Abstract
Objectives
Our aim was to evaluate the feasibility and safety of routine transradial approach (TRA) percutaneous coronary intervention (PCI) for chronic total occlusion (CTO) lesions using the sheathless technique with standard guiding catheters.
Background
Transradial approach PCI was applied for CTO lesions. A major limitation of TRA CTO PCI is the inability to use large guiding catheters because of the relatively small size of the radial artery. Therefore, the sheathless technique for TRA PCI has been recently developed. However, reports on TRA CTO PCI using the sheathless technique are still lacking.
Methods
Sixty-eight patients with CTO lesions were enrolled for TRA PCI using the sheathless technique with standard guiding catheters. The baseline characteristics, coronary angiographic characteristics and major procedure or access site related complications were compared between procedure success and procedure failure group to determine the predictors of success in sheathless CTO PCI. In-hospital and 30-day clinical outcomes were also evaluated in this study. Routine assessments of radial artery occlusion via Doppler ultrasound and pulse oximeter were recorded during one-year clinical follow-up.
Results
The mean duration of CTO by history was 31.8 ± 42.3 months. The 7 Fr standard guiding catheter was used with the sheathless technique in 91.2%, and bilateral sheathless approach in 42.6% of the study patients. The procedure-related complications included coronary perforation needing covered stent deployment (2.9%), cardiac tamponade (2.9%), collateral perforation needing coil deployment (4.4%), and contrast induced nephropathy (2.9%). Only 2 patients (2.9%) experienced forearm ecchymosis at the radial artery access sites. In-hospital mortality and 30-day all-cause mortality were 2.9%, and 30-day MACEs were 1.5%. The rate of radial artery occlusion during one-year clinical follow-up was only 3.0%.
Conclusions
It is feasible and safe to routinely use the sheathless technique with standard guiding catheters for TRA CTO PCI, with a low incidence of procedure-related complications and long-term radial artery occlusion.
Keywords
Transradial approach;Percutaneous coronary intervention;Chronic total occlusion;Sheathless guiding catheter
1. Introduction
Chronic total occlusions (CTO) of coronary arteries are widely encountered in patients undergoing coronary angiography, with an incidence rate between 18.4% and 52% [1]; [2] ; [3]. Despite the advance in techniques and technology, percutaneous coronary intervention (PCI) for CTO remains a challenge. Therefore, the attempted rate of CTO PCI was only 10% to 15% in patients with coronary artery disease, for whom CTO was the most common reason for coronary artery bypass surgery [2] ; [3]. Successful CTO PCI seems to be associated with improved short-term and long-term clinical outcomes and mortality [4] ; [5].
The transfemoral arterial approach (TFA) is the worldwide accepted method for CTO interventions because the artery can accommodate a larger guiding catheter, and hence provides better support and a larger working space. However, the conventional TFA has several clinical and anatomical limitations including distal abdominal aortic disease, very tortuous or occluded ilio-femoral arteries, morbid obesity, and long period of bed rest required after the procedure. Significant hemostatic difficulties can occur with the TFA in obese patients, which may increase the risk of groin complications, and lead to longer hospitalization.
Transradial approach (TRA) PCI was introduced two decades ago, and now this technique is being generally applied. The feasibility and safety of TRA PCI have also been approved for acute coronary syndrome, bifurcation lesions, unprotected left main lesions, carotid artery stenting, and CTO [6]; [7]; [8]; [9] ; [10]. Recently, the retrograde approach via collaterals was the most significant advance in PCI techniques for CTO, which needs larger guiding catheters for good backup support and complex devices. However, one of the major limitations regarding TRA CTO PCI is the inability of using larger guiding catheters, due to the relatively smaller size of the radial arteries compared to that of the femoral arteries. In fact, Saito et al. [11] determined that the radial artery lumen is smaller than the 7 Fr introducer sheath in almost one-third of the men and two-thirds of the women in Japan.
The 7Fr standard guiding catheters had the same diameter of the outer lumen compared with a 5Fr artery sheath which was far smaller than the usual radial artery size. Using the sheathless technique could facilitate any 7Fr standard guiding catheter to be available in transradial CTO intervention. Mamas and colleagues [12] reported a pilot study of a 6.5 Fr sheathless guiding catheter system that may resolve the limitation of size in TRA PCI, but cannot change guiding catheters accommodated with each patient when performing CTO PCI. Although the sheathless guiding catheter technique has recently been applied to complex PCI, such as bifurcation lesions and rotational atherectomy [13]; [14] ; [15]. Studies to investigate the feasibility and safety of CTO PCI using the sheathless technique are still lacking.
The aim of the present pilot study was to evaluate the safety and feasibility of the sheathless technique using standard guiding catheters for TRA CTO PCI in routine practice, the predictors of success sheathless CTO PCI and the in-hospital and 30-day clinical outcomes.
2. Materials and methods
2.1. Patient population
Between January 2010 and June 2012, 72 patients who received TRA CTO PCI using the sheathless technique with standard guiding catheters were recruited. Exclusion criteria included: (1) a history of acute or recent ischemic or hemorrhagic stroke (< 2 months), (2) an episode of acute or recent myocardial infarction (MI), (3) major surgery or trauma within the preceding 2 months and (4) end-stage renal disease (ESRD). Four patients with recent MI undergoing staged revascularization for CTO lesions were excluded. Finally, 68 patients were enrolled into this study for evaluation. All patients gave their written informed consent prior to participation in the study, and the study protocol was approved by our hospitals Internal Research Board Committee.
