ABSTRACT
Cardiovascular disease is a leading cause of death throughout the world. The demand for new therapeutic interventions is increasing. Although pharmacological and surgical interventions dramatically improve the quality of life of cardiovascular disease patients, cheaper and less invasive approaches are always preferable. Biomaterials, both natural and synthetic, exhibit great potential in cardiac repair and regeneration, either as a carrier for drug delivery or as an extracellular matrix substitute scaffold. In this review, we discuss the current treatment options for several cardiovascular diseases, as well as types of biomaterials that have been investigated as potential therapeutic interventions for said diseases. We especially highlight investigations into the possible use of conductive polymers for correcting ischemic heart disease-induced conduction abnormalities, and the generation of biological pacemakers to improve the conduction pathway in heart block.
Keywords
Myocardial infarction ; Heart regeneration ; Biomaterial ; Tissue engineering ; Stem cell
1. Introduction
According to the Public Health Agency of Canadas Economic Burden of Illness in Canada report, cardiovascular disease-related physician care, hospital care, and drugs cost 11.7 billion Canadian dollar in 2008, and was the largest burden on the health system [ 1 ]. Likewise, cardiovascular disease is the leading cause of morbidity and mortality worldwide, and significantly impacts quality of life [ 2 ]. Currently, there is no cure for key cardiovascular diseases such as myocardial infarction (MI) and arrhythmia, as the heart has a very limited innate capacity for repair. Available therapies such as pharmacological interventions, coronary artery bypass graft surgery, and ventricular assistant devices have significantly improved patients' quality of life and prolonged their longevity [ 3 , 4 and 5 ]. However, new therapeutic interventions with lower cost and higher efficacy are still required for the future.
The use of regenerative medicine to treat cardiovascular diseases has emerged as a research topic in the past few decades. Furthermore, recent advances in genomics and molecular medicine have brought the concept of personalized treatment for cardiovascular disease to the fore [ 6 ]. The advent of human-induced pluripotent stem cells (iPSCs) opens a new chapter for regenerative medicine because they are a potential cardiomyocyte source [ 7 ]. Indeed, human iPSC-derived cardiomyocytes have been engineered into cell sheets, which significantly improved cardiac function when implanted into infarcted rat hearts [ 8 ]. iPSC-derived cardiomyocytes have also been used to study cardiac ion channel pathology and arrhythmia pathogenesis mechanisms in vitro [ 9 ]. However, the limited survivability of implanted stem cells remains a major obstacle to cardiac regeneration.
Bioengineering has the capacity to alleviate this problem. Biomaterials can be delivered alone, or serve as a scaffold or carrier for cells or growth factors [ 10 , 11 and 12 ]. The therapeutic potential of both natural and synthetic biomaterials has been demonstrated in animal models of MI and peripheral artery disease (PAD) [ 13 ]. Specifically, the application of injectable biomaterials results in decreased left ventricular wall dilatation, increased angiogenesis, enhanced endogenous tissue repair via endogenous stem cell recruitment, and the preservation of cardiac function [ 12 ,14 ].
In this review, we focus on cardiovascular diseases that induce conduction abnormalities and their current interventions; biomaterial types that have been investigated in cardiovascular research; and the potential of these biomaterials for future therapeutic intervention. Since current research focuses largely on improving cardiac function and cardiac tissue regeneration, relatively little has been done in terms of evaluating conductive polymer potential for correcting cardiac event-related arrhythmias and conduction block in vivo . We thus specifically explore the potential of conductive polymers to correct arrhythmias and improve conductivity in the infarcted heart. Future challenges in translating conductive polymer work from bench to bedside will be examined as well.
2. Regenerating the injured heart
2.1. Cell therapy treatment of ischemic heart disease
Coronary artery disease (CAD) is the most common type of heart disease and the leading cause of death around the world [ 2 ]. Coronary arteries supply oxygenated blood and nutrients to the heart muscle. CAD is caused by the deposition of cholesterol-containing plaques in the arterial wall, resulting in vessel wall-thickening and narrowing of the vascular lumen. The narrowing of coronary arteries leads to decreased blood flow and causes cardiomyocyte ischemic injury. Patients with decreased blood flow to the heart experience chest pain (angina), shortness of breath, and limited exercise capacity. A complete blockage of the vessel causes MI, leading to cell necrosis and massive loss of cardiomyocytes [ 15 ]. The current standard intervention for ischemic heart disease centers around lifestyle modification, pharmacological interventions, and surgical interventions [ 16 ]. Patients who do not respond well to all available treatments may progressively develop heart failure, which requires a heart transplant.
Cell therapy (in which cellular material is injected into animals or patients) has emerged as a potential strategy for treating MI. Multiple candidate cell types have been proposed and investigated, such as bone marrow mononuclear cells [ 17 ,18 ], embryonic stem cells [ 19 ,20 ], skeletal myoblasts [ 21 ,22 ], endothelial progenitor cells [ 23 ], and cardiac stem cells [ 24 ,25 ]. Although improved cardiac function was observed in both animal and human studies [ 26 , 27 and 28 ], several obstacles remain, such as low cell retention/engraftment, cell delivery efficiency, electromechanical integration, and long-term safety [ 29 , 30 , 31 , 32 and 33 ]. Improving these elements is critical for future cell therapy research. Biomaterials may be the answer, as they serve as excellent scaffolds for cell delivery and tissue engineering. Cells can be either mixed with biomaterials and delivered to the injured site directly or cultured on biomaterial-based scaffolds for future transplantation [ 34 ,35 ].
2.2. Biomaterials in cardiac repair and regeneration
In order for biomaterials to be feasible for cardiac repair, they must be biocompatible, be biodegradable, reduce local microenvironment hostility, persist for a sufficient time period to facilitate cell engraftment and integration with native tissue, and act as a reservoir for the slow release of bioactive molecules [ 36 , 37 and 38 ]. Both natural (gelatin [ 39 ], collagen [ 40 ], alginate [ 41 ,42 ], chitosan [ 43 ], and fibrin glue [ 44 ]) and synthetic (poly(lactic-co -glycolic acid) or PLGA [ 45 ], carbon nanotubes [ 46 ], and polyurethane [ 47 ]) biomaterials have been investigated as candidates for cardiac regeneration [ 37 ].
2.3. Natural biomaterials
A biomaterial can be introduced to the heart as an injectable hydrogel or a patch [ 48 ]. Collagen, a major component of the extracellular matrix (ECM), is one of the popular natural materials used in cardiac repair. Miyagi et al. [ 10 ] investigated the effect of a collagen patch as a slow-release reservoir of the vascular endothelial growth factor (VEGF)-165 to promote vascularization in a right ventricle defect rat model. They found that the collagen patch with immobilized VEGF enhanced the growth of endothelial and bone marrow cells in vitro . A VEGF-collagen patch, when implanted onto a defective right ventricular free wall of the heart, resulted in elevated angiogenesis and ventricular wall thickness when compared with a control collagen-only patch. These findings indicate that growth factor immobilization onto a collagen patch improves cardiac repair via the promotion of cell recruitment and proliferation [ 10 ]. A collagen patch alone has been shown to preserve infarcted heart contractility, attenuate adverse remodeling, and improve heart function. It did this by diminishing infarct region fibrosis, supporting blood vessel formation between the infarct region and the patch, and attracting various native cells (e.g., smooth muscle cells, epicardial cells, and immature cardiomyocytes) to populate the patch [ 49 ]. Collagen is also used as a cell carrier to deliver various cell types to infarct regions after infarction. Frederick et al. [ 50 ] have shown that vitronectin/collagen scaffold seeded with endothelial progenitor cells induced neovasculogenesis and preserved ventricular function post-MI in a rat.
