Summary
Background/objective
One thousand liver transplantations have been performed at the only liver transplant center in Hong Kong over a period of 22 years, which covered the formative period of living donor liver transplantation. These 1000 transplantations, which marked the journey of liver transplantation from development to maturation at the center, should be educational. This research was to study the experience and to reflect on the importance of technical innovations and case selection.
Methods
The first 1000 liver transplantations were studied. Key technical innovations and surgical therapeutics were described. Recipient survival including hospital mortality was analyzed. Recipient survival comparison was made for deceased donor liver transplantation and living donor liver transplantation indicated by hepatocellular carcinoma and other diseases.
Results
Among the 1000 transplantations, 418 used deceased donor grafts and 582 used living donor grafts. With the accumulation of experience, hospital mortality improved to < 2% in the past 2 years. In the treatment of diseases other than hepatocellular carcinoma, living donor liver transplantation was superior to deceased donor liver transplantation, with a 10-year recipient survival around 90%.
Conclusion
Transplant outcomes have been improving consistently over the series, with a very low hospital mortality and a predictably high long-term survival.
Keywords
deceased donor liver transplantation;experience;living donor liver transplantation
1. Introduction
Liver transplantation (LT) was invented by Starzl et al in the 1960s1 and became a legitimate treatment in the 1980s.2 The impressive improvement in recipient outcomes was the result of four areas of medical research. Cold storage of livers in University of Wisconsin solution has extended the graft preservation time3 and improved long-term graft survival.4 Cyclosporine as the first calcineurin inhibitor used is efficacious in reducing graft rejection.5 Prevention of activation of hepatitis B virus (HBV) with passive immunoprophylaxis6 and then by antiviral agent lamivudine7 has remarkably reduced HBV infection of grafts. Careful selection of patients with hepatocellular carcinoma (HCC) for LT has also contributed to a better rate of recipient long-term survival.8
Asia entered this surgical arena only in the early 1990s. In 1991, Hong Kong saw its first deceased donor LT (DDLT). Two years after Strong et al9 of Brisbane performing the first living donor LT (LDLT), Hashikura et al10 performed the first adult LDLT in 1993 outside Australia. Japan was instrumental in the development of LDLT, a long-waited procedure for Asia where rates of organ donation from the deceased have always been very low.11 Hong Kong had its first LDLT conducted in 1993 for a child; in 1994, it had its first adult LDLT.12 In 1996, the first adult LDLT using the right liver placed Hong Kong on the world map of organ transplantation.13 As one of the worlds leading centers in LT, here we give an account of our first 1000 cases and reflect on this experience. Only with a clear understanding of the past can we advance further in our work.
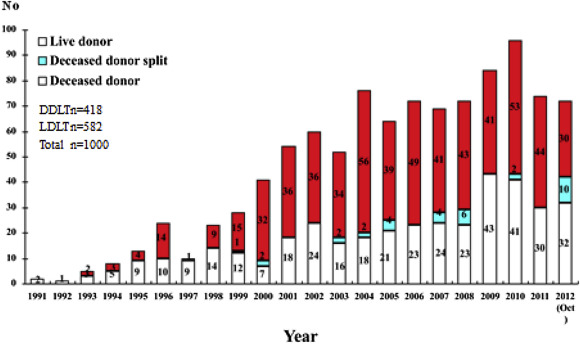
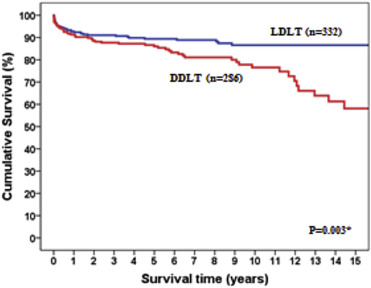
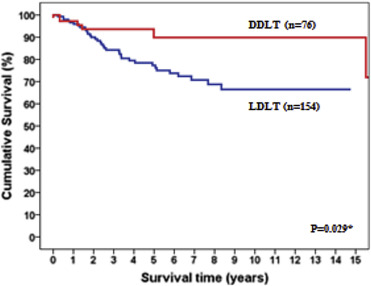
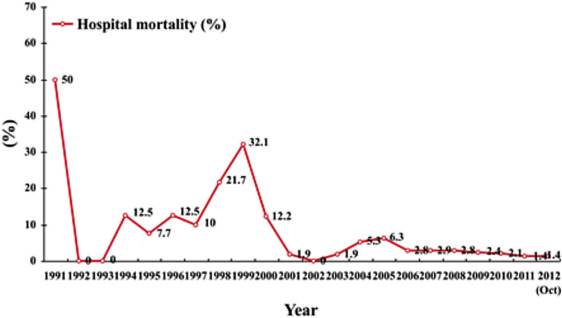
Our first 1000 LTs started in 1991 and were accomplished in October 2012. Among them, 418 were DDLTs and 582 LDLTs (Figure 1). There were 152 pediatric recipients. In the treatment of non-HCC diseases, LDLT surpassed DDLT in recipient survival, with a 10-year survival around 90% (Figure 2), but it was outperformed by DDLT in the treatment of HCC, with a 10-year survival of around 70% only (Figure 3). The hospital mortality of LT had been reduced to < 3% since 2006 and was < 2% in 2011 and 2012 (up to October; Figure 4).
