Abstract
Protozoa and helminths are the two main groups that cause parasitic diseases with a broad spectrum of clinical symptoms. Protozoa are unicellular organisms like the malaria parasite Plasmodium, which is responsible for the majority of deaths associated with parasitic infections. Helminths are alternative parasites that can produce debilitating diseases in hosts, some of which result in chronic infections. The discovery of effective therapeutic drugs is the key to improving health in regions of poverty and poor sanitation where these parasites usually occur. It is very encouraging that the 2015 Nobel Prize in Physiology or Medicine was awarded to Youyou Tu as well as William C. Campbell and Satoshi Õmura for their considerable contributions in discovering artemisinin and avermectin, respectively. Both drugs revolutionized therapies for filariasis and malaria, significantly reducing by large percentages their morbidity and mortality.
Keywords
Artemisinin; Avermectin filariasis; Malaria; 2015 Nobel Prize
Malaria is one of most important parasitic diseases, remaining a major health problem in most tropical developing countries in Africa, Asia, and Latin America. The Plasmodium protozoans P. vivax, P. ovale, P. malariae, P. falciparum, and/or P. knowlesi are the etiological agents of malaria in humans, and all are transmitted by Anopheles mosquitoes in nature [1]. Approximately 3.4 billion people worldwide are exposed annually and 1.2 billion of them are at high risk. Of these, 198 million malaria cases with clinical symptoms were reported in 2013, causing at least 650,000 deaths every year [2]. In addition to being a health threat, endemic malaria is an economic and social burden, particularly in low income countries [3].
|
|
|
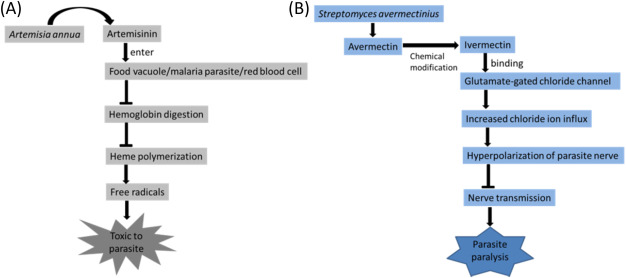
Fig. 1. (A) Artemisinin identified from Artemisia annua contains an endoperoxide bridge reacts with the iron atoms, leading to disruption on the hemoglobin catabolism and heme detoxification systems in the food vacuole of Plasmodium species by forming free radicals. Malaria parasites developing inside the erythrocyte are consequently damaged which treated with artemisinin. (B) Streptomyces avermectinius – originated avermectin possesses activity against nematodes including those cause river blindness and lymphatic filariasis by binding to glutamate-gated chloride channels in treated parasites, causing increased chloride ion influx and hyperpolarization of the parasite nerves. It, in turn, interferes with the transmission of nerve signals, leading to paralysis and death of the parasite in consequence. |
Nematodes or roundworms like whipworm, pinworm, hookworms, and filariae are also important human parasites that currently or formerly occurred widely in tropical regions of the world. A number of nematodes are intestine-dwelling; however, filariae mostly live in other tissues, such as lymphatics and skin. Onchocerca volvulus, which causes onchocerciasis, is one important species naturally transmitted by black flies (Simulium spp.). Adults of O. volvulus usually develop a nodular form under the skin, frequently resulting in disfiguring skin conditions, visual impairment, and even permanent blindness (river blindness). Onchocerciasis is geographically distributed in sub-Saharan Africa and parts of Latin America. Meanwhile, Wuchereria bancrofti and Brugia malayi/Brugia timori are the species transmitted by mosquitoes and cause clinical symptoms similar to those of elephantiasis (lymphatic filariasis); i.e., impaired lymphatic drainage, fibrotic skin tissue, and scrotal hydrocele [4] and [5]. Most cases occur in the tropics and it has been identified by the World Health Organization as the second leading cause of permanent or long-term disability [6].
