Abstract
Introduction and objectives
Different percutaneous interventional procedures are needed to reach and maintain adequate anatomical and physiological conditions for the Fontan circulation. We aim to describe the experience gained at a childrens hospital in such interventions, and to analyze the clinical outcomes.
Methods
Retrospective study of all patients with Fontan circulation completed between 1995 and 2013. We analyzed the clinical characteristics and the different types of percutaneous interventions performed, considering three different periods of time: before Glenn surgery, between Glenn and Fontan surgeries, and after Fontan was completed. Survival and time to indication of percutaneous interventions in each period were analyzed, as well as the clinical situation at last follow-up.
Results
Of the 91 patients analyzed, 46 (50.5%) required percutaneous interventions. The most frequent procedures were pulmonary artery angioplasty and angioplasty of the Fontan conduit. Estimated survival at 10, 20 and 30 years of age was 96.2%, 94.7% and 89.4%, respectively. There were no significant differences in survival of patients undergoing percutaneous interventions or not. Overall survival and time to indication of percutaneous interventions were significantly lower in the group of patients with right morphology systemic ventricle. Patients with fenestrated Fontan required interventions more frequently. At the end of follow-up, 66 patients (72.5%) were asymptomatic, without significant differences between patients who underwent or did not undergo percutaneous interventions.
Conclusions
Interventional catheterization procedures are often necessary to reach and maintain the fragile Fontan circulation, mainly in patients with right morphology systemic ventricles and fenestrated Fontan conduits.
Abbreviations
CHD, congenital heart disease;IVC, inferior vena cava;NYHA, New York Heart Association;PI, percutaneous intervention;SVC, superior vena cava;TCD, total cavopulmonary derivation
Keywords
Fontan procedure;Percutaneous intervention;Congenital heart disease;Cardiac catheterization
1. Introduction
Total cavopulmonary derivation (TCD) surgery consists, regardless of the changes emerged since the first description of the technique by Fontan in 1971 [1], in the connection of the systemic venous flow to the pulmonary arteries passively, bypassing the ventricular impulse. This is performed through the direct connection of the superior vena cava (SVC) to the pulmonary branches in a first staged surgery (Glenn operation), and of the inferior vena cava (IVC) in a second staged procedure (Fontan operation), achieving so a complete venous return derivation into the pulmonary circuit.
For such univentricular physiology, favorable anatomic and hemodynamic parameters are needed. These are frequently achieved only with previous percutaneous or surgical procedures when necessary. In general, these determinants include a sufficient but not excessive pulmonary flow in early stages, optimal ventricular function, absence of significant stenosis or regurgitation of the systemic atrioventricular valve, sinus rhythm, low pulmonary resistance and pulmonary arteries of adequate size [2].
In recent years, given the increased survival of patients with Fontan circulation, percutaneous techniques have emerged for new indications or to solve complications, which aim to keep this fragile physiology in optimal circumstances. We present in this paper the cumulative work experience in structural percutaneous procedures performed at our institution over the past 18 years in patients with univentricular physiology.
2. Materials and methods
2.1. Study population and analyzed variables
All patients who had a TCD completed at La Paz Childrens University Hospital in Madrid from 1995 to October 2013, and at least one follow-up visit, were included. No patients with cavopulomonary bypass pending completion or who were considered candidates for biventricular physiology were included. The variables were collected retrospectively from medical reports and paper or computerized clinical history of patients. In addition to demographic data, information on the type of congenital heart defect (CHD), the date of Glenn surgery and Fontan surgery, the date of each interventional catheterization, and the type of procedure performed were obtained. Moreover, information on the need of other than Glenn or Fontan surgery during follow-up was collected. Three periods of time were considered for presentation of the results regarding the performance of percutaneous interventions (PIs): prior to Glenn surgery (pre-Glenn), between Glenn and Fontan surgery, and after Fontan surgery (post-Fontan). The last date of follow-up of each patient, and the date of death in patients who died, were recorded. Also, the overall clinical status of each patient at follow-up, according to their functional New York Heart Association (NYHA) class, and the presence of relevant issues related to the failure of Fontan circulation, such as protein-losing enteropathy, plastic bronchitis, desaturation (arterial oxygen saturation below 90%) or inclusion on heart transplant list, were recorded. To assess the clinical outcome and mortality of the patients with follow-up in other centers, a telephone contact was made in October 2013, by the end date of the overall monitoring of the cohort.
All data were recorded and used in accordance with the applicable data protection laws and ethical principles of the Declaration of Helsinki.
2.2. Indication of percutaneous interventional procedures
Indications of the different types of PI were mainly proposed by the clinical cardiologist responsible for each patients follow-up in our high-resolution (clinical evaluation + electrocardiogram + echocardiography) outpatient clinics. Other times, it is the interventional cardiologist who proposed a specific PI based on the hemodynamic or anatomic findings in pre-Glenn and pre-Fontan diagnostic catheterizations that, most of the times, all patients on the way to univentricular physiology undergo. In any case, all PI proposed were discussed and finally indicated during Children Heart Team meetings where clinicians, interventional cardiologists and surgeons responsible for the care of each patient participated.
2.3. Cardiac catheterization
All catheterizations were performed under general anesthesia and mechanical ventilation with endotracheal intubation. The route of venous access was femoral or jugular and arterial access was femoral.
2.4. Statistical analysis
Qualitative variables were expressed as percentages and the relationships between them were analyzed using the χ2 test or Fishers exact test. Quantitative variables were expressed as mean ± standard deviation for normally-distributed variables, or as median [interquartile range] for variables which were not normally distributed. To test the hypothesis of normal distribution of quantitative variables, the Kolmogorov–Smirnov test was used. Relationship between continuous variables of normal distribution was analyzed according to the number of existing categories, using the Student t test or ANOVA with post-analysis according to the Bonferroni method; continuous variables not normally distributed were analyzed using Mann–Whitney or Kruskal–Wallis tests, depending on the number of categories. Significant value of p < 0.05 (two-tailed) was considered statistically significant. For the overall survival analysis and time to the indication of PI, the Kaplan–Meier method was used. Comparisons were performed using the log-rank test. The statistical analysis was performed using SPSS PASW statistics 15.0 package (SPSS Inc., Chicago, Illinois, United States).