|
|
|
Figure 1. The first 1000 liver transplants at Queen Mary Hospital, the teaching hospital for The University of Hong Kong, with yearly numbers of transplant types. DDLT = deceased donor liver transplant; LDLT = living donor liver transplant. |
|
|
|
Figure 2. Adult recipient survival of transplants for indications other than hepatocellular carcinoma. |
|
|
|
Figure 3. Adult recipient survival of transplants for hepatocellular carcinoma. |
|
|
|
Figure 4. Yearly hospital mortality of liver transplants at Queen Mary Hospital, Hong Kong. |
2. Innovations
2.1. Inclusion of the middle hepatic vein in the right liver graft
In 1990, Tanaka et al14 used the right liver in a pediatric LDLT to avoid a precarious anatomy of the left hepatic artery. In 1996, our center pioneered the use of the right liver in adult LDLT to provide a graft adequate in size for the recipient and to avoid small-for-size syndrome.13 As the right anterior section represents about half the volume of the right liver, the middle hepatic vein was usually included in the right liver graft to prevent congestion of segment 5 and segment 8,15 giving the recipient enough functional liver. The donor should have a remnant liver at least 30% of the original liver. Meticulous preservation of the segment 4b hepatic vein helps to minimize the risk of venous congestion in segment 4.16 Even in the situation of a rather low insertion of the vein into the middle hepatic vein, the latter is divided caudal to the insertion, and merging of it to the right hepatic vein is feasible.17
Implantation of the right and middle hepatic veins is direct and requires no interpositional vascular graft. Merging of the two hepatic veins into a single cuff using venoplasty expedites vascular anastomosis.18 Following anastomosis of the hepatic vein to the inferior vena cava (IVC), patency of the IVC and hemodynamic stability can be restored. Back flow of venous blood through the right portal vein of the graft can be controlled with a bulldog vascular clamp during portal vein anastomosis. As the graft has been preserved in cold histidine-tryptophan-ketoglutarate solution with low potassium content, flushing of the graft before clamp release is not necessary.19 The shortening of cross-clamping time of the IVC by a median of 16 minutes (unpublished data) also renders venovenous bypass unnecessary.20
Conversance with hepatic venoplasty and direct anastomosis between the IVC and the hepatic vein provides convenience in sequential (or domino) LT, in which an amyloid liver is used as a graft. An amyloid liver graft usually does not contain the IVC, so the short hepatic veins can first be merged as a single cuff, which is then anastomosed to the IVC of the recipient.21
In the combined heart–liver transplant and the following sequential LT our center performed a couple of years ago, the single-cuffed hepatic vein of the amyloid liver graft was, with the same principle, anastomosed to the infrahepatic IVC obtained from the deceased heart and liver donor on the back table for the recipient of the amyloid graft. The donor of the amyloid graft did not provide the IVC as side-to-side IVC anastomosis was safer for her because her newly implanted heart would not be able to tolerate IVC cross-clamping.22
2.2. Monoagent nucleoside/nucleotide prophylaxis without hepatitis B immunoglobulin
Passive immunization with parenteral hepatitis B immunoglobulin significantly reduces the chance of HBV infection of transplanted livers.23 However, the cost and the inconvenience of such a regimen are well known. Lamivudine, the first available orally administered effective agent against HBV, has been used, usually in combination with hepatitis B immunoglobulin.24 Our center pioneered lamivudine monotherapy on patients transplanted for hepatitis B,25 and the rate of hepatitis B recurrence from emergence of mutants has been standing at 4–40%. Adoptive immunity to HBV is sometimes found in our patients who have received liver grafts from donors positive for hepatitis B surface antibodies. However, the production of hepatitis B surface antibodies varies in incidence and duration after LT,26 and the effect can only be beneficial and synergistic with lamivudine. Patients who develop lamivudine-resistant mutations can be given add-on adefovir.27
Entecavir, a much more potent nucleoside/nucleotide analogue for the control of HBV, can also be used as a monotherapy agent for LT recipients. It can bring about a 91% loss of hepatitis B surface antigens, with 98.8% having undetectable HBV DNA.28 A long-term study of 362 LT patients with chronic hepatitis B on oral nucleoside/nucleotide analogue therapy without hepatitis B immunoglobulin demonstrated a virological relapse rate at 3 years of 0% for entecavir versus 17% for lamivudine (p < 0.001), highlighting the importance of using a drug with a high barrier to resistance. The regimen without hepatitis B immunoglobulin resulted in excellent long-term survival, with an 8-year survival of 83% without any HBV-related death. 29
2.3. Donor interchange
Exchanging donated organs to overcome blood group incompatibility has taken place nearly a decade ago in kidney transplantation.30 The issue, however, is more complicated in LT, with two major problems. In LDLT, the estimated mortality of donor left hepatectomy is 0.1% and that of donor right hepatectomy is 0.5%,31 which are many times higher than the 0.02% estimated mortality of living kidney donation.32 For the recipient, LT is usually life-saving. Graft failure and the lack of a timely retransplant means death. Thus, donor interchange in LDLT carries a higher donor risk and a higher recipient risk, but with a bigger benefit.
