Abstract
The blocking problems in equipment and flowlines caused by precipitation of wax solids is a well-known problem and large research efforts have been focused on developing procedures dealing with wax deposition. Among the different alternatives, the use of chemicals is frequently used to inhibit wax precipitation. The presence of these compounds modifies the size and/or the shape of the paraffinic solids, thus affecting their flow behavior and cold properties. In this work, a methodology based on different experimental techniques was developed to study the effect of different additives on the crude oil behavior at low temperature. Four commercial inhibitors were added to three crude oils in different concentrations. The effect of the inhibitors on wax precipitation was studied using differential scanning calorimetry (DSC) and cross-polarized microscopy (CPM). Obtained results for wax appearance temperature (WAT) using DSC do not show remarkable differences regarding the presence of inhibitor. However, CPM analyses revealed differences in both size and shape of paraffin solids. The results obtained in this work reveal that in some cases the inhibitor does not modify the WAT values, but the morphology of the precipitated particles. In these cases, it is necessary to combine DSC and CPM analyses to obtain reliable results.
Introduction
Paraffins can precipitate during crude oil production operations when the oil is cooled because of heat losses to the surroundings. The precipitation of waxes from petroleum mixtures at low temperatures is an important flow assurance problem in oil production as it can cause important economic losses, which may even lead to abandoning wells [1, 6, 10, 18, 25].
The main variables to evaluate wax precipitation are wax appearance temperature (WAT), the amount of precipitated solid as a function of operating conditions (wax precipitation curve, WPC), pour point, gel point, etc. The knowledge of these variables is crucial to estimate wax precipitation potential and to determine the best strategy to control and mitigate the problem. There is a wide variety of experimental techniques to determine these parameters, as reported elsewhere [12], [14].
The paraffin deposition and the variables involved have been deeply investigated. Thus, Burger et al. [3] studied the mechanism of wax deposition which occurred as a result of lateral transport by diffusion, shear dispersion, and Brownian motion. The rate of deposition is affected by a number of factors such as temperature of the pipe, flow rate, residence time, and wax concentration among others [24]. In this sense, different models have been developed to describe wax deposition in pipelines [13].
Different techniques have been developed to mitigate the paraffin deposition, which are commonly classified into five categories: mechanical, thermal, bacterial, chemical and electromagnetic treatments. In the practice, the use of a single method is not effective and therefore a combination of them is rather applied [26]. Among the different treatments, the use of chemical inhibitors has increased in the crude oil industry as there are a number of chemicals with paraffin inhibition properties. However, there is not an inhibitor equally effective in all wells and it is usual to find a chemical which performs correctly in a well but it fails in another one even in the same basin [24]. Therefore, the application of chemical inhibitors is frequently restricted to each well.
Wax inhibitors are usually grouped in three main categories [20]:
- Wax crystal modifiers
- Detergents
- Dispersants
Detergents and dispersants are basically surface-active agents as, for example, polyesters and amine ethoxylates. They prevent or limit sedimentation of crystals by keeping them dispersed as separate particles, thus reducing their trend to interact with and to adhere to solid surfaces [17].
Wax crystal modifiers are chemicals capable of growing into wax crystals and to alter their growth and surface properties. These chemicals reduce the affinity of crystals to interlock and form three-dimensional networks, thereby lowering the pour point and the viscosity. For that, they are commonly named pour point depressants (PPD) [20, 26]. Pretreatment of crude oils with those chemicals is an attractive alternative to solve wax deposition problems during transport of crude oils along pipelines [4]. However, the performance of these compounds is not clear. The most accepted theory proposes a combination of mechanisms involving nucleation, cocrystallization, and adsorption [21], but different observations have been made regarding the influence on the crystal size and shape and its effects on lowering the pour point [8, 9, 22]. The experimental observations reported in the literature indicate that there is not a single mechanism but a combination of them. The most accepted theories suggest that the decrease of pour point in the presence of crystal modifiers is frequently accompanied by the transition from single crystals to aggregates [20].
A number of experimental techniques have been reported elsewhere to study the action of PPD for the control of wax crystallization. Thus, X-ray diffraction [23], IR spectrometry [27], DSC [28], and rheometry [15] have been applied to investigate into the interaction between PPD and waxes.
As described earlier, the mechanism of inhibitors on wax is widely studied in the literature, although the results are not fully conclusive.
