Abstract
Objective
The aim of this study was to investigate the effects of low-dose fractionated radiotherapy on cartilage degeneration after distal femoral fresh massive osteochondral allograft transplantation.
Methods
Twenty-four New Zealand White rabbits were divided into three groups of 8 rabbits each. All rabbits underwent distal femoral medial condyle fresh massive osteochondral allograft transplantation from California rabbits. The group 1 underwent transplantation without any preliminary process. The group 2 underwent fractionated local radiotherapy of 100 cGy for five days starting on the transplantation day. The group 3 included the rabbits to which the grafts transplanted after radiating in vitro by a single dose radiation of 1500 cGy. The hosts were sacrificed twelve weeks later. Anteroposterior and lateral radiographs were taken. Synovial tissue, cartilaginous tissue, and subchondral bone were assessed histopathologically.
Results
Nonunion was present in three cases of group 2 and one of group 3 in which cartilage degeneration was more severe. Synovial hypertrophy and pannus formation were more obvious in non-radiated rabbits. Hypocellularity and necrosis of the subchondral bone were rare in group 2. More cartilage tissue impairment was present in group 3 compared to group 1.
Conclusion
In osteochondral massive allograft transplantations, the immune reaction of the host could be precluded with radiotherapy, and the side-effects can be prevented by low-dose fractionated regimen. The total dose of fractionated radiotherapy for an immune suppression should be adjusted not to damage the cartilage tissue, but to avoid articular degeneration in the long term.
Keywords
Osteochondral allograft ; Radiotherapy ; Cartilage degeneration ; Animal study
Introduction
Osteochondral allografts (OCA) are used as biological implants in reconstruction following bone tumor resection, osteonecrosis, osteochondritis dissecans, osteoarthritis, and post-traumatic cartilage defects.1 ; 2 ; 3 Complications following OCA application include fracture in the graft, nonunion, subchondral collapse, and osteoarthritis. These complications are usually thought to be secondary to immune response to the bony component of the graft. Cartilage viability is important for the success of OCA. Freezing and freeze-drying methods used to suppress immunity in bony allografts cause chondrocyte death in OCA. Even use of cryoprotectants such as glycerol and dimethyl sulfoxide is unable to provide chondrocyte survival in more than 50% of cases.4 ; 5 In response, fresh application of OCA without preservation has become more popular.
Fractionated low-dose radiotherapy has been used as a painkiller in some degenerative joint diseases, although the mechanism is still unknown.6 ; 7 An experimental model has been used to investigate the anti-inflammatory effects of fractionated low-dose radiotherapy on osteoarthritis. This model had a similar pathway to osteoarthritis developed after OCA transplantation.8 Correspondingly, the present study was designed to reveal whether fractionated low-dose radiotherapy can prevent articular degeneration following fresh massive OCA transplantation.
Materials and methods
The present study was performed in an experimental and medical research laboratory, and the samples harvested for histopathological investigation were assessed by the Gazi University School of Medicine Department of Pathology. Radiotherapy applications were performed by members of the Hacettepe University School of Medicine Department of Radiation Oncology. Grafts were received by 4-month-old female New Zealand White rabbits weighing 2700–3200 g each. Donors were equally weighted California rabbits of the same age.
Twenty-four New Zealand White rabbits who underwent massive OCA transplantation had been divided into 3 groups. Group 1 underwent transplantation without preliminary process. Group 2 underwent fractionated radiotherapy for 5 days, beginning on the day of transplantation. Group 3 included rabbits that received grafts following in vitro, single-dose radiation.
Surgical technique
Rabbits were fasted 6 h prior to surgery, and were anesthetized by intramuscular injection of 50 mg/kg ketamine hydrochloride (Ketalar® ; Pfizer, Inc., New York City, NY, USA) and 10 mg/kg xylazine hydrochloride (Rompun® ; Bayer, Inc., Leverkusen, Germany). Infection prophylaxis (25 mg/kg cefazolin sodium; Sefazol® ; Mustafa Nevzat, Inc., İstanbul, Turkey) was administered immediately prior to surgery. After the fur was shaved, the skin was sterilized with gauze pads soaked in 7.5% povidone iodine (Polyod® ; Droğsan, Inc., Ankara, Turkey).
