1 Introduction
Over the past decades, much research has been focussed on the development of cellulose reinforced poly(lactic acid) (PLA) nanocomposites. This is due do to the need of fully bio-based materials with a more environmentally friendly life cycle [1]. Among all cellulose-based nanoreinforcements, cellulose nanofibers (CNF) got into the focus of investigation as reinforcement material for applications in food packaging, automotive, construction, among others [2]. This is due to their large aspect ratio, togheter with their outstanding mechanical properties and their low percolation threshold. Numerous works reported that CNF loadings as low as 0.5 to 5 wt.% can positively influence PLA thermal, mechanical, rheological and foaming properties [3, 4].
Nonetheless, the well-established self-agglomeration issues of cellulose-based materials into polymers restrict the full CNF development to specific processing methods. Recall that CNF are obtained as a hydrophilic gel-like suspension. Accordingly, solvent casting/evaporation from a liquid medium is nowadays the frontrunner technique to obtain a satisfactory CNF dispersion degree, leading to a percolating network at relatively low contents (0.5-1 wt.%) [2, 3]. The modification of the CNF surface is another route to enhance filler dispersion as well as compatibility with the polymeric matrix [5]. However, both methodologies are organic solvent-dependent and time expensive, hence limiting their expansion for mass production.
To overcome these shortcomings, the production of PLA/CNF composites through conventional melt-processing methods is under investigation [6, 7]. Although these methods are faster and solvent-free, CNF aggregation and degradation issues have been reported. Additionally, the continuous shearing applied during processing prevents the formation of a percolating network and higher CNF contents are needed to obtain similar properties as compared to their solvent casting counterparts. In order to provide solutions to CNF aggregation on drying before extrusion, Kizitlas et al. [8] reported the use of a masterbatch prepared through melt-blending as carrier system to introduce CNF into PLA. The aqueous suspension of CNF is first melt mixed with a third polymer (polyhydroxybutyrate, PHB) and then the resulting masterbatch is diluted into PLA. Despite that a satisfactory CNF dispersion was observed in the sample holding 5 wt.%, the masterbatch preparation through melt blending is somewhat hazardous (CNF wet feeding into PHB at 170 ºC). Furthermore, the hydrophobicity of PHB can hinder hydrophilic fillers from dispersing well in the masterbatch.
Our effort to overcome the abovementioned shortcomings focused on the use of a biodegradable water-soluble biopolymer as carrier system (i.e. polyethylene glycol, PEG), that can be rapidly and straightforwardly melt-blended with PLA. The cellulose nanocrystal surface physical PEGylation has already reported as a successful technique to avoid CNC self-agglomeration on drying [9, 10]. However, to the best of the author’s knowledge, no researches have been conducted to investigate the effect of a PEG carrier system on the CNF dispersion using industrial scalable melt-processing techniques. Accordingly, this study aimed at investigating the evolution of the dispersion of 5 wt.% of CNF into PLA nanocomposites using different percentages of PEG (0, 10 and 20 wt.%) as carrier system though microscopy and rheological analyses. Finally, the load-bearing capacity of CNF was studied via thermo-mechanical experiments.
2 Experimental section
2.1 Materials
An extrusion grade polylactic acid (Ingeo PLA 4032D, D-lactic content ≈ 2%) was supplied by Natureworks LLC (Arendonk, Belgium) and used as received. Polyethylene glycol (PEG) (MW ≈ 2000 g.mol-1) was purchased from Alfa Aesar (ThermoFisher, Karlsruhe, Gemany). An antioxidant (Irganox 1010) was kindly supplied by BASF (Barcelona, Spain) to counteract PLA degradations during melt blending. Enzymatic-cellulose nanofibers (CNF) were kindly supplied by the Chalmers University of Technology [11]. The diameter distribution of the nanofibrils was about 11.5 nm ± 4.0 nm [12].
2.2 PLA/PEG/CNF nanocomposites processing
Initially, two PEG-based masterbatches with a nominal content of dried CNF set at 20 and 33 wt.% were prepared by dissolving PEG in distilled water at room temperature. Then a desired content of the the gel-like suspension of CNF was added to the solution and stirred for 3h at RT. The final mixtures were poured onto glass Petri dishes and dried at 35ºC for 3 days. For comparison purposes, CNF alone were subjected to the same experimental protocol.