Treatment for patients is essential in order to prevent morbidity and mortality, and conventional drugs have been developed against both important parasites. Quinine/chloroquine was identified as effective against malaria decades or even centuries ago. In contrast, diethylcarbamazine (N, N-diethyl-4-methyl-1-piperazine carboxamide; DEC or Hetrazan) was developed and has been used to treat filariasis since the last mid-century. Unfortunately, their efficacies have largely decreased due to induced resistance caused by the prevalent use of these drugs over time. Discoveries of artemisinin and avermectin have recently opened a new era of chemotherapy for these two important parasitic diseases, not only relieving a number of diseases but also saving many lives from infections.
Discovery of artemisinin
The malaria parasite was not identified until the military doctor Alphonse Laveran described his finding in “New parasite found in the blood of several patients suffering from marsh fever” in 1802. Its natural cycle via transmission by Anopheles mosquitoes between humans was subsequently demonstrated by Ronald Ross a few years later [7]. Humans and female mosquitoes both provide adequate environments for the development of malaria parasites in their complex life cycle [8]. In humans, the parasite forms male (micro-) and female (macro-) gametocytes as the final developmental stage, which are ingested by mosquitoes along with a blood meal. The opposite gametocyte sexes subsequently mate in the lumen of the midgut where infective sporozoites form within the resultant oocysts. They then migrate to the salivary glands, from which they can be passed along with saliva to another human when infected female mosquitoes bite again [9]. After injection into humans via mosquito bites, sporozoites rapidly multiply in the liver to form merozoites, which then invade red blood cells in the bloodstream. Infected red blood cells usually burst and release merozoites, which cyclically enter and infect other red blood cells. This feature results in typical clinical symptoms such as chills and fever, as well as anemia in most cases. Complications like cerebral malaria, acute renal failure, black water fever, hypotension, shock, and even death also occur in many cases of P. falciparum infection [10].
Quinine was identified from the bark of Cinchona (Family: Rubiaceae) as early as 1820, while its first derivate compound, chloroquine, was first synthesized in 1934 and has since been used as a therapeutic for malaria [11]. Unfortunately, resistance to chloroquine appeared in most of the areas harboring malaria by the late 1960s while the incidence of the disease continued to rise [12]. This makes it urgent to develop new approaches to replace chloroquine in malaria treatment and control. Artemisinin was first identified in 1972 by Youyou Tu and her team in the Academy of Traditional Chinese Medicine, China (ScienceNet.cn; http://tinyurl.com/gkw7mjh). After graduating from Peking University Medical School/Beijing Medical College with a major in Pharmaceutical Sciences, Youyou Tu started working on traditional herbal medicine in 1955. During the Cultural Revolution period in China, Youyou Tu was appointed the director of Project 523 in 1969, which strove to develop novel therapeutic drugs to heal chloroquine-resistant malaria. In a large-scale screening of herbal medicines, Artemisia spp. (Family: Asteraceae), or sweet wormwood, was selected from more than 640 traditional herb medicine compounds for its potential antimalarial effects (Science Net; http://tinyurl.com/gkw7mjh).
Artemisia has been used as an ancient Chinese medicine to relieve fever for more than 2000 years [13]. The active ingredient extracted from Artemisia annua but not Artemisia apiacea (both species of plants grow commonly in China), later called artemisinin, was demonstrated to be effective against Plasmodium infections. In order to overcome inconsistent experimental results, Youyou Tu revisited a Chinese ancient manuscript called “The handbook of prescriptions for emergency treatments (肘後備急方 in Chinese)” written in 340 BC by Ge Hong (葛洪 in Chinese). Upon inspiration from that book, she changed the protocol for component extraction by using ether, which has a low boiling point (35 °C), in place of water and ethanol [14]. Youyou Tu was ultimately able to obtain artemisinin from the new extraction process, from which an ingredient highly effective against the development of malaria parasites was obtained in 1972 [15]. Later, water-soluble derivative compounds such as artesunate and OZ 277 were synthesized for use in injections [16] and [17].
