptake = N in tissue × ΔBiomass) [21]. Lemna minor, the current gold standard for aquatic phytoremediation, exhibits a relative growth rate of 0.42 d⁻¹ (dry weight basis) under optimal conditions, corresponding to a biomass doubling time of approximately 1.65 days [22].
By contrast, nitrogen preference, defined as the degree to which a plant species absorbs and assimilates one inorganic nitrogen form more effectively than another, is an important but often neglected factor in phytoremediation research. Although understudied, nitrogen preference has been shown to play a key role in plant physiology, growth, nitrogen uptake efficiency, and competitive dominance [23]. Most aquatic macrophytes prefer ammonium, owing to the lower metabolic energy required for its uptake and assimilation, which generally translates to faster growth [10]. Paradoxically, ammonium can also cause severe toxicity when it is the sole nitrogen source, especially at elevated concentrations, and plants adapted to nitrate-dominant environments tend to perform poorly in ammonium-rich media [23].
As it relates to phytoremediation, ammonium is the predominant nitrogen-containing component in aquaculture wastewater, suggesting that ammonium-preferring species will be most effective in treatment systems [20]. Research on the nitrogen preference of Lemna minor confirmed its ammonium preference and subsequently informed the development of a sustainable duckweed-based remediation system for aquaculture [20]. If Phyllanthus fluitans were to exhibit a similarly favorable preference, it could prove equally significant, given the two species’ shared free-floating morphology. However, wastewater rarely contains a single nitrogen form; effluent typically includes both ammonium and nitrate, despite being ammonium-dominant [24]. Thus, ideal phytoremediation candidates should be capable of rapidly assimilating both inorganic nitrogen forms while exhibiting a preference for ammonium.
Lastly, several practical considerations that do not directly relate to nitrogen uptake are nonetheless consequential for phytoremediation suitability, including tolerance to suboptimal water conditions, resistance to diseases and pests, and the utility of harvestable biomass [25].
1.3 Free-floating macrophytes and Phyllanthus fluitans
Phyllanthus fluitans, commonly known as red root floater, is a free-floating aquatic macrophyte of the family Phyllanthaceae, native to freshwater habitats in tropical and subtropical South America. Free-floating macrophytes are the most commonly used plant group in aquatic phytoremediation, owing to their numerous advantages, including utility in wastewater treatment, biofuel production, ecotoxicological assessment, and aquaculture [26].
The physiology of free-floating macrophytes aligns well with the key markers for phytoremediation suitability outlined in Section 1.3. Previous research indicates that floating macrophytes can adjust their root length in response to ambient nitrogen concentrations, elongating roots at low concentrations and shortening them at high concentrations, which suggests adaptability [27]. Such resilience is highly desirable for phytoremediation, as plants that wither under unfavorable conditions risk further polluting already eutrophic waters [11]. Moreover, free-floating macrophytes can assimilate atmospheric CO₂ directly while drawing nutrients from the aquatic environment, which contributes to their characteristically rapid growth [2,15]. Numerous studies confirm that floating macrophytes achieve higher growth rates than their emergent or fully submerged counterparts [9].
Currently, Lemna minor is the most widely used macrophyte in phytoremediation. Its rapid growth rate, environmental resilience, free-floating morphology, and ammonium preference make it highly effective at remediating nitrogen-rich waters. However, L. minor has significant drawbacks: its small frond size makes it highly susceptible to displacement by wind and water flow, increasing the risk of infiltration into natural water bodies where it is recognized as a prominent invasive species [28]. Additionally, under conditions of stress, L. minor can exhibit toxicity symptoms and significantly reduced remediation efficiency [29]. These limitations have led researchers to advocate strongly for studying alternative species, particularly those lacking adequate research that may perform better under specific conditions [30].
The present study therefore investigated P. fluitans, a free-floating macrophyte with very few published reports and virtually no peer-reviewed studies. Most existing information on P. fluitans comes from aquarium hobbyist blogs and forums, where users describe a growth profile, including rapid growth and nutrient uptake, similar to species known to be suitable for phytoremediation. These anecdotal observations are corroborated by the USDA APHIS Weed Risk Assessment, which classified P. fluitans as high risk, assigning it a 53.4% probability of becoming a major invader, a 44.0% probability of becoming a minor invader, and only a 2.6% probability of being non-invasive [31]. Management efforts for P. fluitans have so far totaled $15,559.73 in direct costs, representing 272.5 hours of labor and 67 treated acres along the Peace River (Sowinski, 2017a, as cited in [31]). Notably, P. fluitans has significantly larger fronds than L. minor, which reduces the risk of escape from contained phytoremediation systems.
P. fluitans also possesses two additional qualities that enhance its appeal for phytoremediation. First, it accumulates anthocyanins, appearing green under nitrogen-sufficient conditions and red under nitrogen limitation. Anthocyanin accumulation is a well-documented adaptive response to nitrogen deficiency across plant taxa [32], and thus serves as a visual bioindicator of nitrogen status. Second, P. fluitans can be used as animal feed after harvest, and in European markets, suppliers receive approximately €1 per 67.3 g sold, meaning harvested biomass may help offset system start-up costs or even generate revenue [13].
1.4 Research Aims and Hypothesis
The present study has the following primary objectives:
- Assess phytoremediation suitability. Evaluate P. fluitans as a candidate for nitrogen phytoremediation by measuring growth rate (a proxy for nitrogen uptake), root plasticity (a marker of resilience), and growth response to different ammonium-to-nitrate ratios (a marker of nitrogen preference).
- Address gaps in current literature. Contribute to the phytoremediation and plant physiology literature by supplying the first controlled data on P. fluitans growth across varying ammonium-to-nitrate ratios, an approach that better reflects real wastewater conditions than studies using single nitrogen forms in isolation.
- Inform invasive species management. Generate physiological data on P. fluitans that environmental agencies can use to refine weed risk assessments and develop management strategies.