Hwang et al33 of ASAN Medical Center, Seoul, South Korea, and Soin of Medanta Institute, Gurgaon, India, have been active in donor interchange in LDLT. In Hong Kong, we conducted the first donor interchange in 2009. It helped out a B-to-A pair and an A-to-B pair.34 Our second donor interchange involved an altruistic donation from an O-to-AB pair, helping an A-to-B pair.
3. Evolution
3.1. Acute liver failure
When first started, LDLT was for patients with acute liver failure but without a deceased donor liver graft (DDLG).35 LDLT was considered undesirable in the West36 where the supply of DDLGs is not as tight as that in the East. Our LDLT program has been extended to serve patients with less urgent needs, and acute and elective cases have comparable survival outcomes after LDLT.37 As the failure of LDLT for acute liver failure is a consequence of delay in treatment,38 a proactive approach to living donor workup is crucial.39
3.2. Minimum graft size requirement
The ratio of the left liver to the right liver is generally 1:3. If the left liver has a bigger volume in relation to the total liver volume, it is more likely to be adequate as a remnant liver. A larger left liver also means a safer donor right hepatectomy, and is a justification for a right liver LDLT. The result would be an adequate right liver graft and an adequate left liver remnant.
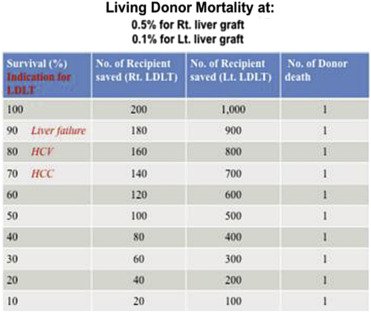
In the case where a donor has a very large body size in relation to the recipient and thus a very large liver, a left liver LDLT is most favorable. Although the workable minimum graft size requirement is 35%, lowering of the requirement with a tiny increase in the recipient mortality rate should be the logical move for LDLT. This could give the ratio of recipient benefit to donor risk a 5-fold increase. In fact, right liver donors are five times more likely to die from the donation than left liver donors (Figure 5). It has been estimated that for every 5% reduction in the minimum graft size requirement, twice as many left liver LDLTs could be feasible.40
|
|
|
Figure 5. The numbers of patients who would be saved with every donor death in left liver and right liver LDLTs for various indications. HCC = hepatocellular carcinoma; HCV = hepatitis C; LDLT = living donor liver transplant; Lt = left; Rt = right. |
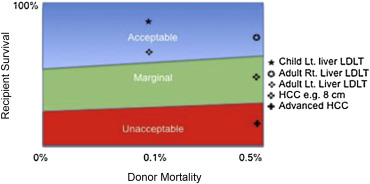
Extending the patient selection criteria for LDLT treating HCC might result in a lower recipient success rate, but the ratio of recipient benefit to donor risk can still be the same if the donor risk is also lowered (Figure 6). Lowering of the donor risk can be attained by using the left liver instead of the right liver. Of course, any graft used must be of adequate size for the recipient. The absence of significant portal hypertension in patients is a factor favoring the adoption of left livers. A high graft portal flow of over 400 mL/100 g/min calls for portal manometry by insertion of a catheter via the inferior mesenteric vein, and a portal pressure of over 20 mmHg calls for portal inflow modulation with splenic artery ligation.41
|
|
|
Figure 6. Equipoise between recipient survival and donor mortality in left liver and right liver LDLTs for different indications. HCC = hepatocellular carcinoma; HCV = hepatitis C; LDLT = living donor liver transplant; Lt = left; Rt = right. |
3.3. Wait and transplant for HCC
DDLGs are a public resource while living donor liver grafts are dedicated gifts. Judicious use of DDLGs is on a utilitarian ground whereas donor autonomy governs the use of living donor liver grafts. Both kinds of grafts are precious and therefore a predictably high recipient survival is to be expected. The graft allocation policy is conservative for HCC patients since DDLGs are scarce and survival outcomes are excellent in DDLT recipients with indications other than HCC. Our center has adopted a new policy since October 2009 to benefit HCC patients who are on the waiting list for DDLT. Under the policy, patients whose HCC has remained at ≤ Stage 2 in the past 6 months are given extra points. The test of time, although reliable, does have a price.42 Patients with less aggressive HCC, as demonstrated by the waiting time, could have been transplanted at least 6 months earlier when the lesions were smaller and perhaps fewer. Survival of these patients could only be better because of a less advanced, perhaps also less aggressive, disease.
