Summary
Background
The technical complexity of endoscopic submucosal dissection (ESD) demands adequate training to lower the surgical risks. The diameter and mucosal structure of the porcine colon is similar to the human colon making the pig a good animal for colonoscopic procedure training. However, a standardized animal bowel preparation used in colonic ESD training has not been established.
Methods
Colonoscopic procedures were performed in 12 pigs, divided into four groups. The control group (Group 1) fasted and received no preprocedure preparation. Group 2 received a single dose of sodium phosphate 2 mL/kg; Group 3 and Group 4 received split doses of sodium phosphate (2 mL/kg and 4 mL/kg, respectively). An experienced endoscopist, blinded to the preparation method, assigned a score from excellent (4 points) to poor (1 point) at five regions of the colon. The final mean bowle cleansing score was calculated from five regions in each pig. Serum biochemistry and electrolyte levels were analyzed.
Results
The different doses of sodium phosphate preparations did not change the serum glucose, creatinine, sodium, potassium, calcium, or phosphorus levels. The colonic cleansing scores in Group 1 (1.3 ± 0.4; mean ± standard deviation) and Group 2 (1.5 ± 0.2) were lower than those of Group 3 (2.6 ± 0.6) and Group 4 (3 ± 0.2).
Conclusion
The use of oral sodium phosphate is easy and safe in porcine bowel preparation for ESD training. Bowel preparation using a split dose of 2 mL/kg or 4 mL/kg sodium phosphate produces an adequate bowel cleansing. The optimal dosage still needs to be established in a large-scale study.
Keywords
Bowel preparation ; Endoscopic submucosal dissection ; Live pig model ; Sodium phosphate
Introduction
Endoscopic submucosal dissection (ESD) is a standard, widely accepted procedure for performing en bloc resection of early gastrointestinal neoplasms [1] . The technique is very operator-dependent, involving special skills and instrumentation. The learning curve from novice level to full competency should be supported systematically at every stage with adequate training programs. A panel of experts from Europe reached a consensus that hands-on experience with live pigs should be a major part of any structured training program [2] . At present, training in endoscopy or developing new endoscopic techniques relies mainly on the pig model [3] , as the porcine colon is comparable in size and structure to the human colon. In our previous report, we examined the depth of ESD specimens from the resected specimens. Histological examination of these specimens showed that the muscularis mucosa and superficial submucosa were present in the specimens; this observation demonstrated the feasibility of the ESD technique for resection [4] .
A gradually increasing number of publications have described ESD training using the porcine colon [5] ; [6] ; [7] ; [8] ; [9] . The bowel cleansing protocols currently used in pigs were initially extrapolated from protocols used in humans or dogs [10] ; [11] ; [12] . The regimens include dietary restrictions, administration of stimulant laxatives, enemas, oral polyethylene glycol lavage, and sodium phosphate. Researchers should be aware of the range of options and the advantages of various colonoscopy preparations, as well as their limitations [13] , to determine the ideal bowel preparation for experimental animals used in ESD training.
In contrast with human medicine, few researches have been performed to evaluate the safety and efficacy in bowel preparation for pigs. Polyethylene glycol (PEG) has been extensively used in humans with good efficiency; however, it requires a substantially large volume of solution to achieve the optimal result. Sodium phosphate is a low-volume, hyperosmolar laxative and has been found to be effective; it is a well-accepted bowel preparation used before a colonoscopic procedure in humans. However, few literatures discussed the use of sodium phosphate solution in experimental animals, and a standardized regimen of bowel preparation has not been established.
The primary goal of this study was to access the difference in efficacy between different doses of sodium phosphate in bowel preparation. The secondary goal was to determine the safety of sodium phosphate use through serum biochemistry and electrolyte analysis.
Methods
Preparation of pigs
All pigs, weighing between 26 kg and 28 kg, were supplied by our institutions Animal Center. The study was conducted over three sessions, with four pigs randomly used in each session. The animals just received boiled rice 2 days before study. Food was withheld, but water was allowed during the preparation for colonoscopy. In the study groups, each pig received 2 mL/kg or 4 mL/kg sodium phosphate, diluted with the same volume of water, via orogastric intubation. All procedures and animal treatments were conducted in accordance with the Guide for the Care and Use of Laboratory Animals and were approved by the Animal Care and Use Committee and The Institutional Review Board of Mackay Memorial Hospital, Taipei, Taiwan (MMH-A-S-100-34).