The aim of this work is not to explore the capabilities of DSC and CPM techniques applied to wax precipitation, but to develop a methodology, based on both techniques, to select the most appropriate inhibitors. Four chemical inhibitors of unknown composition and nature were added in different proportions to several crude oils. Differential scanning calorimetry (DSC) and cross-polarized microscopy (CPM) were used to observe the effect of the different additives on the crude oil waxes at low temperatures. The results obtained by DSC indicated a slight decrease of the WAT in presence of some inhibitors, but it was not conclusive. The CPM results, however, show clear differences in the size particle distribution regarding the inhibitor used. Finally, the pour point of crude oils and the resulting mixtures was determined, showing agreement with the results obtained by DSC and CPM analyses. The results obtained for the studied inhibitors indicate the possibility to use a combination of DSC and CPM analyses to study the effect of inhibitors, which do not greatly affect the WAT values, but mainly modify the morphology of the precipitated particles.
The novelty of our work is to present a methodology which allows the optimal selection of inhibitors and combines both the WAT value variation and the change in solid morphology of the precipitated solid from crude oil in the presence of wax inhibitor. When the wax inhibitor slightly affects the WAT value, additional information such as particle size and shape distributions is needed. These data are not commonly reported in the literature from CPM analyses and for that, this work can be of importance in this field. In conclusion, this work proposes the combination of data related to WAT (DSC) with those related to solid morphology (CPM) to study the performance of wax inhibitors.
Experimental Section
Materials
Three dead crude oils (crude oil D-3, D-13, and G-3) and four wax inhibitors (I1, I2, I3, and I4) of unknown composition and nature provided by Repsol (Repsol Technology Centre, E-28931 Móstoles, Madrid, Spain) were used in this work. The main features of the studied crude oils are reported in Table 1.
| Crude oil D-3 | Crude oil D-13 | Crude oil G-3 | |
|---|---|---|---|
| Origin | South America | South America | Europe |
| Type | Medium | Medium | Medium |
| Base | Naphthenic | Paraffinic | Paraffinic |
| API | 31 | 27 | 31 |
Differential scanning calorimetry
The DSC technique is widely used to study wax solidification, [5, 7, 16] and the interaction between inhibitors and waxes [28]. This technique has the advantage of its simplicity and fast response which makes it appropriate for routine analyses.
DSC calibration was carried out by using pure n-alkane within the range of n-C7 to n-C18, thus covering a temperature range between −90 and 30°C. The obtained results suggest the possibility to linearly correlate the pure n-alkane melting temperature determined by DSC and the reference values [5].
In this work, the experimental apparatus used was a DSC Mettler–Toledo DSC822e and the temperature profile used was as follows:
- The sample is heated at 3°C/min from 25 to 80°C to completely dissolve the solid phase and to remove any thermal history.
- The sample is cooled down from 80 to −120°C at 3°C/min.
- The sample is heated up from −120 to 80°C at 3°C/min.
A procedure previously developed for thermogram integration using properties of n-paraffins as reference was used, thereby yielding the wax appearance temperature (WAT) [5].
Cross-polarized microscopy
This technique is frequently used to determine the WAT values [11, 19] and to characterize the waxes resulting from the precipitation process [2, 16]. By using this method, well-crystallized regions can be determined as they present different refractive index from the hydrocarbon matrix.
A Nikon Eclipse 80i microscope was used in cross-polarized mode to observe wax crystallization at a magnification of 20× and 50×. Cooling and heating rates were provided by a temperature control stage connected to the microscope, which allows covering the temperature from −120 to 350°C. The calibration for temperature was carried out using the melting point of pure n-paraffins (n-C14 to n-C38). A linear relationship between theoretical melting point of pure n-paraffins and the WAT determined by CPM was obtained.
The samples were analyzed as follows:
- The sample is heated for 1 h at 50°C in an oven to complete dissolution.
- The sample is cooled down from 50 (fixed as reference) to 0°C at 3°C/min.
The WAT values were determined when the first paraffin wax crystals were visually identified at a magnification of 20×.
The images for further treatment were recorded at 0°C using a digital camera incorporated to the system. The analysis of the images was carried out using Nis-ElementsTM software, thus obtaining the particle size and shape distribution.
Pour point analysis
This property is defined as the lowest temperature at which oils flow freely at specific test conditions. It was determined following the ASTM D97 standard test method. In this work, it was used as benchmark of the results obtained by DSC and CPM analyses.