3. Results
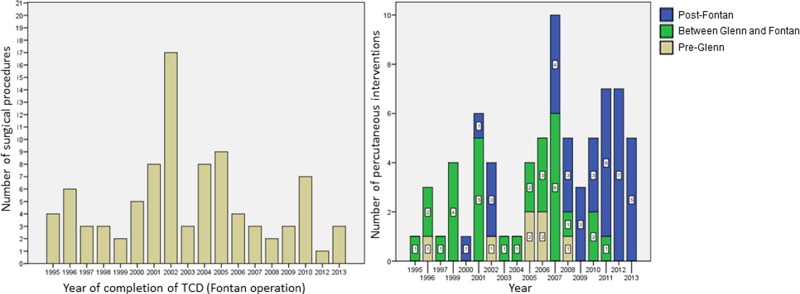
From 1995 to October 2013, a total of 91 patients with CHD underwent a TCD in two stages: first the superior cavopulmonary connection (Glenn surgery), and in a second time the inferior cavopulmonary connection (Fontan). In 15 patients (16.5%) our team opted for a fenestration of the Fontan conduit into the right atrium (fenestrated Fontan). In only 2 patients, an atriopulmonary connection was performed, and in 2 patients a Kawashima procedure (derivation of the hepatic veins to the pulmonary artery when absence of the hepatic portion of the IVC is patent) was performed. 46 out of 91 patients (50.5%) had at least one interventional cardiac catheterization performed. Demographic characteristics, underlying CHD and the age at Glenn and Fontan surgeries are shown in Table 1. Patients with right morphology systemic ventricle needed PI more frequently than patients with left morphology systemic ventricle (61.2% vs. 38.1%, p = 0.028). The underlying CHD differed significantly between patients undergoing or not PI (p = 0.015), being particularly more frequent in patients with hypoplasia/atresia of the left ventricle/valves (p = 0.014). There were no significant differences in the age at which Glenn or Fontan surgeries were done between patients undergoing or not PI. In the 46 patients needing PI, a total of 73 procedures were done. The median number of interventional catheterizations per patient was 1 [1] ; [2]. The description of the procedures performed and the timing of them are shown in Table 2. Angioplasty procedures described include both simple (balloon) angioplasties and angioplasties with stent placement. The yearly distribution of Fontan operations (time when the TCD is completed) performed along the period of review and corresponding PI, is represented in Fig. 1.
| Total cohort (n = 91) | Patients with percutaneous interventions (n = 46) | Patients without percutaneous interventions (n = 45) | p | |
|---|---|---|---|---|
| Demographic characteristics | ||||
| Age at last follow-up or death (years) | 15.5 ± 5.4 | 14.6 ± 5.8 | 16.4 ± 4.9 | 0.107 |
| Age at Glenn surgery (years) | 1.6 [0.9–3.1] | 1.5 [0.8–3.0] | 2.0 [1.3–3.2] | 0.249 |
| Age at Fontan surgery (years) | 7.0 ± 2.8 | 6.8 ± 2.7 | 7.3 ± 3.0 | 0.409 |
| Males (%) | 57 (62.6) | 30 (65.2) | 27 (60.0) | 0.607 |
| Cardiopathy | ||||
| Hypoplasia/atresia of left ventricle/valves (%) | 37 (40.7) | 26 (56.5) | 11 (24.4) | 0.014 |
| – Hypoplastic left ventricle⁎ | 13 | 11 | 2 | |
| – Double outlet RV with ventricular disbalance | 12 | 5 | 7 | |
| – Mitral atresia | 5 | 4 | 1 | |
| – Shone syndrome | 4 | 4 | ||
| – Mitral-aortic atresia | 2 | 2 | ||
| – Criss-cross heart | 1 | 1 | ||
| Atresia or stenosis of right valves (%) | 37 (40.7) | 15 (32.6) | 22 (48.9) | 0.250 |
| – Tricuspid atresia | 20 | 6 | 14 | |
| – Pulmonary atresia with intact septum | 6 | 2 | 4 | |
| – Hypoplastic RV⁎ | 5 | 3 | 2 | |
| – L-TGA with pulmonary atresia | 3 | 1 | 2 | |
| – Tricuspid and pulmonary atresia | 2 | 2 | ||
| – D-TGA with tricuspid stenosis | 1 | 1 | ||
| Left morphology single ventricle (%) | 9 (9.9) | 3 (6.5) | 6 (13.3) | 0.317 |
| Right morphology single ventricle (%) | 8 (8.8) | 2 (4.3) | 6 (13.3) | 0.157 |
| Systemic ventricle | ||||
| Left morphology (%) | 42 (46.2) | 16 (34.8) | 26 (57.8) | 0.028 |
| Right morphology (%) | 49 (53.8) | 30 (64.2) | 19 (42.2) | |
Continuous variables are presented as mean ± standard deviation (normal distribution) or as median [interquartile range] (non-normal distribution). Right column shows the p value for comparisons between patients with and without percutaneous interventions. RV: right ventricle; L-TGA: L-transposition of the great arteries; D-TGA: D-transposition of the great arteries.
