Abstract
Aim
The aim of this study was to evaluate the relationship between coronary artery calcification (CAC) assessed by multi-detector computed tomography (MDCT) and myocardial perfusion assessed by cardiac magnetic resonance imaging (CMR) in a group of symptomatic patients.
Method
Retrospective analysis of 120 patients (age 65.1 ± 8.9 years, 88 males) who presented with atypical chest pain to Bethanien Hospital, Frankfurt, Germany, between 2007 and 2010 and who underwent CAC scoring using MDCT, CMR, and conventional coronary angiography. Patients were divided into those with high-grade (HG) stenosis (n = 67, age 65.1 ± 9.4 years) and those with no-HG stenosis (n = 53, age 65.1 ± 8.6 years).
Results
There were more males with HG stenosis (82.1% vs. 62.3%, p = 0.015), in whom the percentage and number of abnormal perfusion segments were higher at rest (37.3% vs. 17%, p = 0.014) but not different with stress (p = 0.83) from those with no-HG stenosis. Thirty-four patients had myocardial perfusion abnormalities at rest and 26 patients developed perfusion defects with stress. Stress-induced myocardial perfusion defects were 22.4% sensitive and 79.2% specific for detecting HG stenosis. The CAC score was lower in patients with no-HG stenosis compared to those with HG stenosis (p < 0.0001). On the ROC curve, a CAC score of 293 had a sensitivity of 71.6% and specificity of 83% in predicting HG stenosis [(AUC 0.80 (p < 0.0001)]. A CAC score of 293 or the presence of at least 1 segment myocardial perfusion abnormality was 74.6% sensitive and 71.7% specific in detecting HG stenosis, the respective values for the 2 abnormalities combined being 19.4% and 90.6%. The severity of CAC correlated with the extent of myocardial perfusion in the patient group as a whole with stress (r = 0.22, p = 0.015), particularly in those with no-HG stenosis (r = 0.31, p = 0.022). A CAC score of 293 was 31.6% sensitive and 87.3% specific in detecting myocardial perfusion abnormalities.
Conclusion
In a group of patients with exertional angina, coronary calcification is more accurate in detecting high-grade luminal stenosis than myocardial perfusion defects. In addition, in patients with no stenosis, the incremental relationship between coronary calcium score and the extent of myocardial perfusion suggests coronary wall hardening as an additional mechanism for stress-induced angina other than luminal narrowing. These preliminary findings might have a clinical impact on management strategies of these patients other than conventional therapy.
Keywords
Coronary calcification;CMR;Coronary CT;Exertional angina and myocardial perfusion
1. Introduction
Cardiac magnetic resonance imaging (CMR) myocardial perfusion has high diagnostic accuracy for coronary artery disease (CAD), even superior to single-photon emission computed tomography [1]; however, it is known for its limitations. Coronary artery calcium (CAC) score assessed by multi-detector computed tomography (MDCT) has also been shown to have high specificity in excluding obstructive CAD [2]. The diagnosis of CAD by the two techniques is based on different concepts; while CMR assesses myocardial perfusion as a consequence of coronary disease, MDCT analyzes the arterial disease morphology and allows for quantification of coronary wall calcification. In addition, MDCT non-invasive coronary angiography has shown higher accuracy than CMR in determining coronary stenosis [3].
Coronary calcification itself generally reflects atherosclerosis and its extent correlates with the overall plaque burden, in the form of luminal stenosis [4]. However, many symptomatic patients might present with coronary calcification in the absence of significant luminal stenosis, suggesting that arterial wall hardening could be associated with ischemic and compromised myocardial blood supply as a cause of symptoms. The aim of this study was to evaluate the potential relationship between CAC assessed by MDCT and myocardial perfusion assessed by CMR in a group of symptomatic patients, irrespective of the presence of luminal stenosis.
2. Methods
This is a retrospective analysis of 120 patients (mean age 65.1 ± 8.9 years, 88 males) who presented with atypical chest pain, defined as inconsistent exertional chest discomfort, to Bethanien Hospital, Frankfurt, Germany, between 2007 and 2010 and who underwent CAC scoring using MDCT and myocardial perfusion scanning using CMR. All patients subsequently underwent conventional coronary angiography, which was performed not more than 1 month after the MDCT and CMR perfusion scans. None of the patients had acute coronary syndrome, heart failure, valvular heart disease, thyroid and parathyroid diseases, inflammatory disease, or chronic kidney disease (creatinine > 130 mmol/L). Significant obstructive coronary disease was considered present when there was clear evidence for at least one high-grade (HG) stenosis with ≥ 50% lumen narrowing on the conventional angiogram.
