Summary
Background
Several cases of successful percutaneous endoscopic gastrostomy (PEG) through the transnasal route have been reported, and Pseudomonas aeruginosa infection in transnasal PEG was described earlier. This study was conducted to investigate the difference between transnasal PEG and conventional PEG.
Methods
A retrospective case–control study was conducted to compare pull-type transnasal (T-PEG) and peroral (O-PEG) placement of a 20 Fr PEG tube in a community hospital. Thirty-eight T-PEG and 38 O-PEG were analyzed in 76 chronic dysphagic patients from homes or nursing homes. The operating time, occurrence of choking during PEG, stomal site infection, bacterial pathogens, and post-PEG complication were recorded and analyzed.
Results
The mean age was 76.3 ± 10.3 years for T-PEG versus 79.3 ± 6.9 years for O-PEG; 67% were male versus 48% female; operating time was 14.6 ± 4.0 minutes for T-PEG versus 11 ± 3 minutes for O-PEG (p = 0.0028), and choking occurred in three patients in the T-PEG group versus five in the O-PEG group. There were 10 stomal site infections (9 with P. aeruginosa) in the T-PEG group and 14 (8 with P. aeruginosa) in the O-PEG group (p < 0.001). One systemic infection of the urinary tract, one buried bumper, and one stomal soiling were observed in the T-PEG and O-PEG groups. No PEG-related mortality occurred within 3 months after all PEG procedures.
Conclusion
Transnasal insertion of PEG using a pull method is a feasible and safe alternative when conventional pull-method PEG is not possible. However, P. aeruginosa infection is common when using T-PEG; therefore, more studies focusing on prophylaxis of T-PEG-associated P. aeruginosa infection are required.
Keywords
Gastrostomy site infection; Percutaneous endoscopic gastrostomy; Pseudomonas aeruginosa; Transnasal percutaneous endoscopic gastrostomy
Introduction
Percutaneous endoscopic gastrostomy (PEG) is a world-wide standard enteral feeding procedure for patients with swallowing dysfunction [1]. PEG is safe and has fewer minor adverse events compared with conventional surgical gastrostomy [2], [3] and [4]. However, peroral PEG (O-PEG) is a big challenge in some patients who have trismus or oral cancer, and historically, surgical gastrostomy with its accompanying anesthetic risk has been the only alternative for these patients. Successful transnasal insertions of a PEG tube with a 5.9-mm pediatric endoscope and even a regular adult scope have been reported by pioneers in the field, with good results [5], [6], [7], [8], [9], [10] and [11]. Dominant Pseudomonas aeruginosa infection of the stomal site has been reported in a series of cases of transnasal PEG (T-PEG) [12]. However, there have been no case–control studies comparing T-PEG and O-PEG. Thus, this work is the first to compare the clinical differences between transnasal and peroral insertion of PEG using the pull method.
Methods
A retrospective, case–control study was conducted and approved by the Institutional Review Board of Pingtung Christian Hospital, Pintung, Taiwan (IRB458A). PEG is not popular in Southern Taiwan due to a lack of understanding among the general population. In Taiwan, nasogastric (NG) tube rather than PEG is a standard feeding method for patients with dysphagia and PEG is recommended only in patients who remove NG tubes frequently, or in whom NG tube insertion is difficult or fails. So, all of the PEG procedures performed from 2009 to 2013 at Pingtung Christian Hospital were enrolled into this study. However, patients meeting the following criteria were excluded from the study: estimated survival time < 2 months, aged < 20 years, hospitalized for an acute disease or infection, receiving ongoing treatment with warfarin or clopidogrel, had undergone tracheostomy, required mechanical ventilation, complex comorbidity, or an inadequate record of medical information.
T-PEG was performed in patients who tolerated nasal intubation or who had difficulty with oral intubation due to trismus or oral malignancy. O-PEG was performed in patients who rejected nasal intubation. Neither the nasal cavity nor the oral cavity was decolonized prior to either type of PEG; prophylactic cefazolin 1 g was intravenously administered prior to each PEG. Additional spraying of lidocaine solution and epinephrine solution into the nasal cavity and using an ultrathin 5-mm endoscope (GIF-N-260; Olympus, Tokyo, Japan) were applied to the T-PEG. The other premedication and procedure were the same as those used in conventional pull-method PEG to insert a 20 Fr PEG tube (MIC PEG kits; Kimberly–Clark, Roswell, GA, USA) for enteral feeding. All of the patients were hospitalized for the PEG procedure, observed for 1–2 days in hospital after the PEG procedure, and followed-up in the outpatient department at 7 days after PEG. All of the adverse events and stomal site examination results were recorded routinely. A gastrostomy site infection was defined as wound erythema with discharge or pus discharge with/without wound erythema. Bacterial culture was obtained routinely if stomal site infection occurred.