⁎. Cases of ventricular hypoplasia without a primary valvular cause.
| Number (%) | Age of patients (years) | Time from last surgery (years)⁎ | |
|---|---|---|---|
| Pre-Glenn procedures | 7 | 0.81 ± 0.97 | |
| Coarctation/recoarctation of aorta angioplasty | 5 (71.4%) | 1.01 ± 1.12 | |
| Angioplasty of both pulmonary arteries | 1 (14.3%) | 0.35 | |
| Angioplasty of right pulmonary veins | 1 (14.3%) | 0.33 | |
| Pre-Fontan procedures | 30 | 4.34 ± 3.06 | 2.44 ± 2.54 |
| Pulmonary artery/arteries angioplasty | 19 (63.3%) | 4.50 ± 2.76 | 2.65 ± 2.12 |
| Angioplasty of the Glenn “conduit” | 2 (6.7%) | 1.64 and 8.90 | 0.13 and 1.00 |
| Coarctation/recoarctation of aorta angioplasty | 2 (6.7%) | 2.38 and 7.39 | 1.21 and 6.22 |
| Embolization of collateral vessels | 2 (6.7%) | 0.69 and 3.51 | 0.12 and 1.92 |
| Angioplasty of the Glenn “conduit” + both PAB | 1 (3.3%) | 0.51 | 0.08 |
| Angioplasty of LPA + embolization of LSVC | 1 (3.3%) | 11.39 | 10.22 |
| Angioplasty of left pulmonary veins | 1 (3.3%) | 0.65 | 0.08 |
| Angioplasty of superior vena cava | 1 (3.3%) | 3.30 | 0.42 |
| Angioplasty of the aortopulmonary anastomosis (Damus–Kaye–Stansel surgery) | 1 (3.3%) | 4.30 | 1.42 |
| Post-Fontan procedures | 36 | 14.09 ± 4.90 | 7.38 ± 4.50 |
| Angioplasty of the Fontan conduit | 9 (25.0%) | 18.32 ± 2.40 | 11.61 ± 2.21 |
| Pulmonary artery/arteries angioplasty | 8 (22.2%) | 10.64 ± 4.30 | 4.65 ± 2.68 |
| Embolization of collateral vessels | 5 (13.9%) | 13.89 ± 7.15 | 6.98 ± 6.52 |
| Angioplasty of the Fontan conduit + PAB | 2 (5.5%) | 13.35 and 17.30 | 4.00 and 12.32 |
| Fontan conduit fenestration closure | 2 (5.5%) | 6.44 and 17.78 | 2.42 and 11.17 |
| Fontan conduit angioplasty + fenestration closure | 2 (5.5%) | 15.91 and 18.02 | 8.93 and 9.84 |
| Enlargement of Fontan conduit fenestration | 2 (5.5%) | 7.29 and 14.26 | 1.98 and 9.92 |
| Embolization of modified BT fistula | 2 (5.5%) | 7.43 and 13.10 | 0.06 and 3.67 |
| Angioplasty of PAB + collateral vessels embolization | 1 (2.8%) | 15.05 | 8.55 |
| Glenn angioplasty + collateral vessel embolization | 1 (2.8%) | 11.94 | 5.33 |
| Embolization of LSVC | 1 (2.8%) | 15.75 | 9.36 |
| Occlusion of pulmonary main artery with device | 1 (2.8%) | 13.46 | 1.59 |
| All procedures | 73 | 9.43 ± 6.63 | 5.15 ± 4.46 |
Continuous variables are presented as mean ± standard deviation. PAB: main pulmonary artery branches; LPA: left pulmonary artery; LSVC: left superior vena cava; BT: Blalock–Taussig.
⁎. In pre-Fontan procedures, time from Glenn surgery is shown; in post-Fontan procedures, time from Fontan surgery is shown; in all procedures, the age of the patient was considered for pre-Glenn procedures.
|
|
|
Fig. 1. Temporal distribution Fontan operations (left) and interventional catheterizations (right) performed in the cohort of patients. For each year, the number of interventional catheterizations performed pre-Glenn surgery, between Glenn and Fontan surgeries, and post-Fontan surgery, is exposed. TCD: Total cavopulmonary derivation. |
Before Glenn surgery, 6 patients underwent PI. A single procedure was performed in 5 patients, whereas the remaining one needed two consecutive aortic coarctation balloon angioplasties. The most common procedure along this period was the angioplasty of coarctation or recoarctation of the aorta.
Between Glenn and Fontan surgeries, 21 patients underwent PI. In 13 patients a single procedure was performed, in 7 patients two procedures were done, and in one patient three procedures were carried out: aortic coarctation and recoartation balloon angioplasties, and a complex procedure which included left pulmonary artery angioplasty and embolization of left superior vena cava. Globally, the most common procedure was pulmonary artery angioplasty.
Finally, after Fontan surgery, 30 patients underwent PI. In 25 patients a single procedure was necessary, in 4 patients two procedures were performed, and in one patient three procedures (angioplasty of pulmonary arteries, and two consecutive angioplasties of the Fontan conduit) were done. Along this period of time, the most common PI was the angioplasty of the Fontan conduit and pulmonary artery branches. When comparing both pulmonary artery branches and Fontan conduit angioplasties, pulmonary angioplasties were performed in younger patients (p < 0.001) and sooner since Fontan surgery (p < 0.001). In patients in whom a fenestrated Fontan had been done, PI was needed more frequently than in patients with non-fenestrated Fontan (60% vs. 27.6%, p = 0.015). 4 fenestration closure procedures were performed during follow-up.