According to the coronary angiography results, patients were divided into two groups: HG stenosis group (n = 67, mean age 65.1 ± 9.4 years) and no-HG stenosis group (n = 53, mean age 65.1 ± 8.6 years). Being a retrospective comparison of imaging methods which had been ordered due to clinical indications by the cardiologists responsible for the patients' management. Therefore, an ethical vote did not appear to be necessary, according to the hospital policy.
2.1. CMR perfusion scan
CMR studies were performed using a 1.5-Tesla MRI system (Magnetom Sonata Maestro Class, Siemens AG, Erlangen, Germany), with the patient in the supine position, and additional ECG electrodes connected with external system (Magnitude 3150, InVivo Research Inc., Orlando, FL, USA) for continuous heart rate monitoring [5]. Blood pressure was also monitored. Both a six-channel body phased-array coil and a two-channel spine phased-array coil were used. Sequences acquired during breath-hold were performed during quiet expiration.
After localizers and anatomical images, perfusion imaging was performed. Typically, 3 short-axis slices, each with 10 mm slice thickness, were acquired at the basal, mid papillary, and apical levels of the left ventricle. Patients were stressed using conventional adenosine protocol. Adenosine stimulates A2 receptors in the microvasculature, leading to relaxation of the arterioles. In normal myocardium, this leads to increased perfusion without changes in blood volume [6]. With coronary stenosis, the magnitude of the increased perfusion during vasodilation is compromised [6]. The pressure drop results in capillary closure, reduced perfusion, and reduced blood volume, which is demonstrated as slower arrival and lower contrast agent concentration in the ischemic segment [6].
A single shot prospectively gated balanced Turbo Field Echo (TFE) sequence with a typical in-plane resolution of 2.5 × 2.5 mm was used. Patients were then allowed to rest until the hemodynamic effects of the adenosine had subsided (typically 5 min). The location and distribution of myocardial perfusion defects in the left ventricle were described using the American Heart Association 16-segment model [7].
For the stress study, intravenous adenosine was started 3 min before contrast injection. Twenty short-axis images were taken at every level of myocardium before, during, and after contrast injection. Myocardial perfusion was measured during adenosine infusion using high dose of Gadolinium-DTPA (0.06 mmol/kg). Adenosine was injected at a rate of 0.14 mg/kg/min, for 3–6 min for a total dose of 0.48–0.84 mg/kg. To avoid risk of large bolus drug, adenosine and contrast were administered through separate IVs [8].
Acquired images were subsequently transferred to a dedicated computer for analyzing changes in the myocardial signal intensity [9]. Two experienced observers, blinded to the MDCT results, decided by visual assessment on the myocardial perfusion and the blood supply of the 6 conventionally studied segments.
Rest and adenosine stress scans were magnified and displayed at the same time for visual assessment [10]. In normal scans, the first pass into the myocardium changed its colour uniformly from black to gray. A slowly changing colour to gray suggested impaired perfusion and hence was considered as a perfusion defect either at rest or induced, if it occurred at peak stress. The CMR system employed quantitative parametric tissue analysis [5].
2.2. Coronary artery calcium (CAC) score
CAC was measured using 64 MDCT (Somatom Sensation Cardiac 64; Siemens Medical Solutions, Forchheim, Germany) with a gantry rotation time of 330 ms (collimation 64 × 0.6 mm, reconstruction increment 0.3 mm). Images were acquired with the patient in quiet expiratory pause. Oral beta-blockers (bisoprolol 5 mg or metoprolol 50 mg) were given 1 h before the scan if the resting heart rate was > 60 beats/min. Calcification was described as the presence of > 2 contiguous pixels with > 130 Hounsfield Units. The workstation software automatically detected calcified areas and marked it in colour. The individual lesion scores were automatically summed to calculate the total Agatston score for each of the epicardial coronary artery territories as well as for the total coronary tree [4].