The age, sex, cause of dysphagia, type of residence, comorbidity, details of the PEG including the route of insertion and the occurrence of choking or aspiration during PEG, cardiopulmonary function monitoring results, and post-PEG adverse events including gastrostomy site infection, bacterial pathogens, systemic infection, tube dislodgement or migration were also recorded and analyzed. The results were expressed as the mean (standard deviation) for quantitative variables and frequency for categorical variables. Normally distributed quantitative variables were analyzed by the Student t test. The categorical variables were analyzed using the χ2 test.
Results
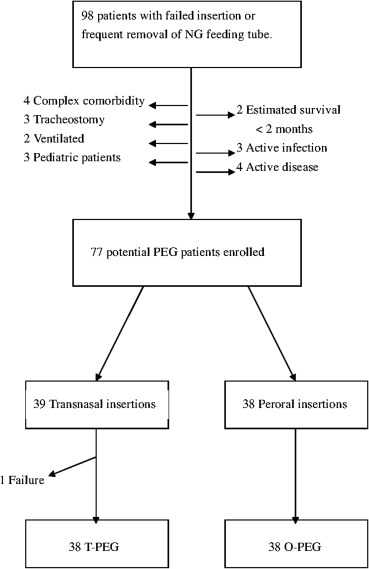
Ninety-eight patients with dysphagia who had removed their NG tube frequently or who experienced a difficult insertion of an NG tube, and therefore underwent PEG, were enrolled. Twenty-one participants were excluded; four with complex comorbidity, three with tracheostomy, two with mechanical ventilation, three aged < 20 years, two with an estimated survival time < 2 months, three with active infection, and four with active disease. Additionally, T-PEG failed in one patient. In all, 38 patients with dysphagia who underwent T-PEG and 38 who underwent O-EPG (from home or a nursing home) were included in the study (Fig. 1). Comparing the T-PEG and O-PEG patients, the mean age was 76.3 ± 10.3 years versus 79.3 ± 6.9 years, the percentage male patients was 67% versus 48%, length of surgery was 14.6 ± 4.0 minutes versus 11 ± 3 minutes (p = 0.0028), and choking occurred in three patients versus five patients (no occurrence of subsequent pneumonia). One systemic infection of the urinary tract, one buried bumper, and one soiling of the stoma were observed in both the T-PEG and O-PEG groups. No PEG-related mortality occurred within 3 months after PEG. There were 10 gastrostomy site infections in T-PEG and 14 stomal site infections in O-PEG. Although stomal site infection was less common with T-PEG than O-PEG (10/38 vs. 14/38; p < 0.001), for those patients in whom infection did occur, P. aeruginosa was more commonly associated with T-PEG than O-PEG (9/10 vs. 8/14; p < 0.001; Table 1. The pathogens of gastrostomy site infections by these two types of PEG are shown in Table 2. However, all the stomal site infections were managed uneventfully with dressing and medical therapy regardless of the pathogen.
|
|
|
Figure 1. Flow chart of the selection process for study participants. |
Table 1.
Characteristics and clinical outcomes of patients who underwent PEG.