Results and Discussion
Differential scanning calorimetry
DSC analysis is commonly used for the crude oil characterization, especially to determine its behavior at low temperatures. In this work, this experimental technique was used to detect changes in crude oil behavior at low temperature due to the presence of inhibitor. The additives tested (I1, I2, I3, and I4) were added in different concentrations (300, 1000, and 6000 ppm) to the studied crude oils. The raw crude oils and the resulting mixtures (crude oil + additive) were analyzed by DSC. No qualitative differences were detected in the thermograms of the different samples. In order to quantify the effect of the selected inhibitors, the WAT values of the raw crude oils and their mixtures with the inhibitors were determined following a method reported elsewhere [5]. The obtained results are summarized in Table 2. As can be seen, only the mixture of crude oil plus I4 additive at concentration of 1000 ppm shows a noticeable decrease on the WAT (4°C in the case of crude oil G-3), but the obtained results are not conclusive themselves to confirm the best inhibitor. However, these results indicate that 1000 ppm is the concentration at which the effect of the additives can be observed with this technique.
| Inhibitor | Concentration (ppm) | Crude oil D-3 | Crude oil D-13 | Crude oil G-3 |
|---|---|---|---|---|
| WAT (°C) | ||||
| 20.7 | 36.4 | 43.0 | ||
| I1 | 300 | 21.3 | 37.0 | 42.1 |
| 1000 | 21.6 | 36.5 | 41.1 | |
| 6000 | 19.4 | 36.4 | 41.1 | |
| I2 | 300 | 20.8 | 37.4 | 42.1 |
| 1000 | 20.1 | 38.9 | 42.1 | |
| 6000 | 20.0 | 37.4 | 42.1 | |
| I3 | 300 | 20.7 | 36.7 | 41.1 |
| 1000 | 20.0 | 36.4 | 41.1 | |
| 6000 | 19.8 | 36.3 | 41.1 | |
| I4 | 300 | 21.4 | 35.9 | 42.1 |
| 1000 | 19.4 | 34.9 | 39.2 | |
| 6000 | 18.2 | 34.4 | – | |
Cross-polarized microscopy
One of the purposes of this work is to quantitatively detect the effect of additives in different mixtures by means of CPM.
The analysis procedure was optimized to correctly detect wax precipitation. Figure 1 shows the micrographs of the raw crude oil G-3 at three temperatures (30, 15, and 0°C). As can be seen, the amount of precipitated waxes is much higher at 0°C, and consequently the obtained data at that temperature seem to be more reproducible. Therefore, this temperature was set in order to carry out further CPM analyses.
|
|
|
Figure 1. Micrographs (20×) of raw crude oil G-3 at (A) 30°C; (B) 15°C; and (C) 0°C. |
The effect of the inhibitor concentration was also studied to confirm the results previously obtained by DSC analysis. As an example, Figure 2 shows the micrographs for the crude oil G-3 mixed with additive I1 and I4 at different concentrations (300, 1000, and 6000 ppm). It can be seen how the solid wax morphology changes as a function of the inhibitor concentration. Thus, in the case of additive I4, the particles look like agglomerates dispersed in the organic matrix at an additive concentration value of 1000 ppm. This effect is also observed in the case of additive I1, although in a minor extent. These results suggest, in agreement to those form DSC, that an inhibitor concentration of at least 1000 ppm is necessary to detect significant differences.
|
|
|
Figure 2. Micrographs (20×) of raw crude oil G-3 mixed with the selected additives at different concentrations. |
Figure 3 shows the micrographs of the raw crude oil G-3 and its mixtures with each additive at 0°C and 1000 ppm. As can be observed, the presence of additive I4 affects in a larger extent than the presence of the rest of additives. Thus, the particle morphology confirms the change from the needle-like shape present in the raw crude oil to agglomerate particles dispersed in the oil matrix when adding additive I4.