To address these objectives, the effects of different ammonium (NH₄⁺) to nitrate (NO₃⁻) ratios on the growth of P. fluitans were tested using ratios of 1:3, 3:1, 1:0, 0:1, 1:1, and 0:0 (control). Based on the finding that L. minor exhibited its highest growth rate at a 25:75 nitrate-to-ammonium ratio [33] (one of the only previous studies to employ nitrogen form ratios) and the physiological and morphological similarities between the two species, the researchers of this study hypothesized that P. fluitans would exhibit the highest growth rate in the 1:3 (nitrate to ammonium) treatment. This hypothesis was further supported by the general observation that aquatic macrophytes tend to prefer ammonium but exhibit toxicity symptoms when it is the sole nitrogen source [9,10,23]. Additionally, based on previous reports of invasion and its free-floating morphology, P. fluitans was expected to exhibit rapid, exponential growth and resilience to environmental stressors, the latter assessed through variation in root length.
2 METHODOLOGY
The growth rate (and subsequent nitrogen preference, and vitality) of the plant species was observed for the duration of the 28 days for Trial 1 and 24 days for Trial 2. The data obtained from the investigation regarding changes in leaf growth, root growth, and wet weight was then compared to the control group. A 1:1 ratio of ammonium to nitrate acted as a control for Trial 1, but was replaced by a ratio of 0:0 water for Trial 2. The concentration of the nitrogen provided in treatments was based on the average nitrogen concentration in domestic wastewater due to this study’s focus on phytoremediation.
2.1 Preparing stock solution
Stock solutions were added before being poured into individual containers for experimentation. Two food-grade plastic containers with a volume of at least 2 liters, were used for making and holding the stock solutions. To organize the experimental setup, two sets of tape (with the text “stock solution 1”, “stock solution 2” and “stock solution 3 (Trial 2)” written on them) were created and stuck onto each of the 2 containers with the corresponding group name. Stock solution 1 represented the ammonium solution, stock solution 2 represented the nitrate solution, and stock solution 3 represented the water solution. Both tanks were filled with 1.9 liters of distilled water and 2.22 mL of micronutrient mix (Tropica Plant Growth Premium Fertilizer; link: https://tropica.com/en/plant-care/liquid-fertilizers/premium-nutrition/), 2.5 mL of “Seachem Flourish Phosphorus” (a potassium phosphorus-based fertilizer; link: https://www.seachem.com/flourish-phosphorus.php) (removed for Trial 2), 7.0 g of “Seachem Alkaline buffer” (a commercially available sodium bicarbonate-based supplement; Link: https://www.seachem.com/alkaline-buffer.php), and 20 g of “Seachem equilibrium” (changed to 2.5 g for Trial 2) (a commercially available supplement/fertilizer derived from potassium sulfate, calcium sulfate, magnesium sulfate, ferric sulfate, & manganese sulfate; link: https://www.seachem.com/equilibrium.php). Both batches of solution were mixed with a stirring rod to dissolve solutes and make both solutions as homogeneous as possible. In the nitrate solution (stock solution 2) 3.3 grams of potassium nitrate were added. To the ammonium solution (stock solution 1) 2.02 grams of ammonium chloride was added (concentrations displayed in Table 1).
After both stock solutions were made (following the concentrations shown in Table 2) they were used to create separate groups using 100% virgin plastic containers (L 175 mm x H 80 mm x W 117 mm) to test the different ratios. To distinguish between each of the tested groups each had tapes at their sides with a corresponding text stating either “0.002M nitrate”, “0.002M ammonium”, “1:3 ammonium to nitrate”, “3:1 ammonium to nitrate”, “1:1 ammonium to nitrate” (as the control for Trial 1), and “water” (as the control for Trial 2). The number of solutions from the original stock solution and distilled water that were used for these separate ratios are stated in Table 2.
| Stock Solution | Amount of Distilled Water (L) | Concentration of Nitrate (M) | Concentration of Ammonium (M) | Mass of Ammonium chloride (g) | Mass of Potassium Nitrate (g) |
| 1 | 1.9 | --------------- | 0.02 | 2.02 | --------------- |
| 2 | 1.9 | 0.02 | --------------- | -------------- | 3.3 |
| 3 * | 1.9 | --------------- | --------------- | --------------- | --------------- |
| Ratio of Ammonium to Nitrate | Amount of solution from stock solution 1 (mL) | Amount of solution from stock solution 2 (mL) | Amount of solution from stock solution 3 (mL) * | Distilled water used to fill each tank (mL) |
| * Water (control for Trial 2) | 0 | 0 | 85 | 765 |
| 0.002M nitrate (0:1) | 0 | 85 | 0 | 765 |
| 1:3 | 21.25 | 63.75 | 0 | 765 |
| 1:1 (control for Trial 1) | 42.5 | 42.5 | 0 | 765 |
| 3:1 | 63.75 | 21.25 | 0 | 765 |
| 0.002M ammonium (1:0) | 85 | 0 | 0 | 765 |
2.2 Preparing specimens
All the plants that were used in the experiment were sourced from an online retailer and quarantined (with the same corresponding setup for the control) for 7 days to allow plants to heal from the damage of being shipped. Afterward, plants were chosen per container based on their separate examinations for health conditions regarding visible signs of nutrient deficiency, physical damage, invasion by epiphytes such as algae, possible signs of disease, etc. In Trial 1 researchers used 6 samples per group, in account for P. fluitans reputation as an exceedingly fast growing plant. However, in Trial 2 this changed to 12 samples per group. This was done in consideration for P. fluitans delicate nature and to avoid damage occurring when handling as interpretations for treatments effects.
When measured for statistical data all groups of plants were then gently tapped with a dry paper towel to ensure the removal of all surface liquids on the plants before measurement. Plants selected for all groups had a leaf amount of 2 to 6 leaves. The mass of the plants was taken using an analytical balance. The resulting mass was ensured to not differ more than ± 0.2 g between groups; this was done to ensure that the plants picked are made into equal groups with a similar mass.
2.3 Growth Conditions
To ensure that the only independent variable throughout the experiment was the tested ratios, outside factors were controlled through various methods. All the samples were placed in a three-tiered light rack alongside each other. Two long LED lights acted as the light source, controlled using an electrical timer to properly time the light from 9 am (EST) - 7 pm (EST) so that each group was provided with a controlled 10 hours of light per day. Additionally, factors that might influence the results such as plant weight, pH/nutrient levels, and temperature were controlled respectively by measuring all plants to create groups with similar mass, changing the water for a periodic amount of time, and maintaining all groups near each other to simulate similar temperatures conditions.
