Summary
The most common causes of acute gastric outlet obstruction (GOO) are duodenal and type 3 gastric ulcers. However, mechanical or functional causes may also lead to this pathology. Acute GOO is characterized by delayed gastric emptying, anorexia, or nausea accompanied by vomiting. Herein we report a 56-year-old man diagnosed with GOO secondary to paraesophageal hiatal herniation of gastric antrum after laparoscopic fundoplication. Because of the rarity of this disease, common gastrointestinal complaints may mislead the emergency physician to diagnose a nonsurgical gastrointestinal disease if a detailed history and physical examinations are not obtained.
Keywords
antral herniation;gastric outlet obstruction;laparoscopic fundoplication;paraesophageal hiatal hernia
1. Introduction
The stomach is divided into four parts: the cardia, the body, the antrum, and the pylorus. Inflammation, scarring, or infiltration of the antrum and pylorus are associated with the development of gastric outlet obstruction (GOO). In general, clinical entities that can cause GOO are categorized into two well-defined groups—benign and malignant. This classification facilitates the discussion on the management and treatment of GOO. The major benign causes of GOO are peptic ulcer disease, gastric polyps, ingestion of caustics, pyloric stenosis, congenital duodenal webs, gallstone obstruction (Bouveret syndrome), pancreatic pseudocysts, and bezoars.1; 2 ; 3 Here, we presented a patient diagnosed with benign GOO secondary to paraesophageal herniation (PEH) of gastric antrum after laparoscopic fundoplication.
2. Case report
A 56-year-old man was admitted to our emergency department with complaints of upper abdominal pain for 12 hours, nausea, and nonbilious vomiting. He had a history of antireflux surgery at about 11 months ago. On physical examination, epigastric tenderness and severe abdominal distension were noted. Dilated stomach was seen in plain abdominal radiography and paraesophageal herniation of the antrum was detected on abdominal computed tomography imaging (Figure 1, Figure 2 ; Figure 3). The patients complaints were relieved when a nasogastric tube was inserted and 3 L of gastric fluid was decompressed. A laparoscopic revision surgery was performed on the 3rd day of hospital admission. Fundoplication disruption, a large diaphragmatic defect, opened sutures over the diaphragmatic cruris, several adhesions, and antral herniation of the stomach were observed intraoperatively. We speculated that the mobile antrum was herniated through the openings of the diaphragmatic cruris into the thorax. During the revision surgery, dissection of adhesions, repair of diaphragmatic defect using a Proceed mesh (Ethicon, Inc., a Johnson & Johnson company), and re-Nissen fundoplication were performed. No complication was observed during the 12-month follow-up of the patient.
|
|
|
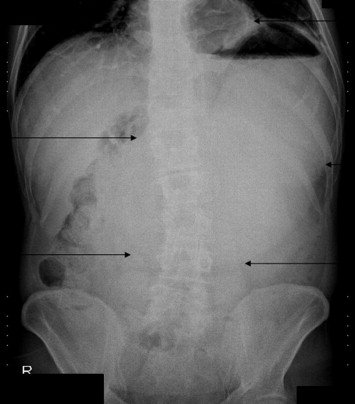
Figure 1. Upright abdominal X-ray of the patient illustrating a dilated stomach and intrathoracic gases. |
|
|
|
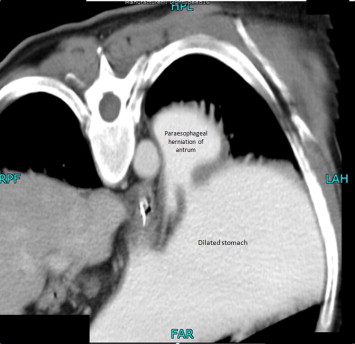
Figure 2. Computed tomography scan illustrating paraesophageal herniation of the antrum and dilated stomach. |
|
|
|
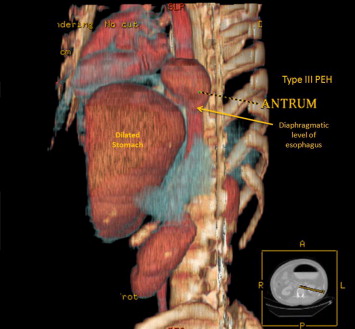
Figure 3. Three-dimensional image from tomography scan demonstrating herniation of the antrum. Intraoperatively, type III PEH was corrected. PEH = paraesophageal herniation. |
3. Discussion
Intrinsic or extrinsic obstruction of the pyloric channel or duodenum is the usual pathophysiology of GOO; the mechanism of obstruction depends upon the underlying etiology. Vomiting is the cardinal symptom. Vomiting is usually described as nonbilious and characteristically contains undigested food particles, as in case of our patient.1; 2 ; 3 In the present case, acute onset of the complaints led the physician to suspect gastrointestinal obstruction, which was supported by the radiological examinations revealing complete obstruction at the level of antrum with dilated stomach.
A flat and upright X-ray should be obtained for patients who had symptoms related to GOO.4 In our case, plain radiography revealed a dilated stomach and gas formation behind the heart (Fig. 1). The gas formation behind the heart led the physician suspect PEH. Despite the limited information obtained from upright abdominal radiography, plain radiography is the first preferred diagnostic tool for the diagnosis of diaphragmatic hernia.4 Multislice thoracoabdominal computed tomography scan is a very useful and reliable tool for the diagnosis and detection of complications of both PEH and GOO.4 ; 5 In our case, GOO secondary to PEH of gastric antrum was shown on thoracoabdominal computed tomography images (Figs. 2 and 3).