The radiopaque marker method to evaluate colonic transit time
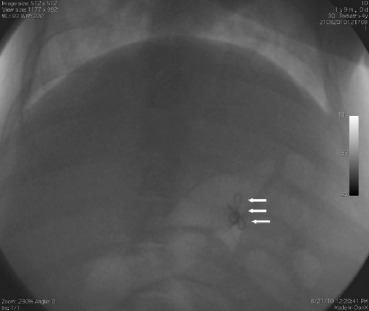
Because bowel preparations have rarely been tested in pigs, three additional pigs were used for a preliminary study to evaluate colonic transit time. The pigs were administered a capsule containing handmade radiopaque markers with food every 8 hours. After an additional 8 hours, a series of abdominal radiographs were taken every 8 hours to evaluate the average colonic transit time until a pig passed the first mark (Fig. 1 ).
|
|
|
Figure 1. The pigs were administered a capsule containing handmade radiopaque markers (arrows) every 8 hours. A series of abdominal radiographs were taken to evaluate the average colonic transit time. |
Colonic cleansing methods
An oral sodium phosphate solution (Fleet Phospho-soda; C.B. Fleet Co., Inc., Lynchburg, VA, USA) was administered in either a single or split dose of 2 mL/kg, or a split dose of 4 mL/kg. The single dose of sodium phosphate was given in the evening (6:00 PM) on the day before the examination. The split doses were divided into two portions, with half of the dose given in the evening (6:00 PM) on the day before the examination and the second dose given the next morning (8:00 AM) before the examination (12:00 noon). Each group comprised three pigs. Group 1, the control group, fasted and received no other preparation. Group 2 received a single dose of sodium phosphate of 2 mL/kg. Group 3 received a split dose of sodium phosphate of 2 mL/kg. Group 4 received a split dose of sodium phosphate of 4 mL/kg. Each portion of sodium phosphate solution was administered with an equal volume of water, and followed by a liberal feeding of water throughout the entire period before the colonoscopy. The time interval between initiation of colonic preparation and colonoscopy was approximately 16 hours.
Biochemistry and electrolyte analysis and evaluation of adequacy of colonic cleansing
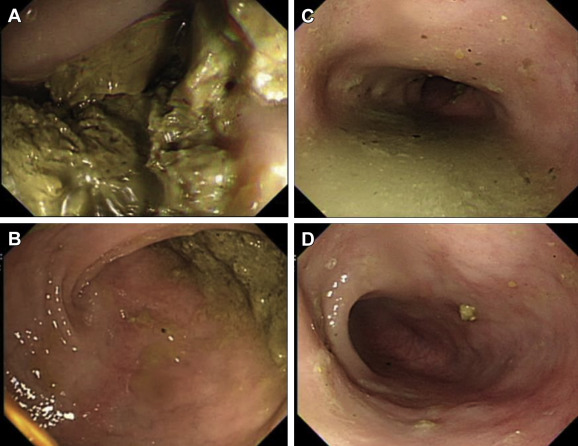
Serum glucose, sodium, potassium, chloride, calcium, phosphate, and creatinine were measured at the beginning of the colonoscopy procedure. All colonoscopic examinations were performed by a single experienced endoscopist who was unaware of the bowel preparation regimens. Examinations were conducted using an Olympus CF Q240 colonoscope (Olympus Optical Co. Ltd., Tokyo, Japan), which is for exclusive use in animals. Cleansing was scored on a 4-point scale as follows: 1 point, poor, large amounts of solid fecal matter, unable to be suctioned (Fig. 2 A); 2 points, fair, moderate amount of thick liquid to semisolid fecal matter, able to be suctioned (Fig. 2 B); 3 points, good, small amount of thin liquid fecal matter, easily suctioned (Fig. 2 C); 4 points, excellent, no fecal matter, or almost none (Fig. 2 D).
|
|
|
Figure 2. Cleansing was scored on a 4-point scale as follows: (A) 1 point, poor, large amounts of solid fecal matter, not able to be suctioned; (B) 2 points, fair, moderate amount of thick liquid to semisolid fecal matter, able to be suctioned; (C) 3 points, good, small amount of thin liquid fecal matter, easily suctioned; (D) 4 points, excellent, no fecal matter, or almost none. |
The endoscopist rated the condition of colonic preparation by using this scale at five regions: distal descending colon, mid-portion descending colon, proximal descending colon, transverse colon, and ascending colon (pigs have no sigmoid colon). The farthest region was estimated to be approximately 100 cm from the anus, which is adequate for ESD training.
Statistical analysis
Given the small number of pigs in each group, statistical analysis of the data would be of questionable validity. For the purposes of this pilot study, however, we used descriptive statistics, reporting the mean [± standard deviation (SD)] of the cleansing scores and serum biochemistry and electrolyte levels. These data were used for a rough comparison of the different sodium phosphate preparations, to assess their safety profile and efficacy of colonic cleansing.