Chitosan is the second most abundant polysaccharide in nature, and has been widely used in agriculture, food and nutrition, environmental protection, and material sciences [ 51 ]. The chitosan gel has a porous sponge structure [ 52 ], and can be used as a cell scaffold [ 53 ] or carrier for controlled and localized drug delivery [ 54 ]. Liu et al. [ 43 ] have shown that an injectable chitosan hydrogel enhanced stem cell engraftment and survival by improving oxidative stress in the ischemic heart through reactive oxygen species scavenging and chemokine recruitment. A chitosan-collagen hydrogel has also been used to deliver an angiopoietin-1 analogue to enhance endothelial cell function and survival. Indeed, the angiopoietin-1 analogue-bound hydrogel mitigated endothelial cell apoptosis and stimulated endothelial cell tube-like structure formation [ 55 ]. Finally, implantation of a chitosan-hyaluronan/silk fibroin patch in an infarcted rat left ventricle (LV) resulted in reduced LV dilatation, increased wall thickness, and improved heart function [ 56 ].
Aside from collagen and chitosan, other natural biomaterials have also been investigated as candidates for cardiac regeneration therapy. Alginate is a polysaccharide derived from seaweed [ 57 ] and has been extensively investigated in wound healing, drug delivery, and tissue engineering [ 58 ]. Research investigating the effect of alginate implantation in MI models has shown that an injectable aliginate-hydrogel injected into the myocardium can be completely absorbed and replaced by connective tissue within 6 weeks. It was effective in reinforcing scar thickness, attenuating adverse ventricular dilatation, and improving cardiac function, whether injected 7 days or 60 days after MI [ 41 ]. These results could be a useful reference for administration timing in future studies. The peri-infarct zone injection of an alginate-chitosan hydrogel was also shown to be effective in preventing LV remodeling and promoting tissue repair by reducing cell apoptosis and increasing angiogenesis in an acute MI rat model [ 59 ]. Fibrin glue, which is primarily made of fibrinogen and thrombin, is used to create a fibrin clot. It is usually used to repair left ventricular wall rupture secondary to acute MI. The glue can seal the ruptured myocardium immediately and be naturally absorbed [ 60 , 61 and 62 ]. Christman et al. [ 44 ] investigated the effect of fibrin glue scaffold implantation on cardiac function preservation post-MI. Scaffold implantation resulted in the preservation of infarct wall thickness and cardiac function 5 weeks after injection in a rat model of ischemia-reperfusion injury [ 44 ].
2.4. Synthetic biomaterials
Synthetic biomaterials are generally constructed from synthetic polymers, metals, or a combination of both. Synthetic biomaterials possess excellent strength and durability, but present increased toxicity, creating biocompatibility issues [ 48 ]. Like their natural counterparts, synthetic biomaterials are generated and used with the aim of serving as a reservoir for gradual and controlled drug delivery, and acting as a scaffold to support cell engraftment and integration.
Polylactic acid (PLA), poly-glycolide (PLG), and their copolymer PLGA are commonly used synthetic materials. Mukherjee et al. [ 63 ] employed a nanostructured matrix made of poly(L -lactic acid), poly(ε-caprolactone), and collagen to mimic the native microenvironment of the myocardium. A nanoscale PLA-co-poly(ε-caprolactone)/collagen biocomposite scaffold was used to culture and support isolated rabbit cardiomyocytes. The results showed that adult rabbit cardiomyocytes attached to the scaffold exhibited growth and cell organization comparable to that found in native myocardium [ 63 ]. Insulin-like growth factor (IGF)-1 bound to PLGA nanoparticles was delivered to the peri-infarcted area immediately after MI. The results showed that IGF-1-complexed PLGA nanoparticles prolonged IGF-1 retention in the tissue, reduced cardiomyocyte apoptosis, and improved LV function [ 45 ].
Carbon nanofibers have also been investigated for cardiac engineering. Martins et al. [ 64 ] mixed conductive carbon nanotubes with chitosan to form a chitosan/carbon scaffold with similar elastic properties to native myocardium. This conductive scaffold not only supported neonatal rat cardiomyocytes survival in vitro, but also increased the expression of myosin heavy chain, troponin T, and connexin-43, which are involved in muscle contraction and electrical coupling and are important for the organization of cell-cell electrical signal transmission [ 64 ]. Zhou et al. [ 65 ] also developed a carbon nanofiber/gelatin hydrogel scaffold that supported in vitro culture of neonatal rat cardiac cells and integrated with the host myocardium when implanted post-MI in the rat heart. Heart function as assessed by fractional shortening and ejection fraction evaluated by echocardiography was improved after hydrogel injection while progression of pathological deterioration (e.g., ventricular dilation) was inhibited [ 65 ].
Synthetic peptide-based biomaterials have also been investigated. Synthetic peptides are able to self-assemble into three-dimensional (3D) hydrogels with adjustable fiber dimensional properties and potential nanoscaling [ 66 ]. They can be directly injected into myocardium to improve the microenvironment for cell recruitment and survival [ 67 ], but also can act as a carriers for drug and cell delivery [ 68 ]. Tokunaga et al. [ 69 ] used the self-assembling peptide Puramatrix to deliver cardiac progenitor cells into border-zone myocardium after MI to ensure effective cell delivery and improve cell survival. In addition, self-assembling peptide nanofibers were used to deliver platelet-derived growth factor (PDGF) in a sustained manner in order to protect cardiomyocytes from apoptosis and preserve cardiac function post-MI [ 70 ].
Polyurethane, a durable and flexible synthetic polymer, was synthesized into an elastomeric patterned film to match the physical and mechanical properties of native cardiac tissue and investigated as a scaffold for tissue engineering. Cardiomyocytes seeded on patterned polyurethane film were able to grow following the film pattern and form a multilayered contractile tissue construct [ 71 ].
Despite the fact that clinical trials examining the application of biomaterial to cardiac diseases are scarce, a recent clinical study did report the clinical outcomes following 6 months of follow-up [ 72 ]. The authors found that alginate-hydrogel in addition to standard medical therapy (SMT) for patients with advanced chronic heart failure (HF) was more effective than SMT alone for improving exercise capacity and symptoms. The results from a year of extended follow-up for this clinical trial were also recently reported [ 73 ]. It was shown that alginate-hydrogel in addition to SMT was more effective than SMT alone for providing sustained 1-year benefits in exercise capacity, symptoms, and clinical status for patients with advanced HF. These data support further larger clinical evaluations of this novel therapy.