3.4. Transplanting the resectable and resecting the transplantable
Providing patients with the treatment that has the best survival outcome is an easily understood policy. However, from the health policy point of view, there are always competitions for resources, and hence resources have to be distributed according to specific criteria, one of which has to be cost-effectiveness. In practice, a treatment is cost-effective if it provides a survival benefit of world standard.
An ideal transplant candidate with HCC and cirrhosis is a patient in whom two or more tumors have been diagnosed and for whom, in the absence of macrovascular and/or lymph node invasion, secure R0 resection of a large tumor cannot be guaranteed.43
LT is offered not only as a primary treatment but also to two groups of patients who have undergone other treatments in advance. The first group is patients whose HCCs have been successfully downstaged using transarterial chemoembolization. Downstaging tumors to a stage within standard criteria, e.g., Milan44 or UCSF (University of California, San Francisco)45, can bring about good transplant outcomes. These tumors are less aggressive as a result of transarterial chemoembolization. The other group is patients failed by local ablative therapy, which typically uses radiofrequency. This treatment policy selects patients having tumors with more aggressive behavior for salvage LT. Poor recipient outcomes are expected.46
If the price paid by liver donors is disregarded, LT is the best treatment for HCCs even if they are resectable, as long as they are intrahepatic, because LT provides the best survival outcome. Nevertheless, using DDLGs for this purpose is out of the question since the benefit is to improve survival by double. For every two DDLGs used, one could have been spared should the patient have undergone liver resection which turned out to be curative.
Donation of liver as a dedicated gift by a living donor is not unreasonable if both the patient and the donor wish to have the best recipient survival. However, one cannot deny that one out of two donations from living donors is unnecessary since one out of two patients can be cured with liver resection.47 It could also be seen as using two donors instead of one donor to save one patient with resectable HCC. The doubling of donor risk or the cost paid by the donors could be offset in a more favorable situation if the left liver of the donor is adequate for the patient who does not have established portal hypertension. This is based on the fact that donor left hepatectomy is five times safer than donor right hepatectomy. Similarly, for a patient with HCC beyond the standard criteria, the recurrence rate is higher. If a left liver is adequate for an LDLT, the ratio of recipient benefit to donor risk is not worse than that in a case using a right liver graft for HCC within the standard criteria. This double equipoise requires careful assessment of recipient survival and donor risk.48
HCC as a cancer does have different degrees of aggressiveness. HCC without vascular invasion often does not metastasize within or outside the liver. Resection is adequate and effective enough if the lesion is resectable. More aggressive HCC often recurs within the liver, resulting in poor disease-free survival. LT includes complete removal of the native liver and thus precludes disease recurrence within the remnant liver after hepatectomy. The most aggressive and advanced HCC, with major vascular invasion as a manifestation, will metastasize within and beyond the liver. Treating such HCC using a precious liver graft, be it from a deceased donor or from a living donor, is not justifiable.
In an intention-to-treat analysis, the rate of salvage LT was only 28%. When a follow-up plan is followed strictly, the rate of recurrence with disease beyond standard criteria for transplantation is particularly high. The recurrence of HCC to a stage beyond standard criteria is predictable if the patient has three of these four features: microvascular invasion, satellite nodules, poor differentiation of tumor cells, and liver cirrhosis. Preemptive LT before recurrence should be considered.49 Nevertheless, the transplant outcome would not be good if the tumor cells are poorly differentiated.50 For unresectable HCCs, as long as the lesions are not large and there are no numerous or major vascular invasions, LT is acceptable and should have a best survival outcome. It has been shown that microvascular invasion per se does not compromise survival as long as the disease is within the up-to-7 criteria. 51
3.5. Rescue liver transplantation
We have performed four cases of rescue LDLT. With careful recipient assessment and expeditious donor workup, all four recipients were saved.52 If a voluntary and suitable living donor is available, a cirrhotic HCC patient who would have a remnant liver marginal in size and quality if liver resection was conducted should not be denied LT. The presence of microvascular invasion, even if confirmed with histopathological examination, is not a contraindication to transplantation if the disease is within the up-to-7 criteria.51 For more advanced HCC, LDLT can still be acceptable if the tumor cells are not poorly differentiated.50 However, given the uncertainty of recipient outcomes and the possibility of post-transplant liver failure, sometimes in addition to a disease stage too advanced for transplantation, LDLT should be conducted only if the donor is anxious to donate. Timeliness is crucial – transplantation must be conducted before sepsis appears.
4. Reflections
4.1. Donor risks
The early postoperative outcomes of right liver donors are markedly different from those of left liver donors as the remnant liver of the former is only half the size of that of the latter. On average, a left liver remnant is only one third of the whole liver. When this ratio gets smaller, the postoperative peak level of serum total bilirubin and international normalized ratio get higher. Nevertheless, the complication rates are similar among groups of donors with different sizes of left liver remnant.53
It is the right of every potential living donor to have a clear understanding of the potential or possible complications as well as the complication rate before consent to donation is given. Although the time needed for a donors liver function to return to normal seems to have no correlation with complication, it is a recovery index. Nonetheless, this is usually not included in the discussion before liver donation. One reason is the absence of studies documenting this. The other reason is the uncertainty of the potential donor being able to understand this rather technical aspect of the surgery. However, potential donors need to keep abreast of the latest knowledge about this major surgery. When more than one volunteer are suitable for a donation, this issue should be raised for all stakeholders to discuss before deciding who is to be the donor. This is on the basis that the volunteers have similar enthusiasm but their remnant liver after donation would be different in size. This principle is also applicable to factors like donor age, body mass index, and physical condition. It is not possible to give an accurate weight on each and every factor, but the balance ought to be made by the donation advocate.