The acidic food vacuole is one of the essential organelles in the intra-erythrocyte stages of malaria parasites [18]. This vacuole has a role in the digestion of host hemoglobin, the iron-containing oxygen-transport metalloprotein in the red blood cells of all vertebrates [19]. Malaria parasites developing in erythrocytes generally transport a large quantity of erythrocytic cytoplasm to the food vacuole, within which hemoglobin is degraded [20]. During hemoglobin catabolism, potentially toxic heme moieties are polymerized to become hemozoin, or malaria pigment [21], which also provides iron to the parasite [20]. Structurally, artemisinin contains two oxygen atoms linked together and known as an endoperoxide bridge, which reacts with iron atoms [22] and leads to the disruption of hemoglobin catabolism and heme detoxification systems in Plasmodium species by forming free radicals [23]. Ultrastructural studies have revealed that artemisinin frequently causes disruption of the food vacuole membrane and extensive loss of organelle structures at longer incubation times, suggesting that the food vacuole is an important initial site of endoperoxide antimalarial activity [24]. As a result, artemisinin becomes toxic to malaria parasites when it reacts with the high iron content of the parasites to generate free radicals [[[#fig1|Fig. 1]]A], which damage the parasite [25]. Furthermore, the regular function of the electron transport chain within the mitochondria of the malaria parasite may also be damaged [26] and [27]. Perhaps it is caused by the disruption of the mitochondrial membrane in response to artemisinin used against malaria parasites [24]. Since mitochondria do not exist in erythrocytes, artemisinin does not cause such damages to the erythrocyte and thus could be specifically effective against malaria parasites.
Discovery of avermectins
A number of nematode parasites are accommodated in the intestine. Such intestine-dwelling nematodes include Ascaris lumbricoides, Trichuris trichiura, Enterobius vermicularis, and Strongyloides stercoralis, which occur particularly commonly in tropical regions [28]. Four chemotherapeutic anthelmintic drugs in two main groups have long been recommended by WHO for treating these infections. Albendazole and mebendazole belong to the benzimidazoles group, while levamisole and pyrantel are in the imidazothiazoles/tetrahydropyrimidines group [29]. These anthelmintics are basically safe with few or no side effects in the treatment of nematode parasites within the human intestine [29]. A large proportion of infections of intestinal nematode parasites have evidently been reduced after extensive mass treatment.
There are many nematodes that regularly live outside the intestine with different environments; e.g., filariae. As a result, traditional anthelmintics that work by expelling intestinal nematodes are useless in the treatment of filariae [30]. Filariae are naturally transmitted by blood-feeding insects. Adult parasites develop and mate in specific tissues and produce microfilariae in the host. Among them, W. bancrofti and B. malayi are the main species occurring in tropical and subtropical countries [31]. Both reside in lymphatics and frequently cause lymphatic filariasis with a variety of clinical manifestations, including lymphedema of the limbs, and genital diseases such as hydrocele, chylocele, and swelling of the scrotum [32]. In the mosquito vector, the microfilariae ingested with blood meals develop mostly in thoracic muscles, migrate to the mosquitos mouthparts, and then infective larvae (L3) enter a new host via blood-feeding [32]. O. volvulus causes river blindness, the commonest infectious blindness in the world, which occurs primarily in Africa and with limited distribution in Latin America. This parasite is naturally transmitted by black flies (Simulium spp.), whose larval stages are spent in swift streams [33]. The L3 of O. volvulus are deposited when infected black flies feed again, subsequently developing into adult filariae at several locations on a persons skin, generating nonspecific symptoms and severe pruritus, acute and chronic dermatitis, vitiligo-like hypopigmentation, and atrophy [34]. Migration of microfilariae into eyes can cause ocular disease that leads to blindness in severe cases [35].