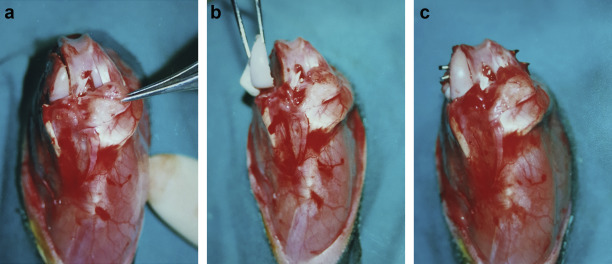
The extremity was draped in a sterile fashion, and an anterior longitudinal incision was made. Joint approach was made by medial parapatellar capsular incision, and medial collateral ligament was horizontally incised in the center. Condyles were entirely exposed with the knee in flexion. The medial femoral condyle was osteotomized with an electric bone saw from the medial aspect of the intercondylar notch to the medial femoral cortex, 0.5 cm proximal to the attachment of the medial collateral ligament. Anterior cruciate ligament and medial meniscus were secured intact (Fig. 1 a). Fresh and similarly dimensioned medial femoral condyle allograft harvested from a California rabbit, using the same technique, was orthotopically transplanted. Fixation of the grafts was established with 2 k wires (Fig. 1 b, c). Transected stumps of the medial collateral ligament were repaired in an end-to-end fashion with 5-0 polypropylene (Prolene® ; Ethicon, Inc., Edinburgh, Scotland). The joint capsule was then sutured in a continuous manner with 4-0 vicryl (Ethicon, Inc., Edinburgh, Scotland), as was the skin, with 4-0 catgut (Droğsan, Inc., Ankara, Turkey).
|
|
|
Fig. 1. Surgical technique of osteochondral allograft transplantation. a. Oblique osteotomy was performed between the medial aspect of the intercondylar notch and the medial femoral cortex, using an electric bone saw. b. Position of fresh osteochondral allograft. c. Fixation of the osteotomy with 2 k wires. |
Implementation of radiotherapy
Group 2 underwent fractionated low-dose local radiotherapy with Cobalt-60 external beam radiation following surgery. Five times, 100 cGy exposure to radiotherapy was administered on a per-day basis. A total of 500 cGy was administered, with the first dose administered the evening following surgery. The animals were anesthetized with 10 mg/kg of ketamine hydrochloride prior to radiotherapy. In Group 3, the harvested grafts were placed in a sterile jar with 0.5% NaCl and wrapped with sterile drapes. Grafts were exposed to a single dose of 1500 cGy radiotherapy and were transplanted after 6 h.
Postoperative follow-up
Anteroposterior and lateral radiographs were postoperatively obtained. No immobilization was performed for the knees. The animals were allowed to move freely in cages under relative humidity of 55 ± 5% and temperature of 21 ± 3 °C. They were fed with standard rabbit granule ad libitum, with free access to water. Hosts were sacrificed after 12 weeks by injection of air into the heart. The femur and the tibia were osteotomized at mid-shaft level in order to obtain the entire joint.
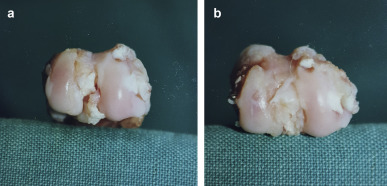
Samples were fixed with 10% formalin solution after radiographs were obtained. Following macroscopic examination (Fig. 2 ), samples were decalcified in a 10% formic acid solution for 1 week. Samples were embedded in paraffin blocks and sagittally sectioned into 6-μm slices. Slices were stained with Hematoxylin and Eosin and Safranin-O, and were examined under light microscope. Histological features of the synovial tissue, cartilaginous tissue, and subchondral bone were assessed by Hematoxylin and Eosin staining, while those of the proteoglycans of the cartilage matrix (chondroitin sulfate, keratan sulfate), were assessed by Safranin-O staining.