2.2. Operator selection
The CTO intervention procedures were limited to one experienced operator (C.J.W.) in our institution. The experienced operator met the criteria of “CTO operators” who had specific proctored experience of more than 100 CTOs [16].
2.3. Definitions
CTO lesion was defined according to the thrombolysis in myocardial infarction grade (TIMI) of 0 for more than 3 months, and with the presence of typical angina or reversible myocardial ischemia in a thallium stress study. The duration of occlusion was estimated by the history of angina, history of MI in the same territory, or previous coronary angiography. The definition of procedure success was that successful guidewire and balloon were crossed with or without final stenting, with a residual diameter stenosis of < 30%, and the presence of a final TIMI 3 flow. Major adverse cardiac events (MACEs) included cardiovascular death, recurrent MI, or urgent revascularization during the same hospitalization. Contrast induced nephropathy (CIN) was defined as an increase of at least 0.5 mg/dL or at least 25% of baseline serum creatinine concentration after exposure to contrast media within 48 to 72 h. Acute stent thrombosis was defined as an episode occurring within 24 h, and subacute stent thrombosis within 30 days after the index procedure.
2.4. Procedure and protocol
2.4.1. Radial arterial puncture and sheathless technique steps
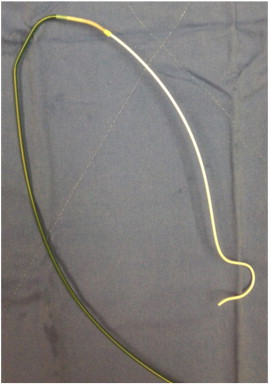
Both radial arteries were evaluated by either Allens test or portable Doppler ultrasound before the PCI procedures. A 5 Fr or 6 Fr conventional arterial sheath was inserted via the usual radial artery access site, defined as a puncture site about 3–5 cm above the styloid process. To prevent vasospasm and thromboembolic events, a cocktail solution consisting of 200 μg nitroglycerin and 60 I.U. per kilogram heparin was administered via the artery sheath. Before performing CTO PCI, a standard guiding catheter was used for diagnostic coronary angiography. A 0.035″ 260 cm J-tipped Teflon wire was inserted and reached the proximal ascending aorta, and then the 5 Fr artery sheath was exchanged to a 7r Fr standard guiding catheter which was loaded by a 5 Fr 125 cm VTK catheter (VTK, Cook Inc., Bloomington, IN, USA) in order to taper down the distal tip (Fig. 1), with the sheathless technique following From et al.s report [17]. After the procedures, the standard guiding catheter was removed along with the J-tip Teflon wire, and the access site was compressed by a TR band (Terumo, Tokyo, Japan).
|
|
|
Fig. 1. A 7 Fr Kimny guiding catheter (Boston Scientific, Grove, MN, USA) was loaded by a 5 Fr 125 cm VTK catheter (VTK, Cook Inc., Bloomington, IN, USA) in order to taper down the distal tip. |
2.5. Selection of standard guiding catheters
At first, a 5 Fr Ikari IL 3.5 guiding catheter (Terumo, Tokyo, Japan), or a 6 Fr Kimny Mini-radial guiding catheter (Boston Scientific, Grove, MN, USA) was used for diagnostic coronary angiography. Standard nonhydrophilic guiding catheters consisting of a 7 Fr Kimny Mini-radial guiding catheter (Boston), a 7 Fr 100 cm long BL (backup left) 3.5–4 guiding catheter (Terumo), or a 7 Fr 100 cm short AL (Amplatz left) 1 guiding catheter (Terumo) was selected for the antegrade or retrograde approach according to each individual patient. Before PCI for CTO lesions, a second intracoronary bolus of 5000 I.U. heparin was administered via the guiding catheters to keep activated clotting time 250 to 300 s.
2.6. Selection of antegrade or retrograde guidewires
Guidewire selection for the antegrade approach was decided by operator after diagnostic coronary angiography. The guidewires ranged from those with usual stiffness as Runthrough Floppy (Terumo, Japan) or Rinato wires (Asahi, Tokyo, Japan), hydrophilic wires such as Pilot (Abbott, Abbott Park, IL, USA) or Fielder wires (FC or XT; Asahi) or SION (Asahi), and stepwise harder-tip wires such as Miracle (3, 4.5, 6, 12 g; Asahi) or Conquest wires (9, 12, or 20/8 g; Asahi). The selected guidewire for the antegrade approach was placed at the proximal cap of the CTO site with a 1.8 Fr microcatheter (Finecross; Terumo). For the retrograde approach, we super-selected a suitable collateral vessel with a microcatheter, and then selected guidewires including either Fielder-FC (Asahi), Fielder-XT (Asahi), or SION (Asahi) wires under a microcatheter support to advance into the distal part of the CTO lesion. Externalization was completed with a 0.010″ long RG-3 300 cm guidewire (Asahi).
2.7. Bilateral transradial approach for simultaneous coronary injections
If antegrade approach failed or was infeasible due to unfavorable anatomy, the retrograde approach using bilateral simultaneous coronary injections was tried through suitable retrograde collaterals consisting of septal collaterals, epicardial collaterals, saphenous vein grafts, or the left internal mammary artery. Recanalization by retrograde approach required one or more complex techniques, such as kissing wire technique, controlled antegrade and retrograde subintimal tracking (CART) technique, reverse CART technique, knuckle wire technique, or parallel-wire technique. After the retrograde guidewire had passed the occluded segment, the retrograde guidewire was manipulated into the antegrade guiding catheter. Then, the original 190 cm guidewire was exchanged for a long RG-3 300 cm guidewire through a microcatheter. Stepwise balloon angioplasty was done and was followed by the evaluation of intravascular ultrasound study (IVUS) for the preparation of subsequent stenting to target CTO lesions. After successful balloon dilatation, either bare-metal stents (BMS), or drug-eluting stents (DES), were deployed, based on the patients preference. (Since general health insurance guidelines in Taiwan only reimburse for BMS, the additional cost of DES needs to be paid by the patient.)