The obtained flakes of the different PEG-based masterbatches were melt blended with PLA using a DSM Xplore Micro 15cc twin screw compounder to reach a CNF nominal content of 5 wt.%. The melt temperature was set to 180ºC, the screw speed at 100 rpm and the residence time was fixed at 2.5 min under a N2 blanket. Prior to processing, PLA pellets were vacuum-dried at 60ºC overnight. During mixing, 0.5 wt.% of Irganox 1010 was added to minimize PLA degradation. The nominal PLA/PEG/CNF ratios were 95/0/5; 85/10/5 and 75/20/5 and the samples were codified as PLA/CNF, PLA/10PEG/CNF and PLA/20PEG/CNF, respectively.
Rectangular (45 x 12 x 2 mm) and disk-shaped (diameter: 25 mm; thickness: 1.5 mm) samples were prepared by injection-moulding using a DSM Xplore Micro 5cc injection moulding machine with a barrel temperature of 190ºC and a mold temperature of 30ºC. Reference PLA and PLA/PEG blends (nominal ratios: 90/10 and 80/20 wt/wt) were prepared using the same processing procedure for the sake of comparison.
2.3 Characterization
Optical miscroscopy (Nikon, Optiphot-Pol XTP-11) equipped with a cross-polarizer was used to evaluate the effect of the PEG content on the degree of CNF dispersion within PLA.
DSC analyses were carried out using a MDSC Q2000 instrument (TA Instruments) under a dry N2 atmsphere. 5-6 mg of the samples were sealed in standard aluminium pans and subjected to a heating/cooling/heating procedure over a temperature range from -80 to 200 ºC at a heating/cooling rate of 10 ºC.min-1. The degree of cristallinity (Xc) was calculated using a melting enthalpy for a 100% crystalline PLA of 93.6 J.g-1 [13].
The possible thermal degradation of CNF was evaluated via wide angle X-ray scattering analysis using a Bruker D8 Advance diffractometer (Billerica) with CuKα radiation of wavelength of 0.154 nm. Scans were recorded in the range 5-50º (step size= 0.02, scanning speed= 1 s/step).
The rehological behaviour was evaluated using an AR-G2 rheometer (TA Instruments) in parallel plate (25 mm) configuration with a constant gap of 1.4 mm at 180 ºC under a dry N2 atmosphere. SAOS tests were carried out in the angular frequency range 1 <ω< 623 rad.s-1 at a fixed 3% strain (L.V.R.). To ensure sufficient data in the terminal regime, creep-recovery experiments were performed using fresh samples. The dynamical mechanical spectra were obtained combining the results from both methodologies.
Dynamic mechanical thermal analysis (DMTA) was performed on rectangular samples using a DMTA Q800 equipment (TA Instruments). Samples were tested in dual cantilever bending mode from -100 to 80ºC at a heating rate of 2ºC.min-1 at 0.02% of deformation (L.V.R.) and a frequency of 1 Hz.
3 Results and discussion
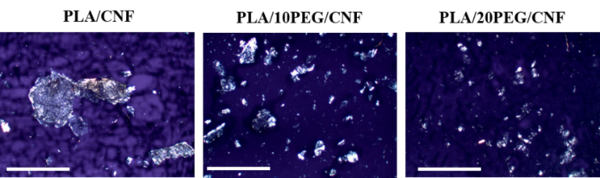
After melt-processing, the CNF dispersion within PLA was investigated using cross-polarized light microscopy and the micrographs are depicted in Figure 1. PLA/CNF sample revealed the expected coarse suspension of large CNF aggregates, which is in line with the well documented self-agglomeration issue of CNF on drying before melt blending [14].
In contrast, a more homogeneous morphology was seemingly obtained for both PLA/PEG/CNF bio-composites. Polarized light microscopy evidenced the presence of a larger number of smaller CNF aggregates whose size apparently gradually decreased when CNF were pre-mixed with PEG first and then melt blended with PLA. Accordingly, the ability of PEG to reduce CNF aggregation through the masterbatch approach suggested that PEG acted as dispersant.