|
|
|
Fig. 2. Macroscopical evaluation of the articular surface of the osteochondral allograft. a. Normal articular cartilage of the graft. b. Degeneration of the articular cartilage of the graft. |
Synovial tissue assessment was made using a grading system that included villus formation, neovascularization, and myxoid changes in the stroma (Table 1 ). Cartilaginous tissue assessment was macroscopically and microscopically performed using the modified Mankin grading system described by Shapiro and Glimcher9 (Table 2 ).
| Evaluation criteria | Synovial hypertrophy | |||
|---|---|---|---|---|
| None | Mild | Moderate | Advanced | |
| Villus formation | 0 | 1 | 2 | 3 |
| Myxoid changes in stroma | 0 | 1 | 2 | 3 |
| Neovascularization | 0 | 1 | 2 | 3 |
| Evaluation criteria | Degeneration | |||
|---|---|---|---|---|
| Macroscopical appearancea | Normal | Mild | Moderate | Severe |
| Brightness of articular surface | ||||
| Fissures | 0 | 1 | 2 | 3 |
| Osteophytes | ||||
| Histological appearance | ||||
| Cartilage structure | None | Mild | Moderate | |
| Surface irregularity | 0 | 1 | 2 | |
| Fissures extending to transitional zone | 0 | 1 | 2 | |
| Fissures extending to deep zone | 0 | 1 | 2 | |
| Transverse fissures | 0 | 1 | ||
| Transverse fissures with tissue loss | 2 | |||
| Cellular properties | None | Mild | Moderate | Severe |
| Clonning | 0 | 1 | 2 | 3 |
| Hypocellularity | 0 | 1 | 2 | 3 |
| Decrease in Safranin-O staining | Normal | Mild | Moderate | Severe |
| 0 | 1 | 2 | 3 | |
a. Scoring was the result of the evaluation of 3 parameters.
Statistics
Statistical analyses were performed using SPSS version 21.0 (SPSS Inc., Chicago, USA). Ratio of pannus formation was analyzed with Fishers exact chi-square test. Comparison of synovial hypertrophy scores and cartilage degeneration scores of the 3 groups were analyzed by Kruskal–Wallis one-way analysis of variance. Multiple comparisons of unequal variances were performed in order to compare cartilage degeneration scores of the groups. Comparison of cartilage degeneration scores of Group 2 (with or without delayed union) was analyzed by Mann–Whitney U test, and p values less than 0.05 were considered statistically significant.
Results
During 12-week follow-up, 2 rabbits in Group 1 died of unknown cause, and 1 rabbit in Group 2 developed septic arthritis. Displacement of implanted graft due to migration of a k wire developed in 1 rabbit from Group 1 and 1 rabbit from Group 3. These 5 rabbits were excluded from the study.
Radiographic evaluation
Radiographic evidence of nonunion was found in 3 rabbits from Group 2 and 1 from Group 3. Rabbits in Group 2 showed no pathologic motion in the graft-host interface. These grafts were assumed to be delayed-union.
Evaluation of the synovial hypertrophy
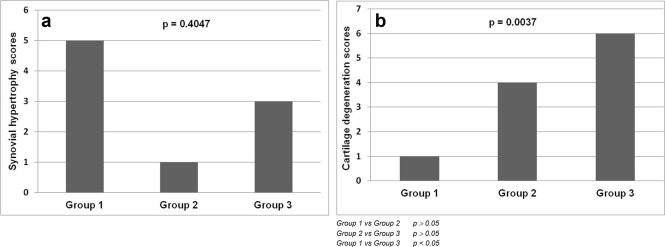
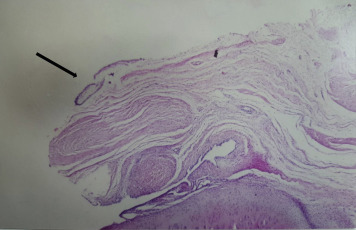
Synovial hypertrophy was macroscopically present in all knees, particularly in Group 1 rabbits. Pannus formation was also observed in 3 knees in Group 1. Median values for synovial hypertrophy scores for Groups 1, 2, and 3 were 5, 1, and 3, respectively. No statistical difference between these values was found (p = 0.4047) (Fig. 3 a). Pannus tissue formation in Group 1 was also confirmed histologically (Fig. 4 ), and statistical difference between Group 1 and the other groups was observed (p = 0.0103).