2.8. Medications
Patients received medical treatments including oral aspirin (100 mg/day) and anti-ischemic agents. The pre-medication with a loading dose of clopidogrel (300 or 600 mg) was given 4 to 12 h before the index procedure. A post-procedure dose of clopidogrel (300 mg) was also administered following the maintenance dose of clopidogrel (75 mg per day) for at least 9 months after the implantation of DES, or at least 3 months after BMS deployment or balloon angioplasty.
2.9. Assessment of the predictors of success in sheathless CTO PCI
To determine the predictors of success in sheathless CTO PCI, the enrolled patients were divided into procedure success and procedure failure groups. Baseline characteristics, coronary angiographic characteristics and major procedural or access site related complications were compared between procedure success and procedure failure group.
2.10. Assessment of in-hospital outcomes and 30-day clinical outcomes
In-hospital outcomes were classified as in-hospital mortality, cardiac death, recurrent myocardial infarction (MI), stent thrombosis, arrhythmia or stroke, in-hospital MI was defined by elevation of cardiac troponin values > 5 times upper reference limit in patients with normal baseline values or a rise of cardiac troponin values > 20% if the baseline values are elevated as 3rd universal definition with type 4b PCI related MI. Death from uncertain causes was also classified as cardiac death. 30-day MACEs were defined as above.
2.11. Assessment of radial artery occlusion
All patients were followed up to evaluate radial artery occlusion at the access sites, physical examination for radial pulsation, Doppler ultrasound for blood flow of the radial arteries at access sites, and pulse oximeter were performed during one-year clinical follow-up. Radial artery occlusion was defined as the absence of a radial pulsation with the absence of blood flow detected by Doppler ultrasound probe. In order to eliminate the blood supply from collaterals, the ipsilateral ulnar artery was compressed for 30 s before and during radial artery assessment.
2.12. Statistical analysis
Data were presented as means ± SD. Categorical variables between the successful procedure group and the failed procedure group were compared using chi-square test or Fisher exact test as appropriate. Moreover, continuous variables between the two groups of study patients were compared by Mann–Whitney U test. Finally, statistical analyses were performed using a statistical software program (SPSS version 19.0; SPSS Inc.; Chicago, Illinois, U.S.A.). All P values were two-sided, and the level of statistical significance was set at 0.05.
3. Results
3.1. Baseline demographic characteristics of study patients
A total of 68 patients were enrolled in this study, with the procedure success rate of up to 85.3% (58 successful cases, and 10 failed cases separately). Table 1 reveals the baseline demographic characteristics of the study patients. The mean age of all patients was 62.8 ± 11.4 years, and the prevalence of male patients was 83.8%. There were no significant differences in age, body mass index, cholesterol level, low-density lipoprotein (LDL) level and hemoglobin A1C (HbA1C) between the procedure successful group and the procedure failed group. When comparing with the procedural success group, the procedure failed group had fewer males (60.0% vs. 87.9%, P = 0.049). After excluding patients with end-stage renal disease, the creatinine level in the procedure successful group was lower than that in the procedure failed group without a significant difference (1.0 ± 0.3 mg/dL vs. 1.6 ± 0.9 mg/dL, P = 0.066). The prevalence of risk factors for coronary artery disease, including hypertension, diabetes mellitus, dyslipidemia, and smoking habit did not differ between the two groups. Moreover, the incidence of comorbidities including previous myocardial infarction (MI), previous coronary artery bypass graft history, previous stroke, peripheral vascular disease, and end-stage renal disease was similar between the two groups. The duration of CTO and the left ventricular ejection fraction was also similar between the procedure successful group and the procedure failed group (32.7 ± 43.0 months vs. 26.4 ± 39.7 months, P = 0.547; 58.2 ± 15.8% vs. 54.3 ± 13.5%, P = 0.393, respectively).
| All (n = 68) | Success (n = 58) | Failure (n = 10) | P value | |
|---|---|---|---|---|
| Demographics | ||||
| Age (years) | 62.8 ± 11.4 | 62.3 ± 10.8 | 65.5 ± 14.8 | 0.298⁎ |
| Male | 57 (83.8) | 51 (87.9) | 6 (60.0) | 0.049 |
| Body mass index (kg/m2) | 25.7 ± 3.8 | 25.8 ± 3.8 | 25.2 ± 4.0 | 0.641 |
| Creatinine (mg/dL)§ | 1.1 ± 0.4 | 1.0 ± 0.3 | 1.6 ± 0.9 | 0.066§ |
| Cholesterol (mg/dL) | 150.9 ± 35.5 | 148.2 ± 30.3 | 167.5 ± 58.6 | 0.323 |
| LDL (mg/dL) | 114.7 ± 36.1 | 111.5 ± 32.8 | 117.1 ± 47.3 | 0.508 |
| HbA1C | 7.7 ± 1.8 | 7.5 ± 1.6 | 8.1 ± 2.4 | 0.781 |
| Risk factors | ||||
| Hypertension | 55 (80.9) | 46 (79.3) | 9 (90.0) | 0.673 |
| Diabetes mellitus | 27 (39.7) | 23 (39.7) | 4 (40.0) | 1.000 |
| Dyslipidemia | 28 (41.2) | 22 (37.9) | 6 (60.0) | 0.297 |
| Smoking habit | 15 (22.1) | 15 (25.9) | 0 (0) | 0.102 |
| Comorbidity | ||||
| Previous MI | 19 (27.9) | 18 (31.0) | 1 (10.0) | 0.262 |
| Previous CABG | 4 (5.9) | 3 (5.2) | 1 (10.0) | 0.479 |
| Previous stroke | 9 (13.2) | 9 (15.5) | 0 (0) | 0.336 |
| Peripheral vascular disease | 4 (5.9) | 4 (6.9) | 0 (0) | 1.000 |
| ESRD | 5 (7.4) | 3 (5.2) | 2 (20.0) | 0.153 |
| Duration of CTO (months) | 31.8 ± 42.3 | 32.7 ± 43.0 | 26.4 ± 39.7 | 0.547 |
| LVEF (%) | 57.7 ± 15.5 | 58.2 ± 15.8 | 54.3 ± 13.5 | 0.393 |
LDL indicates low-density lipoprotein; HbA1C, hemoglobin A1C; MI, myocardial infarction; CABG, coronary artery bypass graft; ESRD, end-stage renal disease; CTO, chronic total occlusion; and LVEF, left ventricle ejection fraction.