2.3. Coronary angiography
The Judkins technique was used with at least four views of the left system and two views of the right system. Angiography was performed within 1 month after the CT scan in all patients. Analysis of the coronary angiograms was performed by an independent experienced observer. Significant stenosis was defined as ≥ 50% lumen narrowing of any epicardial coronary artery.
2.4. Statistical analysis
A standard statistical software package (SPSS 20, IBM, Armonk, NY, USA) was used for the statistical analyses. Categorical variables were expressed as absolute number and percentage (%). Normally distributed continuous data were expressed as mean ± standard deviation. The comparison between the HG stenosis and the no-HG stenosis groups was analyzed using chi-squared test. Spearman rank correlation was used to define the correlation between different CAC levels and myocardial perfusion on CMR. The null hypothesis was rejected on p values < 0.05.
3. Results
Coronary risk factor distribution in the total study population and subgroups are listed in Table 1. The cardiovascular risk factors did not differ between the two groups, except for a higher proportion of males in the HG lesions group (p = 0.015).
| Risk factors | Total n = 120 | HG stenosis n = 67 | no-HG stenosis n = 53 | p value |
|---|---|---|---|---|
| Males, n(%) Age group (over 60 y) n(%) | 88 (73.3) 86 (71) | 55 (82.1) 49 (73.1) | 33 (62.3) 37 (69.8) | 0.015 0.421 |
| Hypertension, n(%) | 45 (37.5) | 24 (35.8) | 21 (39.6) | 0.669 |
| Smoking, n(%) | 18 (15.0) | 11 (16.4) | 7 (13.2) | 0.625 |
| Diabetes, n(%) | 14(11.7) | 8(11.9) | 6(11.3) | 0.916 |
| Obesity, n(%) | 3(2.5) | 1(1.5) | 2(3.8) | 0.427 |
| Family history of CVD, n(%) Prior MI, n(%) | 20(16.7) 34 (28.3) | 11(16.4) 25 (37.3) | 9(17.0) 9 (17) | 0.934 0.014 |
3.1.1. CMR perfusion between HG stenosis and no-HG stenosis (Table 2 and Fig. 1)
The percentage and number of perfusion segments were significantly higher in patients with HG stenosis with more than 1 segment perfusion defect at rest (p = 0.014), but there was no difference with stress (p = 0.83). Thirty-four patients had myocardial perfusion abnormalities at rest, and 26 patients developed perfusion defects with stress. Stress-induced myocardial perfusion defects were 22.4% sensitive and 79.2% specific for detecting HG coronary stenosis.
| CMR perfusion defect (n, %) | |||
|---|---|---|---|
| 0 segment | > 1 segment | p value | |
| At rest | |||
| HG stenosis | 42 (62.7) | 25 (37.3) | 0.014 |
| no-HG stenosis | 44 (83.0) | 9 (17) | |
| At stress | |||
| HG stenosis | 52 (77.6) | 15 (22.4) | 0.83 |
| no-HG stenosis | 42 (79.2) | 11 (20.8) | |
|
|
|
Fig. 1. The difference in myocardial perfusion between the two groups at rest and with stress |
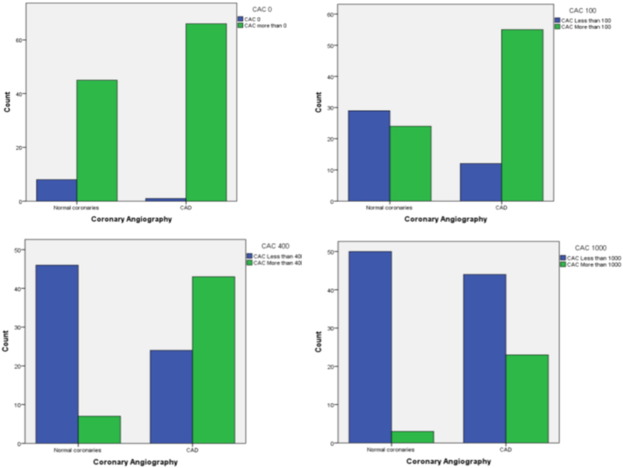
3.1.2. CAC score between HG stenosis and no-HG stenosis (Table 3 and Fig. 2)
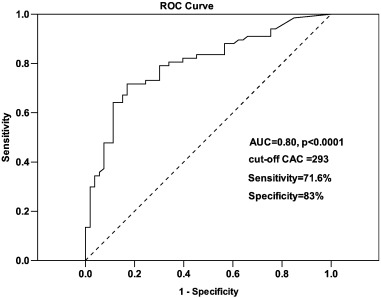
The patient number and percentage were significantly different between the two groups with a lower CAC score in patients with no-HG stenosis and a higher CAC score in those with HG stenosis (p < 0.0001). On the ROC curve, the CAC cut-off value of 293 had a sensitivity of 71.6% and specificity of 83% in predicting HG coronary stenosis, giving an area under the curve of 0.80 (p < 0.0001) (Fig. 3).