2.4 Maintaining plants/experimenting
The sample size for Trial 1 was 6 samples per group, while Trial 2 had a sample size of 12 per group. After being measured, plants with 2 to 6 leaves were gently placed on the water's surface of each of the groups in their corresponding separate containers. Throughout the experiment, all groups were provided with 10 hours of light per day for 28 days while placed near one another to prevent inconsistencies in temperature for Trial 1. The same measures were taken for Trial 2, but in a 24-day duration. The plant medium/solution was replaced every Monday to maintain proper levels of nutrients with the same process repeated on maintained on the prepared pots and conditions. Plants were periodically removed and wrapped with a paper towel soaked in distilled water while this procedure was occurring. Throughout this procedure, all unexpected occurrences (deteriorating plant health, abnormal amount of dead plant matter, chlorosis, etc.) were recorded.
2.5 Measuring Root Length & Leaf Numbers
The number of leaves of the different treatments was recorded at the beginning of the experiment to be later compared to the final results. This was followed by a weekly counting of the leaves manually every Monday, Wednesday, and Friday, with a separate smaller section on written notes on dying leaves, leaves lost (the number of dead leaves in each group was documented before taking measurements), leaves damaged, and buds growing. This acted as a precaution considering the delicateness of the plants and only samples of floating plants, with at least 1 healthy leaf, and a root system considered in the graph showing the numbers of total leaves per treatment. Additionally, the measurement of the longest root of each plant per treatment was taken using a ruler and recorded in centimeters using an Excel spreadsheet every Monday and Friday for Trial 1, but only every Monday for Trial 2 of the experiment's duration.
2.6 Measuring Fresh and Dry Weight
Fresh weight was measured multiple times a week for a longitudinal representation of growth across the length of the study. The “fresh weight” of individual plants (separated by groups) was taken (every Monday and Friday for Trial 1, but every Monday for Trial 2), using an analytical balance. Aside from the recording of wet weight measurements from the damaged plants, this process involved carefully removing the separate groups of plants from their respective tanks and placing them on a wet paper towel which was soaked in plant growth medium to minimize shock. Plants were wrapped with a paper towel when measurements weren’t taking place. Preparing plants for measurement involved carefully removing the separate groups of plants from their growth medium and gently removing the liquids or foreign debris from the plants using a paper towel. Lastly, the dry weight was taken post-study using an electronic balance after the plants had been completely dried on a paper towel using an incubator at 37 to 40°C for 10 hours. The dry weight was taken separately between groups to provide a precise measurement in comparing the biomass between groups.
2.7 Measuring Survival Number
The number of plants that were alive was counted every Monday, Wednesday, and Friday of the study period. They were distributed into a Microsoft Excel spreadsheet with their corresponding treatments and sample sizes. They were then calculated as a survival number percentage in which the same size number was divided by the initial sample size and the quotient was multiplied by 100. This was done by Microsoft Excel’s basic functions.
2.8 Safety
Although most substances that were used in the experiment were mostly commercial and “not hazardous” under the criteria of the federal OSHA Hazard Communication Standard 29CFR 1910.1200, and Regulation (EC) No 1272/2008 (GHS), they can still be dangerous if exposed to for a long duration of time. These substances that can pose a threat include Seachem Flourish Phosphorus, Tropica Plant Growth Premium Fertilizer, Seachem Alkaline buffer, and Seachem equilibrium. However, ammonium chloride and potassium nitrate received a 2 and 1 NEPA health rating, respectively. To reduce the exposure risk, researchers washed exposed skin before and after experimentation, wore protective clothing and gloves when in contact with the material, and ensured that skin protection, eye protection, and protective clothing were put on precisely. During the experimental process, or when researchers came into contact with the material, researchers refrained from consuming any liquids or foods. It was also ensured that eye wash stations or safety showers were within the area in which the experiment was conducted. The products used were kept away from any heat in the case of gases being released and inhaled. Safety precautions and procedures were taken referencing the MSDS sheets. The plant used in this experiment poses no known health risks to humans (assuming it is not digested in excessive amounts).
3.0 DATA ANALYSIS
Data was collected and stored using Microsoft Excel. Images were taken using a smartphone and cropped to only exhibit the extent of plant material. The mean values were analyzed using Microsoft Excel. Data was shown using scatter plots and bar graphs in which the x-values were time in days and the y-values were mean fresh weight in grams, dry weight in grams, mean root length in centimeters, total leaf amount, and survival number as a percentage. Standard deviation, standard error of the mean, and error bars were added and determined using Microsoft Excel. Trial 1 figures used standard deviation as error bars while Trial 2 figures used standard error of the mean as error bars. To calculate significance, one-way ANOVA followed by Tukey HSD was done using https://astatsa.com/OneWay_Anova_with_TukeyHSD/. Significance was determined by p-values, which were considered significant when the p-value was less than 0.05. Large error bars in select figures are a result of plants' reproduction within the trial, reflecting differences between samples, not to be confused with 'error' that reflects the probability of findings not being reproducible.
4.0 DATA AND RESULTS
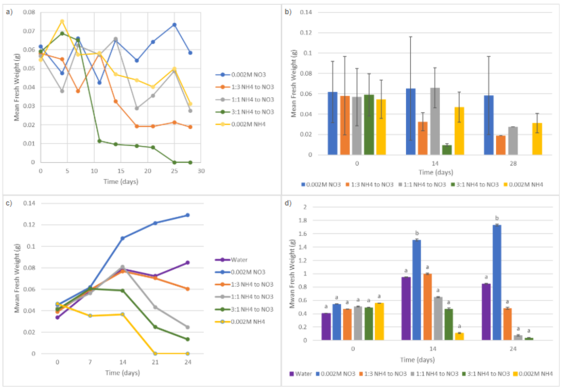
In Trial 1, Fig. 1a & Fig. 1b, total fresh weight generally decreased over time across most treatment groups, with the exception of the 0.002M NO₃⁻ treatment (0.3499 g by Day 28). Ammonium-containing treatments showed a substantial decline through Day 28. In Trial 2, Fig. 1c & Fig. 1d, total fresh weight changed significantly over time depending on the source of nitrogen. Plants treated with sole nitrate had the greatest increase in biomass from Day 0 (0.546 g) to Day 24 (1.730), with a significant increase by Day 14, resulting in the highest overall fresh weight. Treatments containing ammonium showed reduced growth rates over time. Both the 1:3 and 1:1 NH₄⁺ to NO₃⁻ treatments initially increased by Day 14 (0.999 g and 0.649 g, respectively) and declined by Day 24. By Days 14 and 24, nitrate-induced plants were significantly greater in biomass than ammonium-induced plants, suggesting that nitrate supports biomass accumulation while ammonium negatively affects long-term growth.