3.1. Overall survival of the cohort of patients
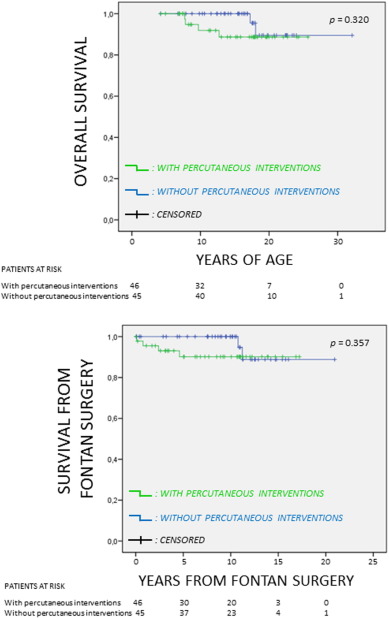
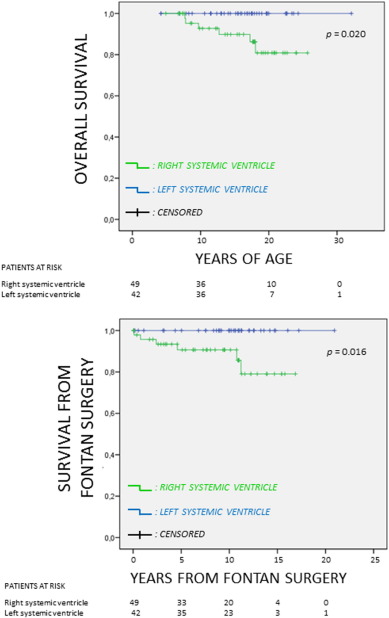
After a mean follow-up of 15.5 ± 5.4 years (median follow-up 16.8 [11.4 to 19.5] years), in the cohort of 91 patients there were 6 deaths (total mortality 6.6%), one of which was caused by complications during the early post-operative period of Fontan surgery, and another in the post-operative period of cardiac transplantation (performed shortly after the Fontan operation). All deaths occurred in the group of patients with right morphology systemic ventricle. There were 4 deaths in the group of patients undergoing PI, and 2 in the group without intervention (p = 0.320). No death was directly related to or occurred during a catheterization, or was due to complications of it. Among the 15 patients who had undergone fenestrated Fontan surgery, no deaths were observed. Causes of death and underlying CHD of deceased patients are shown in Table 3. Estimated survival at 10, 20 and 30 years of age was 96.2% (95% CI: 92.1%–100%), 94.7% (95% CI: 94.2%–99 8%) and 89.4% (95% CI: 80.8%–98.0%), respectively. There was no significant difference between survival of patients undergoing or not PI (Fig. 2). Survival was significantly lower in patients with right morphology systemic ventricle than in those with left morphology systemic ventricle (p = 0.02) (Fig. 3). Estimated survival after TCD (Fontan operation) was completed, at 10 and 20 years of follow-up, was 95.1% (95% CI: 90.4%–99.8%) and 89.1% (95% CI: 79.9%–98.3%), respectively.
| Cardiopathy | Percutaneous interventions performed | Age (years) | Time from Fontan surgery (years) | Cause of death | |
|---|---|---|---|---|---|
| 1 | Hypoplastic left ventricle | Yes | 7.71 | 2.30 | Sudden death. |
| 2 | Shone syndrome | Yes | 7.80 | 0.77 | Cardiogenic shock in the early post-operative period of heart transplant. |
| 3 | Single right ventricle | Yes | 12.72 | 0.10 | Cardiogenic shock and sepsis in the early post-operative period of Fontan surgery. |
| 4 | Mitral atresia | Yes | 9.69 | 4.61 | Sudden death. |
| 5 | Single right ventricle | No | 18.04 | 10.78 | Sudden death. |
| 6 | Single right ventricle | No | 17.24 | 11.21 | Sudden death. |
|
|
|
Fig. 2. Overall survival and survival after Fontan operation among patients undergoing or not undergoing PI. |
|
|
|
Fig. 3. Overall survival and survival after Fontan operation in patients with right or left systemic ventricle. |
3.2. Time to the indication of percutaneous intervention
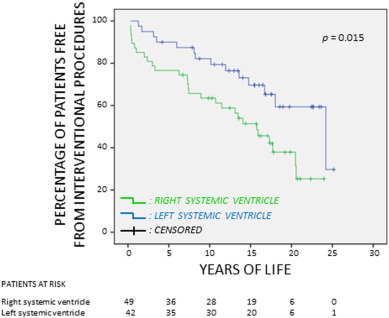
Mean time to the first PI was 16.1 years (95% CI 14.2 to 18.1 years). This time was shorter in patients with right morphology systemic ventricle (13.7 years, 95% CI: 11.1 to 16.3 years vs. 18.87 years, 95% CI: 16.2 to 21.5 years, p = 0.015) (Fig. 4). The estimated percentage of patients free of PI at 1, 5 and 10 years of age was 93.1% (95% CI: 87.9%–98.4%), 82.8% (95% CI: 75.0%–90.6%) and 72.1% (95% CI: 62.7%–81.5%), respectively.
|
|
|
Fig. 4. Time to first percutaneous intervention in patients with right or left systemic ventricle. |
In patients undergoing interventional catheterization, time to PI was also analyzed considering three periods of time: prior to Glenn surgery, between Glenn and Fontan surgeries and after Fontan surgery, when TCD was completed (Fig. 5). For the first two periods of time, follow-up was only considered until Glenn surgery and Fontan surgery, respectively. This explains the small number of patients with longer follow-up than 2 years in the pre-Glenn period, or 4 years in the period of time between Glenn and Fontan surgeries. Mean time to PI after the completion of Fontan circulation was lower for patients with fenestrated Fontan than for non-fenestrated patients (8.88 years, 95% CI: 7.1 to 10.6 years vs. 12.32 years, 95% CI: 10.8 to 13.8 years, p = 0.013).
|
|
|
Fig. 5. Patients undergoing percutaneous interventions. Time to first percutaneous intervention along three different time periods: before Glenn surgery (pre-Glenn), between Glenn and Fontan surgeries, and after Fontan surgery (post-Fontan). |
3.3. Need for surgery other than Glenn or Fontan
In addition to superior and inferior cavopulmonary connections, and excluding early pre-Glenn palliative procedures, that are often necessary in these patients (such as systemic-to-pulmonary shunts), 4 patients (4.4%) required a surgical fenestration opening, and 9 patients (10%) required other operations (one surgery for each patient, except for 2 patients requiring 3 and 4 surgeries, respectively), mainly to treat thrombotic problems or complications related to the Glenn or Fontan operations (Table 4). In addition, 2 patients received a heart transplant but one died in the immediate postoperative period.