At our center, the donation advocate is a transplant surgeon who has no direct involvement in the management or treatment of the patient, and thus has no conflict of interest. Nevertheless, the advocate ought to have a good knowledge of the chances of success and failure of the proposed LDLT, as well as the 10-week mortality rate of living right liver donors.54 The ratio of recipient benefit to donor risk has to be estimated as accurate as possible so as to enable the patient and the potential donor to come to an informed decision.
4.2. Safe donor surgery
It took 22 years for LDLT to come into clinical practice9 after DDLT had been successfully performed in 1967.51 It then took another 7 years for a right liver LDLT to be applied to adults.13 A prerequisite for an LDLT program is the safe performance of donor hepatectomy. Most budding centers in the late 1990s developed adult LDLT on a solid base of an outstanding hepatobiliary surgery service. The surgeons were already masters of liver surgery and thus were competent at donor hepatectomy.54 This pattern was most recognizable in Japan,55 Taiwan,56 Korea,57 and Hong Kong.58 Some centers in Germany and France59 also had a similar development. Centers in the United States also caught up in the move later.
Although donor hepatectomy is by definition performed on a normal liver, the standard of practice ought to be superb to justify the conduct of this major surgery on a healthy person who has absolutely no medical indication for it. The logical answer to this is to provide the best facilities and the best liver surgeons for the procedure. In the formative years of donor hepatectomy, particularly donor right hepatectomy, controversies arose over whether the middle hepatic vein should be included in the graft or preserved in the remnant liver. It is a matter of balance between providing the most and the best for the recipient and leaving the most and the best with the donor. Donor hepatectomy mandates dedication, technical expertise, and anatomical knowledge of the liver. Although surgeons who carry out this procedure are experts, it is also a learning process for them. Hence a steep learning curve is observed at every transplant center.
For this procedure to be performed by newer surgeons, the transfer of knowledge and skills ought to be smooth, and no significant extra risk should be posed to the donors. Delegation of the surgical duties, as well as the privilege, is a gradual process and should be under guidance and continuous auditing.
At our center, newer surgeons were included as operators in donor right hepatectomies after the first 200 cases. They participated under the guidance of expert surgeons. The involvement of newer surgeons was accompanied by a temporary increase in blood loss and in operation time, evident but well within the acceptable range. These two factors and complication rate, the ultimate outcome measure, are important parameters for assessment of the standard of service. The complication rate did not increase after the first 200 cases. We managed to maintain a high standard of donor right hepatectomy while training a new generation of surgeons.60
5. Conclusion
5.1. Accountable liver transplant service
Altruistic donor interchange enables an ABO-compatible LDLT for an ABO-incompatible pair of donor and recipient. The altruistic O and AB pair deserve a transplant service par excellence; they should not bear any extra risks. Their additional satisfaction from helping the other pair cannot be denied yet is difficult to quantify. Technical successes in LT and the generosity of altruists do have a positive impact on organ donation. Ensuring excellent transplant results is the least the LT community can do in promoting organ donation, be it from the living or from the deceased.61
5.2. Ways ahead
In a rat model, the minimum regenerative threshold of subtotal hepatectomy was a 10% remnant liver, with attention to operative techniques and surgical therapeutics.62 ; 63 The minimum remnant liver size requirement for donor survival after a major hepatectomy on a normal liver is around 25%.64 ; 65 However, an LDLT using a small-for-size graft should produce results comparable with those achieved by liver resection, which include minimal injuries resulting from ischemia, preservation, and reperfusion. Translational studies followed by carefully conducted clinical trials are the way to verify this hypothesis.
It should be considered whether to accept a slightly lower rate of recipient survival when the rate of donor mortality can be lowered significantly from 0.5% to 0.1%. In right liver LDLT, the mortality is one in 200 and one in 50 for donors and recipients, respectively, whereas the corresponding ratios in left liver LDLT are one in 1000 and one in 50. It takes hundreds of donors to really see the difference in donor mortality, and thus the different safety profiles, of the two donor operations. Of course, multicenter studies and surrogate parameters of donor morbidities enable objective appraisal of the difference. It is also easy to appreciate the more speedy recovery of recipients of liver grafts that are adequate in size. This is particularly attractive to patients who are very ill before transplantation. In DDLT, a bad graft for a bad recipient is bad. Thus, transplantation of a small-for-size graft to a very ill patient is usually avoided. Our experience of transplanting bad DDLGs to patients topping the transplant waiting list has been a good lesson for such practice. It is the clear concept of maximizing the ratio of recipient benefit to donor risk that encourages the use of left liver grafts which are smaller yet adequate.