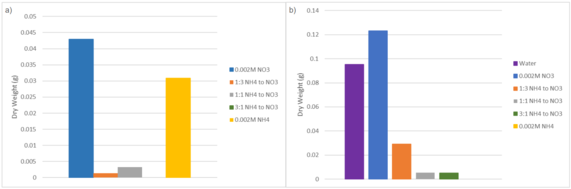
Final dry weight measurements support the trend observed in Fig. 1, indicating that nitrogen form influenced biomass accumulation. In Fig. 2a, The 0.002M NO₃⁻ had the highest overall dry weight (0.0429 g), followed by the 0.002M NH₄⁺ treatment (0.0309 g). Treatments containing both nitrate and ammonium yielded considerably lower dry mass. The 3:1 NH₄⁺ to NO₃⁻ treatment group had no remaining samples at Day 28, preventing measurement of dry weight. In Trial 2, Plants treated with 0.002M NO₃⁻ produced the greatest final dry weight (0.1231 g) in comparison to all other treatment groups. The control showed less dry weight than the sole nitrate treatment (0.0953 g). Treatments containing ammonium yielded comparably lower dry weights (1:3 NH₄⁺ to NO₃⁻, 0.0292 g; 1:1 NH₄⁺ to NO₃⁻, 0.0055 g; 3:1 NH₄⁺ to NO₃⁻, 0.0052 g). The 0.002M NH₄⁺ treatment produced no measurable dry weight by the end of the trial.
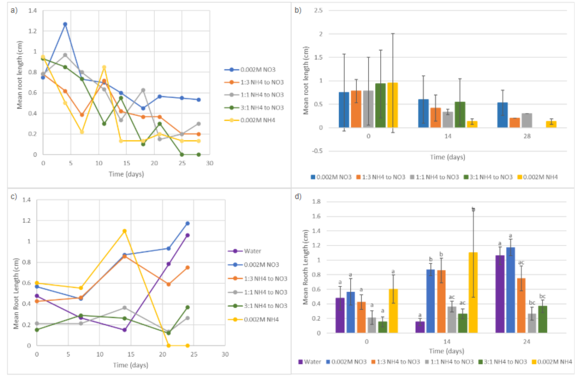
In Trial 1, Fig. 3a & Fig. 3b, mean root length generally decreased over time across all nitrogen treatments. The 0.002M NO₃⁻ group had the highest average root length by Day 28 compared to other treatments. In Trial 2, Fig. 3c & Fig. 3d, plants grown with 0.002M NH₄⁺ initially exhibited the greatest root elongation, having the highest mean root length by Day 14 (1.100 cm), but declined by Day 24. In contrast, 0.002M NO₃⁻ showed steady root growth, resulting in one of the significantly higher mean root lengths by Day 24 (1.436 cm). The control and 1:3 NH₄⁺ to NO₃⁻ also indicated relatively high root elongation by Day 24 (0.750 and 1.060 cm, respectively). Treatments containing higher amounts of ammonium (1:1 and 3:1 NH₄⁺ to NO₃⁻) consistently produced shorter roots (0.267 and 0.367 cm, respectively).
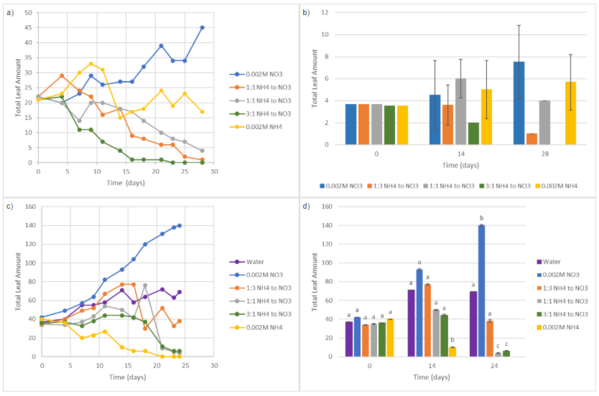
In Trial 1, Fig. 4a & Fig. 4b, total leaf amount generally declined with combined nitrate and ammonium treatments. By Day 28, 0.002M NO₃⁻ showed the greatest increase in total leaf count relative to other treatments (45) followed by 0.002M NH₄⁺ (17). In contrast, combined treatments (1:3, 1:1, and 3:1 NH₄⁺ to NO₃⁻) showed reductions in leaf number over time (1, 4, and 0 by Day 28).In Trial 2, Fig. 4c & Fig. 4d, total leaf amount increased over time, primarily in the 0.002M NO₃⁻ group, showing a statistically significant increase from Day 0 (42) to Day 24 (140), and had the highest overall leaf number. The control group showed a moderate increase by Day 14, while the 1:3 NH₄⁺ to NO₃⁻ group showed some growth at Day 14 (77), then decreased by nearly 50% at Day 24 (38). Conversely, the 1:1 and 3:1 NH₄⁺ to NO₃⁻ treatments significantly reduced total leaf amount over time, and the 0.002M NH₄⁺ consistently had the lowest leaf count.
Similar to Figures 1-5, survival rates depended on the nitrogen sources over time. In Trial 1, Fig. 6a, survival generally remained stable in the 0.0022M NO₃⁻ treatment, maintaining 100% survival by Day 28. The 0.002M NH₄⁺ treatment group initially increased to 116.67% and remained stable through Day 11, but declined to 50% of the original sample size at Day 14. Conversely, mixed nitrogen sources showed a reduction in survival percentage over time, with 3:1 NH₄⁺ to NO₃⁻ treatment reaching complete mortality by Day 28. In Trial 2, Fig. 6b, plants treated with sole NO₃⁻ maintained the highest survival throughout the trial, at 150% by Day 24. The control and 1:3 NH₄⁺ to NO₃⁻ fluctuated in the survival number but generally declined towards the end. Conversely, higher ammonium treatments, 1:1 and 3:1 NH₄⁺ to NO₃⁻, showed steep declines following Day 16, and sole NH₄⁺ treatment exhibited complete mortality. Overall, nitrate treatments sustained plant viability, while increasing ammonium treatments negatively influenced survival over time.