| T-PEG (n = 38) | O-PEG (n = 38) | p | |
|---|---|---|---|
| Age | 76.3 (10.3) | 79.5 (6.9) | |
| Male | 67 | 48 | |
| Cause of dysphagia | |||
| Stroke | 21 | 20 | |
| Trismus | 5 | 0 | |
| Head and neck cancer | 3a | 4 | |
| Neurological disorder | 12 | 14 | |
| Residence | |||
| Home | 13 | 10 | |
| Nursing home | 25 | 28 | |
| Cardiopulmonary function | |||
| Pre-PEG heart rate | 91 (17) | 96 (14) | NS |
| Post-PEG heart rate | 93 (15) | 99 (15) | |
| Pre-PEG PaO2 | 96 (2) | 97 (2) | |
| Post-PEG PaO2 | 96 (2) | 97 (2) | |
| PEG | |||
| Operating time, min | 14.6 (4.0) | 11 (3.1) | 0.028 |
| Choking during PEG | 3 | 5 | |
| Bleeding event | 2 (epistaxis) | 0 | |
| Post-PEG adverse events | |||
| Stomal site infection | 10 (26.3) | 14 (36.8) | <0.001 |
| P. aeruginosa | 9/10 (90) | 8/14 (57) | <0.001 |
| Systemic infection | 1 | 1 | |
| Buried bumper | 1 | 1 | |
| PEG related mortality | 0 | 0 | |
Data are presented as %, n (%), or mean ± SD.
NS = not significant; O-PEG = oral PEG; PaO2 = pulse arterial oxygen saturation; PEG = percutaneous endoscopic gastronomy; T-PEG = transnasal PEG.
a. Three cases of head and neck cancer are included among the patients with trismus.
| T-PEG (n = 10) | DM | O-PEG (n = 14) | DM | |
|---|---|---|---|---|
| # 1 | Pseudomonas aeruginosa | No | P. aeruginosa + Enterococcus spp. | Yes |
| # 2 | P. aeruginosa + CNS Providencia rettgeri | Yes | P. aeruginosa + Klebsiella pneumoniae | No |
| # 3 | P. aeruginosa, Serratia marcescens | No | P. aeruginosa + K. pneumoniae'Citrobacter kosera | No |
| # 4 | P. aeruginosa Streptococcus viridans | Yes | P. aeruginosa | No |
| # 5 | P. aeruginosa | No | P. aeruginosa + K. pneumoniae'Morganella morganii spp. | No |
| # 6 | P. aeruginosa'K. pneumoniae + ORSA | No | P. aeruginosa | No |
| # 7 | P. aeruginosa | Yes | P. aeruginosa + Strep. viridans | No |
| # 8 | P. aeruginosa | No | P. aeruginosa | No |
| # 9 | P. aeruginosa'Strep. viridans + CNS | No | K. pneumoniae + Strep. viridans | No |
| #10 | CNS | No | Ser. marcescens | No |
| #11 | K. pneumoniae + Strep. viridans | No | ||
| #12 | K. pneumoniae + Enterococcus + Strep. viridans | No | ||
| #13 | K. pneumoniae + Strep. viridans | No | ||
| #14 | K. pneumoniae + Strep. viridans | Yes |
CNS = coagulase-negative Staphylococcus; DM = diabetes mellitus; ORSA = oxacillin-resistant Staphylococcus aureus.
Discussion
There are three methods of PEG tube insertion; pull, push, and introducer. The pull/push PEG techniques have the advantage of less tube dislodgement due to the bumper type device inside the stomach, but these methods are not aseptic procedures. Thus, local stomal infection is not unusual due to oropharyngeal contamination, and prophylactic broad-spectrum antibiotics are recommended [13]. The introducer method has lower wound infection because of its adherence to aseptic procedure but this method has the disadvantages of increased risk of bleeding, due to the large trocar and higher probability of catheter dislodgement if the balloon bumper is used [14].
Conventional PEG is a challenge for patients with dysphagia who have difficulty with oral intubation. In 1996, Counihan et al [5] reported the first successful T-PEG using a pull method in a patient with intermaxillary fixation. Additional studies then confirmed that the pull-method T-PEG using a smaller or ultrathin endoscope was a feasible and safe procedure [7], [8], [9], [10], [11] and [12]. The introducer technique was also applied to ultrathin T-PEG, with no local stomal infection noted because it is an aseptic procedure, but the long-term durability of a balloon-type internal binder was questionable given that tube displacement was observed in 47.8% and gastric contents leakage was noted in 9% of the patients undergoing this type of procedure [15]. As with conventional PEG, the majority of T-PEGs were performed using pull techniques because the procedure is simple and the internal binder is durable. Therefore, gastrostomy site infection is inevitable in T-PEG owing to the bacterial contamination in the nasopharyngeal cavity.