The thermal properties of injected samples obtained from the first heating scan are compiled in Table 1. The incorporation of CNF alone did not modify the PLA thermal behaviour. Both samples can be considered as amorphous. The plasticizing effect attributed to low MW PEG on PLA led to a clear improvement of the PLA/PEG blend crystallization ability [15]. The cold crystallization temperature (Tcc) decreased while XC increased with the PEG content. Interestingly, the melt temperature (Tm) remained almost constant. According to the processing conditions used, adding CNF did not greatly influence PLA/PEG thermal behaviour.
The second heating scan revealed that PEG is only partially miscible within PLA when adding 20 wt.% PEG. The measured Xc values of PLA and PLA/CNF samples indicated that aggregated CNF did not improve the poor crystallization ability of PLA. By contrast, XC values increased up to 16 % between PLA/10PEG and PLA/10PEG/CNF samples, suggesting that an improvement in the CNF dispersion could enhance PLA crystallization. Indeed, their large specific area could enhance the heterogeneous crystallization of PLA [2]. Such effect was not clearly identified between samples with 20 wt.% of PEG, possibility due to an advanced plasticizing effect.
| Sample nomenclature | Tg
(ºC) |
Tcc
(ºC) |
Tm
(ºC) |
Xc
(%) |
| PLA | 60 | 108.5 | 167.6 | 4 |
| PLA/CNF | 61 | 107.7 | 167.6 | 2 |
| PLA/10PEG | - * | 75.7 | 165.9 | 22 |
| PLA/10PEG/CNF | - * | 76.9 | 166.0 | 19 |
| PLA/20PEG | - * | 88.1 | 166.9 | 32 |
| PLA/20PEG/CNF | - * | 87 | 166.8 | 29 |
*Signal overlaps with the melting transition of PEG
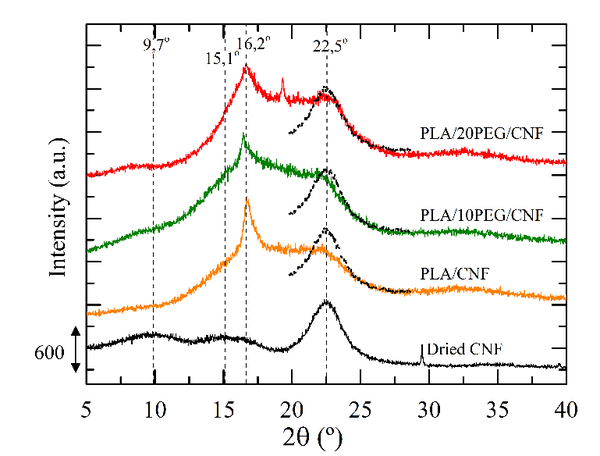
Figure 2 shows the X-ray diffractograms of dried CNF and all the CNF-reinforced (nano)composites. Dried CNF show peaks around 2θ= 9.7, 15.1, 16.2 and 22.5o, which are characteristic of the semi-crystalline cellulose I structure [16]. Since the peak intensity correlates with the crystallinity degree, a qualitative evaluation of the CNF crystallinity can be done by comparing the magnitude of the diffraction peak at 2θ= 22.5º between dried CNF and CNF-reinforced composites, as shown in Figure 2. Results suggested that melt processing showed a lower effect on the original integrity of cellulose I crystal polymorphism when the CNF surface was PEGylated first.