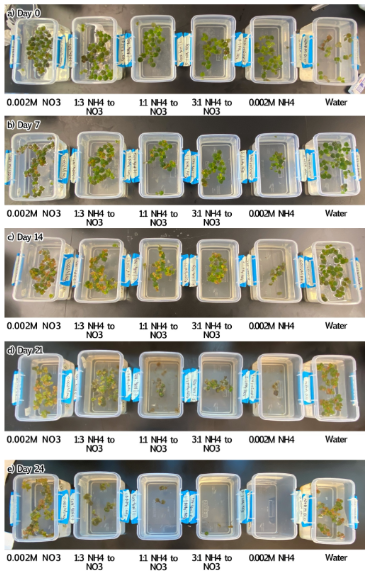
Algae growth was seen in 0.002M NO₃⁻, 1:1 NH₄⁺ to NO₃⁻, and 3:1 NH₄⁺ to NO₃⁻. Comparing treatment group photos from Day 0 (A) to Day 28 (E), sample sizes decreased as plant conditions worsened.
Algae growth was seen in 1:3 NH₄⁺ to NO₃⁻, 1:1 NH₄⁺ to NO₃⁻, and sole NH₄⁺ on Day 21. As the study progressed, treatment group photos from Day 0 to Day 24 demonstrated a decreasing sample size shown through surface area and worsening plant conditions in all groups other than 0.002M NO₃⁻ and the control.
5.0 DISCUSSION
The findings of this study suggest that P. fluitans has a strong preference for nitrate and an equally strong ammonium intolerance, with plants in both independent trials, especially Trial 2, exhibiting the highest average fresh weight and total dry weight when nitrate was the sole nitrogen source.
Although Trial 1 provided substantial insight into P. fluitans’ physiology, its high mortality rate across most groups prohibited definitive conclusions regarding nitrogen preference. As a result, the researchers initially inferred that P. fluitans may prefer media containing a sole nitrogen form. Although Trial 1’s data supported this preliminary hypothesis, it was made less credible when mechanisms established in previous studies and evolutionary adaptation were questioned. Nitrogen preference is primarily determined by the predominant nitrogen source in a plant’s native environment [34]. Ecologically, it is highly unlikely that a given environment undergoes pH shifts so dramatic as to swing the predominant nitrogen form from one extreme to the other. Moreover, no comprehensive review of nitrogen preference has documented this type of dual-extreme preference in any species [35].
These inconsistent results likely stemmed from the addition of “Seachem Equilibrium” to the growth medium in Trial 1, an additive that was significantly reduced in Trial 2. Although intended to supply calcium and magnesium, Seachem Equilibrium contains a substantial amount of potassium. Previous research indicates that potassium at sufficiently high concentrations can alleviate ammonium toxicity and may even produce synergistic effects [36]. A study on rice seedlings [37] attributed this phenomenon to potassium’s ability to restrict ammonium overaccumulation. Within plant roots, potassium actively competes with ammonium for access to transport channels. Even when ammonium overaccumulates and disrupts auxin distribution, a hormone responsible for root growth and gravitropism, potassium can partially restore proper hormone distribution. Together, these mechanisms allow potassium to meaningfully blunt ammonium toxicity while permitting the plant to assimilate this metabolically efficient nitrogen form, thereby creating synergy.
Trial 2’s data were more robust owing to three factors: a high mortality rate occurring only in selective groups, the emergence of a clear dose-dependent pattern, and the reduction of confounding variables (i.e., Seachem Equilibrium). The group receiving only nitrate (0.002M NO₃⁻) showed significantly higher fresh weight, dry weight, root length, and leaf number than all other groups, indicating a strong nitrate preference. Conversely, the group receiving solely ammonium showed the opposite pattern, with complete die-off by the end of the trial. The conclusion of ammonium intolerance is further strengthened by Fig. 6, which shows a dose-dependent, direct relationship between increasing ammonium concentration and declining survival rate. This intolerance can also be observed visually in Fig. 5, where groups receiving ammonium exhibit classic signs of toxicity: leaf chlorosis (yellowing), necrosis (browning and tissue death), leaf curling or wilting, and stunted shoot growth [38]. Although P. fluitans exhibits the root plasticity characteristic of macrophytes with high adaptability (as evidenced by differences in root length both between groups and over time), it was insufficient to meaningfully prevent toxicity. The strength of this nitrate preference is notably unusual among free-floating macrophytes. A review of the literature reveals that the vast majority of tested floating species (approximately 29 of 33) prefer ammonium as their primary nitrogen source [10,23]. The strong nitrate preference observed in P. fluitans thus places it among a small minority of floating macrophytes.
The addition of Seachem Equilibrium may have also confounded results for reasons beyond its potassium content. Although most plants perform well in environments with substantial levels of hardness-increasing ions such as calcium and magnesium [39], some species are intolerant of these minerals at high concentrations. Using Trial 1 data, the researchers hypothesized that P. fluitans may be sensitive to moderate and high levels of general water hardness. To evaluate this hypothesis and to reduce mortality, the addition of Seachem Equilibrium was decreased from 10 grams to 2.5 grams in Trial 2. As shown by the contrast between Fig. 6 and Fig. 12, survival rates markedly improved for most groups in Trial 2, allowing for a clear pattern of ammonium intolerance to emerge.
This sensitivity to water hardness is further corroborated by both anecdotal and mechanistic evidence. Multiple accounts from aquatic hobbyists on online forums describe P. fluitans’ intolerance to high general hardness, a characteristic also observed by the researchers when cultivating these plants for the present study. From a mechanistic and evolutionary perspective, P. fluitans is likely a “soft-water specialist.” Like nitrogen preference, water hardness tolerance is shaped by conditions in a species’ native environment. The freshwaters of tropical South America, P. fluitans’ native habitat, are characteristically low in dissolved minerals; the Amazon and Orinoco basins, which encompass the species’ range, have reported conductivities as low as 5–30 µS/cm and calcium concentrations often below 5 mg/L [40,41]. This suggests that P. fluitans may not have developed the robust efflux pumps that hard-water-adapted plants use to reduce cytosolic calcium accumulation. When introduced to hard water, excess calcium may cause cytotoxicity by chelating negatively charged molecules such as phosphates and proteins [42].
