Summary
A 51-year-old man was referred to our clinic for recurrent obstructive jaundice and underwent pylorus-preserving pancreaticoduodenectomy for a suspected malignancy. The pathology showed immunoglobulin G4 positive plasma cell infiltrated at the pancreas and the gallbladder. We discuss the cost-effectiveness of serum immunoglobulin G4 level prior to arranging for a pancreaticoduodenectomy, which would reduce the possibility of surgical complications as well as costs.
Keywords
Immunoglobulin G4 ; Obstructive jaundice ; Pancreaticoduodenectomy ; Pancreatitis
Introduction
Immunoglobulin G4 (IgG4)-related pancreatitis, also called sclerosing pancreatitis, lymphoplasmacytic sclerosing pancreatitis, or autoimmune pancreatitis, is associated with serum IgG4 level and tissue infiltration of IgG4-positive plasma cells [1] . Typically, IgG4-related pancreatitis clinically presents with chronic inflammation of the pancreas, tumefactive lesions, and obstructive jaundice, which is easily confused with malignancy. IgG4-related pancreatitis is considered a systemic disease, and its extrapancreatic involvement has been recognized since 2003 [2] . In general, the treatment of IgG4-related disease begins with steroids; subsequently, disease-modifying antirheumatic drugs are administered to patients with remission and maintenance statuses, and rituximab is used to treat recurrent or refractory disease [3] .
Case report
A 51-year-old Taiwanese man was referred to our clinic for recurrent obstructive jaundice. The patients aspartate aminotransferase, alanine aminotransferase, and total bilirubin levels had risen intermittently for 6 months.
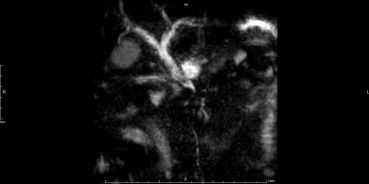
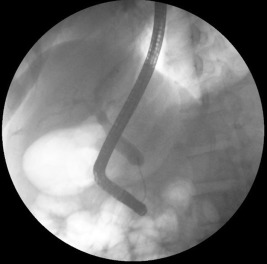
He had a history of benign prostatic hyperplasia that was not controlled medically, but had no history of major operations or allergies. The patient smoked one and a half packs of cigarettes per day for > 20 years, quit drinking alcohol 10 years prior, and socially chewed betel nuts. He is a judo coach who experiences frequent abdominal contusions. Physical examinations revealed mild icteric sclera, a birth mark approximately 3 cm in size in the right upper quadrant of the abdomen, and no abdominal tenderness, rebounding pains, or Murphys sign. In a review of systems, the patient complained of anorexia, fatigue, nausea, vomiting, itchy skin, and weight loss (from 79 kg to 68.2 kg in the past 6 months). Magnetic resonance (MR) cholangiopancreatography was scheduled while the patient was hospitalized and revealed distal common bile duct (CBD) obstruction (Fig. 1 ). In addition, the patient received endoscopic retrograde cholangiopancreatography, but cannulation of the ampulla of Vater failed initially. Fistulotomy was performed by inserting a stent into the CBD. The cholangiography revealed an absence of distal CBD (Fig. 2 ). Based on the tentative impression of stricture of distal CBD with obstructive jaundice, pylorus-preserving pancreaticoduodenectomy was arranged to treat a possible malignancy.
|
|
|
Figure 1. Recombinant MR image showing mild dilatation of both lobes' IHDs, CHD, and CBD, with abrupt luminal narrowing over the distal CBD. CBD = common bile duct; CHD = common hepatic duct; IHD = intrahepatic duct; MR = magnetic resonance. |
|
|
|
Figure 2. Cholangiopancreatography shows absence of distal common bile duct (CBD). |
An indurate mass approximately 4 cm in size was intraoperatively observed over the pancreatic head, causing distal CBD obstruction and enlarged lymph nodes over Station 8A. The pathology showed prominent sclerosing fibrosis with marked lymphoplasmacytic cell infiltration in the pancreas, CBD, and gallbladder as well as reactive hyperplasia of the lymph nodes. The immunohistochemical stains showed that infiltrated plasma cells were CD138 and IgG4 positive, proliferative small bile ducts were CK7 positive, no P53 overexpression occurred, and Ki-67 mildly increased. The final diagnosis was IgG4-related disease with pancreatitis, cholangitis, and cholecystitis.