Results
Safety and colonic cleansing scores
Anesthesia and colonoscopy were performed without complications in all pigs. No animal experienced any vomiting or hematochezia after administration of sodium phosphate. No statistically different results were observed between groups in the serum biochemistry or electrolyte measurements of glucose, creatinine, sodium, potassium, calcium, or phosphorus (Table 1 ). The colonic cleansing score in Group 1 (1.3 ± 0.4; mean ± SD) and Group 2 (1.5 ± 0.2) showed a trend toward lower scores than that observed in Group 3 (2.6 ± 0.6) and Group 4 (3 ± 0.2) (Table 1 ).
| Group a | Mean cleansing score | Glucose (mg/dL) | Creatinine (mg/dL) | Na (mEq/L) | K (mEq/L) | Cl (mEq/L) | Ca (mg/dL) | P (mg/dL) |
|---|---|---|---|---|---|---|---|---|
| Control (n = 3) | 1.3 ± 0.4 | 113.5 ± 43.1 | 0.9 ± 0.1 | 140.0 ± 0 | 4.75 ± 1.1 | 102.5 ± 2.1 | 10.1 ± 1.2 | 9.15 ± 0.4 |
| Group 2 (n = 3) | 1.5 ± 0.2 | 101.0 ± 5.7 | 1.2 ± 0 | 140.0 ± 0 | 3.65 ± 0.2 | 103.0 ± 2.8 | 9.0 ± 0.5 | 10.3 ± 0.9 |
| Group 3 (n = 3) | 2.6 ± 0.6 | 102.2 ± 9.6 | 1.1 ± 0.2 | 142.2 ± 4.3 | 3.48 ± 0.3 | 103.4 ± 1.9 | 10.4 ± 2.3 | 10.1 ± 2.0 |
| Group 4 (n = 3) | 3.0 ± 0.2 | 128.0 ± 12.3 | 1.1 ± 0.2 | 139.3 ± 2.1 | 3.93 ± 0.5 | 102.7 ± 2.1 | 10.2 ± 1.0 | 10.3 ± 0.7 |
Data are expressed as mean ± standard deviation.
a. Group 1: control without preparation; Group 2: oral 2 mL/kg single dose of sodium phosphate; Group 3: oral 2 mL/kg split doses of sodium phosphate; Group 4: oral 4 mL/kg split doses of sodium phosphate.
Discussion
The benefits of en bloc resection with ESD for colon neoplasms, including less invasiveness and potential cure, are increasingly being recognized. However, the colon is a technically complex location for ESD because the thinner wall, luminal angulations, and small caliber of the colon reduce maneuverability. For this reason, animal models are invaluable as training resources to improve performance and reduce risk of complications, and to evaluate when the learning curve has been completed and a trainee is ready to perform ESD in patients. Successful ESD training relies on adequate bowel preparation in pigs to improve the model. We conducted this prospective, blinded trial to evaluate the safety profile and efficacy of three different sodium phosphate doses for bowel preparation. We aimed to establish a standardized bowel preparation protocol and improve the model of colonic ESD in pigs.
Bowel preparation is relatively difficult in animals and the commonly used protocols for human bowel preparation have some drawbacks in animals. Enema administration is time consuming and may be uncomfortable for the animals [14] . Large-volume PEG is widely used and has proven efficacy in humans. When orogastric intubation is used to administer PEG, the large amount of solution takes several minutes to infuse, leading to discomfort; furthermore, rapid, vigorous gastric distention can also result in vomiting or regurgitation [15] ; [16] . Sodium phosphate is a buffered saline laxative that has been extensively used in humans with equal efficiency to PEG, but it requires a substantially smaller solution volume. Many human studies have tried to reduce the amount of fluid intake required for bowel preparations and allow for minimal dietary restrictions by splitting the dose [17] . This also can decrease the stress and discomfort of animals by reducing the duration of orogastric intubation and avoiding rapid gastric distention for prevention of aspiration. We selected sodium phosphate as a bowel preparation agent to simplify the bowel preparation procedure. As a result, in our study, we reported no vomiting or hematochezia in animals after administration of sodium phosphate.
Although similar, some differences exist in the precise structure of the pig colon compared with the human colon. The pig colon is much thinner than the human colon, and it has more fat tissue and blood vessels. Nevertheless, the pig colon is still the model of choice for colonoscopy training and colonic ESD training. The live pig model simulates a more realistic endoscopy setting and provides the opportunity to deal with bleeding and perforations.