LDLT is here to stay. Its cost and benefit vary according to donor risk, recipient survival, and the availability of DDLGs.66 Donor risk will not increase as long as the current standard of care is maintained. Recipient survival has improved with experience in surgery and surgical therapeutics, as well as improved patient selection. The proportion and the number of LDLTs cannot be reduced without an increase in DDLGs. When the ratio of recipient benefit to donor risk is concerned, donor left hepatectomy is preferable to donor right hepatectomy. The adoption of a lower minimum graft size requirement without compromising recipient survival will encourage the use of left liver lobes as grafts in LDLT.
Acknowledgments
See Ching Chan received research funding from the Li Shu Fan Medical Foundation, Hong Kong.
References
- 1 T.E. Starzl, C.G. Groth, L. Brettschneider, et al.; Orthotopic homotransplantation of the human liver; Ann Surg, 168 (1968), pp. 392–415
- 2 R.W. Busuttil, J.O. Colonna 2nd, J.R. Hiatt, et al.; The first 100 liver transplants at UCLA; Ann Surg, 206 (1987), pp. 387–402
- 3 F.O. Belzer; Clinical organ preservation with UW solution; Transplantation, 47 (1989), pp. 1097–1098
- 4 R.J. Porte, R.J. Ploeg, B. Hansen, et al.; Long-term graft survival after liver transplantation in the UW era: late effects of cold ischemia and primary dysfunction. European Multicentre Study Group; Transplant Int, 11 (1998), pp. S164–716
- 5 R.Y. Calne, D.J. White; The use of cyclosporin A in clinical organ grafting; Ann Surg, 196 (1982), pp. 330–337
- 6 D. Samuel, R. Muller, G. Alexander, et al.; Liver transplantation in European patients with the hepatitis B surface antigen; N Engl J Med, 329 (1993), pp. 1842–1847
- 7 C.M. Lo, S.T. Fan, C.L. Lai, et al.; Lamivudine prophylaxis in liver transplantation for hepatitis B in Asians; Transplant Proc, 31 (1999), pp. 535–536
- 8 V. Mazzaferro, E. Regalia, R. Doci, et al.; Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis; N Engl J Med, 334 (1996), pp. 693–699
- 9 R.W. Strong, S.V. Lynch, T.H. Ong, et al.; Successful liver transplantation from a living donor to her son; N Engl J Med, 322 (1990), pp. 1505–1507
- 10 Y. Hashikura, M. Makuuchi, S. Kawasaki, et al.; Successful living-related partial liver transplantation to an adult patient; Lancet, 343 (1994), pp. 1233–1234
- 11 K. Tanaka, S. Uemoto, Y. Tokunaga, et al.; Surgical techniques and innovations in living related liver transplantation; Ann Surg, 217 (1993), pp. 82–91
- 12 C.M. Lo, P. Gertsch, S.T. Fan; Living unrelated liver transplantation between spouses for fulminant hepatic failure; Br J Surg, 82 (1995), p. 1037
- 13 C.M. Lo, S.T. Fan, C.L. Liu, et al.; Extending the limit on the size of adult recipient in living donor liver transplantation using extended right lobe graft; Transplantation, 63 (1997), pp. 1524–8152
- 14 Y. Yamaoka, M. Washida, K. Honda, et al.; Liver transplantation using a right lobe graft from a living related donor; Transplantation, 57 (1994), pp. 1127–1130
- 15 S. Lee, K. Park, S. Hwang, et al.; Congestion of right liver graft in living donor liver transplantation; Transplantation, 71 (2001), pp. 812–814
- 16 S.C. Chan, C.M. Lo, C.L. Liu, et al.; Tailoring donor hepatectomy per segment 4 venous drainage in right lobe live donor liver transplantation; Liver Transpl, 10 (2004), pp. 755–762
- 17 S.C. Chan, C.M. Lo, C.L. Liu, et al.; Versatility and viability of hepatic venoplasty in live donor liver transplantation using the right lobe with the middle hepatic vein; Hepatobiliary Pancreat Dis Int, 4 (2005), pp. 618–621
- 18 C.M. Lo, S.T. Fan, C.L. Liu, et al.; Hepatic venoplasty in living-donor liver transplantation using right lobe graft with middle hepatic vein; Transplantation, 75 (2003), pp. 358–360
- 19 S.C. Chan, C.L. Liu, C.M. Lo, et al.; Applicability of histidine-tryptophan-ketoglutarate solution in right lobe adult-to-adult live donor liver transplantation; Liver Transpl, 10 (2004), pp. 1415–1421
- 20 S.T. Fan, B.H. Yong, C.M. Lo, et al.; Right lobe living donor liver transplantation with or without venovenous bypass; Br J Surg, 90 (2003), pp. 48–56