|
|
|
Fig. 3. Comparison of synovial hypertrophy and cartilage degeneration scores. a. Median synovial hypertrophy scores. b. Median cartilage degeneration scores. |
|
|
|
Fig. 4. Pannus formation expanding toward the cartilage tissue (black arrow). Hematoxylin and Eosin staining with original magnification of ×10. |
Evaluation of the subchondral bone
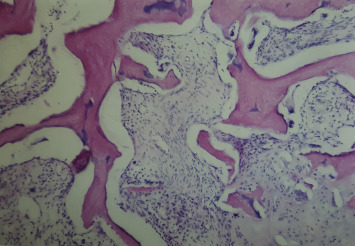
Hematoxylin and Eosin-stained subchondral bone was examined histologically. Hypocellularity and necrosis were seen in all graft bone marrow (Fig. 5 ). However, these factors were more limited in Group 2 than in Groups 1 and 3. Furthermore, in Groups 2 and 3, connective tissue hypertrophy was observed in the bone marrow and subchondral bone. No rabbit developed lymphocyte infiltration in the subchondral bone.
|
|
|
Fig. 5. Hypocellularity and necrosis in bone marrow. Hematoxylin and Eosin staining with original magnification of ×20. |
Evaluation of the cartilaginous tissue
According to cartilage degeneration scores9 obtained by microscopical and macroscopical examination, median values of Groups 1, 2, and 3 were 1, 4, and 6, respectively. These values were significantly different (p = 0.0037) (Fig. 3 b). Cartilage scores were not statistically different between Groups 1 and 2 (p > 0.05), and Groups 2 and 3 (p > 0.05), whereas Group 3 had more scores than Group 1 (p < 0.05). Median value of the rabbits in Group 2 with and without delayed union was 6 and 3, respectively, and was significantly different (p = 0.0477).
Hypocellularity, and thereof reduction in Safranin-O staining, were also more obvious in Group 3 than in Groups 1 and 2. If only these parameters were taken into account, median values for Groups 1, 2, and 3 would be 0, 1, and 3, respectively. These values were significantly different (p = 0.0175). Results of these 2 parameters were significantly higher in Group 3, compared to Group 1 (p < 0.05), while no significant difference between Groups 1 and 2 or between Groups 2 and 3 were found (p > 0.05).
Discussion
Viability of cartilage cells and osseous union of transplanted graft are necessary for satisfactory result of OCA transplantation. As it lacks vascular supply and is supplied by diffusion from the synovial fluid, the chondrocytes can survive following fresh OCA transplantation.10 However, bone-to-bone union slowly evolves, due to the immune reaction of the host to the bone and bone marrow of the graft.10 ; 11 This undesired reaction affects healing and blood supply to a graft by impairing revascularization and activating bone-resorbing osteoclasts.12 ; 13 Delayed union of an OCA may be a portent of fracture, nonunion, collapse in the subchondral bone, and chondral degeneration. Chondrocytes usually do not constitute an immune reaction, as they lurk in the matrix.10 ; 14 However, immune reaction to the bony component of the allograft impairs the cartilage indirectly by inducing excretion of cytokines in the synovial fluid, pannus formation, and subchondral bone collapse.13 ; 15 ; 16 ; 17 ; 18 Immune reaction to allograft can be suppressed by freezing, freeze-drying, use of systemic immune-suppressor drugs, and total lymphoid radiation (TLI).12 ; 19 ; 20 ; 21 ; 22 ; 23 ; 24 However, even when dimethyl sulfoxide or glycerol solutions are used as cryoprotectants, 50% of cartilage chondrocytes cannot survive.4 ; 5 ; 12 ; 25 In addition, immunosuppression and TLI cause serious side effects.
For local suppression of the immune response, irrigation of the subchondral bone with cytotoxic agents and covering of the subchondral bone with bone cement has been described in cases of fresh OCA.26 ; 27 The aim of the present study was to understand the effects of low-dose radiation on graft-host incorporation and fate of the cartilage.