3. Conduction abnormalities and arrhythmia
3.1. Ventricular arrhythmias
MI is often associated with increased incidence of arrhythmias, particularly ventricular arrhythmias [ 74 ,75 ]. Due to the limited regenerative ability of the heart, massive loss of cardiomyocytes post-MI leads to the formation of scar tissue, which is primarily composed of fibroblasts and collagen. Scar tissue has significantly decreased contractility and electrical signal conduction, leading to the formation of abnormal conduction pathways and the development of conduction block. Ventricular arrhythmias, either ventricular tachycardia (VT) or ventricular fibrillation (VF), are very common in MI patients, and are the major cause of sudden cardiac death (SCD) [ 76 ]. In acute MI, the ischemic cardiomyocytes in and around the infarct zone have altered electrophysiological properties, which serve as a potential risk for developing re-entrant loop (a circular electrical pathway), a mechanism for sustaining ventricular arrhythmias. As the MI passes the acute phase and the scar starts to stabilize, the scar tissue actually serves as an anatomical block promoting the development of re-entrant loop, which is a trigger for SCD [ 76 ]. Although medical interventions significantly decrease morbidity and mortality, SCD accounts for 70% of deaths of outpatients with CAD and more than 50% of deaths in the total patient population [ 77 ,78 ]. Dyssynchronized contraction (a difference in the timing, or lack of synchrony, of ventricular contraction in the heart) resulting from VF leads to decreased hemodynamics, circulatory collapse, and the loss of cerebral function.
Current therapies for ventricular arrhythmias are based on an established understanding of arrhythmia triggers, such as electrolyte imbalance [ 79 ], anatomical changes, ischemia [ 74 ], hypoxia [ 80 ], and physical and mental exertion [ 81 ]. The primary goal of current therapy is to restore normal heart rhythm. Immediate defibrillation and cardioversion is required for VF or pulseless sustained VT. The early administration of β-blockers (drugs designed to stabilize cell membranes and minimize the overall metabolic demands of the heart) is associated with a reduced incidence of VF [ 82 ,83 ]. Another clinical intervention is the use of an implantable cardioverter defibrillator (ICD), an electrical device designed to stop fibrillation and restore normal rhythm, which has been routinely used in patients with cardiomyopathy and survivors of sudden cardiac arrest (SCA) [ 78 ,84 ]. Despite the clinical use of ICDs for terminating arrhythmias in patients, VT or VF can still occur, and are associated with increased mortality and HF risk [ 85 ].
3.2. Atrioventricular (AV) block
Heart rate and cardiac contraction is controlled by the sinoatrial (SA) node located in the right atrial wall, which is a natural pacemaker that generates and sends electrical impulses through the atria. This signal is then slowed by the atrioventricular (AV) node, located in the inferior posterior region of the interatrial septum, before reaching the ventricles to allow for complete atrial contraction. The impulse then reaches the ventricles via the bundle of His, which branches into right and left bundle branches and terminates in the Purkinje fiber system, facilitating synchronized ventricular contraction [ 86 ]. An abnormality in any of these components will cause a conduction disorder, which affects the pumping function of the heart [ 87 ].
AV block is a conduction disease that occurs when the electrical signal from the atria is partially or completely blocked by the AV node so that ventricular rhythm is decreased. Conditions such as neuromuscular disorders (e.g., muscular dystrophy) [ 88 ,89 ], systemic diseases (e.g., cardiac sarcoidosis [ 90 ] and amyloidosis [ 91 ]), myocardial ischemia/infarction [ 92 ,93 ], neoplastic disorders (e.g., primary cardiac lymphoma [ 94 ]), and catheter ablation [ 95 ,96 ] can all cause abnormal AV conduction [ 86 ]. Disruption of electrical signal conduction can occur at the AV node and at the infra- or supra-AV node structures, as well as at the bundle of His and the bifurcation of the left and right bundle [ 97 , 98 , 99 and 100 ]. The consequence is that the atrial electrical impulse is either delayed or only partially propagated to the ventricles, which potentially results in HF and SCD if untreated [ 101 ].
Currently employed treatments for AV block vary depending on the etiology and symptomatology. While asymptomatic AV block requires no intervention, symptomatic AV block (either innate or exacerbated during exercise) is treated with pacemaker implantation [ 100 ,101 ]. Indeed, permanent cardiac pacing is recommended in patients with AV block unless there is a reversible cause of AV block or a contraindication for pacemaker implantation [ 101 ].
Improving cardiac function and controlling arrhythmias are equally important. The efficacy of current therapies is limited by the different disease presentation of patients [ 102 ]. Therefore, new therapies are required to terminate abnormal conduction pathways and improve patients' survival. Conduction disorders, such as AV block and ventricular arrhythmias, may benefit from conductive biomaterials−polymers that exhibit high electrical conduction. Preliminary investigations have already revealed that conductive biomaterials have great potential for improving conduction abnormalities induced by ischemic heart diseases and conduction block.
3.3. Conductive biomaterials
Polyacetylene, an organic conductive polymer, was first discovered and described in 1977 by Alan J. Heeger, Alan G. MacDiarmid, and Hideki Shirakawa. This discovery was recognized by the Nobel Prize in 2000 [ 103 ]. Conductive polymers are appealing not only because they exhibit electrical properties, but also because they are easy to synthesize, and are also biocompatible and biodegradable [ 104 ]. Conductive polymers have been used successfully as materials for biosensors, neural implants, drug delivery devices, and tissue-engineering scaffolds [ 105 ]. The conductivity of these polymers comes from their unique structure: a conjugated backbone formed by a series of alternating single and double bonds. These alternating bonds allow electrons to move freely between and within the chains [ 106 ]. The ability of conductive polymers to promote the generation of electrically active tissues, such as nerve tissue and heart tissue, has also been investigated [ 107 ,108 ].
There are many conductive polymers, such as polypyrrole (PPy), polyaniline (PANI), poly(3,4-ethylenedioxythiophene) (PEDT, PEDOT), polyazulene (PAz), and polythiophene derivatives [ 105 ]. Of these, PPy is one of the better-characterized polymers, having been extensively investigated in neuroscience and cardiac research.
George et al. [ 109 ] generated a PPy-containing conductive implant designed for neural prosthetics to guide neural stem cell (NSC) differentiation and neurite elongation. They found that PPy-containing conductive implants promoted the growth of neurons and glial cells around the implant when sutured in a rat cerebral cortex [ 109 ]. A conductive laminin/PPy film was able to guide human embryonic stem cells into neural lineages for nerve regeneration and repair [ 110 ]. In addition, PPy mixed with the counter-ion of dodecylbenzenesulfonate (DBS) was found to support the survival of NSCs and guide NSC differentiation toward neural lineages [ 111 ].
Potential applications for PPy in cardiac research have also been investigated. Kai et al. [ 112 ] created a nanofibrous membrane, composed of PPy, poly(ε-caprolactone), and gelatin, designed to mimic the ECM, for cardiac tissue engineering. They demonstrated that this membrane promoted human cardiac myocyte attachment, proliferation, interaction, and expression of cardiac-specific proteins [ 112 ]. A 3D PPy-coated PLGA polymer scaffold has been generated for potential stem cell transplant after MI. This conductive polymer scaffold is biocompatible, supporting the growth and proliferation of both cardiac progenitor cells and iPSCs. In addition, the PPy-coated PLGA scaffold could be electrically stimulated to modulate cell behavior [ 113 ]. PPy has also been shown to stimulate angiogenesis post-MI. Mihardja et al. [ 114 ] generated an alginate-PPy polymer and injected it into the infarct area, where increased angiogenesis was observed compared with saline-treated animals 5 weeks post-injection. In addition, myofibroblast infiltration was also increased in the infarct area [ 114 ].
To summarize, PPy conductive polymers not only provide suitable ECM support for cardiac cells, but also affect the biological behavior of cells. In tissue engineering combined with cell therapy, a conductive polymer-based cardiac tissue construct could be implanted in the heart post-MI to facilitate electrical signal conduction and integrate implanted cells with host cells.