| Diagnosis | Surgical treatment | Age (years) | Time from Glenn surgery⁎ | Time from Fontan surgery⁎ | |
|---|---|---|---|---|---|
| 1 | Thrombosis of the superior cavopulmonary derivation (Glenn). | Thrombectomy. | 3.44 | 58 days | |
| 2 | Persistent LSVC draining into the coronary sinus. Sinus node dysfunction. | LSVC ligation. Epicardial pacemaker implant. | 4.47 | 1.4 years | |
| Pacemaker battery end-of-life. | Pacemaker generator replacement. | 8.15 | 5.0 years | ||
| Fontan Kawashima with desaturation. Pacemaker battery end-of-life. | Inclusion of suprahepatic veins into Fontan circulation. Pacemaker generator replacement. | 11.57 | 19 days | ||
| Median sternotomy infection. | Desbridement and aspiration drain insertion. | 11.63 | 39 days | ||
| 3 | Thrombosis of the inferior vena cava. | Thrombectomy. | 12.97 | 10 days | |
| 4 | Postsurgical pericardial effusion. | Aspiration drain insertion. | 7.45 | 29 days | |
| 5 | Acute postsurgical mediastinitis. | Closed vacuum-drainage system insertion. | 6.63 | 42 days | |
| 6 | Chronic retrosternal fistula retroesternal crónica. | Fistulectomy. | 13.10 | 7.7 months | |
| Retrosternal membrane infection. | Resection of the infected retrosternal membrane. | 13.31 | 10.3 months | ||
| Retrosternal fistula relapse | Fistulectomy. | 17.59 | 5.1 years | ||
| 7 | Tricuspid patch dehiscence. | Resuture of the tricuspid patch. | 4.93 | 10 months | |
| 8 | Diffuse thrombosis of the Fontan prosthetic conduit. | Thrombectomy. | 11.68 | 3.2 years | |
| 9 | Fontan conduit severe stenosis. | Extracardiac Fontan conduit replacement. | 17.97 | 11.0 years |
LSVC: left superior vena cava.
⁎. For surgeries performed between Glenn and Fontan surgeries, time from Glenn surgery is displayed. For surgeries performed after Fontan operation, time from Fontan surgery is shown.
3.4. Clinical outcomes
At the end of clinical follow-up, 66 patients (72.5%) were asymptomatic, 19 patients (20.9%) had symptoms and 6 patients had died. Among symptomatic patients, 9 of them (9.8% of the total cohort) had NYHA functional class ≥ 2. Clinical outcomes are listed in Table 5. All 3 patients on a waiting list for heart transplantation have NYHA functional class III, and one of them also suffer from protein-losing enteropathy. The surviving transplanted patient had NYHA functional class IV and protein-losing enteropathy. Among patients with follow-up in other centers, 6 were unable to contact, so only time until their last outpatient visit reported was considered (Table 6).
| Clinical situation | Total cohort (n = 91) | Patients with percutaneous interventions (n = 46) | Patients without percutaneous interventions (n = 45) | p |
|---|---|---|---|---|
| Asymptomatic (%) | 66 (72.5) | 31 (67.4) | 35 (77.7) | 0.550 |
| Symptomatic/complications (%) | 19 (20.9) | 11 (23.9) | 8 (17.8) | |
| – NYHA functional class ≥ II (%) | 9 | 4 | 5 | |
| – Cyanosis | 10 | 6 | 4 | |
| – Protein-losing enteropathy | 5 | 3 | 2 | |
| – Plastic bronchitis | 4 | 2 | 2 | |
| – Inclusion on heart transplant list | 3 | 3 | ||
| – Heart transplantation (alive) | 1 | 1 | ||
| Deceased (%) | 6 (6.6) | 4 (8.7) | 2 (4.4) | 0.320 |
NYHA: New York Heart Association.
| Last follow-up date | Age (years) | Time from Fontan surgery (years) | |
|---|---|---|---|
| 1 | June 2001 | 14.35 | 5.02 |
| 2 | November 2001 | 7.80 | 0.77 |
| 3 | June 2002 | 6.72 | 0.38 |
| 4 | September 2002 | 8.26 | 1.10 |
| 5 | February 2006 | 9.84 | 0.07 |
| 6 | October 2006 | 15.51 | 12.51 |
At the end of follow-up, there were no significant differences in the clinical status of patients according to whether they underwent PI or not (p = 0.550 for the presence of symptoms or complications). There were no significant differences between patients with fenestrated or non-fenestrated Fontan (p = 0.744 for the presence of symptoms or complications). Among the 10 patients with cyanosis, 3 patients had a fenestrated Fontan and another 3 were included in those 4 who required surgical fenestration opening. Table 7 shows the clinical status at follow-up of patients with right and left morphology ventricles. There were no statistically significant differences in the proportion of symptomatic patients (excluding deaths), but in the group of patients with right morphology ventricle we found 8 out of 10 cases of cyanosis, and all cases of plastic bronchitis.
| Clinical situation | Right morphology systemic ventricle (n = 49) | Left morphology systemic ventricle (n = 42) | p |
|---|---|---|---|
| Asymptomatic (%) | 31 (63.3) | 35 (83.3) | 0.385 |
| Symptomatic/complications (%) | 12 (24.5) | 7 (16.7) | |
| – NYHA functional class ≥ II (%) | 4 | 5 | |
| – Cyanosis | 8 | 2 | |
| – Protein-losing enteropathy | 2 | 3 | |
| – Plastic bronchitis | 4 | ||
| – Inclusion on heart transplant list | 2 | 1 | |
| – Heart transplantation (alive) | 1 | ||
| Deceased (%) | 6 (14.3) | 0 |
NYHA: New York Heart Association.