Fractionated low-dose radiotherapy has been used as a painkiller in some degenerative joint diseases,6 ; 7 and has been shown to cease osteoarthritis progression in experimental models.28 In such a study, zymosan and inactivated tubercle bacillus were used in rat knees to produce osteoarthritis by means of complement system activation and pannus tissue formation,8 similar to the development of chondral damage due to the immune response to the bony component of OCA. Thus, the present study was designed with the suggestion that low-dose radiotherapy may suppress immune response and prevent cartilage damage in cases of fresh OCA.
In Group 2, fractionated radiotherapy regimen was used as described by Trott.8 In the literature, the lowest doses of radiotherapy applied to allografts to suppress immune response have mostly been between 1200 and 1500 cGy.24 ; 29 ; 30 Still, no study has sought to explain the effects on cartilage degeneration when used following OCA transplantation. A dosage of 1500 cGy is reportedly sufficient to generate permanent cartilage damage.31 However, due to the need to suppress immune response and serve as an alternative radiotherapy regimen to compare to Group 2, 1500 cGy of radiotherapy was applied to Group 3.
Synovial hypertrophy was more obvious in Group 1, though no significant difference in synovial hypertrophy scores was found. Synovial hypertrophy, observed in all groups, may have been an immune reaction to the graft or a post-surgical reaction. It was not possible to discriminate between these entities in the present study.
Pannus formation was found in 3 Group 1 rabbits, in contrast with other groups. These findings were statistically significant, and revealed that pannus formation, which indicates an immune response to fresh OCA, could be precluded by radiotherapy. However, cartilage structure scores of Groups 1 and 2 were statistically similar, which may have been caused by the inhibiting effect of radiotherapy on the bony union. A delayed union was radiographically observed in 3 Group 2 rabbits. Cartilage structure scores of those rabbits were higher than those of others in the same group. It is believed that radiotherapy resulted in a delayed union, which facilitated cartilage degeneration by adversely affecting the subchondral bone, as a result of which, the in vivo immune-suppressing effect of low-dose radiotherapy was balanced by an adverse effect on the bony union. However, over a longer period of time, pannus formation in Group 1 may have changed the balance in favor of Group 2, by degenerating the articular cartilage.
Cartilage scores of Group 3 were higher than those of Group 1, possibly due to impairment of chondrocytes by a single high dose of radiotherapy, as hypocellularity of the chondral tissue and reduction in Safranin-O staining were more obvious in Group 3 than Group 1.
Overall cartilage structure scores, hypocellularity, and Safranin-O staining scores were not statistically different in Groups 2 or 3, possibly due to short follow-up period. Long-term effects of fractionated radiotherapy on chondrocytes are different from a single high dose of radiotherapy. The latter results in permanent cartilage damage in the long term.31 In light of what is now known, it is expected that cartilage degeneration in Group 3 would have worsened in the long term, while it would have reached a plateau in Group 2. Similar results observed by hypocellularity and Safranin-O staining of Groups 1 and 2 showed that low-dose fractionated radiotherapy regimen did not adversely affect chondrocytes.
Subchondral hypocellularity and necrosis were histopathologically observed in all groups, an expected finding after avascular bone graft transplantation, and even after autogenous bone graft transplantation. Hypocellularity and necrosis were more extensive in Groups 1 and 3 than in Group 2, which may be due to pannus formation in Group 1 and high-dose radiotherapy in Group 3. Fair results in Group 1 show that immune response to OCA does not always correlate with clinical outcome. Genetically different species of donor and host may not always bring about histoincompatibility or graft incorporation trouble, since major histocompatibility complexes are members of highly polymorphic gene group.22 Furthermore, it has been suggested that immune response antibodies may play a role as protector from humoral immunity,20 which may explain the lack of lymphocyte infiltration observed in Group 1.
A primary limitation of the present study was the low number of rabbits in each group, making statistical analysis difficult. However, increasing the number of experimental subjects in an animal study bears ethical problems. Another limitation was short follow-up period. A more precise evaluation of results, regarding effect of pannus formation in Group 1 and long-term effect of fractionated radiotherapy in Group 2, would have been possible if the follow-up period had been longer. However, 2 radiotherapy protocols were presently implemented, providing information regarding correct dosage and application of radiotherapy for immune suppression in cases of OCA. To the authors' knowledge, the present is the first study to use radiotherapy to attempt to prevent complications following fresh OCA application.