⁎. Data are presented as mean ± SD or number (%) of patients.
§. Creatinine level was calculated after excluding patients with end-stage renal disease.
3.2. Coronary angiographic characteristics of CTO lesions
Table 2 shows the coronary angiographic characteristics of study patients. Ad hoc PCI was performed in 27 patients (39.7%), but no significant difference was found between the procedure successful group and the procedure failed group. The total procedural time, fluoroscopy time, and contrast volume were similar between the two groups. The average number of access time of the radial arteries was 1.8 ± 1.0, and in the procedure failed group radial arteries were frequently punctured when compared with the procedure successful group, although the difference did not reach a level of significance (2.3 ± 1.6 vs. 1.7 ± 0.9, P = 0.375). A 7 Fr standard guiding catheter for sheathless technique was used in 62 patients (91.2%) during TRA CTO PCI. Contralateral injection for antegrade/retrograde approaches and simultaneous injection for CTO lesions were applied in 72.1% of the study patients, and the bilateral sheathless approach was used in 42.6%. The most common target CTO lesion was the right coronary artery (54.4%), followed by the left anterior descending artery (39.7%), and left circumflex artery (5.9%), without relationship to procedure success. Collaterals were used in 52 patients (76.5%). The most frequently used collaterals were septal collaterals (48.5%), followed by epicardial collaterals (23.5%), and bypass grafts (4.4%). The occlusion lesion length of CTO was about 74.7 ± 26.8 mm, and there was no difference between the two groups. When comparing with the procedural successful group, Syntax score in the procedure failed group was higher than that in the procedure successful group, although the difference did not reach a level of significance (25.1 ± 11.0 vs. 18.5 ± 9.9, P = 0.091). The procedure successful group received more DES implantation for CTO lesions than those of the procedure failed group (70.7% vs. 10.0%, P < 0.001). Furthermore, IVUS-guided PCI was also more frequently used in the procedure successful group than in the procedure failed group (74.1% vs. 30.0%, P = 0.010). In addition, rotational atherectomy was performed on 5.9% of the study patients.
| All (n = 68) | Success (n = 58) | Failure (n = 10) | P value | |
|---|---|---|---|---|
| Ad hoc PCI | 27 (39.7) | 22 (37.9) | 5 (50.0) | 0.502⁎ |
| Total procedure time (minutes) | 156.7 ± 65.4 | 153.6 ± 69.3 | 173.8 ± 34.9 | 0.252 |
| Fluoroscopy time (minutes) | 77.4 ± 41.2 | 76.0 ± 43.7 | 84.7 ± 25.8 | 0.395 |
| Contrast volume (mL) | 440.9 ± 173.0 | 437.1 ± 170.9 | 461.4 ± 197.1 | 0.695 |
| Access time of radial arteries | 1.8 ± 1.0 | 1.7 ± 0.9 | 2.3 ± 1.6 | 0.375 |
| Assess time ≧ 2 | 34 (50.0) | 29 (50.0) | 5 (50.0) | 1.000 |
| 7 Fr sheathless guiding catheter | 62 (91.2) | 52 (89.7) | 10 (100.0) | 0.581 |
| Contralateral injection | 49 (72.1) | 41 (70.7) | 8 (80.0) | 0.714 |
| Bilateral sheathless approach | 29 (42.6) | 25 (43.1) | 4 (40.0) | 1.000 |
| Multiple-vessel disease | 61 (89.7) | 52 (89.7) | 9 (90.0) | 1.000 |
| Target CTO lesion | ||||
| LAD | 27 (39.7) | 22 (37.9) | 5 (50.0) | 0.502 |
| LCX | 4 (5.9) | 4 (6.9) | 0 (0) | 1.000 |
| RCA | 37 (54.4) | 32 (55.2) | 5 (50.0) | 1.000 |
| Collaterals use | 52 (76.5) | 45 (77.6) | 7 (70.0) | 0.689 |
| Septal | 33 (48.5) | 28 (48.3) | 5 (50.0) | 1.000 |
| Epicardial | 16 (23.5) | 14 (24.1) | 2 (20.0) | 1.000 |
| Graft | 3 (4.4) | 3 (5.2) | 0 (0) | 1.000 |
| Retrograde success | 31 (45.6) | 28 (48.3) | 3 (30.0) | 0.326 |
| Occlusion lesion length (mm) | 74.7 ± 26.8 | 74.1 ± 26.1 | 77.5 ± 31.5 | 0.733 |
| Syntax score | 19.5 ± 10.3 | 18.5 ± 9.9 | 25.1 ± 11.0 | 0.091 |
| Stent use | ||||
| Drug-eluting stent | 42 (61.8) | 41 (70.7) | 1 (10.0) | < 0.001 |
| Bare metal stent | 18 (26.5) | 17 (29.3) | 1 (10.0) | 0.270 |
| Balloon angioplasty | 5 (7.4) | 0 (0) | 5 (50.0) | < 0.001 |
| IVUS-guided PCI | 46 (67.6) | 43 (74.1) | 3 (30.0) | 0.010 |
| Rotational atherectomy | 4 (5.9) | 3 (5.2) | 1 (10.0) | 0.479 |
PCI indicates percutaneous coronary intervention; CTO, chronic total occlusion; LAD, left anterior descending artery; LCX, left circumflex artery; RCA, right coronary artery; IVUS, intra-vascular ultrasound.