The present study is believed to be the first case–control study to compare T-PEG and O-PEG using the pull method. The significantly longer operating time in the T-PEG group compared with that of the O-PEG group is expected because T-PEG involves a longer intubation route and narrower space. Compared with the O-PEG group, local gastrostomy site infection was less common but the presence of P. aeruginosa in cases of stomal site infection was more common in the T-PEG group. A previous study indicated that bacterial colonization occurred more frequently in the oropharynx than in the nasal cavity; the oropharyngeal and nasal colonization rates were 68% and 19%, respectively [16]. This may explain why more frequent gastrostomy site infection was found in the O-PEG group compared with the T-PEG group. Earlier studies demonstrated that the oropharyngeal ecosystem changed significantly in tube-fed patients. Pathogenic bacteria were isolated from the oropharynx in 54% of NG tube-fed elderly patients, and the most frequent Gram-negative bacteria (GNB) isolated were Proteus spp. (21%) and P. aeruginosa (18%) [17]. GNB, including P. aeruginosa, colonized 64% of NG tube-fed elderly, and P. aeruginosa was isolated in 60% of the patients colonized with GNB. In orally fed elderly patients, only 8% were colonized with GNB, and no P. aeruginosa was isolated [18]. The lack of mechanical clearance of the mouth provided by chewing and swallowing [19], combined with the ability of P. aeruginosa to adhere and form a biofilm on plastic tubes [20] and [21] may explain the high prevalence of P. aeruginosa in tube-fed patients.
In our T-PEG group, the recent NG-intubated nostril was selected as the insertion route to ensure the passing of the endoscope and gastrostomy tube; this may be the other cause of dominant P. aeruginosa contamination, which then colonized the intubated nostril through the NG tube. Staphylococcus aureus was common in the nasal cavity, and the nose might carry methicillin-resistant S. aureus (MRSA) in hospital workers and patients [22]. Hull et al [23] reported that nasopharyngeal colonization by MRSA invariably led to stomal colonization after PEG; stomal site infection occurred in 26% of their cases, and 88% of these infections were caused by MRSA. However, MRSA was not evident in our T-PEG patients. The lower nasal MRSA carriage of our healthy residents at home or in nursing homes may explain the rare MRSA infections in our T-PEG patients, despite the fact that no decolonization of the nasal cavity was performed prior to PEG.
The main limitation of this study was that the follow-up period was only 1 week after PEG and patients visited the outpatient department if adverse events were noted. This case–control study suggests that T-PEG does not differ from O-PEG in terms of feasibility, safety, or PEG-related adverse events, and is associated with a lower rate of gastrostomy infection. However, when infection does occur, P. aeruginosa is the dominant pathogen in T-PEG. In conclusion, transnasal insertion of PEG using the pull method is a feasible and safe alternative when conventional pull-method PEG is not possible. However, P. aeruginosa infection is common when using T-PEG; therefore, more studies focused on the prophylaxis of T-PEG-associated P. aeruginosa infection are required.
Conflicts of interest
The author declares no conflict of interest.
References
- [1] M.W. Gauderer; Percutaneous endoscopic gastrostomy-20 years later: a historical perspective; J Pediatr Surg, 36 (2001), pp. 217–219
- [2] D.E. Larson, D.D. Burton, K.W. Schroeder, E.P. DiMagno; Percutaneous endoscopic gastrostomy. Indications, success, complications, and mortality in 314 consecutive patients; Gastroenterology, 93 (1987), pp. 48–52
- [3] J.L. Ponsky, M.W. Gauderer; Percutaneous endoscopic gastrostomy: indications, limitations, techniques, and results; World J Surg, 13 (1989), pp. 165–170