In conclusion, fresh OCA triggers an immune reaction characterized by pannus formation. This response can be precluded by low-dose, in vivo fractionated, or high-dose, in vitro single-fraction radiotherapy. Low-dose fractionated radiotherapy has adverse effects on graft-host incorporation. However, total radiotherapy dose administered in fractions will result in less chondrocyte damage in the long term. More severe articular degeneration can be expected with fresh OCA, due to pannus formation. Single-fraction high-dose radiotherapy is not suitable for immune suppression, as it has adverse effects of on chondrocytes, with permanent cartilage damage. Optimal dose regimen of fractionated radiotherapy for immune suppression without any incorporation trouble should be the focus of further investigation.
Acknowledgement
The authors would like to thank Prof. Ömür Ataoğlu, M.D. for his support in histopathologic evaluation, Prof. Faruk Zorlu, M.D. for his support in radiotherapy application, and Prof. Erdal Cila, M.D. for his contribution to the study design.
References
- 1 S.L. Sherman, J. Garrity, K. Bauer, J. Cook, J. Stannard, W. Bugbee; Fresh osteochondral allograft transplantation for the knee: current concepts; J Am Acad Orthop Surg, 22 (2014), pp. 121–133
- 2 F. De Caro, S. Bisicchia, A. Amendola, L. Ding; Large fresh osteochondral allografts of the knee: a systematic clinical and basic science review of the literature; Arthroscopy, 31 (2015), pp. 757–765
- 3 W. Mnaymneh, T. Malinin; Massive allografts in surgery of bone tumors; Orthop Clin North Am, 20 (1989), pp. 455–467
- 4 W.W. Tomford, G.P. Duff, H.J. Mankin; Experimental freeze-preservation of chondrocytes; Clin Orthop Relat Res, 197 (1985), pp. 11–14
- 5 N.S. Schachar, L.E. McGann; Investigations of low-temperature storage of articular cartilage for transplantation; Clin Orthop Relat Res, 208 (1986), pp. 146–150
- 6 H.J. Feldmann; Painful degenerative and inflammatory diseases of joints and soft tissue: X-ray instead of pharmaceutics?; [Article in German] MMW Fortschr Med, 155 (2013), p. 42 [Abstract]
- 7 S. Keller, K. Müller, R.D. Kortmann, et al.; Efficacy of low-dose radiotherapy in painful gonarthritis: experiences from a retrospective East German bicenter study; Radiat Oncol, 8 (2013), p. 29
- 8 K.R. Trott, R. Parker, M.P. Seed; The effect of X-rays on experimental arthritis in the rat; [Article in German] Strahlenther Onkol, 171 (1995), pp. 534–538 [Abstract]
- 9 F. Shapiro, M.J. Glimcher; Induction of osteoarthrosis in the rabbit knee joint; Clin Orthop Relat Res, 147 (1980), pp. 287–295
- 10 C.M. Capeci, M. Turchiano, E.J. Strauss, T. Youm; Osteochondral allografts: applications in treating articular cartilage defects in the knee; Bull Hosp Jt Dis, 71 (2013), pp. 60–67
- 11 D.G. Poitout; Future of bone allografts in massive bone resection for tumor; [Article in French] Presse Med, 25 (1996), pp. 527–530 [Abstract]
- 12 C. Wingenfeld, R.J. Egli, A. Hempfing, R. Ganz, M. Leunig; Cryopreservation of osteochondral allografts: dimethyl sulfoxide promotes angiogenesis and immune tolerance in mice; J Bone Jt Surg Am, 84-A (2002), pp. 1420–1429
- 13 J.D. Bastian, R.J. Egli, R. Ganz, W. Hofstetter, M. Leunig; Chondrocytes within osteochondral grafts are more resistant than osteoblasts to tissue culture at 37°C; J Invest Surg, 24 (2011), pp. 28–34