Albendazole has ever been used as a drug to eliminate lymphatic filariasis; unfortunately, it has little effect on reducing microfilariae abundance in the host [36]. Diethylcarbamazine (DEC), discovered by Yellapragada Subbarow, was subsequently used against filarial infections from the early 1950s [37]. Pharmacologically, DEC is an inhibitor of arachidonic acid metabolism in filarial microfilaria [38] that facilitates the loss of the microfilarial sheath and results in body damage [39]. Treated microfilariae but not adult worms become more susceptible to attack by the innate immunity of the host [38]. Adult worms remaining in tissues stay alive and reproductive, persistently producing microfilariae that may be picked up when the host is bitten again by black flies. Ingested microfilariae then migrate within the host, possibly causing clinical outcomes that include blindness in severe cases. In addition, DEC administration frequently causes a wide spectrum of side effects such as fever, headache, dizziness, nausea, and muscle or joint pain [40]. It may also worsen eye disease caused by O. volvulus (CDC website, http://tinyurl.com/zn7kbca). Such disadvantages remained until the time Satoshi Õmura, a Japanese microbiologist and a professor in the School of Pharmaceutical Sciences, Kitasato University, isolated avermectins from Streptomyces (a part of Actinobacteria) (Nobelprize.org: http://tinyurl.com/od8efal). As with many important breakthroughs in medical research, the finding of avermectin is astonishing and undoubtedly a serendipity [41]. The journey of the discovery started in 1973 by a unique collaboration between international laboratories or sectors affiliated to governments, non-governmental organizations, and industry.
A team was then formed to discover a drug effective against helminth parasites to reduce their impacts on the poverty-stricken. Within the program, Satoshi Õmura proposed research activities to search for novel antibiotic agents from natural organisms [42]. Not long afterward, a new strain of Streptomyces avermectinius, an actinomycete, was isolated from soil samples and successfully cultured in the laboratory. Streptomyces is a group of Gram-positive bacteria resembling filamentous fungi in shape from which a number of secondary metabolites with the bioactivity of antibiotics and related products have been obtained [43]. Among such cultures, Satoshi Õmura selected about 50 that were promising against harmful microorganisms [41]. William C. Campbell, a professor at Drew University, Madison, NJ, USA, and an expert in parasite biology, obtained Streptomyces cultures from Satoshi Õmura and found that a component from one of the cultures was remarkably efficient against parasites commonly seen in domestic and farm animals in 1974 [42]. The component was then purified and named Avermectin, and subsequently was determined to be a series of macrocyclic lactone derivatives [44]. Despites lacking significant antibacterial or antifungal activity, in contrast, avermectin was shown to have activity against helminths, especially nematodes, through a screening system conducted at Merck Sharp & Dohme Research Laboratories in Rahway, NJ [45]. It is particularly useful in treating the adult stage of O. volvulus and W. bancrofti/B. malayi, which are not responsive to DEC in most cases nowadays [46].
After chemically modifying avermectin, a more effective compound called ivermectin (a dihydro derivate of avermectin) was obtained and found effective in killing filariae in humans [45]. Ivermectin has proven to be one of the most successful therapeutic drugs for filariasis [47], usually by causing the paralysis of susceptible parasites [48]. More specifically, avermectins' mode of action is to bind with high affinity to the parasites glutamate-gated chloride channels, increasing the chloride ion influx due to the changed permeability of the cell membrane and hyperpolarization of the parasite nerves [49]. The drug eventually interferes with the transmission of nerve signals by its effect on neurotransmitter y-amino-butyric acid (GABA), which is important in nematodes [45]. As a result, it could play a critical role leading to the paralysis and death of parasites treated with avermectins [[[#fig1|Fig. 1]]B].