- [4] M.D. Grant, M.A. Rudberg, J.A. Brody; Gastrostomy placement and mortality among hospitalized Medicare beneficiaries; JAMA, 279 (1998), pp. 1973–1976
- [5] T. Counihan, L.M. Napolitano, S.O. Heard; Transnasal insertion of percutaneous endoscopic gastrostomy in a patient with intermaxillary fixation: case report; J Trauma, 41 (1996), pp. 530–532
- [6] A. Taller, E. Horvath, L. Harsanyi, Z. Balatoni, L. Ilias; Transnasal percutaneous endoscopic gastrostomy; Endoscopy, 29 (1997), p. 337
- [7] A. Lustberg, A.S. Fleisher, P.E. Darwin; Transnasal placement of percutaneous endoscopic gastrostomy with a pediatric endoscope in oropharyngeal obstruction; Am J Gastroenterol, 96 (2001), pp. 936–937
- [8] S. Roman, A. Pereira, L. Caruso, P. Sagnard, T. Ponchon, J. Dumortier; Transnasal percutaneous endoscopic gastrostomy; Gastroenterol Clin Biol, 25 (2001), pp. 106–107
- [9] A.M. Lustberg, P.E. Darwin; A pilot study of transnasal percutaneous endoscopic gastrostomy; Am J Gastroenterol, 97 (2002), pp. 1273–1274
- [10] J. Dumortier, M.G. Lapalus, A. Pereira, J.P. Lagarrigue, A. Chavaillon, T. Ponchon; Unsedated transnasal PEG placement; Gastrointest Endosc, 59 (2004), pp. 54–57
- [11] M.A. Vitale, G. Villotti, L. D'Alba, M.A. De Cesare, S. Frontespezi, G. Iacopini; Unsedated transnasal percutaneous endoscopic gastrostomy placement in selected patients; Endoscopy, 37 (2005), pp. 48–51
- [12] L.F. Lin, H.C. Shen; Unsedated transnasal percutaneous endoscopic gastrostomy carried out by a single physician; Dig Endosc, 25 (2013), pp. 130–135
- [13] L. Gossner, J. Keymling, E.G. Hahn, C. Ell; Antibiotics prophylaxis in percutaneous endoscopic gastrostomy (PEG): a prospective randomized clinical trial; Endoscopy, 31 (1999), pp. 119–124
- [14] H. Ogino, H. Akiho; Usefulness of percutaneous endoscopic gastrostomy for supportive therapy of advanced aerodigestive cancer; World J Gastrointest Pathophysiol, 15 (2013), pp. 119–125
- [15] H.S. Lee, C.H. Lim, E.Y. Park, W.H. Lee, J.H. No, B.Y. Jun, et al.; Usefulness of the introducer method for percutaneous endoscopic gastrostomy using ultrathin transnasal endoscopy; Surg Endosc, 28 (2014), pp. 603–606
- [16] S. Faias, M. Cravo, I. Claro, P. Lage, C. Nobre-Leitao; High rate of percutaneous endoscopic gastrostomy site infection due to oropharyngeal colonization; Dig Dis Sci, 51 (2006), pp. 2384–2388
- [17] A. Leibovitz, G. Plotnikov, B. Habot, M. Rosenberg, R. Segal; Pathogenic colonization of the oral flora in frail elderly patients fed by naso-gastric tube or percutaneous entero-gastric tube; J Gerontol Biol Sci Med Sci, 58A (2003), pp. 52–55
- [18] A. Leibovitz, M. Dan, J. Zinger, Y. Cameli, B. Habot, R. Segal; Pseudomonas aeruginosa and the oropharyngeal ecosystem of tube-fed patients; Emerg Infect Dis, 9 (2003), pp. 956–959
- [19] L.B. Palmer, K. Albulak, S. Fields, A.M. Filkin, S. Simon, G.C. Smaldone; Oral clearance and pathogenic oropharyngeal colonization in the elderly; Am J Respir Crit Care Med, 164 (2001), pp. 464–468
- [20] L. Martinez-Martinez, A. Pascual, E.J. Perea; Effect of three plastic catheters on survival and growth of Pseudomonas aeruginosa; J Hosp Infect, 16 (1990), pp. 311–318
- [21] L. Martinez-Martinez, A. Pascual, E.J. Perea; Kinetics of adherence of mucoid and non-mucoid Pseudomonas aeruginosa to plastic catheters; J Med Microbiol, 34 (1991), pp. 7–12
- [22] Z. Rashid, K. Farzana, A. Sattar, G. Murtaza; Prevalence of nasal Staphylococcus aureus and methicillin-resistant Staphylococcus aureus in hospital personnel and associated risk factors; Acta Pol Pharm, 69 (2012), pp. 985–991
- [23] M. Hull, A. Beane, J. Bowen, C. Settle; Methicillin-resistant Staphylococcus aureus infection of percutaneous endoscopic gastrostomy site; Aliment Pharmocol Ther, 15 (2001), pp. 1883–1888
Document information
Published on 01/01/2017
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?