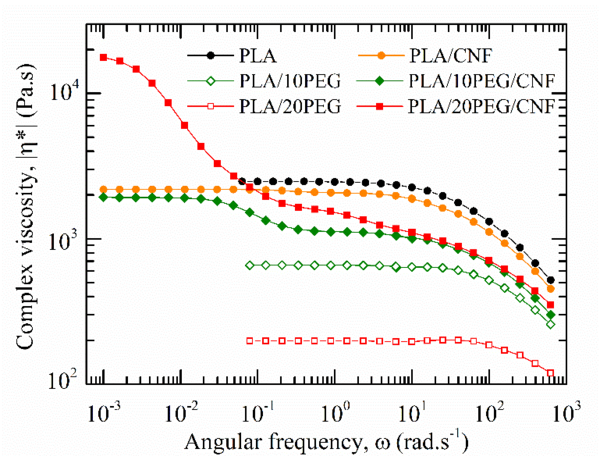
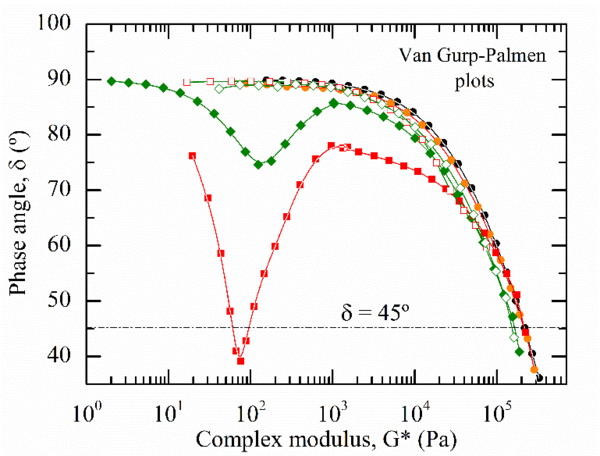
The rheological behaviour of the CNF-reinforced samples were investigated to have an insight in the evolution of the CNF dispersion as a function of the PEG content. Figure 3 shows the frequency dependence of the complex viscosity, |η*|, together with the Van-Gurp Palmen plots where the phase angle δ=arctan(G’/G’’) is plotted against the complex modulus, G*.
Pure PLA exhibits the typical pseudoplastic fluid behaviour. By adding PEG, a gradual drop in |η*| values was observed due to its plasticizing effect on PLA. PLA/CNF composites exbited a lower viscosity than PLA over the whole experimental window. This behaviour might be explained by a possible disruption of the molecular network induced by the large CNF aggregates that lower the PLA viscous resistance
Drastic changes in the PLA/CNF rheological behaviour were observed when CNF were added to PLA via the PEG masterbatch approach. Both PLA/PEG/CNF samples exhibited the characteristic rheological behaviour of structured fluids. The magnitude of the yield stress in |η*(ω)| increased with the PEG content, suggesting that a stronger CNF network was formed in PLA/20PEG/CNF. These observations are supported by the Van Gurp-Palmen plots. While a purely viscous behaviour is characterized by δ=90°, a purely elastic melted material exhibits a δ value of 0°. The PLA/20PEG/CNF sample displays δ values lower than 45°, implying that the elastic component of the melt exceeded the viscous one. This transition is characteristic of solid-like composites and corroborates the formation of a three-dimensional CNF network, also defined as rheological percolating CNF network in PLA/20PEG/CNF samples [17]. This results proves the successfulness of the CNF surface PEGylation to promote filler dispersion within PLA through melt blending.
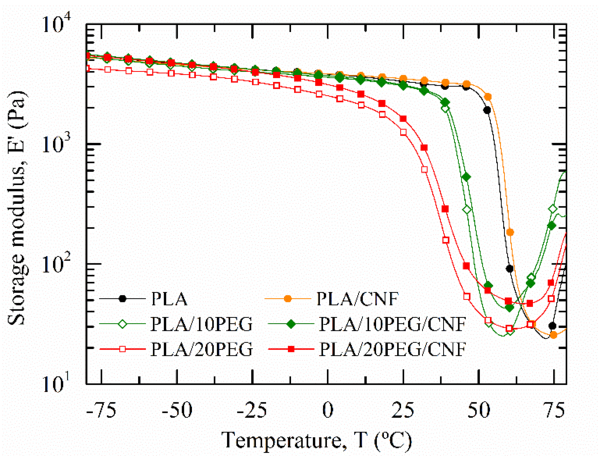
The effect of the CNF dispersion degree within PLA on the thermo-mechanical properties was investigated through DMTA. Figure 4 displays the temperature variation of the storage modulus, E’. Table 2 compiles the E’ at 25 ºC, the minimum E’ value in the rubbery plateau and the α-relaxation temperature, Tα.