Conclusion
Parasitic infections usually cause a broad of spectrum of clinical outcomes depending on the species or stage of invading parasites. Malaria is an ancient and deadly human infectious disease. The discovery of artemisinin opened a new era in the battle against chloroquine-resistant malaria. Following the use of artemisinin-based combination therapies (ACTs) recommended by WHO [50], this newly discovered medicine has significantly reduced malarial disease and deaths, particularly the severe form caused by P. falciparum[51]. The parallel discovery of avermectins has been of immeasurable value in the control of onchocerciasis and lymphatic filariae. It is particularly useful in killing the adult stages of these two devastating diseases in association with infections by filariae. Generally, filarial infections do not directly cause mortality in most cases. However, they affect the health of individuals and families and their socioeconomic condition. The 2015 Nobel Prize in Physiology or Medicine honors the discoveries of antiparasitics by Youyou Tu, William C. Campbell, and Satoshi Õmura, not only in science but also in humanity.
References
- [1] B. Singh, L. Kim Sung, A. Matusop, A. Radhakrishnan, S.S. Shamsul, J. Cox-Singh, et al.; A large focus of naturally acquired Plasmodium knowlesi infections in human beings; Lancet, 363 (2004), pp. 1017–1024
- [2] WHO. Achieving the malaria MDG target: reversing the incidence of malaria 2000–2015. Global Malaria Programme, World Health Organization. pp. 1–32.
- [3] R.I. Chima, C.A. Goodman, A. Mills; The economic impact of malaria in Africa: a critical review of the evidence; Health Policy, 63 (2003), pp. 17–36
- [4] D.N. Udall; Recent updates on onchocerciasis: diagnosis and treatment; Clin Infect Dis, 44 (2007), pp. 53–60
- [5] S. Knopp, P. Steinmann, C. Hatz, J. Keiser, J. Utzinger; Nematode infections: filariases; Infect Dis Clin North Am, 26 (2012), pp. 359–381
- [6] L.K. Das, S.P. Pani, S.K. Vinod; Locomotor disability in bancroftian filarial lymphoedema patients; J Commun Dis, 40 (2008), pp. 13–19
- [7] E. Bendiner; Ronald Ross and the mystery of malaria; Hosp Pract, 29 (1994 15), pp. 95–97
- [8] W. Ceusters, B. Smith; Malaria diagnosis and the Plasmodium life cycle: the BFO perspective; Interdisciplinary ontology. Proceedings of the third interdisciplinary ontology meeting (Tokyo, Japan, February 27–28, 2010), Keio University Press, Tokyo (2010), pp. 25–34
- [9] H.H. Chang, E.L. Moss, D.J. Park, D. Ndiaye, S. Mboup, S.K. Volkman, et al.; Malaria life cycle intensifies both natural selection and random genetic drift; Proc Natl Acad Sci USA, 110 (2013), pp. 20129–20134
- [10] D.N. Patel, P. Pradeep, M.M. Surti, S.B. Agarwal; Clinical manifestations of complicated malaria – an overview; J Indian Acad Clin Med, 4 (2003), pp. 323–331
- [11] W.S. Johnson, B.G. Buell; A new synthesis of chloroquine; J Am Chem Soc, 74 (1952), pp. 4513–4516
- [12] T.E. Wellems, C.V. Plowe; Chloroquine-resistant malaria; J Infect Dis, 184 (2001), pp. 770–776
- [13] Y. Zhou; Ancient Chinese anti-fever cure becomes panacea for malaria; Bull WHO, 87 (2009), pp. 743–744
- [14] E. Hsu; Reflections on the ‘discovery’ of the antimalarial qinghao; Br J Clin Pharmacol, 61 (2006), pp. 666–670