PEH is present in 14% of all hiatal hernias. Esophageal hernias can be classified as follows: type I—sliding hiatal hernia; type II—paraesophageal hernia; and type III—combined sliding and paraesophageal hernias. Sliding or type I hernia, in which the gastroesophageal junction (GEJ) migrates above the esophageal hiatus, is the most common type of hiatal hernia (95%).6 Paraesophageal hernias occur when defects in the diaphragm allow varying degrees of abdominal contents to herniate cephalad into the mediastinum. Types II and III are paraesophageal hernias, and they account for approximately 2–5% of all hiatal hernias.6 True paraesophageal hernias are characterized by a normally positioned GEJ and an intrathoracically migrated stomach.6 If both the GEJ and the stomach have migrated cranially into the mediastinum, a mixed or type III hiatal hernia is present.6 In this case, we report PEH of antrum as an unusual type 3 hiatal hernia.
The clinical symptoms caused by PEHs are mostly related to the impaired gastric emptying (postprandial distress and fullness, nausea, bloating, dysphagia, and retching) caused by mechanical obstructions.6 Most patients with PEH have minimal symptoms.4 However, these symptoms are often nonspecific and do not provide an early diagnosis in the emergency department.4 PEH can lead to severe complications such as incarceration, volvulus, complete obstruction, or strangulation, which are true emergencies in the emergency department.4 Although life-threatening complications of PEH are uncommon, it requires urgent correction.6
All patients with GOO require preoperative nasogastric decompression for several days, antisecretory therapy, and correction of fluid and electrolyte imbalances.5 If the patient is acutely obstructed, nasogastric decompression, intravenous fluid replacement, nutritional support, and acid-suppressive therapy should be performed.5 In our case, 3 L of gastric fluid was decompressed initially, and the patient was followed up for 3 days for correction of fluid imbalances and antisecretory therapy.
Surgical therapy choices are reduction of the dislocated stomach, excision of the hernia sac, and closure of the hiatal defect.6 In our case, the hernia was reduced after dissecting the adhesions and the diaphragmatic defect was repaired using a Proceed mesh.
In the present case, the patient had a history of large amount of alcohol and fat consumption 15 hours previously. It can be speculated that alcohol and fatty dietary intake might have caused fermentation and formation of gas. Formation of gases might have caused dilation and dysmotility, and induced hypomotility of stomach.
The patient also had a history of laparoscopic Nissen fundoplication operation 11 months ago. In a fundoplication operation, the gastric fundus (upper part) of the stomach is wrapped, or plicated, around the lower end of the esophagus and stitched in place, reinforcing the closing function of the lower esophageal sphincter. Whenever the stomach contracts, it also closes the esophagus instead of squeezing stomach acids into it. This prevents the reflux of gastric acid.7 This rotational operation most probably cause predisposition to rotational movement of the stomach. In our case, GOO secondary to PEH of antrum can be defined as a late complication of laparoscopic antireflux surgery.
In fact, hiatal herniation represents one of the major reasons for re-do surgery after laparoscopic fundoplication. Frantzides et als8 intraoperative findings for a re-do surgery were as follows: slipped fundoplication and hiatal hernia 33.8%, hiatal hernia alone 10.2%, and malpositioned fundoplication and hiatal hernia 7.3%.
4. Conclusion
Because of the rarity of this disease, common gastrointestinal complaints may mislead the emergency physician to diagnose a nonsurgical gastrointestinal disease if a detailed history and physical examinations are not obtained. Emergency department physicians should maintain a high level of suspicion for this disease when treating patients with acute abdominal pain and seek surgical intervention immediately for this condition.
References
- 1 S.K. Khullar, J.A. DiSario; Gastric outlet obstruction; Gastrointest Endosc Clin N Am, 6 (1996), pp. 585–603
- 2 P.T. Cherian, S. Cherian, P. Singh; Long-term follow-up of patients with gastric outlet obstruction related to peptic ulcer disease treated with endoscopic balloon dilatation and drug therapy; Gastrointest Endosc, 66 (2007), pp. 491–497
- 3 A. Avan, D.E. Johnston, M.M. Jamal; Gastric outlet obstruction with benign endoscopic biopsy should be further explored for malignancy; Gastrointest Endosc, 48 (1998), pp. 497–500
- 4 C.C. Chang, C.L. Tseng, Y.C. Chang; A surgical emergency due to an incarcerated paraesophageal hernia; Am J Emerg Med, 27 (2009), pp. 134.e1–134.e3
- 5 K.K. Yau, W.T. Siu, H.Y. Cheung, et al.; Acute gastric volvulus: an unusual cause of gastric outlet obstruction; Minim Invasive Ther Allied Technol, 14 (2005), pp. 2–5
- 6 L. Krähenbühl, M. Schäfer, J. Farhadi, et al.; Laparoscopic treatment of large paraesophageal hernia with totally intrathoracic stomach; J Am Coll Surg, 187 (1998), pp. 231–237
- 7 S. Horgan, C.A. Pellegrini; Surgical treatment of gastroesophageal reflux disease; Surg Clin North Am, 77 (1997), pp. 1063–1082
- 8 C.T. Frantzides, A.K. Madan, M.A. Carlson, et al.; Laparoscopic revision of failed fundoplication and hiatal herniorraphy; J Laparoendosc Adv Surg Tech A, 19 (2009), pp. 135–139
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?