4. Discussion
This paper summarizes the review of a cohort of patients with or in the way to univentricular circulation with structural percutaneous procedures. Fontan circulation is based on a fragile physiology, where discrete anatomical variations of size or pressure gradients imply large variations in cardiac output, arterial oxygen saturation and functional class.[3] Improvement of surgical techniques and the increasingly earlier age at which the TCD is completed have enabled the Fontan circulation as a real alternative for a growing number of complex congenital or acquired heart diseases in our environment. In addition, the extracardiac TCD or modified Fontan, which has replaced the classic atriopulmonary shunt, has significantly reduced complications such as arrhythmias and mortality in the most recent series.[4] The sheer size of anatomic structures in children with CHD and their longer survival have increasingly made more necessary different PI techniques that aim to keep Fontan circulation in optimum condition. In past decades, the fenestration of the inferior cavopulmonar derivation (Fontan conduit) was considered beneficial in the immediate postoperative management of these patients because it ensured a greater systemic output and a lower incidence of postoperative pleural effusions, at the expense of a permissive degree of desaturation. In our cohort, only 16.5% of patients had this fenestration performed, probably because the patients in our study cohort represent a more recent cohort, in which the candidates have been better selected and the vast majority of them had one or more diagnostic catheterizations at which we demonstrated favorable hemodynamic parameters for univentricular circulation to be possible.
This series includes only patients with CHD led to univentricular physiology. Although new technical possibilities such as the pulmonary valvular perforation with radiofrequency, or surgical, as the double switch, allow to achieve a biventricular circulation in patients who would have undergone univentricular palliantion decades ago, there are still cases of extreme hypoplasia of one of the ventricles where this is not possible. Thus, our study included 6 cases of pulmonary atresia with intact ventricular septum, in which the Z score of right ventricular dimensions was below − 4 in all cases (all required a neonatal systemic-to-pulmonary fistula), or 3 cases of L-transposition of the great arteries with pulmonary stenosis or atresia where the subpulmonary left ventricle was extremely hypoplastic and biventricular correction was not contemplated.
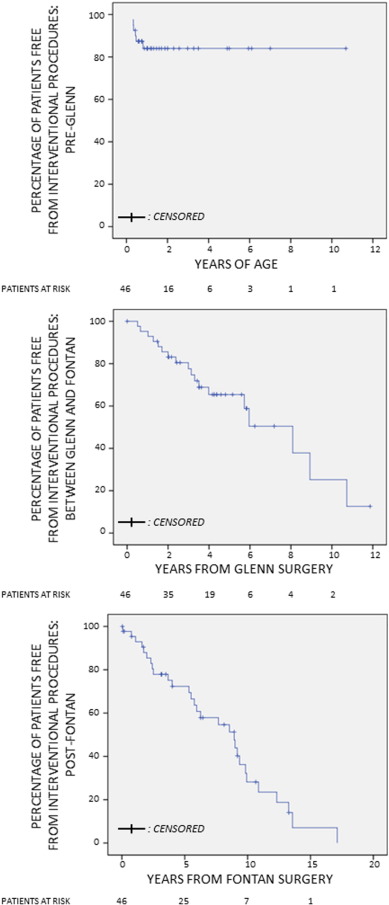
From a technical point of view, we can consider two groups of patients who may benefit from PI in Fontan circulation. The first includes patients with symptoms related to low cardiac output state and high venous pressures. The second includes those patients with progressive desaturation, due either to residual right-to-left shunts (fenestration, LSVC …), or the emergence of new shunts as veno-venous collaterals (between systemic vein and pulmonary vein) or pulmonary arteriovenous fistulae. The first group would benefit from percutaneous techniques such as fenestration opening [5], angioplasty of stenotic pathways such as Fontan conduit, pulmonary arteries, aortic coarctation [6] or the anastomosis between the SVC and corresponding pulmonary artery (Glenn connection). In the second one, however, we should think of techniques such as fenestration closure (Fig. 6), veno-venous collateral embolization, embolization of the persistent SVC or symmetric conduction of suprahepatic drainage flow into both lungs to prevent the development or to promote involution of arteriovenous fistulae [7].
|
|
|
Fig. 6. Fenestration closure device in a patient who had undergone previous angioplasty in the left pulmonary artery with stent (solid arrow). A: Fontan conduit in initial angiography, where contrast flow through the fenestration (asterisk) to right atrium was observed. B: An Atrial Septal Occluder device (Amplatzer™) (dotted arrow) was implanted for fenestration closure, showing no residual shunt in control angiography. |
Approximately, one half of the patients required at least one catheterization with structural PI, being more common in those with systemic right ventricle and fenestrated Fontan, and without significant effects in terms of mortality. This may be because patients with hypoplasia of left cavities or valves require more complex operations (aortic advance, Norwood with systemic-to-pulmonary shunt or Norwood–Sano, Damus–Kaye–Stansel, arterial switch [8] …) and at younger ages than patients with left morphology systemic ventricle. Another added reason has to do with the appearance of symptoms or failure of the Fontan circulation much earlier in the group of patients with a single right ventricle [9].