Many factors affect the quality of bowel preparation, including dietary restriction, cleansing agent used, dosing of the purgative (e.g., single dose or split dose), and the time interval between bowel preparation and the start of colonoscopy. Dietary restriction of fiber or residue is advised in human bowel preparation protocols, and a low-residue diet for 2 days before colonoscopy improves bowel cleansing [18] . Therefore, our animals were fed a low-residue diet of boiled rice from 2 days before the procedure.
Because bowel preparations have never been tested in pigs, three additional animals were used for a preliminary pilot study to evaluate porcine colonic transit time. A capsule containing handmade radiopaque markers was given to these pigs; the average colonic transit time determined from this preliminary study was approximately 16 hours. Residual material in the small bowel may pass into the large bowel if colonoscopy is scheduled more than 8–12 hours after the administration of a single-dose lavage solution; a second dose of medication resolves this issue [19] . Based on the hypothesis that a split dose might be preferable, we designed a split dose of sodium phosphate to compare it with the same amount of medication given as a single dose. In our study, the mean colonic cleansing score was higher in Group 3 and Group 4 using split doses of sodium phosphate (2 mL/kg and 4 mL/kg, respectively) than in Group 2 using a single dose.
When using oral sodium phosphate solutions, hyperphosphatemia with hypocalcemia or hypokalemia occurred in some human case series [20] ; [21] . In our study, the different doses of sodium phosphate preparations did not change the serum levels of glucose, creatinine, sodium, potassium, calcium, or phosphorus. Even with the relatively high sodium phosphate dose of 4 mL/kg (Group 4), no significant difference in laboratory values was observed. Anesthesia and colonoscopy were performed without complications in all pigs. The mean colonic cleansing scores were (2.6 ± 0.6) in Group 3 and (3 ± 0.2) in Group 4, which is an adequate cleansing level; small amounts of liquid fecal matter can easily be suctioned out for ESD training. The associated cost involving the pig, veterinarian, and other manpower is an important issue, and may be a limiting factor for some training programs that utilize live animals. The animals and animal facility charges, including manpower, were approximately $300 USD (2 trainees could share an animal). Therefore, we believe that this live porcine simulator should be used only for achieving the competent level to gain the experience of clinical practice.
In a prospective randomized trial, we compared three doses of sodium phosphate for bowel preparations in a pig model. We demonstrated that oral administration of sodium phosphate solution is efficacious and safe in pigs when the dosing regimen is adjusted for weight. Bowel preparation with a split dose of 2 mL/kg or 4 mL/kg of sodium phosphate results in adequate bowel cleansing for ESD training. The optimal dose still needs to be established in a large-scale study. Whether this approach in pig bowel preparations will achieve improved training outcomes with a lower risk of complications in humans, shortened procedure times, and higher en bloc resection rates during ESD is yet to be determined. In the future, we need well-designed large clinical trials to evaluate these points.
Conflicts of interest
All authors declare no conflicts of interest.
Acknowledgments
The authors wish to thank their colleagues at the Animal Center of Mackay Memorial Hospital for preparing the pigs and providing technical assistance.
References
- [1] T. Gotoda, H. Yamamoto, R.M. Soetikno; Endoscopic submucosal dissection of early gastric cancer; J Gastroenterol, 41 (2006), pp. 929–942
- [2] P.H. Deprez, J.J. Bergman, S. Meisner, T. Ponchon, A. Repici, M. Dinis-Ribeiro, et al.; Current practice with endoscopic submucosal dissection in Europe: position statement from a panel of experts; Endoscopy, 42 (2010), pp. 853–858
- [3] A. Parra-Blanco, D. Nicolas, M.R. Arnau, A.Z. Gimeno-Garcia, L. Rodrigo, E. Quintero; Gastric endoscopic submucosal dissection assisted by a new traction method: the clip-band technique. A feasibility study in a porcine model (with video); Gastrointest Endosc, 74 (2011), pp. 1137–1141