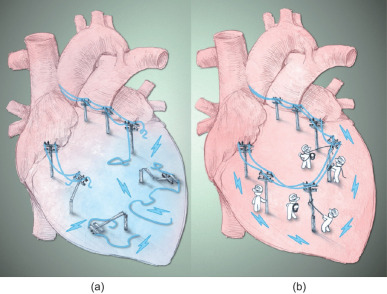
Another potential application of conductive polymers is as a biological pacemaker. Patients with conduction block or HF are treated by the implantation of an electronic pacemaker, which controls heart rhythm and contraction in order to maintain heart pumping function [ 115 ]. Although such devices have shown potential in reducing mortality and hospitalization of patients, the fundamental problem−conduction block−is not yet solved because non-conductive tissue or an abnormal conductive pathway still exists in the heart. Research interest in developing a biological pacemaker began two decades ago in response to challenges and limitations facing electronic pacemakers, such as battery changes, chronic infection, high surgical cost, and device adaptation in developing pediatric patients (e.g., chest and vascular size, child growth, and congenital heart defects) [ 116 , 117 and 118 ]. In a normal heart, the SA node is the trigger that sends an electrical signal to the substrate−cells that sense and receive this signal. A trigger-substrate connection is critical for organized pacing and conduction. A biological pacemaker would be able to integrate with the heart, and respond to endogenous stimuli calling for increased/decreased cardiac activity [ 119 ]. In addition, implantation of a biological pacemaker would be minimally invasive, which is suitable for patients who have contraindications for electronic pacemakers [ 116 ]. Biomaterials play a role in biological pacemaker research by establishing communications between implanted pacemaker cells and their substrate cells, integrating implanted cells, and labeling and tracking transplanted cells in vivo [ 120 , 121 and 122 ]. Conductive polymers, as excellent biomaterial candidates, can act as an ECM to support cell survival while establishing electrical connections between cells (Fig. 1 ). As such, the development of a conductive polymer-pacemaker cell combination has great potential to be the first step in creating a biological pacemaker to be used in future therapeutics.
|
|
|
Fig. 1. Reconnecting disrupted electrical signals in the infarcted heart. (a) Myocardial infarction induces a massive loss of cardiomyocytes, leading to scar tissue formation. Scar tissue disrupts electrical signal propagation, predisposing patients to increased risk of arrhythmia development. (b) The introduction of conductive polymers can reconnect damaged areas with existing healthy myocardium, thus repairing disrupted electrical conduction. |
4. Future challenges and directions
Biomaterials have great potential in cardiac repair and regeneration. The biocompatibility and biodegradation rate of most available biomaterials have been tested in different approaches by different groups. The future of biomaterials and tissue engineering is moving toward finding the ideal biomaterial-cell type combination for tissue regeneration. Selection of optimal cell types/combinations is vital for the success of tissue engineering, as the heart is a carefully balanced milieu of cardiomyocytes, fibroblasts, endothelial cells, and smooth muscle cells. Many questions remain to be answered. The major concern of biomaterial application to cardiac disease is the possibility of creating a substrate for arrhythmia. Although there are few studies that specifically address this risk, a recent study did reveal that the degree of the electrophysiological changes depends on the spread characteristics of the biomaterials [ 123 ]. This study found that hearts injected with highly spread biomaterials showed no conduction abnormalities. However, the injection of a biomaterial exhibiting minimal interstitial spread may create a substrate for arrhythmia shortly after injection by causing LV activation delays and reducing gap junction density at the site of the injection. This work demonstrates that the site of delivery and interstitial spread characteristics are important factors in biomaterial application-associated arrhythmias. In addition, generating biomaterials with elastic and strength properties similar to the native myocardium is critical [ 124 ]. Minimizing the immune response to prevent the encapsulation of biomaterials or tissue constructs is another concern, as encapsulation prevents proper integration of transplanted cells with the native environment [ 125 ]. Since the heart is the largest bioelectrical source in the body [ 126 ], synthesizing conductive polymers, which may facilitate the synchronous beating of cardiomyocytes, will enhance communications between transplanted tissue constructs and the native myocardium. This will be critical, as electrical field stimulation increases protein organization, facilitates cell polarization, and enhances electrical signal propagation [ 107 ]. Finally, the optimal timing for introducing biomaterials or tissue constructs remains unclear, and is vital for potentially minimizing inflammatory responses while limiting scar tissue formation.
Despite these challenges, biomaterial and tissue-engineering research holds great promise and has achieved much progress within the past two decades. The ultimate goal is to use biomaterial scaffolds in combination with appropriate cell types for partial or whole organ (re)generation. This will ultimately reduce the need for organ transplant, correct currently irreversible pathologies, and improve quality of life.
Acknowledgements
The authors thank Dr. Leigh Botly for help with manuscript preparation and editing. The illustration shown in Fig. 1 was conceived by Prof. Ren-Ke Li, with image art created by Benjamin Pakuts, Research Communications Officer at the University Health Network. This work was supported by a grant from the Heart and Stroke Foundation of Ontario (No. G140005765) to Ren-Ke Li. Ren-Ke Li holds a Canada Research Chair in cardiac regeneration. This work was also supported by a generous contribution by Eileen Mercier.
Compliance with ethics guidelines
Zhi Cui, Baofeng Yang, and Ren-Ke Li declare that they have no conflict of interest or financial conflicts to disclose.