| CAC (n, %) | |||||
|---|---|---|---|---|---|
| 0–99 | 100–399 | 400–999 | ≥ 1000 | p | |
| HG stenosis | 12 (17.9) | 12 (17.9) | 20 (29.9) | 23 (34.3) | < 0.0001 |
| No-HG stenosis | 29 (54.7) | 17 (32.1) | 4 (7.5) | 3 (5.7) | |
|
|
|
Fig. 2. The difference in CAC between the two groups |
|
|
|
Fig. 3. the ROC curve for calcium score to predict the coronary artery stenosis, AUC = 0.80 (95% CI 0.82–0.88, p < 0.0001), cut-off value is CAC = 293 with sensitivity 71.6% and specificity 83%. |
3.1.3. CAC or/and myocardial perfusion in predicting HG coronary stenosis
A CAC score of 293 or the presence of at least 1 segment showing myocardial perfusion was 74.6% sensitive and 71.7% specific in detecting HG coronary stenosis. The respective values for the two abnormalities combined were 19.4% sensitivity and 90.6% specificity.
3.1.4. CAC score versus CMR myocardial perfusion (Table 4)
As shown in Table 4, the severity of CAC correlated with the extent of myocardial perfusion in the patient group as a whole with stress (r = 0.22, p = 0.015), particularly in patients with no-HG stenosis (r = 0.31, p = 0.022). A CAC score cut-off value of 293 was 31.6% sensitive and 87.3% specific in detecting myocardial perfusion abnormalities.
| Rest | Stress | |||
|---|---|---|---|---|
| All | r = 0.066 | p = 0.476 | r = 0.221 | p = 0.015 |
| HG stenosis | r = 0.049 | p = 0.696 | r = 0.189 | p = 0.125 |
| No-HG stenosis | r = 0.149 | p = 0.288 | r = 0.314 | p = 0.022 |
4. Discussion
CMR myocardial perfusion has been shown to have high accuracy in detecting coronary artery disease and related events [10]; [11]; [12]; [13]; [14]; [15]; [16]; [17]; [18] ; [19]. This has been superseded by the greater accuracy of CTCA in excluding significant CAD [3]. The exact accuracy of the two techniques in identifying significant CAD (> 50%) coronary stenosis in daily practice remains controversial [20]. Although CMR perfusion is considered a factual reflection of myocardial blood supply, as an accurate functional test, CTCA demonstrates the anatomical phenotypic manifestation of the disease and its implications on the coronary circulation. However, the main limitation of CTCA accuracy is in patients with severe calcification because of its masking of the true plaque size and relative narrowing of the lumen [3] ; [21]. In fact, current guidelines recommend conventional angiography in patients with more than intermediate degree of coronary calcification and a calcium score > 200. Despite that, a subgroup of patients with either severe calcification but no significant stenosis or with impaired myocardial perfusion but no significant stenosis remains, representing a clinical dilemma. There is currently no study that has shown an ideal way of describing these patients or proposed a strategy for managing them. The purpose of this study was to assess the relationship between CAC and CMR myocardial perfusion in patients with insignificant coronary stenosis.