- [4] T.E. Wang, H.Y. Wang, C.C. Lin, T.Y. Chen, C.W. Chang, C.J. Chen, et al.; Simulating a target lesion for endoscopic submucosal dissection training in an ex vivo pig model ; Gastrointest Endosc, 74 (2011), pp. 398–402
- [5] M. Yamasaki, K. Kume, I. Yoshikawa, M. Otsuli; A novel method of endoscopic submucosal dissection with blunt abrasion by submucosal injection of sodium carboxymethylcellulose: an animal preliminary study; Gastrointest Endosc, 64 (2006), pp. 958–965
- [6] N. Yahagi, H. Neuhaus, B. Schumacher, A. Neuqebauer, G.F. Kaehler, M. Schenk, et al.; Comparison of standard endoscopic submucosal dissection (ESD) versus an optimized ESD technique for the colon: an animal study; Endoscopy, 41 (2009), pp. 340–345
- [7] G. Fernańdez-Esparrach, E.L. Matthes, D. Maurice, M. Enderle, C.C. Thompson, D.L. Carr-Locke; A novel device for endoscopic submucosal dissection that combines water-jet submucosal hydrodissection and elevation with electrocautery: initial experience in a porcine model; Gastrointest Endosc, 71 (2010), pp. 615–618
- [8] A. Moss, M.J. Bourke, V. Kwan, K. Tran, C. Godfrey, G. McKay, et al.; Succinylated gelatin substantially increases en bloc resection size in colonic EMR: a randomized, blinded trial in a porcine model ; Gastrointest Endosc, 71 (2010), pp. 589–595
- [9] A. Moss, M.J. Bourke, A.J. Metz, D. McLeod, K. Tran, C. Godfrey, et al.; Beyond the snare: technically accessible large en bloc colonic resection in the West: an animal study; Dig Endosc, 24 (2012), pp. 21–29
- [10] D.I. Park, S.H. Park, S.K. Lee, Y.H. Baek, D.S. Han, C.S. Eun, et al.; Efficacy of prepackaged, low residual test meals with 4L polyethylene glycol versus a clear liquid diet with 4L polyethylene glycol bowel preparation: a randomized trial; J Gastroenterol Hepatol, 24 (2009), pp. 988–991
- [11] A. Amato, F. Radaelli, S. Paggi, V. Terruzzi; Half doses of PEG-ES and senna vs. high-dose senna for bowel cleansing before colonoscopy: a randomized, investigator-blinded trial; Am J Gastroenterol, 105 (2010), pp. 675–681
- [12] S. Tongprasert, A. Sobhonslidsuk, S. Rattanasiri; Improving quality of colonoscopy by adding simethicone to sodium phosphate bowel preparation; World J Gastroenterol, 15 (2009), pp. 3032–3037
- [13] K.E. Occhipinti, J.A. Di Palma; How to choose the best preparation for colonoscopy; Nat Rev Gastroenterol Hepatol, 6 (2009), pp. 279–286
- [14] K.P. Richter, M.B. Cleveland; Comparison of an orally administered gastrointestinal lavage solution with traditional enema administration as preparation for colonoscopy in dogs; J Am Vet Med Assoc, 195 (1989), pp. 1727–1731
- [15] C.F. Burrows; Evaluation of a colonic lavage solution to prepare the colon of the dog for colonoscopy; J Am Vet Med Assoc, 195 (1989), pp. 1719–1721
- [16] M.A. Daugherty, M.S. Leib, J.H. Rossmeisl, F.S. Almy, D.L. Ward; Safety and efficacy of oral low-volume sodium phosphate bowel preparation for colonoscopy in dogs; J Vet Intern Med, 22 (2008), pp. 31–36
- [17] A.A. Siddiqui, K. Yang, S.J. Spechler, B. Cryer, R. Davila, D. Cipher, et al.; Duration of the interval between the completion of bowel preparation and the start of colonoscopy predicts bowel-preparation quality; Gastrointest Endosc, 69 (2009), pp. 700–706
- [18] E. Aoun, H. Abdul-Baki, C. Azar, F. Mourad, K. Barada, Z. Berro, et al.; A randomized single blind trial of split-dose PEG-electrolyte solution without dietary restriction compared with whole dose PEG-electrolyte solution with dietary restriction for colonoscopy preparation; Gastrointest Endosc, 62 (2005), pp. 213–218
- [19] H.M. Xu, J.G. Han, Y. Na, B. Zhao, H.C. Ma, Z.J. Wang; Colonic transit time in patient with slow-transit constipation: comparison of radiopaque markers and barium suspension method; Eur J Radiol, 79 (2011), pp. 211–213
- [20] M. Sabri, C. Di Lorenzo, W. Henderson, W. Thompson, E. Barksdale Jr., S. Khan; Colon cleansing with oral sodium phosphate in adolescents: dose, efficacy, acceptability, and safety; Am J Gastroenterol, 103 (2008), pp. 1533–1539 quiz 1540
- [21] E.H. Seo, T.O. Kim, T.G. Kim, H.R. Joo, M.J. Park, J. Park, et al.; Efficacy and tolerability of split-dose PEG compared with split-dose aqueous sodium phosphate for outpatient colonoscopy: a randomized, controlled trial; Dig Dis Sci, 56 (2011), pp. 2963–2971
Document information
Published on 15/05/17
Submitted on 15/05/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?