References
- 1 Public Health Agency of Canada [Internet]; Economic burden of illness in Canada, 2005–2008. [2014-06-20]; Available from: http://www.phac-aspc.gc.ca/publicat/ebic-femc/2005-2008/index-eng.php
- 2 AS Go, D Mozaffarian, VL Roger, EJ Benjamin, JD Berry, MJ Blaha, American Heart Association Statistics Committee, Stroke Statistics Subcommittee, et al.; Executive summary: heart disease and stroke statistics–2014 update: a report from the American Heart Association; Circulation, 129 (3) (2014), pp. 399–410
- 3 JW Creaser, EC DePasquale, E Vandenbogaart, D Rourke, T Chaker, GC Fonarow; Team-based care for outpatients with heart failure; Heart Fail Clin, 11 (3) (2015), pp. 379–405
- 4 P Heidenreich; Heart failure prevention and team-based interventions; Heart Fail Clin, 11 (3) (2015), pp. 349–358
- 5 PM Larsen, JR Teerlink; Team-based care for patients hospitalized with heart failure; Heart Fail Clin, 11 (3) (2015), pp. 359–370
- 6 FS Collins, H Varmus; A new initiative on precision medicine; N Engl J Med, 372 (9) (2015), pp. 793–795
- 7 J Zhang, GF Wilson, AG Soerens, CH Koonce, J Yu, SP Palecek, et al.; Functional cardiomyocytes derived from human induced pluripotent stem cells; Circ Res, 104 (4) (2009), pp. e30–e41
- 8 H Masumoto, T Ikuno, M Takeda, H Fukushima, A Marui, S Katayama, et al.; Human iPS cell-engineered cardiac tissue sheets with cardiomyocytes and vascular cells for cardiac regeneration; Sci Rep, 4 (2014), p. 6716
- 9 CY Ivashchenko, GC Pipes, IM Lozinskaya, ZJ Lin, XP Xu, S Needle, et al.; Human-induced pluripotent stem cell-derived cardiomyocytes exhibit temporal changes in phenotype; Am J Physiol Heart Circ Physiol, 305 (6) (2013), pp. H913–H922
- 10 Y Miyagi, LL Chiu, M Cimini, RD Weisel, M Radisic, RK Li; Biodegradable collagen patch with covalently immobilized VEGF for myocardial repair; Biomaterials, 32 (5) (2011), pp. 1280–1290
- 11 VF Segers, RT Lee; Biomaterials to enhance stem cell function in the heart; Circ Res, 109 (8) (2011), pp. 910–922
- 12 K Cheng, K Malliaras, DL Shen, E Tseliou, V Ionta, J Smith, et al.; Intramyocardial injection of platelet gel promotes endogenous repair and augments cardiac function in rats with myocardial infarction; J Am Coll Cardiol, 59 (3) (2012), pp. 256–264
- 13 JL Ungerleider, KL Christman; Concise review: injectable biomaterials for the treatment of myocardial infarction and peripheral artery disease: translational challenges and progress; Stem Cells Transl Med, 3 (9) (2014), pp. 1090–1099
- 14 DL Shen, XF Wang, L Zhang, XY Zhao, JY Li, K Cheng, et al.; The amelioration of cardiac dysfunction after myocardial infarction by the injection of keratin biomaterials derived from human hair; Biomaterials, 32 (35) (2011), pp. 9290–9299
- 15 C de Zwaan, MJ Daemen, WT Hermens; Mechanisms of cell death in acute myocardial infarction: pathophysiological implications for treatment; Neth Heart J, 9 (1) (2001), pp. 30–44
- 16 PT O'Gara, FG Kushner, DD Ascheim, DE Casey, MK Chung, JA de Lemos, American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, et al.; 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines; Circulation, 127 (4) (2013), pp. e362–e425
- 17 V Schächinger, S Erbs, A Elsasser, W Haberbosch, R Hambrecht, H Holschermann, REPAIR-AMI Investigators; Improved clinical outcome after intracoronary administration of bone-marrow-derived progenitor cells in acute myocardial infarction: final 1-year results of the REPAIR-AMI trial; Eur Heart J, 27 (23) (2006), pp. 2775–2783
- 18 KC Wollert, GP Meyer, J Lotz, S Ringes-Lichtenberg, P Lippolt, C Breidenbach, et al.; Intracoronary autologous bone-marrow cell transfer after myocardial infarction: the BOOST randomised controlled clinical trial; Lancet, 364 (9429) (2004), pp. 141–148
- 19 O Caspi, I Huber, M Habib, G Arbel, A Gepstein, L Yankelson, et al.; Transplantation of human embryonic stem cell-derived cardiomyocytes improves myocardial performance in infarcted rat hearts; J Am Coll Cardiol, 50 (19) (2007), pp. 1884–1893
- 20 MA Laflamme, KY Chen, A Naumova, V Muskheli, JA Fugate, SK Dupras, et al.; Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts; Nat Biotechnol, 25 (9) (2007), pp. 1015–1024
- 21 P Menasché, O Alfieri, S Janssens, W McKenna, H Reichenspumer, L Trinquart, et al.; The Myoblast Autologous Grafting in Ischemic Cardiomyopathy (MAGIC) trial: first randomized placebo-controlled study of myoblast transplantation; Circulation, 117 (9) (2008), pp. 1189–1200
- 22 P Menasché; Skeletal myoblasts as a therapeutic agent; Prog Cardiovasc Dis, 50 (1) (2007), pp. 7–17
- 23 DG Katritsis, PA Sotiropoulou, E Karvouni, I Karabinos, S Korovesis, SA Perez, et al.; Transcoronary transplantation of autologous mesenchymal stem cells and endothelial progenitors into infarcted human myocardium; Catheter Cardiovasc Interv, 65 (3) (2005), pp. 321–329
- 24 RR Smith, L Barile, HC Cho, MK Leppo, JM Hare, E Messina, et al.; Regenerative potential of cardiosphere-derived cells expanded from percutaneous endomyocardial biopsy specimens; Circulation, 115 (7) (2007), pp. 896–908
- 25 R Bolli, AR Chugh, D D'Amario, JH Loughran, MF Stoddard, S Ikram, et al.; Cardiac stem cells in patients with ischaemic cardiomyopathy (SCIPIO): initial results of a randomised phase 1 trial; Lancet, 78 (9806) (2011), pp. 1847–1857
- 26 JM Hare, JE Fishman, G Gerstenblith, DL DiFede Velazquez, JP Zambrano, VY Suncion, et al.; Comparison of allogeneic vs autologous bone marrow–derived mesenchymal stem cells delivered by transendocardial injection in patients with ischemic cardiomyopathy: the POSEIDON randomized trial; JAMA, 308 (22) (2012), pp. 2369–2379
- 27 JJ Chong, X Yang, CW Don, E Minami, YW Liu, JJ Weyers, et al.; Human embryonic-stem-cell-derived cardiomyocytes regenerate non-human primate hearts; Nature, 510 (7504) (2014), pp. 273–277
- 28 AR Williams, KE Hatzistergos, B Addicott, F McCall, D Carvalho, V Suncion, et al.; Enhanced effect of human cardiac stem cells and bone marrow mesenchymal stem cells to reduce infarct size and restore cardiac function after myocardial infarction; Circulation, 127 (2) (2013), pp. 213–223
- 29 VF Segers, RT Lee; Stem-cell therapy for cardiac disease; Nature, 451 (7181) (2008), pp. 937–942
- 30 D Hou, EA Youssef, TJ Brinton, P Zhang, P Rogers, ET Price, et al.; Radiolabeled cell distribution after intramyocardial, intracoronary, and interstitial retrograde coronary venous delivery: implications for current clinical trials; Circulation, 112 (9 Suppl) (2005), pp. I150–I156