⁎. Data are presented as mean ± SD or number (%) of patients.
Table 3 summarizes data for the major procedural and access site complications during sheathless TRA CTO PCI. In the procedure successful group, 2 patients (2.9%) suffered from cardiac tamponade, and needed emergent pericardiocentesis followed by pig-tail drainage. One patient (1.5%) in the procedure successful group had aortic dissection from the right coronary artery, and donor vessel dissection occurred in another 2 patients (2.9%), who were treated with stent deployment under IVUS guidance. Moreover, 2 patients (2.9%) suffered from type III coronary perforation (the Ellis Classification Scheme) in the procedural successful group. Both of these 2 patients needed the deployment of Cover stent (Boston) and one of them even needed emergent surgery to repair coronary perforation. The perforation of collaterals requiring coil deployment occurred in 3 patients (4.4%). However, there were no differences between the procedure successful group and the procedure failed group with respect to procedure-related complications. Two patients (2.9%) experienced contrast induced nephropathy (CIN), and completely recovered with conservative treatments. Only 2 patients (2.9%) experienced forearm ecchymoses after the procedure without clinical signs of compartment syndrome or need of blood transfusion or surgical intervention.
| All (n = 68) | Success (n = 58) | Failure (n = 10) | P value | |
|---|---|---|---|---|
| Major procedural complications | ||||
| Cardiac tamponade | 2 (2.9) | 2 (3.4) | 0 (0) | 1.000⁎ |
| Aortic dissection | 1 (1.5) | 1 (1.7) | 0 (0) | 1.000 |
| Donor vessel dissection | 2 (2.9) | 2 (3.4) | 0 (0) | 1.000 |
| Coronary perforation needing Cover stent deployment | 2 (2.9) | 2 (3.4) | 0 (0) | 1.000 |
| Collaterals perforation needing coil deployment | 3 (4.4) | 2 (3.4) | 1 (10.0) | 0.384 |
| Contrast-induced nephropathy | 2 (2.9) | 1 (1.7) | 1 (10.0) | 0.274 |
| Access site complications | ||||
| Forearm ecchymosis | 2 (2.9) | 2 (3.4) | 0 (0) | 1.000 |
⁎. Data are presented as number (%) of patients.
3.4. Clinical outcomes and rate of radial artery occlusion
Table 4 presents data for in-hospital adverse events and 30-day clinical outcomes of the study patients. In-hospital mortality happened in 2 patients (2.9%), one of whom had cardiogenic shock and the other had septic shock. Cardiac death occurred in 1 patient (1.5%) who experienced coronary perforation rescued by emergent intra-aortic balloon pump (IABP), extra-corporeal membrane oxygenation (ECMO) and emergent surgery. One patient (1.5%) experienced recurrent MI 3 days later after the index procedure caused by an episode of subacute instent thrombosis, and received urgent revascularization. Two patients (2.9%) had arrhythmia during procedure, consisting of one episode of atrial fibrillation in the procedure successful group, and one episode of ventricular tachycardia in the procedure failed group, which were both successfully converted by cardioversion. Complication from embolic stroke occurred in 1 patient (1.5%). Thirty-day clinical outcomes, similar to in-hospital outcomes, revealed a low incidence of 30-day all-cause mortality (2.9%) and 30-day MACEs (1.5%). Finally, the rate of radial artery occlusion was evaluated in a total of 66 patients (Table 5). Radial artery occlusion during one-year clinical follow-up was found only in 2 patients (3.0%), one of whom was in the procedure successful group and the other one in the procedure failed group. Only 1 patient (1.5%) had poor circulation of the fingers (pulse oximeter < 96%) during compressing ipsilateral ulnar artery.
| All (n = 68) | |
|---|---|
| In-hospital outcomes | |
| In-hospital mortality | 2 (2.9)⁎ |
| Cardiac death | 1 (1.5) |
| Recurrent myocardial infarction | 1 (1.5) |
| Stent thrombosis | 1 (1.5) |
| Arrhythmia | 2 (2.9) |
| Stroke | 1 (1.5) |
| 30-day clinical outcomes | |
| 30-day all-cause mortality | 2 (2.9) |
| 30-day MACEs | 1 (1.5) |
MACEs indicate major adverse cardiac events.
⁎. Data are presented as number (%) of patients.
| All (n = 66) | |
|---|---|
| Radial artery occlusion during one-year follow-up | 2 (3.0)⁎ |
| Absence of radial pulsation (%) | 3 (4.5) |
| Absence of blood flow (%) | 2 (3.0) |
| Pulse oximeter < 96% (%) | 1 (1.5) |
⁎. Data are presented as number (%) of patients.