|
|
|
Figure 3. Micrographs (20×) of raw crude oil G-3 and its mixtures with the selected additives (1000 ppm). (A) Raw crude oil; (B) crude oil + I1; (C) crude oil + I2; (D) crude oil + I3; and (E) crude oil + I4. |
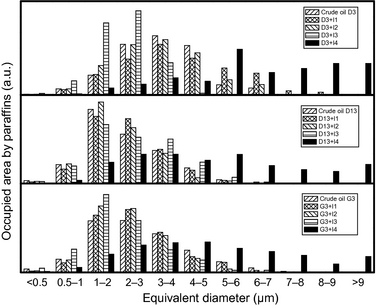
In order to carry out the quantitative analysis of the micrographs, the Nis-ElementsTM software was used. The properties analyzed were the particle size and the particle shape distribution. Figure 4 displays the effect of the different additives on the particle size distribution of precipitates. It is remarkable how the addition of additive I4 yields a particle size distribution different from the rest of inhibitors used. Thereby, the presence of additives I1, I2, and I3 favors equivalent diameters within the range 1–3 microns, whereas the addition of additive I4 makes the particle size distribution shift toward larger values.
|
|
|
Figure 4. Particle size distribution of the raw crude oils and their mixtures with the studied additives. |
This different behavior is in agreement with DSC results, in the sense that inhibitor I4 makes the WAT value decrease. This observation is related to the presence of larger paraffin particles as reported elsewhere [20]. Therefore, among the additives tested, the additive I4 improves the behavior of the crude oil at low temperatures and the effect can be detected using both experimental techniques reported in this work.
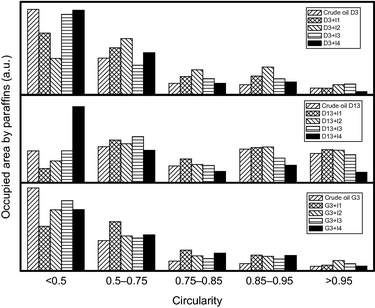
The effect of each additive on the particle shape was also analyzed in this work by means of the “circularity” of the paraffin particles defined as follows:
|
|
(1) |
Figure 5 depicts the variation of this parameter as a function of the inhibitor used. In general, the additives tested in this work do not have a remarkable effect on the circularity of the paraffin particles and therefore, it is not possible to make conclusive remarks.
|
|
|
Figure 5. Shape distribution of the crude oils and their mixtures with the studied additives. |
Pour point analysis
This property is commonly used to evaluate the behavior of crude oil mixtures at low temperatures. The determination of the pour point was used in this work as a benchmark of the results obtained by DSC and CPM analyses. Pour point values were determined for all the crude oils and its mixtures with additives I2 and I4. The former was chosen as its presence does not have important effects on the crude oil behavior, quite the opposite to I4. Table 3 summarizes the obtained results. It can be observed how the addition of inhibitor I4 makes the pour point of the mixture decrease, whereas the addition of I2 does not have a significant effect. These results confirm those obtained by CPM and DSC analyses in the sense that additive I4 shows a better performance on the studied crude oils at low temperature.
| Sample | Pour point (°C) |
|---|---|
| Crude oil D-3 | −24 |
| D-3 + I2 | < −38 |
| D-3 + I4 | < −38 |
| Crude oil D-13 | 6–9 |
| D-13 + I2 | 6 |
| D-13 + I4 | −18 |
| Crude oil G-3 | 6 |
| G-3 + I2 | 12 |
| G-3 + I4 | −15 |
Conclusions
DSC analyses cannot be used itself to evaluate the performance of inhibitors that do not affect the WAT values in a noticeable extent.
CPM analyses allow obtaining valuable information concerning the evolution of paraffin crystals with regard to the crude oil, the type of inhibitor, and its concentration. The combination of DSC and CPM analyses allows the study of the inhibitor influence on WAT values and particle growth and therefore, the obtained results can be used to select of the most adequate inhibitor.
Pour point results confirm that the combination of DSC and CPM analyses can be quantitatively used to determine the effect of additives that do not affect greatly the WAT value of the crude oil.
Conflict of Interest
None declared.
References
- Apte, M. S., A. Matzain, H. Q. Zhang, M. Volk, and J. P. Brill. 2001. Investigation of paraffin deposition during multiphase flow in pipelines and wellbores – part 2: modeling. J. Energy Resour. Technol.123:150–157.
- Bacon, M. M., L. Romero-Zerón, and K. K. Chong. 2010. Determining wax type: paraffin or naphthene?SPE J.15:969–974.
- Burger, E. D., T. K. Perkins, and J. H. Striegler. 1981. Studies of wax deposition in the trans Alaska pipeline. J. Pet. Technol.33:1075–1086.
- Chen, W., Z. Zhao, and C. Yin. 2010. The interaction of waxes with pour point depressants. Fuel89:1127–1132.