- [15] L.H. Miller, X. Su; Artemisinin: discovery from the Chinese herbal garden; Cell, 146 (2011), pp. 855–858
- [16] T. Efferth, M.R. Romero, D.G. Wolf, T. Stamminger, J.J.G. Marin, M. Marschall; The antiviral activities of artemisinin and artesunate; Clin Infect Dis, 47 (2008), pp. 804–811
- [17] A.C. Uhlemann, S. Wittlin, H. Matile, L.Y. Bustamante, S. Krishna; Mechanism of antimalarial action of the synthetic trioxolane RBX11160 (OZ277); Antimicrob Agents Chemother, 51 (2007), pp. 667–672
- [18] N. Abu Bakar, N. Klonis, E. Hanssen, C. Chan, L. Tilley; Digestive-vacuole genesis and endocytic processes in the early intraerythrocytic stages of Plasmodium falciparum; J Cell Sci, 123 (2010), pp. 441–450
- [19] G.A. Biagini, P.G. Bray, D.G. Spiller, M.R. White, S.A. Ward; The digestive food vacuole of the malaria parasite is a dynamic intracellular Ca2+ store; J Biol Chem, 278 (2003), pp. 27910–27915
- [20] P.J. Rosenthal, S.R. Meshnick; Hemoglobin catabolism and iron utilization by malaria parasites; Mol Biochem Parasitol, 83 (1996), pp. 131–139
- [21] M. Aikawa, P.K. Hepler, C.G. Huff, H. Sprinz; The feeding mechanism of avian malarial parasites; J Cell Biol, 28 (1966), pp. 355–373
- [22] S.R. Meshnick, T.E. Taylor, S. Kamchonwongpaisan; Artemisinin and the antimalarial endoperoxides: from herbal remedy to targeted chemotherapy; Microbiol Rev, 60 (1996), pp. 301–315
- [23] A.V. Pandey, B.L. Tekwani, R.L. Singh, V.S. Chauhan; Artemisinin, an endoperoxide antimalarial, disrupts the hemoglobin catabolism and heme detoxification systems in malarial parasite; J Biol Chem, 274 (1999), pp. 19383–19388
- [24] M. del Pilar Crespo, T.D. Avery, E. Hanssen, E. Fox, T.V. Robinson, P. Valente, et al.; Artemisinin and a series of novel endoperoxide antimalarials exert early effects on digestive vacuole morphology; Antimicrob Agents Chemother, 52 (2008), pp. 98–109
- [25] J. Golenser, J.H. Waknine, M. Krugliak, N.H. Hunt, G.E. Grau; Current perspectives on the mechanism of action of artemisinins; Int J Parasitol, 36 (2006), pp. 1427–1441
- [26] M. Fry, J.E. Beesley; Mitochondria of mammalian Plasmodium spp; Parasitology, 102 (1991), pp. 17–26
- [27] J.N. Cumming, P. Ploypradith, G.H. Posner; Antimalarial activity of artemisinin (qinghaosu) and related trioxanes: mechanism(s) of action; Adv Pharmacol, 37 (1997), pp. 253–297
- [28] S. Awasthi, D. Bundy; Intestinal nematode infection and anaemia in developing countries; Br Med J, 334 (2007), pp. 1065–1066
- [29] G. Stepek, D.J. Buttle, I.R. Duce, J.M. Behnke; Human gastrointestinal nematode infections: are new control methods required?; Int J Exp Pathol, 87 (2006), pp. 325–341
- [30] F. Hawking; A review of progress in the chemotherapy and control of filariasis since 1955; Bull WHO, 27 (1962), pp. 555–568
- [31] E.A. Ottesen, C.P. Ramachandran; Lymphatic filariasis infection and disease: control strategies; Parasitol Today, 11 (1995), pp. 129–131
- [32] J.H. Cross; Filarial nematodes; S. Baron (Ed.), Medical microbiology (4th ed.), University of Texas Medical Branch at Galveston, Galveston (TX) (1996) [Chapter 92]
- [33] K.L. Winthrop, J.M. Furtado, J.C. Silva, S. Resnikoff, V.C. Lansingh; River blindness: an old disease on the brink of elimination and control; J Glob Infect Dis, 3 (2011), pp. 151–155