4. Discussion
To the best of our knowledge, this is the first study to investigate the procedural and clinical outcomes of TRA CTO PCI using the sheathless technique with standard guiding catheters. A high procedural success rate was achieved up to 85.3% with minimal procedural complications (10/68, 14.7%), low access site complication rate (2/68, 2.9%), and low in-hospital adverse events (4/68, 5.9%). Moreover, low incidence of radial artery obstruction at access sites during one-year clinical follow-up was only 3.0%. Accordingly, we concluded that TRA CTO PCI using the sheathless technique with standard guiding catheters was applicable and safe.
4.1. Applications and limitations of the TRA CTO PCI
With respect to CTO lesions, our group previously reported the feasibility and safety of bilateral forearm PCI for CTO lesions, in which 95.3% of the patients undergoing TRA had a high procedure success rate (87.1%) and a low access site complication rate (3.5%) [10]; [28] ; [29]. As with previous reports, high successful recanalization rate in our previous study for TRA CTO PCI was dependent on complex devices and new techniques, such as the CART technique [18]; [19] ; [20]. Indeed, the fundamental key point of the CTO PCI procedure is a good backup support from larger guiding catheters. Conventional 6 Fr guide catheter systems may offer enough working space for complex devices or PCI techniques, but is not excellent for backup support for CTO PCI. Therefore, 7 Fr guiding catheters were exchanged commonly during TRA CTO PCI. However, the most critical limitation of TRA PCI is the relatively small size of the radial artery in comparison to the femoral artery. Saito et al. [11] reported that, in a Japanese population, a 7 Fr conventional artery sheaths could not be physically inserted via the radial artery in 28.5% of the male patients and 59.7% of the female patients. During clinical follow-up, 6–11% of the occlusion rate was reported with a 6 Fr artery sheath system, and 1–7% even with a 5 Fr system [21]; [22] ; [23]. Our prior research found that although a conventional 7 Fr artery sheath could be inserted via high radial artery sites with only 3.5% forearm ecchymoses of access sites, puncture via high radial artery site was more difficult than via the usual radial artery site, and required a learning curve [10].
4.2. Routine utilization of sheathless technique for TRA CTO PCI
The recent sheathless guiding catheter technique may resolve the crucial limitation of the radial artery during TRA PCI [13]; [14] ; [15]. In this study, a 7 Fr guiding catheter for sufficient backup support was utilized in 91.2% of study patients. Mamas et al. [12] reported the feasibility and safety of using the 6.5 Fr sheathless guiding catheter system during TRA PCI, in which two thirds of their patients presented with acute coronary syndrome but none had CTO lesions. Moreover, the sheathless guide catheter system is specially designed with a hydrophilic coated device (Sheathless Eaucath, Asahi Intecc, Japan), and has the benefits of a low incidence of radial spasm (5.0%) and radial occlusion at 2 months (2.0%) [12]. In our study, we only enrolled patients with CTO lesions, and tested the sheathless standard guiding catheter to create a larger working space and better backup support. During TRA CTO PCI using the sheathless technique with standard guiding catheters, multiple complex procedures were successfully performed, such as CART or reverse CART technique, externalization, thrombectomy with a distal protection device, IVUS-guided PCI, kissing or anchoring balloon techniques, rotational atherectomy, and even deployment of Cover stent (Boston). Moreover, given the reason of unavailability of the 6.5 Fr sheathless guiding catheter system (Sheathless Eaucath, Asahi Intecc, Japan) in Taiwan, standard guiding catheters were applied for the sheathless technique, which provides the additional benefits of being easily available and manipulated for experienced cardiologic interventionists. In the field of TRA CTO interventions, we successfully down-sized the conventional arterial sheath from 7–8 Fr (transfemoral PCI), 6 Fr (TRA PCI) to 5 Fr (sheathless guiding catheter system) to prevent complications of radial artery access sites. Only 2 patients (2.9%) in this study had forearm ecchymosis. Furthermore, the total procedure time was longer reaching up to 156.7 ± 65.4 min because of CTO interventions. However, no patient suffered from severe radial artery spasm that would have aborted the procedure, and the rate of radial artery occlusion rate in our study was only 3.0%, which is consistent with another published report (2.0%) [12].
In our study, collateral branches were used in 76.5% of patients for retrograde CTO interventions. Three patients (4.4%) in our study had epicardial collateral perforation needing coil deployment, which was slightly higher than the previously reported data of a 3.8% septal collateral perforation rate [24]. In our study, epicardial collaterals (23.5%) were more frequently selected for retrograde approach, thus resulting in more complications than septal collaterals. Despite the use of a larger amount of contrast media (440.9 ± 173.0 mL), the incidence of CIN in our study patients was only 2.9%, which is similar to our previous report (5.4%) and the report in a multicenter CTO registry of Japan (1.2%) [24] ; [25]. About one third of the patients in this study received Ad hoc PCI for CTO lesions [29], which indicates that the sheathless technique for TRA CTO PCI may become a “routine” procedure at our center. However, we must point out the importance of planning and having a clear roadmap for coronary angiography in light of the higher ratio of Ad hoc PCI in the procedure failed group (50.0%). When compared with our previous study [10], there were no significant differences in the procedure time (157 min vs. 162 min) and the fluoroscopy time (77 min vs. 70 min) between sheathless TRA CTO PCI and conventional TRA CTO PCI, which demonstrated that the set-up and manipulation of the sheathless system with standard guiding catheters were not time-consuming. The procedure-related complications such as cardiac tamponade were also similar between the sheathless TRA CTO PCI in this study and the conventional TRA CTO PCI [10]. Although the sheathless guiding catheters appear to provide better backup support, we should keep in mind again that the larger size of a guiding catheter might be more common with aortic dissection or donor vessel dissection. Major in-hospital events, including emergent bypass surgery or PCI, recurrent MI, stroke, cardiac tamponade and cardiac death, were similar to previous reports in CTO studies [18]; [19]; [20]; [26] ; [27].