Taken together, a physiological profile emerges: a strong nitrate preference mismatched with the ammonium-dominant composition of most wastewaters, dose-dependent ammonium toxicity resulting in complete mortality at the highest concentrations, and a probable sensitivity to water hardness that limits survival in mineral-rich effluents. Collectively, these three characteristics render P. fluitans a poor candidate for phytoremediation. Unless unique circumstances favor specific features of P. fluitans (such as anthocyanin accumulation as a visual nitrogen indicator, remediation of solely nitrate-containing or highly acidic waters, or the value of its harvestable biomass), the findings of the present study do not support the use of P. fluitans for wastewater treatment or aquaculture.
Despite not identifying a new phytoremediation candidate, the present study contributes to the literature by providing physiological data on a species with no prior experimental studies but classified by the USDA as “high risk.” Research on understudied but potentially invasive plants is particularly important, as knowledge of their physiology and growth patterns helps environmentalists predict future invasions and develop management strategies [43]. Free-floating macrophytes with exponential growth rates are known to become invasive in environments with ample light and excess nutrients (see Section 1.4) [26]. The present study’s characterization of P. fluitans’ nutrient requirements, stress tolerances, and growth kinetics provides the first experimentally derived data with which to evaluate and refine this risk classification.
Specifically, the results identify several environmental conditions that modulate P. fluitans’ invasive potential. As the plant is highly specialized in its nutrient requirements (a feature more commonly associated with specialist rather than generalist species) [44], P. fluitans is unlikely to pose concern in environments with moderate-to-high ammonium concentrations, which predominate in neutral-to-acidic freshwaters where nitrification is suppressed [45] (unless sufficient potassium is also present), or in waters with high mineral content (particularly calcium). However, P. fluitans retains the potential to become highly problematic where its optimal conditions (low water hardness, nitrate as the primary nitrogen source, elevated macronutrient levels, and ample light) are met, given its capacity for rapid growth as the sole nitrate group more than doubled its biomass over Trial 2’s 24-day period (Fig. 1). Environmentalists can apply these findings to evaluate the invasive potential of P. fluitans using site-specific environmental factors and, where optimal conditions are identified, work to remove the species before severe ecological damage occurs.
5.1 Limitations & Statistical Power
Apart from the addition of Seachem Equilibrium, which elevated Trial 1’s mortality rate but inadvertently yielded some noteworthy findings, two additional limitations are also noteworthy.
A key methodological change in Trial 2 was the increase in sample size from 6 to 12 plants, enabling statistical analysis of both average fresh weight and average root length. However, in standard phytoremediation research, total dry weight is considered a more comprehensive measure of biomass because it accounts for differences in sample size as plants reproduce throughout the trial and is not confounded by water content [46]. Although the methodology of the present study aligns with previous phytoremediation research in which dry weight is typically not statistically analyzed, performing the experiment in triplicate or quadruplicate would permit ANOVA or t-tests and thereby strengthen the conclusions. That said, Trial 2 produced results that so consistently corroborate a strong nitrate preference that this additional level of statistical rigor, while desirable, likely would not alter the conclusion.
A final notable limitation is the absence of an assay measuring nitrogen content in plant tissue, a key component of the standard equation for estimating nitrogen uptake (N uptake = N in tissue × ΔBiomass) [21]. Although integral to fully characterizing nitrogen phytoremediation suitability, this assay could not be conducted due to logistical constraints. This omission is most consequential for the nitrate-only group, which exhibited substantial biomass accumulation: quantifying the tissue nitrogen concentration for this group would have allowed estimation of uptake rates, data that could inform applications such as the remediation of nitrate-dominated waters. Nevertheless, given that P. fluitans’ physiological profile already renders it a poor phytoremediation candidate in the vast majority of cases, this application is highly limited in scope.
6.0 CONCLUSION
Results from this study suggest that Phyllanthus fluitans possesses a strong preference for nitrate, ammonium intolerance, and high mineral sensitivity. As these features run contrary to the established markers of phytoremediation suitability, P. fluitans is likely a poor candidate unless in niche circumstances where one of its unique traits are desired (i.e., anthocyanin accumulation, remediation of nitrate alone, harvestable biomass). However, under optimal conditions, P. fluitans has the potential to become highly invasive, as evidenced by its ability to more than double biomass in just 24 days. More broadly, these findings underscore the importance of studying alternative factors alongside growth rate when evaluating candidate species, as even rapidly growing macrophytes may be functionally unsuitable if their nitrogen preferences are mismatched with the composition of target effluents. As the present study failed to identify a new candidate for phytoremediation, all while limitations of currently used plants still exist, future studies should continue assessing the phytoremediation suitability of promising free-floating macrophytes, not only by investigating conventional markers (i.e., growth rate, nitrogen uptake) but also by investigating ones that are often overlooked, like nitrogen preference. Lastly, environmentalists should apply the physiological profile derived from this study to evaluate the invasive potential of P. fluitans using site-specific ecological conditions and, where optimal conditions are identified, work to remove the species before severe ecological damage occurs.
ACKNOWLEDGEMENTS
We would like to thank Dr. D. Marmor, Mrs. N. Babbar, Dr. L. Wang, Ms. J. Zhu, Mrs. A. MacLeod, Dr. J. Cohen, Dr. X. Lin, Mr. Z. Liang, and Ms. R. DePietro.
REFERENCES
[1] NOAA, “What is eutrophication?,” National Oceanic and Atmospheric Administration, 2017. Retrieved from https://oceanservice.noaa.gov/facts/eutrophication.html.
[2] F. Su, Z. Li, Y. Li, S. Li, H. Chen, P. Zhuang, and F. Wang, “Removal of Total Nitrogen and Phosphorus Using Single or Combinations of Aquatic Plants,” International Journal of Environmental Research and Public Health, vol. 16, no. 23, pp. 4663, 2019.