- 31 T Siminiak, R Kalawski, D Fiszer, O Jerzykowska, J Rzezniczak, N Rozwadowska, et al.; Autologous skeletal myoblast transplantation for the treatment of postinfarction myocardial injury: Phase I clinical study with 12 months of follow-up; Am Heart J, 148 (3) (2004), pp. 531–537
- 32 MG Chang, L Tung, RB Sekar, CY Chang, J Cysyk, P Dong, et al.; Proarrhythmic potential of mesenchymal stem cell transplantation revealed in an in vitro coculture model; Circulation, 113 (15) (2006), pp. 1832–1841
- 33 E Macia, PA Boyden; Stem cell therapy is proarrhythmic; Circulation, 119 (13) (2009), pp. 1814–1823
- 34 D Seliktar; Designing cell-compatible hydrogels for biomedical applications; Science, 336 (6085) (2012), pp. 1124–1128
- 35 GD Prestwich; Hyaluronic acid-based clinical biomaterials derived for cell and molecule delivery in regenerative medicine; J Control Release, 155 (2) (2011), pp. 193–199
- 36 S Pascual-Gil, E Garbayo, P Díaz-Herráez, F Prosper, MJ Blanco-Prieto; Heart regeneration after myocardial infarction using synthetic biomaterials; J Control Release, 203 (2015), pp. 23–38
- 37 P Zammaretti, M Jaconi; Cardiac tissue engineering: regeneration of the wounded heart; Curr Opin Biotechnol, 15 (5) (2004), pp. 430–434
- 38 FJ O'Brien; Biomaterials & Scaffolds for tissue engineering; Mater Today, 14 (3) (2011), pp. 88–95
- 39 P Akhyari, PW Fedak, RD Weisel, TY Lee, S Verma, DA Mickle, et al.; Mechanical stretch regimen enhances the formation of bioengineered autologous cardiac muscle grafts; Circulation, 106 (12 Suppl 1) (2002), pp. I137–I142
- 40 WH Zimmermann, I Melnychenko, T Eschenhagen; Engineered heart tissue for regeneration of diseased hearts; Biomaterials, 25 (9) (2004), pp. 1639–1647
- 41 N Landa, L Miller, MS Feinberg, R Holbova, M Shachar, I Freeman, et al.; Effect of injectable alginate implant on cardiac remodeling and function after recent and old infarcts in rat; Circulation, 117 (11) (2008), pp. 1388–1396
- 42 E Ruvinov, S Cohen; Alginate biomaterial for the treatment of myocardial infarction: progress, translational strategies, and clinical outlook: from ocean algae to patient bedside; Adv Drug Deliv Rev, 96 (2016), pp. 54–76
- 43 Z Liu, H Wang, Y Wang, Q Lin, A Yao, F Cao, et al.; The influence of chitosan hydrogel on stem cell engraftment, survival and homing in the ischemic myocardial microenvironment; Biomaterials, 33 (11) (2012), pp. 3093–3106
- 44 KL Christman, HH Fok, RE Sievers, Q Fang, RJ Lee; Fibrin glue alone and skeletal myoblasts in a fibrin scaffold preserve cardiac function after myocardial infarction; Tissue Eng, 10 (3–4) (2004), pp. 403–409
- 45 MY Chang, YJ Yang, CH Chang, AC Tang, WY Liao, FY Cheng, et al.; Functionalized nanoparticles provide early cardioprotection after acute myocardial infarction; J Control Release, 170 (2) (2013), pp. 287–294
- 46 X Meng, DA Stout, L Sun, RL Beingessner, H Fenniri, TJ Webster; Novel injectable biomimetic hydrogels with carbon nanofibers and self assembled rosette nanotubes for myocardial applications; J Biomed Mater Res A, 101 (4) (2013), pp. 1095–1102
- 47 R Lakshmanan, UM Krishnan, S Sethuraman; Polymeric scaffold aided stem cell therapeutics for cardiac muscle repair and regeneration; Macromol Biosci, 13 (9) (2013), pp. 1119–1134
- 48 MT Lam, JC Wu; Biomaterial applications in cardiovascular tissue repair and regeneration; Expert Rev Cardiovasc Ther, 10 (8) (2012), pp. 1039–1049
- 49 V Serpooshan, M Zhao, SA Metzler, K Wei, PB Shah, A Wang, et al.; The effect of bioengineered acellular collagen patch on cardiac remodeling and ventricular function post myocardial infarction; Biomaterials, 34 (36) (2013), pp. 9048–9055
- 50 JR Frederick, JR Fitzpatrick 3rd, RC McCormick, DA Harris, AY Kim, JR Muenzer, et al.; Stromal cell-derived factor-1 activation of tissue-engineered endothelial progenitor cell matrix enhances ventricular function after myocardial infarction by inducing neovasculogenesis; Circulation, 122 (11 Suppl) (2010), pp. S107–S117
- 51 A Anitha, S Sowmya, PT Sudheesh Kumar, S Deepthi, KP Chennazhi, H Ehrlich, et al.; Chitin and chitosan in selected biomedical applications; Prog Polym Sci, 39 (9) (2014), pp. 1644–1667
- 52 K Song, M Qiao, T Liu, B Jiang, HM Macedo, X Ma, et al.; Preparation, fabrication and biocompatibility of novel injectable temperature-sensitive chitosan/glycerophosphate/collagen hydrogels; J Mater Sci Mater Med, 21 (10) (2010), pp. 2835–2842
- 53 P Roughley, C Hoemann, E DesRosiers, F Mwale, J Antoniou, M Alini; The potential of chitosan-based gels containing intervertebral disc cells for nucleus pulposus supplementation; Biomaterials, 27 (3) (2006), pp. 388–396
- 54 N Bhattarai, J Gunn, M Zhang; Chitosan-based hydrogels for controlled, localized drug delivery; Adv Drug Deliv Rev, 62 (1) (2010), pp. 83–99
- 55 JW Miklas, SM Dallabrida, LA Reis, N Ismail, M Rupnick, M Radisic; QHREDGS enhances tube formation, metabolism and survival of endothelial cells in collagen-chitosan hydrogels; PLoS ONE, 8 (8) (2013), p. e72956
- 56 NH Chi, MC Yang, TW Chung, NK Chou, SS Wang; Cardiac repair using chitosan-hyaluronan/silk fibroin patches in a rat heart model with myocardial infarction; Carbohydr Polym, 92 (1) (2013), pp. 591–597
- 57 S Wee, WR Gombotz; Protein release from alginate matrices; Adv Drug Deliv Rev, 31 (3) (1998), pp. 267–285
- 58 KY Lee, DJ Mooney; Alginate: properties and biomedical applications; Prog Polym Sci, 37 (1) (2012), pp. 106–126
- 59 B Deng, L Shen, Y Wu, Y Shen, X Ding, S Lu, et al.; Delivery of alginate-chitosan hydrogel promotes endogenous repair and preserves cardiac function in rats with myocardial infarction; J Biomed Mater Res A, 103 (3) (2015), pp. 907–918
- 60 M Terashima, S Fujiwara, GY Yaginuma, K Takizawa, U Kaneko, T Meguro; Outcome of percutaneous intrapericardial fibrin-glue injection therapy for left ventricular free wall rupture secondary to acute myocardial infarction; Am J Cardiol, 101 (4) (2008), pp. 419–421
- 61 J Iemura, H Oku, M Otaki, H Kitayama, T Inoue, T Kaneda; Surgical strategy for left ventricular free wall rupture after acute myocardial infarction; Ann Thorac Surg, 71 (1) (2001), pp. 201–204
- 62 T Okonogi, Y Otsuka, T Saito; Repaired left ventricular free wall rupture after acute myocardial infarction by percutaneous intrapericardial fibrin-glue injection therapy; J Invasive Cardiol, 25 (9) (2013), pp. E186–E187
- 63 S Mukherjee, JR Venugopal, R Ravichandran, S Ramakrishna, M Raghunath; Evaluation of the biocompatibility of PLACL/collagen nanostructured matrices with cardiomyocytes as a model for the regeneration of infarcted myocardium; Adv Funct Mater, 21 (12) (2011), pp. 2291–2300