4.4. Access-site vascular complications after transradial PCI
In our study, only 2 access-site vascular complications (ASVC) were recorded during the sheathless TRA CTO PCI. The two cases were categorized as “very early” and “early” diagnoses of ASVC. No need for surgery in our study. Burzotta et al. [30] had concluded that prompt recognition is pivotal as “late” diagnosis which is associated to the need for surgery. Compared with standard arterial sheath technique, the sheathless technique was not associated with higher ASVC in our study. We concluded again that TRA CTO PCI using the sheathless technique with standard guiding catheters was safe without ASVC if prompt recognition was made.
4.5. Limitation
There are some limitations in our study. Firstly, the tip of the standard guiding catheter was tapered down by the VTK catheter, which was not completely fit in the inner diameter of the standard guiding catheters. Although only 2 patients had forearm ecchymoses and another 2 patients had radial artery occlusion, this sheathless technique may increase the potential intimal injury of radial artery. Secondly, following the first limitation, the potential intimal injury or occlusion of the radial artery may be underestimated, because the actual diameter of the radial artery at access sites was not measured. Finally, we did not randomize the patients to compare the sheathless with the conventional guiding catheters, and the number of patient population in this study is relatively small.
5. Conclusions
It is feasible and safe to routinely use the sheathless technique with the standard guiding catheters for TRA CTO PCI with a low incidence of procedure-related complications. The incidence of radial access site complication and the rate of radial artery occlusion were also low.
Conflict of interest
The authors report no relationships that could be construed as a conflict of interest.
References
- [1] R.D. Christofferson, K.G. Lehmann, G.V. Martin, N. Every, J.H. Caldwell, S.R. Kapadia; Effect of chronic total coronary occlusion on treatment strategy; Am J Cardiol, 95 (2005), pp. 1088–1091
- [2] J.A. Grantham, S.P. Marso, J. Spertus, J. House, D.R. Holmes Jr., B.D. Rutherford; Chronic total occlusion angioplasty in the United States; JACC Cardiovasc Interv, 2 (2009), pp. 479–486
- [3] O.M. Jeroudi, M.E. Alomar, T.T. Michael, A. El Sabbagh, V.G. Patel, O. Mogabgab, et al.; Prevalence and management of coronary chronic total occlusions in a tertiary veterans affairs hospital; Catheter Cardiovasc Interv, 84 (2014), pp. 637–643
- [4] S.B. Pancholy, P. Boruah, I. Ahmed, T. Kwan, T.M. Patel, S. Saito; Meta-analysis of effect on mortality of percutaneous recanalization of coronary chronic total occlusions using a stent-based strategy; Am J Cardiol, 111 (2013), pp. 521–525
- [5] M.F. Khan, C.S. Wendel, H.M. Thai, M.R. Movahed; Effects of percutaneous revascularization of chronic total occlusions on clinical outcomes: a meta-analysis comparing successful versus failed percutaneous intervention for chronic total occlusion; Catheter Cardiovasc Interv, 82 (2013), pp. 95–107
- [6] H.K. Yip, C.J. Wu, H.W. Chang, C.Y. Fang, C.H. Yang, S.M. Chen, et al.; Effect of the PercuSurge GuardWire device on the integrity of microvasculature and clinical outcomes during primary transradial coronary intervention in acute myocardial infarction; Am J Cardiol, 92 (2003), pp. 1331–1335
- [7] A. Prasad, C.S. Rihal, R.J. Lennon, H.J. Wiste, M. Singh, D.R. Holmes Jr.; Trends in outcomes after percutaneous coronary intervention for chronic total occlusions: a 25-year experience from the Mayo Clinic; J Am Coll Cardiol, 49 (2007), pp. 1611–1618
- [8] H.Y. Fang, S.Y. Chung, C.K. Sun, A.A. Youssef, A. Bhasin, T.H. Tsai, et al.; Transradial and transbrachial arterial approach for simultaneous carotid angiographic examination and stenting using catheter looping and retrograde engagement technique; Ann Vasc Surg, 24 (2010), pp. 670–679
- [9] C.I. Cheng, C.J. Wu, C.Y. Fang, A.A. Youssef, C.J. Chen, S.M. Chen, et al.; Feasibility and safety of transradial stenting for unprotected left main coronary artery stenosis; Circ J, 71 (2007), pp. 855–861
- [10] C.J. Wu, H.Y. Fang, C.I. Cheng, H. Hussein, S.M. Abdou, A.A. Youssef, et al.; The safety and feasibility of bilateral radial approach in chronic total occlusion percutaneous coronary intervention; Int Heart J, 52 (2011), pp. 131–138
- [11] S. Saito, H. Ikei, G. Hosokawa, S. Tanaka; Influence of the ratio between radial artery inner diameter and sheath outer diameter on radial artery flow after transradial coronary intervention; Catheter Cardiovasc Interv, 46 (1999), pp. 173–178