[3] R. W. Howarth, A. Sharpley, and D. Walker, “Sources of nutrient pollution to coastal waters in the United States: Implications for achieving coastal water quality goals,” Estuaries, vol. 25, no. 4, pp. 656–676, 2002.
[4] J. N. Galloway, J. D. Aber, J. W. Erisman, S. P. Seitzinger, R. W. Howarth, E. B. Cowling, and B. J. Cosby, “The nitrogen cascade,” BioScience, vol. 53, no. 4, pp. 341–356, 2003.
[5] D. Breitburg, L. A. Levin, A. Oschlies, M. Grégoire, F. P. Chavez, D. J. Conley, V. Garçon, D. Gilbert, D. Gutiérrez, K. Isensee, G. S. Jacinto, K. E. Limburg, I. Montes, S. W. A. Naqvi, G. C. Pitcher, N. N. Rabalais, M. R. Roman, K. A. Rose, B. A. Seibel, M. Telszewski, M. Yasuhara, and J. Zhang, “Declining oxygen in the global ocean and coastal waters,” Science, vol. 359, no. 6371, eaam7240, 2018.
[6] U.S. Environmental Protection Agency, Great Lakes National Program Office, U.S. action plan for Lake Erie: Commitments and strategy for phosphorus reduction, 2018. Retrieved from https://www.epa.gov/sites/default/files/2018-03/documents/us_dap_final_march_1.pdf.
[7] NOAA, “Gulf of Mexico ‘dead zone’ is 12th largest on record,” National Oceanic and Atmospheric Administration, 2024. Retrieved from https://www.noaa.gov/news-release/2024-gulf-of-mexico-dead-zone.
[8] W. K. Dodds, W. W. Bouska, J. L. Eitzmann, T. J. Pilger, K. L. Pitts, A. J. Riley, J. T. Schloesser, and D. J. Thornbrugh, “Eutrophication of U.S. freshwaters: Analysis of potential economic damages,” Environmental Science & Technology, vol. 43, no. 1, pp. 12–19, 2009.
[9] A. Jampeetong, H. Brix, and S. Kantawanichkul, “Effects of inorganic nitrogen forms on growth, morphology, nitrogen uptake capacity and nutrient allocation of four tropical aquatic macrophytes (Salvinia cucullata, Ipomoea aquatica, Cyperus involucratus and Vetiveria zizanioides),” Aquatic Botany, vol. 97, no. 1, pp. 10–16, 2012.
[10] A. Jampeetong, T. Sripakdee, T. Khamphaya, and S. Chairuangsri, “The Effects of Nitrogen as NO₃⁻ and NH₄⁺ on the Growth and Symbiont (Anabaena azollae) of Azolla pinnata R. Brown,” Chiang Mai University Journal of Natural Sciences, vol. 15, no. 1, pp. 11–20, 2016.
[11] N. U. M. Nizam, M. M. Hanafiah, I. M. Noor, and H. I. A. Karim, “Efficiency of Five Selected Aquatic Plants in Phytoremediation of Aquaculture Wastewater,” Applied Sciences, vol. 10, no. 8, pp. 2712, 2020.
[12] M. Borin and M. Salvato, “Effects of five macrophytes on nitrogen remediation and mass balance in wetland mesocosms,” Ecological Engineering, vol. 46, pp. 34–42, 2012.
[13] T. Irhayyim, G. Beliczky, and M. Bercsényi, “Nutrient bioremediation efficiency of bacterial biofilms and plant based biofilters in a recirculating common carp (Cyprinus carpio L.) culture system,” Iranian Journal of Fisheries Sciences, vol. 20, no. 3, pp. 828–845, 2021.
[14] H. Gao, Y. D. Scherson, and G. F. Wells, “Towards energy neutral wastewater treatment: methodology and state of the art,” Environmental Science: Processes & Impacts, vol. 16, no. 6, pp. 1223–1246, 2014. [Note: Citation listed as Gao et al., 2020 in original manuscript; exact source requires author verification.]
[15] J. T. Spangler, D. J. Sample, L. J. Fox, J. S. Owen Jr., and S. A. White, “Floating treatment wetland aided nutrient removal from agricultural runoff using two wetland species,” Ecological Engineering, vol. 127, pp. 468–479, 2019.
[16] A. I. Török, A. Moldovan, E. Kovacs, O. Cadar, A. Becze, E. A. Levei, and E. Neag, “Lithium accumulation in Salvinia natans free-floating aquatic plant,” Materials, vol. 15, no. 20, pp. 7243, 2022.
[17] O. Olkhovych, N. Svietlova, Y. Konotop, O. Karaushu, and S. Hrechishkina, “Removal of metal nanoparticles colloidal solutions by water plants,” Nanoscale Research Letters, vol. 11, pp. 1–7, 2016.
[18] U. Rozman, A. J. Kokalj, A. Dolar, D. Drobne, and G. Kalčíková, “Long-term interactions between microplastics and floating macrophyte Lemna minor: The potential for phytoremediation of microplastics in the aquatic environment,” Science of the Total Environment, vol. 831, pp. 154866, 2022.
[19] Y. S. Chang, Y. J. Chang, C. T. Lin, M. C. Lee, C. W. Wu, and Y. H. Lai, “Nitrogen fertilization promotes the phytoremediation of cadmium in Pentas lanceolata,” International Biodeterioration & Biodegradation, vol. 85, pp. 709–714, 2013.
[20] S. Paolacci, V. Stejskal, D. Toner, and M. A. Jansen, “Wastewater valorisation in an integrated multitrophic aquaculture system; assessing nutrient removal and biomass production by duckweed species,” Environmental Pollution, vol. 302, pp. 119059, 2022.
[21] M. Greenway, “Suitability of macrophytes for nutrient removal from surface flow constructed wetlands receiving secondary treated effluent in Queensland, Australia,” Water Science and Technology, vol. 48, no. 2, pp. 211–218, 2003.
[22] P. Ziegler, K. Adelmann, S. Zimmer, C. Schmidt, and K.-J. Appenroth, “Relative in vitro growth rates of duckweeds (Lemnaceae) – the most rapidly growing higher plants,” Plant Biology, vol. 17, pp. 33–41, 2015.