- 21 S.C. Chan, C.M. Lo, K.K. Ng, et al.; Simplifying hepatic venous outflow reconstruction in sequential living donor liver transplantation; Liver Transpl, 15 (2009), pp. 1514–1518
- 22 S.C. Chan, L.C. Cheng, K.L. Ho, et al.; Improvising hepatic venous outflow and inferior vena cava reconstruction for combined heart and liver and sequential liver transplantations; Asian J Surg, 36 (2013), pp. 89–92
- 23 P.J. Johnson, M.H. Wansbrough-Jones, B. Portmann, et al.; Familial HBsAg-positive hepatoma: treatment with orthotopic liver transplantation and specific immunoglobulin; Br Med J, 1 (1978), p. 216
- 24 J.S. Markowitz, P. Martin, A.J. Conrad, et al.; Prophylaxis against hepatitis B recurrence following liver transplantation using combination lamivudine and hepatitis B immune globulin; Hepatology, 28 (1998), pp. 585–589
- 25 C.M. Lo, S.T. Cheung, C.L. Lai, et al.; Liver transplantation in Asian patients with chronic hepatitis B using lamivudine prophylaxis; Ann Surg, 233 (2001), pp. 276–281
- 26 C.M. Lo, J.T. Fung, G.K. Lau, et al.; Development of antibody to hepatitis B surface antigen after liver transplantation for chronic hepatitis B; Hepatology, 37 (2003), pp. 36–43
- 27 C.M. Lo, C.L. Liu, G.K. Lau, et al.; Liver transplantation for chronic hepatitis B with lamivudine-resistant YMDD mutant using add-on adefovir dipivoxil plus lamivudine; Liver Transpl, 11 (2005), pp. 807–813
- 28 J. Fung, C. Cheung, S.C. Chan, et al.; Entecavir monotherapy is effective in suppressing hepatitis B virus after liver transplantation; Gastroenterology, 141 (2011), pp. 1212–1219
- 29 J. Fung, S.C. Chan, C. Cheung, et al.; Oral nucleoside/nucleotide analogs without hepatitis B immune globulin after liver transplantation for hepatitis B; Am J Gastroenterol, 108 (2013), pp. 942–948
- 30 F.L. Delmonico; Exchanging kidneys–advances in living-donor transplantation; N Engl J Med, 350 (2004), pp. 1812–1814
- 31 M.L. Barr, J. Belghiti, F.G. Villamil, et al.; A report of the Vancouver Forum on the care of the live organ donor: lung, liver, pancreas, and intestine data and medical guidelines; Transplantation, 81 (2006), pp. 1373–1385
- 32 A.J. Matas, S.T. Bartlett, A.B. Leichtman, et al.; Morbidity and mortality after living kidney donation, 1999–2001: survey of United States transplant centers; Am J Transplant, 3 (2003), pp. 830–834
- 33 S. Hwang, S.G. Lee, D.B. Moon, et al.; Exchange living donor liver transplantation to overcome ABO incompatibility in adult patients; Liver Transpl, 16 (2010), pp. 482–490
- 34 S.C. Chan, C.M. Lo, B.H. Yong, et al.; Paired donor interchange to avoid ABO-incompatible living donor liver transplantation; Liver Transpl, 16 (2010), pp. 478–481
- 35 C.M. Lo, S.T. Fan, C.L. Liu, et al.; Adult-to-adult living donor liver transplantation using extended right lobe grafts; Ann Surg, 226 (1997), pp. 261–269 discussion 269–270
- 36 D.C. Cronin 2nd, J.M. Millis, M. Siegler; Transplantation of liver grafts from living donors into adults–too much, too soon; N Engl J Med, 344 (2001), pp. 1633–1637
- 37 A.C. Chan, S.T. Fan, C.M. Lo, et al.; Liver transplantation for acute-on-chronic liver failure; Hepatol Int, 3 (2009), pp. 571–581
- 38 C.M. Lo; Living donor liver transplantation for acute liver failure: no other choice; Liver Transpl, 18 (2012), pp. 1005–1006
- 39 S.C. Chan, S.T. Fan, C.L. Liu, et al.; Working up donors for high-urgency and elective adult-to-adult live donor liver transplantation; Liver Transpl, 13 (2007), pp. 509–515
- 40 S.C. Chan, S.T. Fan, K.S. Chok, et al.; Increasing the recipient benefit/donor risk ratio by lowering the graft size requirement for living donor liver transplantation; Liver Transpl, 18 (2012), pp. 1078–1082
- 41 S.C. Chan, C.M. Lo, K.S. Chok, et al.; Modulation of graft vascular inflow guided by flowmetry and manometry in liver transplantation; Hepatobiliary Pancreat Dis Int, 10 (2011), pp. 649–656
- 42 S.C. Chan, W.W. Sharr, K.S. Chok, et al.; Wait and transplant for stage 2 hepatocellular carcinoma with deceased-donor liver grafts; Transplantation, 96 (2013), pp. 995–999
- 43 J. Lerut, H. Mergental, D. Kahn, et al.; Place of liver transplantation in the treatment of hepatocellular carcinoma in the normal liver; Liver Transpl, 17 (2011), pp. S90–97