- Coto, B., C. Martos, J. J. Espada, M. D. Robustillo, and J. L. Peña. 2010. Analysis of paraffin precipitation from petroleum mixtures by means of DSC: iterative procedure considering solid-liquid equilibrium equations. Fuel89:1087–1094.
- Daridon, J. L., J. Pauly, J. A. P. Coutinho, and F. Montel. 2001. Solid-liquid-vapor phase boundary of a North Sea waxy crude: measurement and modelling. Energy Fuels15:730–735.
- Elsharkawy, A. M., T. A. Al-Sahhaf, and M. A. Fahim. 2000. Wax deposition from Middle East crudes. Fuel79:1047–1055.
- Holder, G. A., and J. Winkler. 1965. Wax crystallization from distillate fuels. 1. Cloud and pour phenomena exhibited by solutions of binary n-paraffin mixtures. J. Inst. Pet.51:228.
- Irani, C., and J. Zajac. 1982. Handling of high pour point west-African crude oils. J. Pet. Technol.34:289–298.
- Juyal, P., T. Cao, A. Yen, and R. Venkatesan. 2011. Study of live oil wax precipitation with high-pressure micro-differential scanning calorimetry. Energy Fuels25:568–572.
- Kok, M. V., J. M. Letoffe, P. Claudy, D. Martin, M. Garcin, and J. L. Volle. 1996. Comparison of wax appearance temp-eratures of crude oils by differential scanning calorimetry, thermomicroscopy and viscometry. Fuel75:787–790.
- Kok, M. V., J. M. Letoffe, and P. Claudy. 1999. DSC and rheometry investigations of crude oils. J. Therm. Anal. Calorim.56:959–965.
- Kok, M. V., and R. O. Saracoglu. 2000. Mathematical modelling of wax deposition in crude oil pipelines (comparative study). Petrol. Sci. Technol.18:1121–1145.
- Kok, M. V., J. M. Letoffe, and P. Claudy. 2007. Comparative methods in the determination of wax content and pour points of crude oils. J. Therm. Anal. Calorim.90:827–831.
- Kok, M. V.2014. The effect of pour point depressant on the flow behavior of crude oils. Energ. Source part A2:167–172.
- Letoffe, J. M., P. Claudy, M. V. Kok, M. Garcin, and J. L. Volle. 1995. Crude oils – characterization of waxes precipitated on cooling by DSC and thermomicroscopy. Fuel74:810–817.
- Maithufi, M. N., D. J. Joubert, and B. Klumperman. 2011. Application of gemini surfactants as diesel fuel wax dispersants. Energy Fuels25:162–171.
- Matzain, A., M. S. Apte, H. Q. Zhang, M. Volk, and J. P. Brill. 2002. Investigation of paraffin deposition during multiphase flow in pipelines and wellbores – part 1: experiments. J. Energy Resour. Technol.124:180–186.
- Paso, K., H. Kallevik, and J. Sjöblom. 2009. Measurement of wax appearance temperature using near-infrared (NIR) scattering. Energy Fuels23:4988–4994.
- Pedersen, K. S., and H. P. Rønningsen. 2003. Influence of wax inhibitors on wax appearance temperature, pour point, and viscosity of waxy crude oils. Energy Fuels17:321–328.
- Randolph, A. D., and M. A. Larson. 1988. Theory of particulate processes. Analysis and technique of continuous crystallization. Academic Press, New York, NY.
- Rønningsen, H. P., B. Bjørndal, A. B. Hansen, and W. B. Pedersen. 1991. Wax precipitation from North-sea crude oils. 1. Crystallization and dissolution temperatures, and Newtonian and non-Newtonian flow properties. Energy Fuels5:895–908.
- Srivastava, S. P., R. S. Tandon, P. S. Verma, A. K. Saxena, G. C. Joshi, and S. D. Phatak. 1992. Crystallization behavior of n-paraffins in Bombay-high middle-distillate wax gel. Fuel71:533–537.
- Towler, B. F., and S. Rebbapragada. 2004. Mitigation of paraffin wax deposition in cretaceous crude oils of Wyoming. Pet. Sci. Eng.45:11–19.
- Venkatesan, R., P. Singh, and H. S. Fogler. 2002. Delineating the pour point and gelation temperature of waxy crude oils. SPE J.7:349–352.