Pure PLA exhibited the typical behaviour of amorphous polymers. At low temperatures, the sample stiffness remained fairly constant before experimenting a significant drop around 60ºC due to the glassy-to-rubbery transition. This transition is related to the α-relaxation of the macromolecular chains and corresponds to the dynamic glass transition temperature (Tg) of PLA [18]. The incorporation of CNF alone into PLA did not modify neither the temperature dependence of E’ nor the Tg of PLA within the experimental error, as listed in Table 2.
| Sample nomenclature | E’ at -25 ºC (GPa) | E’ in the RP (MPa) a | Tα (E”)
(ºC) b |
| PLA | 4.1 ±0.3 | 24 ±2 | 54 ±2 |
| PLA/CNF | 4.2 ±0.1 | 22 ±5 | 55.9 ±0.2 |
| PLA/10PEG | 3.8 ±0.2 | 28 ±3 | 41.3 ±0.4 |
| PLA/10PEG/CNF | 4.3 ±0.2 | 38 ±3 | 41.4 ±0.6 |
| PLA/20PEG | 3.7 ±0.5 | 28 ±1 | 27 ±1 |
| PLA/20PEG/CNF | 3.9 ±0.2 | 49 ±4 | 29 ±1 |
a minimum E’ value in the rubbery plateau (RP).
b Tα was determined as the point where the loss modulus, E”, reached a maximum.
By adding PEG, a clear reduction of the elastic-like behaviour of PLA was reported (lower E’ values). The plasticizing effect of PEG on PLA was evidenced by a progressive decrease in Tg. From Figure 4 and table 2, it is evident that the advanced plasticization of both PLA/PEG blends suppressed the load bearing contribution of the crystalline phase above Tg, which could increase the E’ values in the rubbery region [19].
When CNF and PEG were premixed and then melt blended with PLA, both biocomposites exhibited higher E’ values in the glassy region in comparison with their PLA/PEG counterpart. E’ at -25ºC of PLA/10PEG/CNF sample exceeded equally that of pure PLA. In the rubbery region, the previous CNF surface PEGylation resulted in a gradual increase of the rubbery storage modulus as compared to unfilled PLA or PLA/CNF samples. For instance, the minimum E’ value in the rubbery plateau increased by 51 % between PLA/20PEG and PLA/20PEG/CNF samples. This enhancement of the sample stiffness above Tg was ascribed to the tangling effects produced by the internal three-dimensional CNF network which demonstrated the load-bearing capacity of well-entangled CNF [20]. Regarding the PLA/20PEG/CNF sample, a slight increase in Tα was evidenced as compared to PLA/20PEG sample. This was ascribed to the formation of a percolated CNF network that restricted the molecular mobility of PLA macromolecules [2].
4 Conclusions
In the present study, bionanocomposites based on CNF as nanofillers and PLA as polymeric matrix were successfully and straightforwardly prepared through melt processing using a PEG/CNF masterbatch approach. A water-based hydrophilic PEG carrier system was used to counteract the affinity difference between the hydrophobic PLA and hydrophilic CNF, hence minimizing their self-agglomeration on drying. Different PEG concentrations were considered in order to investigate their effect on the final CNF dispersion degree. Finally, surface PEGylated CNF were diluted into PLA using a micro-compounder. Through rheological testing, a clear liquid-solid transition was observed by the masterbatch-based composites holding 20 wt.% of PEG, corroborating the creation of a well-entangled CNF network. Microscopy images confirmed the beneficial effect of an increasing PEG content in achieving a more homogenous morphology. Thermal analysis showed that PEG acted as plasticizer and enhanced the crystallinity of the matrix. However, under the processing conditions used in this work, adding CNF did not impact significantly the final thermal properties. Under thermo-mechanical loading, the elastic response of the rigid interconnected CNF structure mastered the viscoelastic response of the plasticized composites, notably improving PLA rigidity in the glassy as well as in the rubbery state. These promising results showed that PEG can be used as a successful carrier system in order to prepare a highly concentrated CNF masterbatch which then can be rapidly melt-processed using industrial melt-processing technique.