4.1.1. Findings
Our study results concur with some of the above findings in showing only a modest relationship between the presence of significant coronary stenosis and myocardial perfusion abnormalities by CMR. On the other hand, the CAC score was much more sensitive and specific in detecting HG stenosis, giving an area under the ROC curve of 80%. In addition, our findings highlight the relationship between CAC and stress myocardial perfusion defects, with an incremental increase in the number of myocardial segments showing perfusion defects, with stress, parallel to the progressive increase in CAC score, only in patients with no-HG stenosis. This suggests potential development of myocardial ischemia and symptoms as a result of the arterial wall hardening rather than luminal narrowing by a stenosis, suggesting that the CAC score might be reflecting the extent of plaque burden, irrespective of luminal narrowing [22]. We believe that we are the first to demonstrate that extensive CAC correlates with the diffuse pattern of myocardial perfusion defect in the absence of significant coronary stenosis, again suggesting a potential causative relationship. Pellika et al has shown a relationship between CAC and left ventricular wall motion abnormalities using stress echo but made no comment on the extent of obstructive lesions [23]. We too have recently shown parallel subendocardial abnormalities at peak stress in symptomatic patients with no coronary stenosis, particularly in those with significant calcification [24]. This evidence suggests that CAC, particularly with high scores, is likely to compromise coronary blood flow reserve and hence myocardial perfusion at the time of increased demand (peak stress).
4.1.2. Clinical implications
The coronary calcium score remains more accurate in detecting HG luminal stenosis over and above myocardial perfusion defects by CMR. Absolute reliance on luminal narrowing by either CTCA or conventional coronary angiography is likely to miss an important group of patients with limiting angina who do not demonstrate evidence for HG stenosis but suffer from wall hardening which compromises myocardial perfusion. This finding suggests an important role for the routine measurement of the CAC score in angina patients, particularly those with unexplained symptoms by conventional angiography.
4.1.3. Limitations
There was a gender difference between patients with HG stenosis and those without, but this does not seem to have influenced our results, since the relationship between CAC and CMR myocardial perfusion defects was shown in those with no-HG stenosis, negating the potential imbalance of males, who generally have higher incidence of CAC [25]. Assessment of CMR perfusion was semi-quantitative but followed the international recommendations [26]. This study was retrospective in its design therefore subject to potential bias in patient selection. A significantly larger sample volume would have strengthened the relevance of our findings, particularly with the subgroup of patients with extensive calcification who showed clear evidence for myocardial perfusion abnormalities. We relied in our data interpretation on the accuracy of the CAC measurements as previously reported by Achenbach et al [27], who showed non-significant results in the variability of repeating CAC measuring by EBCT as well as the known low variability of the system we used (64-MSCT) [28]. None of the patients we studied was clinically felt to need an FFR assessment, this information is not available. Finally, we did not assess MRI reproducibility, having considered the long experience of the radiologist reported and the lack of potential competitor.
4.1.4. Conclusion
In a group of patients with exertional limiting angina, coronary calcification is more accurate in detecting high-grade luminal stenosis than myocardial perfusion defects. In addition, in patients with no stenosis, the incremental relationship between coronary calcium score and the extent of myocardial perfusion suggests coronary wall hardening as an additional mechanism for stress-induced angina other than luminal narrowing. These preliminary findings might have a clinical impact on management strategies of these patients other than conventional therapy.