[23] S. Boudsocq, A. Niboyet, J. C. Lata, X. Raynaud, N. Loeuille, J. Mathieu, M. Blouin, L. Abbadie, and S. Barot, “Plant Preference for Ammonium versus Nitrate: A Neglected Determinant of Ecosystem Functioning?,” The American Naturalist, vol. 180, no. 1, pp. 60–69, 2012.
[24] Metcalf & Eddy, Inc., G. Tchobanoglous, F. L. Burton, and H. D. Stensel, Wastewater Engineering: Treatment and Reuse, 4th ed. McGraw-Hill, 2003.
[25] A. Yan, Y. Wang, S. N. Tan, M. L. Mohd Yusof, S. Ghosh, and Z. Chen, “Phytoremediation: A Promising Approach for Revegetation of Heavy Metal-Polluted Land,” Frontiers in Plant Science, vol. 11, pp. 359, 2020.
[26] M. J. McCann, “Response diversity of free-floating plants to nutrient stoichiometry and temperature: growth and resting body formation,” PeerJ, vol. 4, pp. e1781, 2016.
[27] Y. Jin, Q. Zhang, L. M. Zhang, N. F. Lei, J. S. Chen, W. Xue, and F. H. Yu, “Distinct responses of frond and root to increasing nutrient availability in a floating clonal plant,” PLoS ONE, vol. 16, no. 10, pp. e0258253, 2021.
[28] P. Skillicorn, W. Spira, and W. Journey, Duckweed aquaculture: A new aquatic farming system for developing countries, World Bank Technical Paper. Washington, D.C.: The World Bank, 1993. ISBN 0-8213-2067-X.
[29] A. O. Ekperusi, F. D. Sikoki, and E. O. Nwachukwu, “Application of common duckweed (Lemna minor) in phytoremediation of chemicals in the environment: State and future perspective,” Chemosphere, vol. 223, pp. 285–309, 2019.
[30] C. Gu, F. Li, J. Xiao, S. Chu, S. Song, and M. H. Wong, “A novel submerged Rotala rotundifolia, its growth characteristics and remediation potential for eutrophic waters,” Scientific Reports, vol. 9, no. 1, pp. 14855, 2019.
[31] USDA APHIS, Weed Risk Assessment for Phyllanthus fluitans Benth. ex Müll. Arg. (Phyllanthaceae) – Red root floater, United States Department of Agriculture, Animal and Plant Health Inspection Service, 2017.
[32] J. Liang and J. X. He, “Protective role of anthocyanins in plants under low nitrogen stress,” Biochemical and Biophysical Research Communications, vol. 498, no. 4, pp. 946–953, 2018.
[33] F. Petersen, J. Demann, D. Restemeyer, A. Ulbrich, H. W. Olfs, H. Westendarp, and K.-J. Appenroth, “Influence of the nitrate-N to ammonium-N ratio on relative growth rate and crude protein content in the duckweeds Lemna minor and Wolffiella hyalina,” Plants, vol. 10, no. 8, pp. 1741, 2021.
[34] L. Wang and S. A. Macko, “Constrained preferences in nitrogen uptake across plant species and environments,” Plant, Cell & Environment, vol. 34, no. 3, pp. 525–536, 2011.
[35] D. T. Britto and H. J. Kronzucker, “Ecological significance and complexity of N-source preference in plants,” Annals of Botany, vol. 112, no. 6, pp. 957–963, 2013.
[36] G. Fang, J. Yang, T. Sun, X. Wang, and Y. Li, “Evidence that synergism between potassium and nitrate enhances the alleviation of ammonium toxicity in rice seedling roots,” PLoS ONE, vol. 16, no. 9, pp. e0248796, 2021.
[37] X. Zhao, Y. Liu, D. Liu, and X. Zhang, “Potassium alleviates ammonium toxicity in rice by restricting its overaccumulation and restoring auxin distribution,” Plant and Soil, vol. 468, pp. 355–370, 2021.
[38] Y. Li, Z. Liu, J. Shi, and G. Zheng, “Phenotype Assessment and Putative Mechanisms of Ammonium Toxicity to Plants,” Plants, vol. 13, no. 3, pp. 325, 2024.
[39] P. J. White and M. R. Broadley, “Calcium in plants,” Annals of Botany, vol. 92, no. 4, pp. 487–511, 2003.
[40] H. Sioli, “The Amazon and its main affluents: Hydrography, morphology of the river courses, and river types,” in The Amazon: Limnology and Landscape Ecology of a Mighty Tropical River and its Basin, H. Sioli, Ed. Dordrecht: Dr W. Junk Publishers, 1984, pp. 127–165.
[41] K. Furch, “Water chemistry of the Amazon basin: The distribution of chemical elements among freshwaters,” in The Amazon: Limnology and Landscape Ecology of a Mighty Tropical River and its Basin, H. Sioli, Ed. Dordrecht: Dr W. Junk Publishers, 1984, pp. 167–199.
[42] A. S. N. Reddy, G. S. Ali, H. Ceber, I. S. Dember, T. Kountouzi, B. W. Poovaiah, and W. Bhalachandran, “Coping with Stresses: Roles of Calcium- and Calcium/Calmodulin-Regulated Gene Expression,” The Plant Cell, vol. 23, pp. 2010–2032, 2011.
[43] J. L. Funk, E. E. Cleland, K. N. Suding, and E. S. Zavaleta, “Restoration through reassembly: plant traits and invasion resistance,” Trends in Ecology & Evolution, vol. 23, no. 12, pp. 695–703, 2008.
[44] D. J. Futuyma and G. Moreno, “The evolution of ecological specialization,” Annual Review of Ecology and Systematics, vol. 19, pp. 207–233, 1988.
[45] U. Falkengren-Grerup, “Interspecies differences in the preference of ammonium and nitrate in vascular plants,” Oecologia, vol. 102, no. 3, pp. 305–311, 1995.
[46] G. Turnage, “Sampling Submersed Aquatic Plant Biomass: Fresh vs. Dry Weight,” GeoSystems Research Institute Report #5093, Mississippi State University, 2022.
Document information
Published on 09/03/26
Submitted on 20/02/25
Volume 8, 2026
Licence: CC BY-NC-SA license
Share this document
Keywords
claim authorship
Are you one of the authors of this document?