- 64 AM Martins, G Eng, SG Caridade, JF Mano, RL Reis, G Vunjak-Novakovic; Electrically conductive chitosan/carbon scaffolds for cardiac tissue engineering; Biomacromolecules, 15 (2) (2014), pp. 635–643
- 65 J Zhou, J Chen, H Sun, X Qiu, Y Mou, Z Liu, et al.; Engineering the heart: evaluation of conductive nanomaterials for improving implant integration and cardiac function; Sci Rep, 4 (2014), p. 3733
- 66 KM French, I Somasuntharam, ME Davis; Self-assembling peptide-based delivery of therapeutics for myocardial infarction; Adv Drug Deliv Rev, 96 (2016), pp. 40–53
- 67 ME Davis, JP Motion, DA Narmoneva, T Takahashi, D Hakuno, RD Kamm, et al.; Injectable self-assembling peptide nanofibers create intramyocardial microenvironments for endothelial cells; Circulation, 111 (4) (2005), pp. 442–450
- 68 AV Boopathy, ME Davis; Self-assembling peptide-based delivery of therapeutics for myocardial infarction; Methods Mol Bio, 1141 (2014), pp. 159–164
- 69 M Tokunaga, ML Liu, T Nagai, K Iwanaga, K Matsuura, T Takahashi, et al.; Implantation of cardiac progenitor cells using self-assembling peptide improves cardiac function after myocardial infarction; J Mol Cell Cardiol, 49 (6) (2010), pp. 972–983
- 70 PC Hsieh, ME Davis, J Gannon, C MacGillivray, RT Lee; Controlled delivery of PDGF-BB for myocardial protection using injectable self-assembling peptide nanofibers; J Clin Invest, 116 (1) (2006), pp. 237–248
- 71 TC McDevitt, KA Woodhouse, SD Hauschka, CE Murry, PS Stayton; Spatially organized layers of cardiomyocytes on biodegradable polyurethane films for myocardial repair; J Biomed Mater Res A, 66 (3) (2003), pp. 586–595
- 72 SD Anker, AJS Coats, G Cristian, D Dragomir, E Pusineri, M Piredda, et al.; A prospective comparison of alginate-hydrogel with standard medical therapy to determine impact on functional capacity and clinical outcomes in patients with advanced heart failure (AUGMENT-HF trial); Eur Heart J, 36 (34) (2015), pp. 2297–2309
- 73 DL Mann, RJ Lee, AJS Coats, G Neagoe, D Dragomir, E Pusineri, et al.; One-year follow-up results from AUGMENT-HF: a multicentre randomized controlled clinical trial of the efficacy of left ventricular augmentation with Algisyl in the treatment of heart failure; Eur J Heart Fail, 18 (3) (2015), pp. 314–325
- 74 AV Ghuran, AJ Camm; Ischaemic heart disease presenting as arrhythmias; Br Med Bull, 59 (1) (2001), pp. 193–210
- 75 B Benito, ME Josephson; Ventricular tachycardia in coronary artery disease; Rev Esp Cardiol (Engl Ed), 65 (10) (2012), pp. 939–955 [English Version]
- 76 RJ Myerburg, MJ Junttila; Sudden cardiac death caused by coronary heart disease; Circulation, 125 (8) (2012), pp. 1043–1052
- 77 D Lloyd-Jones, RJ Adams, TM Brown, M Carnethon, S Dai, G De Simone, WRITING GROUP MEMBERS, American Heart Association Statistics Committee and Stroke Statistics Subcommittee, et al.; Heart disease and stroke statistics–2010 update: a report from the American Heart Association; Circulation, 121 (7) (2010), pp. e46–215
- 78 O Yousuf, J Chrispin, GF Tomaselli, RD Berger; Clinical management and prevention of sudden cardiac death; Circ Res, 116 (12) (2015), pp. 2020–2040
- 79 IA Khan; Clinical and therapeutic aspects of congenital and acquired long QT syndrome; Am J Med, 112 (1) (2002), pp. 58–66
- 80 TD Noakes, L Higginson, LH Opie; Physical training increases ventricular fibrillation thresholds of isolated rat hearts during normoxia, hypoxia and regional ischemia; Circulation, 67 (1) (1983), pp. 24–30
- 81 R Lampert, T Joska, MM Burg, WP Batsford, CA McPherson, D Jain; Emotional and physical precipitants of ventricular arrhythmia; Circulation, 106 (14) (2002), pp. 1800–1805
- 82 The Norwegian Multicenter Study Group; Timolol-induced reduction in mortality and reinfarction in patients surviving acute myocardial infarction; N Engl J Med, 304 (14) (1981), pp. 801–807
- 83 ZM Chen, HC Pan, YP Chen, R Peto, R Collins, LX Jiang, COMMIT (ClOpidogrel and Metoprolol in Myocardial Infarction Trial) Collaborative Group, et al.; Early intravenous then oral metoprolol in 45,852 patients with acute myocardial infarction: randomised placebo-controlled trial; Lancet, 366 (9497) (2005), pp. 1622–1632
- 84 I Goldenberg, J Gillespie, AJ Moss, WJ Hall, H Klein, S McNitt, et al.; Executive Committee of the Multicenter Automatic Defibrillator Implantation Trial II. Long-term benefit of primary prevention with an implantable cardioverter-defibrillator: an extended 8-year follow-up study of the Multicenter Automatic Defibrillator Implantation Trial II; Circulation, 122 (13) (2010), pp. 1265–1271
- 85 P Dorian, SH Hohnloser, KE Thorpe, RS Roberts, KH Kuck, M Gent, et al.; Mechanisms underlying the lack of effect of implantable cardioverter-defibrillator therapy on mortality in high-risk patients with recent myocardial infarction: insights from the Defibrillation in Acute Myocardial Infarction Trial (DINAMIT); Circulation, 122 (25) (2010), pp. 2645–2652
- 86 J Vogler, G Breithardt, L Eckardt; Bradyarrhythmias and conduction blocks; Rev Esp Cardiol (Engl Ed), 65 (7) (2012), pp. 656–667 [English Version]
- 87 T Laske, P Iaizzo; The cardiac conduction system; PA Iaizzo (Ed.), Handbook of cardiac anatomy, physiology, and devices, Humana Press, Totowa (2005), pp. 123–136
- 88 J Finsterer, C Stöllberger; Cardiac involvement in Becker muscular dystrophy; Can J Cardiol, 24 (10) (2008), pp. 786–792
- 89 RE Altekin, A Yanikoglu, M Ucar, C Ermis; Complete AV block and cardiac syncope in a patient with Duchenne muscular dystrophy; J Cardiol Cases, 3 (2) (2011), pp. e68–e70
- 90 JC Lee, J Seiler, R Blankstein, RF Padera, KL Baughman, UB Tedrow; Images in cardiovascular medicine. Cardiac sarcoidosis presenting as heart block; Circulation, 120 (15) (2009), pp. 1550–1551
- 91 SM Banypersad, JC Moon, C Whelan, PN Hawkins, AD Wechalekar; Updates in cardiac amyloidosis: a review; J Am Heart Assoc, 1 (2) (2012), p. e000364
- 92 SM Singh, G FitzGerald, AT Yan, D Brieger, KA Fox, J López-Sendón, et al.; High-grade atrioventricular block in acute coronary syndromes: insights from the Global Registry of Acute Coronary Events; Eur Heart J, 36 (16) (2015), pp. 976–983
- 93 H Hreybe, S Saba; Location of acute myocardial infarction and associated arrhythmias and outcome; Clin Cardiol, 32 (5) (2009), pp. 274–277
- 94 SW Cho, YJ Kang, TH Kim, SK Cho, MW Hwang, W Chang, et al.; Primary cardiac lymphoma presenting with atrioventricular block; Korean Circ J, 40 (2) (2010), pp. 94–98