- [34] C.D. Enk; Onchocerciasis–river blindness; Clin Dermatol, 24 (2006), pp. 176–180
- [35] I.B. Berger, J. Nnadozie; Onchocerciasis and other eye problems in developing countries: a challenge for optometrists; J Am Optom Assoc, 64 (1993), pp. 699–702
- [36] R.K. Shenoy, S. Dalia, A. John, T.K. Suma, V. Kumaraswami; Treatment of the microfilaraemia of asymptomatic brugian filariasis with single doses of ivermectin, diethylcarbamazine or albendazole, in various combinations; Ann Trop Med Parasitol, 93 (1999), pp. 643–651
- [37] R.I. Hewitt, E. White, W.S. Wallace, H.W. Stewart, S. Kushner, S. Subbarow; Experimental chemotherapy of filariasis. II. effect of piperazine derivatives against naturally acquired filarial infections in cotton rats and dogs; J Lab Clin Med, 32 (1947), pp. 1304–1313
- [38] G.A. El-Shahawi, M. Abdel-Latif, A.H. Saad, M. Bahgat; Setaria equina: in vivo effect of diethylcarbamazine citrate on microfilariae in albino rats; Exp Parasitol, 126 (2010), pp. 603–610
- [39] M.S. Florêncio, C.A. Peixoto; The effects of diethylcarbamazine on the ultrastructure of microfilariae of Wuchereria bancrofti; Parasitology, 126 (2003), pp. 551–554
- [40] P.C. Fan; Diethylcarbamazine treatment of bancroftian and malayan filariasis with emphasis on side effects; Ann Trop Med Parasitol, 86 (1992), pp. 399–405
- [41] S. Õmura, A. Crump; The life and times of ivermectin – a success story; Nat Rev Microbiol, 2 (2004), pp. 984–989
- [42] A. Crump, S. Ōmura; Ivermectin, ‘Wonder drug’ from Japan: the human use perspective; Proc Jpn Acad Ser B Phys Biol Sci, 87 (2011), pp. 13–28
- [43] R.E. Procópio, I.R. Silva, M.K. Martins, J.L. Azevedo, J.M. Araújo; Antibiotics produced by Streptomyces; Braz J Infect Dis, 16 (2012), pp. 466–471
- [44] R.W. Burg, B.M. Miller, E.E. Baker, J. Birnbaum, S.A. Currie, R. Hartman, et al.; Avermectins, new family of potent anthelmintic agents: producing organism and fermentation; Antimicrob Agents Chemother, 15 (1979), pp. 361–367
- [45] W.C. Campbell; Ivermectin, an antiparasitic agent; Med Res Rev, 13 (1993), pp. 61–79
- [46] C.A. Bulman, C.M. Bidlow, S. Lustigman, F. Cho-Ngwa, D. Williams, A.A. Rascón Jr., et al.; Repurposing auranofin as a lead candidate for treatment of lymphatic filariasis and onchocerciasis; PLoS Negl Trop Dis, 9 (2015), p. e0003534
- [47] E.D. Deeks; Ivermectin: a review in Rosacea; Am J Clin Dermatol, 6 (2015), pp. 447–452
- [48] D.J. Brownlee, L. Holden-Dye, R.J. Walker; Actions of the anthelmintic ivermectin on the pharyngeal muscle of the parasitic nematode, Ascaris suum; Parasitology, 115 (1997), pp. 553–561
- [49] A.J. Wolstenholme, A.T. Rogers; Glutamate-gated chloride channels and the mode of action of the avermectin/milbemycin anthelmintics; Parasitology, 131 (Suppl Sl) (2005), pp. S85–S95
- [50] T.K. Mutabingwa; Artemisinin-based combination therapies (ACTs): best hope for malaria treatment but inaccessible to the needy!; Acta Trop, 95 (2005), pp. 305–315
- [51] H.M. McIntosh, P. Olliaro; Artemisinin derivatives for treating severe malaria; Cochrane Database Syst Rev, 2 (2000), p. CD000527
Document information
Published on 20/10/16
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?