Discussion
The patient received pylorus-preserving pancreaticoduodenectomy for obstructive jaundice of an unknown cause with a possibility of malignancy. Although no complication occurred because of surgery, the operation could have been avoided if we had confirmed the diagnosis preoperatively. In Taiwan, receiving a test of serum IgG4 levels through the National Health Insurance costs NT$400 (approximately US$13.50). Approximately 2.5% of pancreaticoduodenectomies reveal IgG4-related pancreatitis rather than the suspected malignancy [4] ; [5] ; [6] . Each diagnosis from serum IgG4 screen costs NT$16,000 (approximately US$540.70); by contrast, pancreaticoduodenectomy costs NT$45,670 (approximately US$1543.40). In a review of image findings, focal (24–36%) or diffused (32–48%) parenchymal enlargement of the pancreas was observed on both computed tomography (CT) and MR images [7] ; [8] . Approximately 14–48% of patients with IgG4-related pancreatitis presented with a capsule-like rim around the pancreas, which is specific to IgG4-related pancreatitis [7] ; [8] ; [9] . However, this sign was not observed in the image study of our patient. The capsule-like rim yielded low attenuation on contrast-enhanced CT images and hypointense on both T1- and T2-weighted dynamic MR images with delay enhancement [9] . The capsule-like rim may be a peripancreatic extension of the characteristic inflammatory cell infiltration [9] . For pancreatic malignancy or IgG4-related disease, CT or MR imaging, serology (serum IgG4 and Ca19-9), the involvement of other organs, pancreatic core biopsy, and steroid diagnostic therapy should be considered prior to a surgical resection [10] . Hence, distinguishing IgG4-related disease, which can be treated with steroids rather than surgery, from malignancy early reduces the risk of operation-related morbidity and mortality.
For patients with recurrent obstructive jaundice, physicians should assess CT or MR images and conduct serology studies of liver and pancreatic function as well as IgG4. Furthermore, a pancreatic core biopsy can be performed preoperatively to confirm the diagnosis. The aforementioned examination tool facilitates reducing unnecessary procedures performed to treat benign diseases and reduces the possibility of surgical complications as well as costs.
Conflicts of interest
All authors declare no conflicts of interest.
References
- [1] H. Hamano, S. Kawa, A. Horiuchi, H. Unno, N. Furuya, T. Akamatsu, et al.; High serum IgG4 concentrations in patients with sclerosing pancreatitis; N Engl J Med, 344 (2001), pp. 732–738
- [2] T. Kamisawa, N. Funata, Y. Hayashi, Y. Eishi, M. Koike, K. Tsuruta, et al.; A new clinicopathological entity of IgG4-related autoimmune disease; J Gastroenterol, 38 (2003), pp. 982–984
- [3] J.H. Stone, Y. Zen, D. Deshpande; IgG4-related disease; N Engl J Med, 366 (2012), pp. 539–551
- [4] S.M. Weber, O. Cubukcu-Dimopulo, J.A. Palesty, A. Suriawinata, D. Klimstra, M.F. Brennan, et al.; Lymphoplasmacytic sclerosing pancreatitis: inflammatory mimic of pancreatic carcinoma; J Gastrointest Surg, 7 (2003), pp. 129–137 discussion 137–9
- [5] J.M. Hardacre, C.A. Iacobuzio-Donahue, T.A. Sohn, S.C. Abraham, C.J. Yeo, K.D. Lillemoe, et al.; Results of pancreaticoduodenectomy for lymphoplasmacytic sclerosing pancreatitis; Ann Surg, 237 (2003), pp. 853–858 discussion 858–9
- [6] S.C. Abraham, R.E. Wilentz, C.J. Yeo, T.A. Sohn, J.L. Cameron, J.K. Boitnott, et al.; Pancreaticoduodenectomy (Whipple resections) in patients without malignancy: are they all 'chronic pancreatitis'?; Am J Surg Pathol, 27 (2003), pp. 110–120
- [7] K. Suzuki, S. Itoh, T. Nagasaka, H. Ogawa, T. Ota, S. Naganawa; CT findings in autoimmune pancreatitis: assessment using multiphase contrast-enhanced multisection CT; Clin Radiol, 65 (2010), pp. 735–743
- [8] D.V. Sahani, S.P. Kalva, J. Farrell, M.M. Maher, S. Saini, P.R. Mueller, et al.; Autoimmune pancreatitis: imaging features; Radiology, 233 (2004), pp. 345–352
- [9] H. Irie, H. Honda, S. Baba, T. Kuroiwa, K. Yoshimitsu, T. Tajima, et al.; Autoimmune pancreatitis: CT and MR characteristics; AJR Am J Roentgenol, 170 (1998), pp. 1323–1327
- [10] S.T. Chari, N. Takahashi, M.J. Levy, T.C. Smyrk, J.E. Clain, R.R. Pearson, et al.; A diagnostic strategy to distinguish autoimmune pancreatitis from pancreatic cancer; Clin Gastroenterol Hepatol, 7 (2009), pp. 1097–1103
Document information
Published on 15/05/17
Submitted on 15/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?