Acknowledgements
The authors acknowledge the financial support from the Spanish Ministry of Economy and Competitiveness through the Project MAT2016-80045-R (AEI/FEDER,UE). BASF is thanked for kindly supplying Irganox 1010®. J.-M. Raquez as a FRS-FNRS research associate and LPCM is much indebted to both Wallonia and the European Commission ‘‘FSE and FEDER” for financial support in the frame of H2020-LCFM portofolio.
References
[1] M. A. S. Azizi Samir, F. Alloin, et al., Biomacromolecules, 6, 2, pp. 612-26 (2005)
https://doi.org/10.1021/bm0493685
[2] F. Safdari, D. Bagheriasl, et al., Polym Composite, 39, 5, pp. 1752-62 (2018)
https://doi.org/10.1002/pc.24127
[3] W. Ding, T. Kuboki, et al., Rsc Adv, 5, 111, pp. 91544-57 (2015)
https://doi.org/10.1039/C5RA16901A
[4] S. Y. Cho, H. H. Park, et al., Macromol Res, 21, 5, pp. 529-33 (2013)
https://doi.org/10.1007/s13233-013-1057-y
[5] K. Missoum, M. Belgacem, et al., Materials, 6, 5, pp. 1745-66 (2013)
https://doi.org/10.3390/ma6051745
[6] D. Bondeson, K. Oksman, Compos Interface, 14, 7-9, pp. 617-30 (2007)
https://doi.org/10.1163/156855407782106519
[7] A. P. Mathew, K. Oksman, et al., J Appl Polym Sci, 97, 5, pp. 2014-25 (2005)
https://doi.org/10.1002/app.21779
[8] A. Kiziltas, B. Nazari, et al., Carbohydrate polymers, 140, pp. 393-9 (2016)
https://doi.org/10.1016/j.carbpol.2015.12.059
[9] D. Cheng, Y. Wen, et al., Carbohydrate polymers, 123, pp. 157-63 (2015)
https://doi.org/10.1016/j.carbpol.2015.01.035
[10] P. Zhang, D. Gao, et al., J Appl Polym Sci, 134, 14, pp. (2017)
https://doi.org/10.1002/app.44683
[11] M. Henriksson, G. Henriksson, et al., Eur Polym J, 43, 8, pp. 3434-41 (2007)
https://doi.org/10.1016/j.eurpolymj.2007.05.038
[12] G. Lo Re, J. Engstrom, et al., ACS Applied Nano Materials, pp. (2018)
https://doi.org/10.1021/acsanm.8b00376
[13] R. Hakim, J. Cailloux, et al., J Appl Polym Sci, pp. (2017)
https://doi.org/10.1002/app.45367
[14] Y. Peng, D. J. Gardner, et al., Cellulose, 19, 1, pp. 91-102 (2012)
https://doi.org/10.1007/s10570-011-9630-z
[15] F. J. Li, S. D. Zhang, et al., Polym Advan Technol, 26, 5, pp. 465-75 (2015)
https://doi.org/10.1002/pat.3475
[16] A. D. French, Cellulose, 21, 2, pp. 885-96 (2014)
https://doi.org/10.1007/s10570-013-0030-4
[17] J. Stokes, J. Telford, J Non-Newton Fluid, 124, 1-3, pp. 137-46 (2004)
https://doi.org/10.1016/j.jnnfm.2004.09.001
[18] L. Xiao, Y. Mai, et al., Journal of Materials Chemistry, 22, 31, pp. 15732-9 (2012)
https://doi.org/10.1039/C2JM32373G
[19] T. Tabi, I. Sajó, et al., Express Polym Lett, 4, 10, pp. (2010)
https://doi.org/10.3144/expresspolymlett.2010.80
[20] M. A. Saïd Azizi Samir, F. Alloin, et al., Macromolecules, 37, 11, pp. 4313-6 (2004)
Document information
Published on 24/01/23
Accepted on 27/12/22
Submitted on 27/12/22
Volume 03 - Comunicaciones Matcomp17 (2019), Issue Núm. 3 - Procesos de Fabricación II y Materiales Avanzados, 2023
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?