Conflict of interest
None of the authors have any financial or conflict of interest to disclose.
References
- [1] J.P. Greenwood, N. Maredia, J.F. Younger, J.M. Brown, J. Nixon, C.C. Everett, et al.; Cardiovascular magnetic resonance and single-photon emission computed tomography for diagnosis of coronary heart disease (CE-MARC): a prospective trial; Lancet, 379 (9814) (2012 Feb 4), pp. 453–460
- [2] K. Nieman, T.W. Galema, L.A. Neefjes, A.C. Weustink, P. Musters, A.D. Moelker, et al.; Comparison of the value of coronary calcium detection to computed tomographic angiography and exercise testing in patients with chest pain; Am. J. Cardiol., 104 (11) (2009 Dec 1), pp. 1499–1504
- [3] J.D. Schuijf, J.J. Bax, L.J. Shaw, A. de Roos, H.J. Lamb, E.E. van der Wall, et al.; Meta-analysis of comparative diagnostic performance of magnetic resonance imaging and multislice computed tomography for noninvasive coronary angiography; Am. Heart J., 151 (2) (2006 Feb), pp. 404–411
- [4] R.J. Rennenberg, A.G. Kessels, L.J. Schurgers, J.M. van Engelshoven, P.W. de Leeuw, A.A. Kroon; Vascular calcifications as a marker of increased cardiovascular risk: a meta-analysis; Vasc. Health Risk Manag., 5 (1) (2009), pp. 185–197
- [5] N. Al-Saadi, E. Nagel, M. Gross, A. Bornstedt, B. Schnackenburg, C. Klein, et al.; Noninvasive detection of myocardial ischemia from perfusion reserve based on cardiovascular magnetic resonance; Circulation, 101 (12) (2000 Mar 28), pp. 1379–1383
- [6] A.R. Jayaweera, K. Wei, M. Coggins, J.P. Bin, C. Goodman, S. Kaul; Role of capillaries in determining CBF reserve: new insights using myocardial contrast echocardiography; Am. J. Phys., 277 (6 Pt 2) (1999 Dec), pp. H2363–H2372
- [7] M.D. Cerqueira, N.J. Weissman, V. Dilsizian, A.K. Jacobs, S. Kaul, W.K. Laskey, et al.; Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart. A statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association; Int. J. Card. Imaging, 18 (1) (2002 Feb), pp. 539–542
- [8] B.L. Gerber, S.V. Raman, K. Nayak, F.H. Epstein, P. Ferreira, L. Axel, et al.; Myocardial first-pass perfusion cardiovascular magnetic resonance: history, theory, and current state of the art; J. Cardiovasc. Magn. Reson., 10 (2008 Apr 28) (18-429X-10-18)
- [9] J.H. Cullen, M.A. Horsfield, C.R. Reek, G.R. Cherryman, D.B. Barnett, N.J. Samani; A myocardial perfusion reserve index in humans using first-pass contrast-enhanced magnetic resonance imaging; J. Am. Coll. Cardiol., 33 (5) (1999 Apr), pp. 1386–1394
- [10] M. Hamon, G. Fau, G. Nee, J. Ehtisham, R. Morello, M. Hamon; Meta-analysis of the diagnostic performance of stress perfusion cardiovascular magnetic resonance for detection of coronary artery disease; J. Cardiovasc. Magn. Reson., 12 (2010 May 19) (29-429X-12-29)
- [11] K.R. Nandalur, B.A. Dwamena, A.F. Choudhri, M.R. Nandalur, R.C. Carlos; Diagnostic performance of stress cardiac magnetic resonance imaging in the detection of coronary artery disease: a meta-analysis; J. Am. Coll. Cardiol., 50 (14) (2007 Oct 2), pp. 1343–1353
- [12] P. Gargiulo, S. Dellegrottaglie, D. Bruzzese, G. Savarese, O. Scala, D. Ruggiero, et al.; The prognostic value of normal stress cardiac magnetic resonance in patients with known or suspected coronary artery disease: a meta-analysis; Circ. Cardiovasc. Imaging, 6 (4) (2013 Jul), pp. 574–582
- [13] E. Nagel, H.B. Lehmkuhl, W. Bocksch, C. Klein, U. Vogel, E. Frantz, et al.; Noninvasive diagnosis of ischemia-induced wall motion abnormalities with the use of high-dose dobutamine stress MRI: comparison with dobutamine stress echocardiography; Circulation, 99 (6) (1999 Feb 16), pp. 763–770
- [14] S.N. Falcao, C.E. Rochitte, W.M. Junior, L. Quaglia, P.A. Lemos, J.C. Sbano, et al.; Incremental value of perfusion over wall-motion abnormalities with the use of dobutamine-atropine stress myocardial contrast echocardiography and magnetic resonance imaging for detecting coronary artery disease; Echocardiography, 30 (1) (2013 Jan), pp. 45–54