- 95 MS Schaffer, MJ Silka, BA Ross, JD Kugler, Pediatric Electrophysiology Society; Inadvertent atrioventricular block during radiofrequency catheter ablation. Results of the Pediatric Radiofrequency Ablation Registry; Circulation, 94 (12) (1996), pp. 3214–3220
- 96 B Belhassen, A Glick, R Rosso, Y Michowitz, S Viskin; Atrioventricular block during radiofrequency catheter ablation of atrial flutter: incidence, mechanism, and clinical implications; Europace, 13 (7) (2011), pp. 1009–1014
- 97 DP Rardon, WM Miles, DP Zipes; Atrioventricular block and dissociation; DP Zipes, J Jalife (Eds.), Cardiac electrophysiology: from cells to bedside (2nd ed.), WB Saunders, Philadelphia (1995), pp. 485–489
- 98 Z Issa, JM Miller, DP Zipes; Atrioventricular conduction abnormalities; Clinical arrhythmology and electrophysiology: a companion to Braunwalds heart disease, WB Saunders, Philadelphia (2008), pp. 127–142
- 99 SS Barold, DL Hayes; Second-degree atrioventricular block: a reappraisal; Mayo Clin Proc, 76 (1) (2001), pp. 44–57
- 100 SS Barold, A Ilercil, F Leonelli, B Herweg; First-degree atrioventricular block. Clinical manifestations, indications for pacing, pacemaker management & consequences during cardiac resynchronization; J Interv Card Electrophysiol, 17 (2) (2006), pp. 139–152
- 101 M Brignole, A Auricchio, G Baron-Esquivias, P Bordachar, G Boriani, OA Breithardt, ESC Committee for Practice Guidelines (CPG), et al.; Document Reviewers. 2013 ESC Guidelines on cardiac pacing and cardiac resynchronization therapy: the Task Force on cardiac pacing and resynchronization therapy of the European Society of Cardiology (ESC). Developed in collaboration with the European Heart Rhythm Association (EHRA); Eur Heart J, 34 (29) (2013), pp. 2281–2329
- 102 WG Stevenson, RM John; Ventricular arrhythmias in patients with implanted defibrillators; Circulation, 124 (16) (2011), pp. e411–e414
- 103 JS Miller; The 2000 Nobel Prize in Chemistry–a personal accolade; Chemphyschem, 1 (4) (2000), pp. 229–230
- 104 TJ Rivers, TW Hudson, CE Schmidt; Synthesis of a novel, biodegradable electrically conducting polymer for biomedical applications; Adv Funct Mater, 12 (1) (2002), pp. 33–37
- 105 R Balint, NJ Cassidy, SH Cartmell; Conductive polymers: towards a smart biomaterial for tissue engineering; Acta Biomater, 10 (6) (2014), pp. 2341–2353
- 106 A Guiseppi-Elie; Electroconductive hydrogels: synthesis, characterization and biomedical applications; Biomaterials, 31 (10) (2010), pp. 2701–2716
- 107 M Radisic, H Park, H Shing, T Consi, FJ Schoen, R Langer, et al.; Functional assembly of engineered myocardium by electrical stimulation of cardiac myocytes cultured on scaffolds; Proc Natl Acad Sci USA, 101 (52) (2004), pp. 18129–18134
- 108 T Nakashima, T Ohkusa, Y Okamoto, M Yoshida, JK Lee, Y Mizukami, et al.; Rapid electrical stimulation causes alterations in cardiac intercellular junction proteins of cardiomyocytes; Am J Physiol Heart Circ Physiol, 306 (9) (2014), pp. H1324–H1333
- 109 PM George, AW Lyckman, DA LaVan, A Hegde, Y Leung, R Avasare, et al.; Fabrication and biocompatibility of polypyrrole implants suitable for neural prosthetics; Biomaterials, 26 (17) (2005), pp. 3511–3519
- 110 L Zhang, WR Stauffer, EP Jane, PJ Sammak, XT Cui; Enhanced differentiation of embryonic and neural stem cells to neuronal fates on laminin peptides doped polypyrrole; Macromol Biosci, 10 (12) (2010), pp. 1456–1464
- 111 V Lundin, A Herland, M Berggren, EW Jager, AI Teixeira; Control of neural stem cell survival by electroactive polymer substrates; PLoS ONE, 6 (4) (2011), p. e18624
- 112 D Kai, MP Prabhakaran, G Jin, S Ramakrishna; Polypyrrole-contained electrospun conductive nanofibrous membranes for cardiac tissue engineering; J Biomed Mater Res A, 99 (3) (2011), pp. 376–385
- 113 A Gelmi, JB Zhang, A Cieslar-Pobuda, MK Ljunngren, MJ Los, M Rafat, et al.; Electroactive 3D materials for cardiac tissue engineering; Y Bar-Cohen (Ed.), Proceedings of SPIE Volume 9430: Electroactive Polymer Actuators and Devices (EAPAD) 2015; 2015 Mar 9–12, SPIE, San Diego, CA, USA. Bellingham (2015), p. 94301T
- 114 SS Mihardja, RE Sievers, RJ Lee; The effect of polypyrrole on arteriogenesis in an acute rat infarct model; Biomaterials, 29 (31) (2008), pp. 4205–4210
- 115 KK Witte, RR Pipes, K Nanthakumar, JD Parker; Biventricular pacemaker upgrade in previously paced heart failure patients–improvements in ventricular dyssynchrony; J Card Fail, 12 (3) (2006), pp. 199–204
- 116 HC Cho, E Marbán; Biological therapies for cardiac arrhythmias: can genes and cells replace drugs and devices?; Circ Res, 106 (4) (2010), pp. 674–685
- 117 CI Berul, F Cecchin, American Heart Association, American College of Cardiology; Indications and techniques of pediatric cardiac pacing; Expert Rev Cardiovasc Ther, 1 (2) (2003), pp. 165–176
- 118 MR Rosen, PR Brink, IS Cohen, RB Robinson; Cardiac pacing: from biological to electronic … to biological?; Circ Arrhythm Electrophysiol, 1 (1) (2008), pp. 54–61
- 119 NV Munshi, EN Olson; Translational medicine. Improving cardiac rhythm with a biological pacemaker; Science, 345 (6194) (2014), pp. 268–269
- 120 MR Rosen, RB Robinson, PR Brink, IS Cohen; The road to biological pacing; Nat Rev Cardiol, 8 (11) (2011), p. 656
- 121 AB Rosen, DJ Kelly, AJ Schuldt, J Lu, IA Potapova, SV Doronin, et al.; Finding fluorescent needles in the cardiac haystack: tracking human mesenchymal stem cells labeled with quantum dots for quantitative in vivo three-dimensional fluorescence analysis; Stem Cells, 25 (8) (2007), pp. 2128–2138
- 122 MK Proulx, SP Carey, LM Ditroia, CM Jones, M Fakharzadeh, JP Guyette, et al.; Fibrin microthreads support mesenchymal stem cell growth while maintaining differentiation potential; J Biomed Mater Res A, 96 (2) (2011), pp. 301–312
- 123 SL Suarez, AA Rane, A Muñoz, AT Wright, SX Zhang, RL Braden, et al.; Intramyocardial injection of hydrogel with high interstitial spread does not impact action potential propagation; Acta Biomater, 26 (2015), pp. 13–22
- 124 LA Reis, LLY Chiu, N Feric, L Fu, M Radisic; Biomaterials in myocardial tissue engineering; J Tissue Eng Regen Med, 10 (1) (2016), pp. 11–28
- 125 LG Griffith, G Naughton; Tissue engineering–current challenges and expanding opportunities; Science, 295 (5557) (2002), pp. 1009–1014
- 126 N Tandon, C Cannizzaro, PH Chao, R Maidhof, A Marsano, HT Au, et al.; Electrical stimulation systems for cardiac tissue engineering; Nat Protoc, 4 (2) (2009), pp. 155–173
Document information
Published on 06/04/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?