- [12] M. Mamas, S. D'Souza, C. Hendry, R. Ali, H. Iles-Smith, K. Palmer, et al.; Use of the sheathless guide catheter during routine transradial percutaneous coronary intervention: a feasibility study; Catheter Cardiovasc Interv, 75 (2010), pp. 596–602
- [13] Q. Li, Y. He, R. Jiang, D. Huang; Using sheathless standard guiding catheters for transradial percutaneous coronary intervention to treat bifurcation lesions; Exp Clin Cardiol, 18 (2013), pp. 73–76
- [14] S.A. Harding, N. Shah, N. Briggs, A. Sasse, P.D. Larsen; Complex transradial percutaneous coronary intervention using a sheathless guide catheter; Heart Lung Circ, 22 (2013), pp. 188–192
- [15] G. Kassimis, N. Patel, R.K. Kharbanda, K.M. Channon, A.P. Banning; High-speed rotational atherectomy using the radial artery approach and a sheathless guide: a single-centre comparison with the “conventional” femoral approach; EuroIntervention, 10 (2014), pp. 694–699
- [16] C. Di Mario, G.S. Werner, G. Sianos, A.R. Galassi, J. Büttner, D. Dudek, et al.; European perspective in the recanalisation of chronic total occlusions (CTO): consensus document from the EuroCTO Club; EuroIntervention, 3 (2007), pp. 30–43
- [17] A.M. From, R. Gulati, A. Prasad, C.S. Rihal; Sheathless transradial intervention using standard guide catheters; Catheter Cardiovasc Interv, 76 (2010), pp. 911–916
- [18] S. Rathore, O. Katoh, H. Matsuo, M. Terashima, N. Tanaka, Y. Kinoshita, et al.; Retrograde percutaneous recanalization of chronic total occlusion of the coronary arteries: procedural outcomes and predictors of success in contemporary practice; Circ Cardiovasc Interv, 2 (2009), pp. 124–132
- [19] M. Kimura, O. Katoh, E. Tsuchikane, K. Nasu, Y. Kinoshita, M. Ehara, et al.; The efficacy of a bilateral approach for treating lesions with chronic total occlusions the CART (controlled antegrade and retrograde subintimal tracking) registry; JACC Cardiovasc Interv, 2 (2009), pp. 1135–1141
- [20] J.F. Surmely, E. Tsuchikane, O. Katoh, Y. Nishida, M. Nakayama, S. Nakamura, et al.; New concept for CTO recanalization using controlled antegrade and retrograde subintimal tracking: the CART technique; J Invasive Cardiol, 18 (2006), pp. 334–338
- [21] J.B. Dahm, D. Vogelgesang, A. Hummel, A. Staudt, H. Vo¨lzke, S.B. Felix; A randomized trial of 5 vs. 6 French transradial percutaneous coronary interventions; Catheter Cardiovasc Interv, 57 (2002), pp. 172–176
- [22] P.R. Stella, F. Kiemeneij, G.J. Laarman, D. Odekerken, T. Slagboom, R. van der Wieken; Incidence and outcome of radial artery occlusion following transradial artery coronary angioplasty; Cathet Cardiovasc Diagn, 40 (1997), pp. 156–158
- [23] M. Sanmartin, M. Gomez, J.R. Rumoroso, M. Sadaba, M. Martinez, J.A. Baz, et al.; Interruption of blood flow during compression and radial artery occlusion after transradial catheterization; Catheter Cardiovasc Interv, 70 (2007), pp. 185–189
- [24] Y. Morino, T. Kimura, Y. Hayashi, T. Muramatsu, M. Ochiai, Y. Noguchi, et al.; In-hospital outcomes of contemporary percutaneous coronary intervention in patients with chronic total occlusion insights from the J-CTO Registry (Multicenter CTO registry in Japan); JACC Cardiovasc Interv, 3 (2010), pp. 143–151
- [25] Y.S. Lin, H.Y. Fang, H. Hussein, C.Y. Fang, Y.L. Chen, S.K. Hsueh, et al.; Predictors of contrast-induced nephropathy in chronic total occlusion percutaneous coronary intervention; EuroIntervention, 9 (2014), pp. 1173–1180
- [26] J.T. Hsu, H. Tamai, E. Kyo, T. Tsuji, S. Watanabe; Traditional antegrade approach versus combined antegrade and retrograde approach in the percutaneous treatment of coronary chronic total occlusions; Catheter Cardiovasc Interv, 74 (2009), pp. 555–563
- [27] S. Rathore, O. Katoh, E. Tuschikane, A. Oida, T. Suzuki, S. Takase; A novel modification of the retrograde approach for the recanalization of chronic total occlusion of the coronary arteries intravascular ultrasound-guided reverse controlled antegrade and retrograde tracking; JACC Cardiovasc Interv, 3 (2010), pp. 155–164
- [28] H.Y. Fang, S.Y. Lu, W.C. Lee, Y.S. Lin, C.I. Cheng, C.J. Chen, et al.; The predictors of successful percutaneous coronary intervention in ostial left anterior descending artery chronic total occlusion; Catheter Cardiovasc Interv, 84 (2014), pp. E30–E37
- [29] H.Y. Fang, W.C. Lee, Hussein Hesham, C.Y. Fang, C.I. Cheng, C.H. Yang, et al.; In-hospital and 3-year clinical outcomes following ad hoc versus staged percutaneous coronary interventions in chronic total occlusion — a real world practice; IJC Heart Vessel, 4 (2014), pp. 73–80
- [30] F. Burzotta, L. Mariani, C. Trani, V. Coluccia, M.F. Brancati, I. Porto, et al.; Management and timing of access-site vascular complications occurring after transradial percutaneous coronary procedures; Int J Cardiol, 167 (2013), pp. 1973–1978
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?