- [15] R. Sicari, A. Pingitore, G. Aquaro, E.G. Pasanisi, M. Lombardi, E. Picano; Cardiac functional stress imaging: a sequential approach with stress echo and cardiovascular magnetic resonance; Cardiovasc. Ultrasound, 5 (47) (2007 Dec 4)
- [16] G. Hartlage, M. Janik, A. Anadiotis, E. Veledar, J. Oshinski, D. Kremastinos, et al.; Prognostic value of adenosine stress cardiovascular magnetic resonance and dobutamine stress echocardiography in patients with low-risk chest pain; Int. J. Card. Imaging, 28 (4) (2012 Apr), pp. 803–812
- [17] N. Takahashi, T. Inoue, T. Oka, A. Suzuki, T. Kawano, K. Uchino, et al.; Diagnostic use of T2-weighted inversion-recovery magnetic resonance imaging in acute coronary syndromes compared with 99mTc-Pyrophosphate, 123I-BMIPP and 201TlCl single photon emission computed tomography; Circ. J., 68 (11) (2004 Nov), pp. 1023–1029
- [18] J. Schwitter, C.M. Wacker, N. Wilke, N. Al-Saadi, E. Sauer, K. Huettle, et al.; Superior diagnostic performance of perfusion-cardiovascular magnetic resonance versus SPECT to detect coronary artery disease: the secondary endpoints of the multicenter multivendor MR-IMPACT II (Magnetic Resonance Imaging for Myocardial Perfusion Assessment in Coronary Artery Disease Trial); J. Cardiovasc. Magn. Reson., 14 (61) (2012 Sep 2)
- [19] J.T. Keijer, A.C. van Rossum, M.J. van Eenige, J.J. Bax, F.C. Visser, J.J. Teule, et al.; Magnetic resonance imaging of regional myocardial perfusion in patients with single-vessel coronary artery disease: quantitative comparison with (201)Thallium-SPECT and coronary angiography; J. Magn. Reson. Imaging, 11 (6) (2000 Jun), pp. 607–615
- [20] G. Pundziute, J.D. Schuijf, J.W. Jukema, H.J. Lamb, A. de Roos, E.E. van der Wall, et al.; Impact of coronary calcium score on diagnostic accuracy of multislice computed tomography coronary angiography for detection of coronary artery disease; J. Nucl. Cardiol., 14 (1) (2007 Jan), pp. 36–43
- [21] J. Kefer, E. Coche, G. Legros, A. Pasquet, C. Grandin, B.E. Van Beers, et al.; Head-to-head comparison of three-dimensional navigator-gated magnetic resonance imaging and 16-slice computed tomography to detect coronary artery stenosis in patients; J. Am. Coll. Cardiol., 46 (1) (2005 Jul 5), pp. 92–100
- [22] G. Bajraktari, R. Nicoll, P. Ibrahimi, F. Jashari, A. Schmermund, M.Y. Henein; Coronary calcium score correlates with estimate of total plaque burden; Int. J. Cardiol., 167 (3) (2013 Aug 10), pp. 1050–1052
- [23] G. Ramakrishna, J.F. Breen, S.L. Mulvagh, R.B. McCully, P.A. Pellikka; Relationship between coronary artery calcification detected by electron-beam computed tomography and abnormal stress echocardiography: association and prognostic implications; J. Am. Coll. Cardiol., 48 (10) (2006 Nov 21), pp. 2125–2131
- [24] E. Palmerini, D. Shungin, S. Söderberg, S. Mondillo, M.Y. Henein; Coronary Calcification with No Flow Limiting Lesions: A Potential Cause for Ischaemic Dysfunction in Syndrome X patients; (2015)
- [25] R. Erbel, S. Mohlenkamp, K.H. Jockel, N. Lehmann, S. Moebus, B. Hoffmann, et al.; Cardiovascular risk factors and signs of subclinical atherosclerosis in the Heinz Nixdorf Recall Study; Dtsch. Arztebl. Int., 105 (1-2) (2008 Jan), pp. 1–8
- [26] A.M. Larghat, N. Maredia, J. Biglands, J.P. Greenwood, S.G. Ball, M. Jerosch-Herold, et al.; Reproducibility of first-pass cardiovascular magnetic resonance myocardial perfusion; J. Magn. Reson. Imaging, 37 (4) (2013 Apr), pp. 865–874
- [27] S. Achenbach, D. Ropers, S. Mohlenkamp, A. Schmermund, G. Muschiol, J. Groth, et al.; Variability of repeated coronary artery calcium measurements by electron beam tomography; Am. J. Cardiol., 87 (2) (2001 Jan 15), Article A8
- [28] J. Horiguchi, M. Kiguchi, C. Fujioka, R. Arie, Y. Shen, K. Sunasaka, et al.; Variability of repeated coronary artery calcium scoring and radiation Dose on 64- and 16-slice computed tomography by prospective electrocardiographically-triggered axial and retrospective electrocardiographically-gated spiral computed tomography: a phantom study; Acad. Radiol., 15 (8) (2008 Aug), pp. 958–965
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?