- 14 W.D. Bugbee, A.L. Pallante-Kichura, S. Görtz, D. Amiel, R. Sah; Osteochondral allograft transplantation in cartilage repair: graft storage paradigm, translational models, and clinical applications; J Orthop Res, 34 (2016), pp. 31–38
- 15 C.J. Campbell; Homotransplantation of a half or whole joint; Clin Orthop Relat Res, 87 (1972), pp. 146–155
- 16 M.W. Elves; Newer knowledge of the immunology of bone and cartilage; Clin Orthop Relat Res, 120 (1976), pp. 232–259
- 17 J.J. Rodrigo, L. Sakovich, C. Travis, G. Smith; Osteocartilaginous allografts as compared with autografts in the treatment of knee joint osteocartilaginous defects in dogs; Clin Orthop Relat Res, 134 (1978), pp. 342–349
- 18 S. Stevenson, X.Q. Li, B. Martin; The fate of cancellous and cortical bone after transplantation of fresh and frozen tissue-antigen-matched and mismatched osteochondral allografts in dogs; J Bone Jt Surg Am, 73 (1991), pp. 1143–1156
- 19 D.B. Brooks, K.G. Heiple, C.H. Herndon, A.E. Powell; Immunological factors in homogenous bone transplantation. iv. The effect of various methods of preparation and irradiation on antigenicity; J Bone Jt Surg Am, 45 (1963), pp. 1617–1626
- 20 G.E. Friedlaender, D.M. Strong, K.W. Sell; Studies on the antigenicity of bone. I. Freeze-dried and deep-frozen bone allografts in rabbits; J Bone Jt Surg Am, 58 (1976), pp. 854–858
- 21 H. Burchardt, F.P. Glowczewskie, W.F. Enneking; Allogeneic segmental fibular transplants in azathioprine-immunosuppressed dogs; J Bone Jt Surg Am, 59 (1977), pp. 881–894
- 22 M. Kliman, P.F. Halloran, E. Lee, S. Esses, P. Fortner, F. Langer; Orthotopic bone transplantation in mice. III. Methods of reducing the immune response and their effect on healing; Transplantation, 31 (1981), pp. 34–40
- 23 J.J. Rodrigo, A.M. Schnaser, H.M. Reynolds Jr., et al.; Inhibition of the immune response to experimental fresh osteoarticular allografts; Clin Orthop Relat Res, 243 (1989), pp. 235–253
- 24 R.G. Burwell; Studies in the transplantation of bone. V. The capacity of fresh and treated homografts of bone to evoke transplantation immunity. 1963; Clin Orthop Relat Res (367 Suppl) (1999), pp. 5–11 discussion S2–4
- 25 W.F. Enneking, D.A. Campanacci; Retrieved human allografts : a clinicopathological study; J Bone Jt Surg Am, 83-A (2001), pp. 971–986
- 26 J. Rodrigo, H. Reynolds, E. Thorson, J. Gray, E. Thompson, D. Heitter; Cement coating of osteoarticular allografts in rats to prevent the immune response; Am J Sports Med, 15 (1987), pp. 591–596
- 27 J.J. Rodrigo, E. Heiden, M. Hegyes, N.A. Sharkey; Immune response inhibition by irrigating subchondral bone with cytotoxic agents; Clin Orthop Relat Res, 326 (1996), pp. 96–106
- 28 C. Steffen, C. Müller, K. Stellamor, J. Zeitlhofer; Influence of X-ray treatment on antigen-induced experimental arthritis; Ann Rheum Dis, 41 (1982), pp. 532–537
- 29 M.W. Elves, A. Ferguson; The humoral immune response to allografts of foetal small intestine in mice; Br J Exp Pathol, 56 (1975), pp. 454–458
- 30 R.G. Burwell, G.E. Friedlaender, H.J. Mankin; Current perspectives and future directions: the 1983 invitational conference on osteochondral allografts; Clin Orthop Relat Res, 197 (1985), pp. 141–157
- 31 E.J. Hall; Time dose and fractionation in radiotherapy. A comarison oftwo evaluation systems in clinical use; Br J Radiol, 42 (1969), pp. 427–431
Document information
Published on 31/03/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?