- Yong, F.1996. Paper SPE 31128. Presented at SPE International Symposium on Formation Damage Control, Lafayette, Louisiana.
- Zhang, F. S., and B. Wang. 1995. Studies on the mechanisms involved in pour point depressant and viscosity deduction by some pour point depressants/viscosity reducers. Oilfield Chem.12:347–352.
- Zhang, J. L., C. J. Wu, W. Li, Y. P. Wang, and Z. T. Han. 2003. Study on performance mechanism of pour point depressants with differential scanning calorimeter and X-ray diffraction methods. Fuel82:1419–1426.
References
- Kundur, P.1994. Power system stability and control. McGraw Hill, New York, NY.
- Aziz, T., T. K. Saha, and N. Mithulananthan. 2011. Static and dynamic VAR planning to support widespread penetration of distributed generation in distribution system. Pp. 1–6in21st Australasian Universities Power Engineering Conference (AUPEC), Brisbane, Australia, 25–28 September 2011.
- Elkington, K., V. Knazkins, and M. Ghandhari. 2007. Pp. 213–218inOn the rotor angle stability of power systems with doubly fed induction generators. Power Tech IEEE, Lausanne Stockholm, Sweden.
- Jakus, D., R. Goic, and J. Krstulovic. 2011. The impact of wind power plants on slow voltage variations in distribution networks. Electr. Pow. Syst. Res.81:589–598.
- Lahacani, N. A., D. Aouzellag, and B. Mendil. 2010. Static compensator for maintaining voltage stability of wind farm integration to a distribution network. Renew. Energy35:2476–2482.
- Mariotto, L., H. Pinheiro, G. Cardoso, and M. R. Muraro. 2007. Determination of the static voltage stability region of distribution systems with the presence of wind power generation. Pp. 556–562inInternational Conference on Clean Electrical Power, Capri, 21–23 May 2007.
- Linh, N. T. 2009. Voltage stability analysis of grids connected wind generators. Pp. 2657–2660 in 4th IEEE Conference on Industrial Electronics and Applications, Xi'an, 25-27 May 2009.
- Dong, B., S. Asgarpoor, and W. Qiao. 2009. Voltage analysis of distribution systems with DFIG wind turbines. Pp. 1–5inIEEE Power Electronics and Machines in Wind Applications, Lincoln, NE, 24–26 June 2009.
- Sarkhanloo, M. S., A. S. Yazdankhan, and R. Kazemzadeh. 2012. A new control strategy for small wind farm with capabilities of supplying required reactive power and transient stability improvement. Renew. Energy44:32–39.
- Roy, N. K., H. R. Pota, and M. J. Hossain. 2013. Reactive power management of distribution networks with wind generation for improving voltage stability. Renew. Energy58:85–94.
- Zheng, C., and M. Kezunovic. 2010. Distribution system voltage stability analysis with wind farms integration. PP. 1–6inNorth American Power Symposium, Arlington, TX, 26–28 September 2010.
- Dicorato, M., G. Forte, and M. Trovato. 2012. Wind farm stability analysis in the presence of variable-speed generators. Energy39:40–47.
- Dahal, S., N. Mithulananthan, and T. Saha. 2010. Investigation of small signal stability of a renewable energy based electricity distribution system. Pp. 1–8inIEEE Power and Energy Society General Meeting, Minneapolis, MN, 25–29 July 2010.
- Fernandez, L. M., F. Jurado, and J. R. Saenz. 2008. Aggregated dynamic model for wind farms with doubly fed induction generator wind turbines. Renew. Energy33:129–140.
- Fernandez, R. D., R. J. Mantz, and P. E. Battaiotto. 2007. Impact of wind farms on a power system, an eigenvalue analysis approach. Renew. Energy32:1676–1688.
- Bian, X., C. T. Tse, C. Y. Chung, and K. W. Wang. 2010. Dynamic modeling of large scale power system with FACTS and DFIG type wind turbine. Pp. 753–758in2nd IEEE International Symposium on Power Electronics for Distributed Generation Systems. Hefei, China 16–18 June 2010.
- Aziz, T., S. Dahal, N. Mithulananthan, and T. K. Saha. 2010. Impact of widespread penetrations of renewable generation on distribution system stability. Pp. 338–341in6th International Conference on Electrical and Computer Engineering, Dhaka, Bangladesh, 18–20 December 2010.
Document information
Published on 01/06/17
Submitted on 01/06/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?