- 44 Y.F. Cheng, T.L. Huang, T.Y. Chen, et al.; Impact of pre-operative transarterial embolization on the treatment of hepatocellular carcinoma with liver transplantation; World J Gastroenterol, 11 (2005), pp. 1433–1438
- 45 F.Y. Yao, R.K. Kerlan Jr., R. Hirose, et al.; Excellent outcome following down-staging of hepatocellular carcinoma prior to liver transplantation: an intention-to-treat analysis; Hepatology, 48 (2008), pp. 819–827
- 46 C.M. Lo, S.T. Fan, C.L. Liu, et al.; Living donor versus deceased donor liver transplantation for early irresectable hepatocellular carcinoma; Br J Surg, 94 (2007), pp. 78–86
- 47 S.T. Fan, C. Mau Lo, R.T. Poon, et al.; Continuous improvement of survival outcomes of resection of hepatocellular carcinoma: a 20-year experience; Ann Surg, 253 (2011), pp. 745–758
- 48 E.A. Pomfret, J.P. Lodge, F.G. Villamil, et al.; Should we use living donor grafts for patients with hepatocellular carcinoma? Ethical considerations; Liver Transpl, 17 (2011), pp. S128–132
- 49 D. Fuks, S. Dokmak, V. Paradis, et al.; Benefit of initial resection of hepatocellular carcinoma followed by transplantation in case of recurrence: an intention-to-treat analysis; Hepatology, 55 (2012), pp. 132–140
- 50 D. DuBay, C. Sandroussi, L. Sandhu, et al.; Liver transplantation for advanced hepatocellular carcinoma using poor tumor differentiation on biopsy as an exclusion criterion; Ann Surg, 253 (2011), pp. 166–172
- 51 S.C. Chan, S.T. Fan, K.S. Chok, et al.; Survival advantage of primary liver transplantation for hepatocellular carcinoma within the up-to-7 criteria with microvascular invasion; Hepatol Int, 6 (2011), pp. 646–656
- 52 S.C. Chan, W.W. Sharr, A.C. Chan, et al.; Rescue living-donor liver transplantation for liver failure following hepatectomy for hepatocellular carcinoma; Liver Cancer, 2 (2013), pp. 332–337
- 53 T.E. Starzl, T.L. Marchioro, K.A. Porter, et al.; Homotransplantation of the liver; Transplantation, 5 (1967), pp. S790–803
- 54 R.W. Strong; Surgical resection for cholangiocarcinoma involving the confluence of the major hepatic ducts; Aust N Z J Surg, 57 (1987), pp. 911–915
- 55 M. Makuuchi, H. Hasegawa, S. Yamazaki, et al.; Four new hepatectomy procedures for resection of the right hepatic vein and preservation of the inferior right hepatic vein; Surg Gynecol Obstet, 164 (1987), pp. 68–72
- 56 C.L. Chen, K.M. Fang, Y.L. Hui; Experience with major hepatic resections for hepatocellular carcinoma; Cancer Chemother Pharmacol, 23 (1989), pp. S101–103
- 57 S.G. Lee, Y.J. Lee, K.M. Park, et al.; One hundred and eleven liver resections for hilar bile duct cancer; J Hepatobiliary Pancreat Surg, 7 (2000), pp. 135–141
- 58 S.T. Fan, C.M. Lo, C.L. Liu, et al.; Hepatectomy for hepatocellular carcinoma: toward zero hospital deaths; Ann Surg, 229 (1999), pp. 322–330
- 59 H. Bismuth; Surgical anatomy and anatomical surgery of the liver; World J Surg, 6 (1982), pp. 3–9
- 60 S.C. Chan, A.C. Chan, W.W. Sharr, et al.; Perpetuating proficiency in donor right hepatectomy for living donor liver transplantation; Asian J Surg, 37 (2014), pp. 65–72
- 61 S.C. Chan, W.W. Sharr, K.S. Chok, et al.; Media coverage of liver transplant events promotes donations from the deceased; Hong Kong Med J, 19 (2013), pp. 89–91
- 62 J. Gaub, J. Iversen; Rat liver regeneration after 90% partial hepatectomy; Hepatology, 4 (1984), pp. 902–904
- 63 N. Madrahimov, O. Dirsch, C. Broelsch, et al.; Marginal hepatectomy in the rat: from anatomy to surgery; Ann Surg, 244 (2006), pp. 89–98
- 64 A. Ferrero, L. Vigano, R. Polastri, et al.; Postoperative liver dysfunction and future remnant liver: where is the limit? Results of a prospective study; World J Surg, 31 (2007), pp. 1643–1651
- 65 M.J. Schindl, D.N. Redhead, K.C. Fearon, et al.; The value of residual liver volume as a predictor of hepatic dysfunction and infection after major liver resection; Gut, 54 (2005), pp. 289–296
- 66 C.M. Miller; Ethical dimensions of living donation: experience with living liver donation; Transplant Rev (Orlando), 22 (2008), pp. 206–209
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?