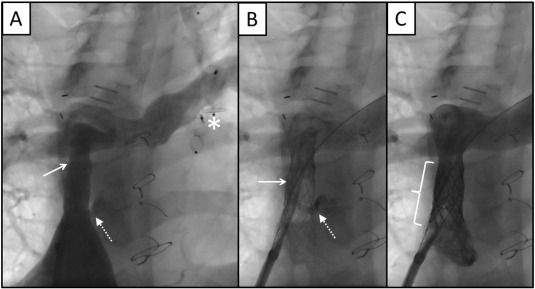
It is predictable that the types of PI more frequently attempted were different in each phase of the cavopulmonary derivation, as hemodynamics in each one is different too. Before Glenn surgery, one should ensure sufficient but not excessive pulmonary flow, in order to avoid cardiac failure symptoms and pulmonary vascular overflow damage, and also resolve the presence of stenosis at any point of the aortic arch. Between Glenn surgery and the completion of TCD, arterial oxygen desaturation and the aim of reaching a sufficient size of the pulmonary arteries are the most common reasons why PI is required, so pulmonary artery angioplasty and right-to-left shunt embolization are the most common procedures. More controversial is the benefit of systematic embolization of systemic-to-pulmonary fistulae that usually appear between Glenn and Fontan surgeries [10]. Upon TCD, symptoms of low cardiac output and failed Fontan (protein losing enteropathy [11] plastic bronchitis [12] …) have sometimes an obstructive underlying mechanism, and therefore, the angioplasty of the Fontan conduit is the most frequently performed procedure (Fig. 7). In the description of the results, we have included under the term “angioplasty” both simple (balloon) angioplasties and angioplasties with stent placement. For reasons of size, angioplasties performed in the pre-Glenn stage are simple angioplasties. Subsequently, stent implantation is mandatory if available for the weight of the patient and if the vascular access size is big enough. This last attitude, however, varies depending on the operator and the technical difficulty. Throughout the years, the smaller size of the introducers and balloons required for stent implantation, and the advent of new low-profile premounted stents with the possibility of postdilation to adapt their diameter to somatic growth, have made the use of stents increasingly more common in angioplasties of the Fontan conduit, pulmonary arteries and aortic arch. Consequently, the number of interventional procedures with stent implantation in the last years of the series is greater than at the beginning. We consider pulmonary artery angioplasty indicated when transarterial gradient is above 1 mm Hg or when pulmonary angiograms show stenosis over 20% of the reference vessel lumen; these sensitive values are used because little hemodynamic variations can cause major clinical deterioration and cardiac output decrease in these patients.
|
|
|
Fig. 7. Fontan conduit angioplasty and fenestration closure. A: Fontan conduit stenosis (solid arrow) and permeable fenestration (dotted arrow) were observed in baseline angiography; previous embolization of the left superior vena cava had been done with a Duct Occluder device (Amplatzer™) and a Vascular Plug II device (Amplatzer™) (asterisk). B: Two open-cell stents were initially implanted in the Fontan conduit (solid arrow), with a mild luminal widening (but sufficient to eliminate the 4 mm Hg gradient recorded), and persistence of contrast flow through the fenestration (dotted arrow). C: Finally, a covered in-stent stent was implanted for fenestration sealing (key), without observing residual shunt in control angiogram. |
The age at which the pulmonary artery angioplasties were performed was significantly lower than the age at which the Fontan conduit angioplasties were done, probably because the growth of the pulmonary arteries between Glenn and Fontan surgeries is relatively slower than that of the IVC, since the effective pulmonary blood flow (Qp) in this phase is less than systemic flow (Qs) with a relationship between both (Qp/Qs) of approximately 0.6. Sometimes, the distortion generated because of the traction of the superior cavopulmonary anastomosis on the right pulmonary branch cannot be completely relieved at the moment of TCD, thus needing earlier percutaneous dilation.
Overall estimated survival up to 30 years of age in our series is slightly higher than that obtained in other less recent series, with a significantly higher mortality in patients with systemic right ventricle, which is consistent with those series [9] ; [13]. This may be related to a worse systolic and diastolic function of the right morphology systemic ventricles when compared with the left morphology ones [9] ; [14], or when compared with right ventricles in biventricular physiology (subpulmonary right ventricle) [15]. Excluding deaths, absence of significant differences in the clinical status of patients at follow-up according to whether they underwent any PI suggests, somehow, that these procedures are effective to maintain adequate functional class and treat clinical complications that would be corrected surgically otherwise. We interpret the absence of deaths or serious complications related to the procedures of PI as an indicator of safety and feasibility.
According to reports from other recent cohort [16], the Fontan conduit fenestration did not seem to influence the clinical status at follow-up in our patients and is a factor associated with increased need for IP itself. Although there are no devices specifically designed for this purpose, the closure of the fenestration can be performed using different closures devices for ventricular septal defects, atrial septal defects, patent ductus arteriosus or patent foramen ovale [17]. In our cohort, fenestration closure was performed in 4 out of 15 (26.7%) patients with fenestrated Fontan, but only 4 among the non-fenestrated patients (5.2%) needed a fenestration to be opened (surgical). At our institution, fenestration closures are indicated in patients with arterial oxygen saturation < 90% and normal pressures in the Glenn–Fontan system. Before implanting the closure device, we perform an occlusion test inflating a balloon in order to exclude pressure increase. The intervention is considered successful when no contrast pass is patent through the fenestration during control angiograms, arterial oxygen saturation increases to > 90% with FiO2 ≤ 0.3, and there are no pressure raise in the Glenn–Fontan system.
We should not forget that all this cumulative experience in PI evolves in parallel with the development of new materials such as bioabsorbable stents [18], and new technology in the catheterization laboratories, as the possibility of acquiring 3D rotational images [19]. In Fontan circulation, new approaches and techniques for electrophysiological studies and ablation of tachycardia when indicated should be considered, once the endovenous access is lost when TCD is completed [20]. Such advances make it possible to perform these relatively expensive and complex procedures, requiring a high level of technology and expertise. Although there are no randomized studies that allow us to comparatively analyze survival, it seems sensible that thanks to these techniques the number of reoperations could be reduced, thereby avoiding the complications inherent in this group of patients. In this sense, it would be useful to create nationwide or multinational multicenter registries that allow pediatric cardiologists to accumulate experience in these procedures. Only in this way it would be possible to compare the effectiveness of the different available techniques to achieve each therapeutic goal, which is not possible with just single-center records given the wide variety of procedures performed and the small number of patients undergoing each of them. Collaborative work with grown up CHD units seems of vital importance in these registries, since only then could we know the final outcome of these patients and the long-term results of the procedures performed. These grown up CHD units will face the challenge of managing complications and deterioration of univentricular physiology in the near future.
5. Limitations
The main limitation of this study is the retrospective nature of the data analysis, although the collection of the data was performed prospectively. Moreover, it has not specifically examined the efficacy of each performed interventional procedure. As a proxy for effectiveness, no patient required surgery as a result of failure or complications of a percutaneous procedure. In terms of security, there were no procedure-related deaths. Another limitation is that this series only included patients with complete TCD, so the results cannot be extrapolated to patients who have only had a partial cavopulmonary derivation done or are still awaiting completion of Fontan circulation.
6. Conclusions
Different types of interventional catheterization procedures are necessary to maintain the fragile univentricular physiology. These techniques can solve many clinical problems and may avoid some operations. Patients with right systemic ventricle are more likely to require interventional procedures, and have a worse prognosis than patients with a left systemic ventricle. Fontan fenestration did not seem to influence the final clinical status at follow-up in our cohort of patients, but is associated with a higher likelihood of needing PI.
Conflicts of interest
None.
References
- [1] F. Fontan, E. Baudet; Surgical repair of tricuspid atresia; Thorax, 26 (1971), pp. 240–248
- [2] A. Mendoza, L. Albert, E. Ruiz, L. Boni, V. Ramos, J.M. Velasco, et al.; Operación de Fontan. Estudio de los factores hemodinámicos asociados a la evolución postoperatoria; Rev. Esp. Cardiol., 65 (2012), pp. 356–362
- [3] B.J. Deal, M.L. Jacobs; Management of the failing Fontan circulation; Heart, 98 (2012), pp. 1098–1104
- [4] M. Cazzaniga, L. Fernández, F. Villagrá; Pérez de León J, Gómez R, Sánchez P, et al. Operación modificada de Fontan o variantes efectuadas en un solo tiempo quirúrgico. Determinantes de la mortalidad; Rev. Esp. Cardiol., 55 (2002), pp. 391–412
- [5] H. Vyas, D.J. Driscoll, A.K. Cabalka, F. Cetta, D.J. Hagler; Results of transcatheter Fontan fenestration to treat protein losing enteropathy; Catheter. Cardiovasc. Interv., 69 (2007), pp. 584–589
- [6] M.J. Del Cerro, A. Fernández, S. Espinosa, F. Benito, M. Burgueros, G.-G. Luis, et al.; Cateterismo intervencionista tras cirugía tipo Norwood; Rev. Esp. Cardiol., 61 (2008), pp. 146–153
- [7] Y. Dori, S. Sathanandam, A.C. Glatz, M.J. Gillespie, J.J. Rome; Catheter approach to redirect hepatic venous return for treatment of unilateral pulmonary arteriovenous malformations after fontan; Catheter Cardiovasc Interv. (2013) http://doi.org/10.1002/ccd.25326
- [8] J.M. Gil-Jaurena, J.I. Zabala, D.C. Albert, R. Castillo, M. González, L. Miró; Switch arterial paliativo como primer tiempo hacia Fontan en pacientes con fisiología univentricular y estenosis subaórtica; Rev. Esp. Cardiol., 66 (2013), pp. 553–555
- [9] P. Anderson, L.A. Sleeper, L. Mahony, S. Colan, A.M. Atz, R.E. Breitbart, et al.; Contemporary outcomes after the Fontan procedure; J. Am. Coll. Cardiol., 52 (2008), pp. 85–98
- [10] P. Banka, L.A. Sleeper, A.M. Atz, C.G. Cowley, D. Gallagher, M.J. Gillespie, et al.; Practice variability and outcomes of coil embolization of aortopulmonary collaterals before Fontan completion: a report from the Pediatric Heart Network Fontan Cross-Sectional Study; Heart J., 162 (2011), pp. 125–130
- [11] G. Butera, D. Marini, S.T. MacDonald; Protein-losing enteropathy resolved by percutaneous intervention; Catheter. Cardiovasc. Interv., 78 (2011), pp. 584–588
- [12] D. Tanase, P. Ewert, A. Eicken; Plastic bronchitis: symptomatic improvement after pulmonary arterial stenting in four patients with Fontan circulation; Cardiol. Young, 18 (2013), pp. 1–3
- [13] P. Khairy, S.M. Fernandes, J. Mayer, J.K. Triedman, E.P. Walsk, J.E. Lock, et al.; Long-term survival, modes of death and predictors of mortality in patients with Fontan surgery; Circulation, 117 (2008), pp. 85–92
- [14] S. Kaneko, N.S. Khoo, J.F. Smallhorn, E.B. Tham; Single right ventricles have impaired systolic and diastolic function compared to those of left ventricular morphology; J. Am. Soc. Echocardiogr., 25 (2012), pp. 1222–1230
- [15] N. Moiduddin, K.M. Texter, A.N. Zaidi, J.A. Hersenson, C.A. Stefaniak, J. Hayes, et al.; Two-dimensional speckle strain and dyssynchrony in single right ventricles versus normal right ventricles; J. Am. Soc. Echocardiogr., 23 (2010), pp. 673–679
- [16] A.C. Fiore, C. Tan, E. Armbrecht, C.B. Huddleston, E. Kim, N. Goel, et al.; Comparison of fenestrated and nonfenestrated patients undergoing extracardiac fontan; Ann. Thorac. Surg., 97 (2014), pp. 924–931
- [17] M. Santisteban, M. Pan, M. Romero, J. Casares, E. Gómez; Suárez de Lezo J. Cierre de fenestración tras la cirugía de Fontan. Aportaciones del intervencionismo percutáneo; Rev. Esp. Cardiol., 66 (2013), pp. 909–911
- [18] C. Zeybek, A. Kırbaş, Y. Yalçın, M.S. Bilal; Successful transcatheter closure of a Fontan fenestration with a bioabsorbable Biostar occluder; Turk Kardiyol. Dern. Ars., 40 (2012), pp. 623–627
- [19] J. Hill, C. Bellotti, A. Golden; Three-dimensional rotational angiography during percutaneous device closure of fontan fenestration; World J. Pediatr. Congenit. Heart Surg., 4 (2013), pp. 324–325
- [20] E. Arana-Rueda, A. Pedrote, J.M. Duran-Guerrero; Santos de Soto J. Abordaje simplificado para la ablación de la taquicardia por reentrada intranodal en paciente con atresia tricuspídea y Fontan extracardiaco; Rev. Esp. Cardiol., 66 (2013), pp. 314–316
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?