Abstract
Catalytic hydrotreating is an attractive method for upgrading bio-derived oils into renewable feedstocks with less oxygen content, suitable for producing valuable hydrocarbons through various petro-refinery processes. This study evaluates the catalytic activity of a commercial alumina (Al2O3) supported NiMo catalyst for hydrotreating tall oil feeds such as crude tall oil (CTO), distilled tall oil (DTO), and tall oil fatty acid (TOFA). Catalytic experiments carried out in a bench-scale fixed bed reactor set-up at different process conditions [space velocity (1–3 h−1), temperature (325–450°C), and H2 pressure (5 MPa)] produced a wide-range of products from tall oil feeds. Hydrotreating of TOFA produced highest yield of n-alkanes (>80 wt%) compared to DTO and CTO hydrotreating. A high conversion of fatty acids and resin acids was obtained in DTO hydrotreating. In CTO hydrotreating, a drop in conversion of fatty acids and resin acids was observed especially at the lowest temperature tested (325°C). The study revealed that there are various deoxygenation pathways preferential at different hydrotreating temperatures. As an example for TOFA, the decarboxylation route is dominant over the hydrodeoxygenation route at high temperatures (>400°C).
Introduction
Catalytic hydrotreating is an essential petro-refinery method to reduce the content of heteroatoms (S, N, and O) from raw materials, which has been widely employed to upgrade refinery feeds prior to such processes as catalytic reforming, catalytic cracking, and steam cracking [1]. In petro-refineries that use sulfur-rich fossil-fuel-based feeds, the catalytic hydrotreating technology is fully developed specifically for the hydrodesulfurization (HDS) of petroleum products and naphtha streams [2-4]. However, the recent research developments prompted by the increasing demand of bio-based fuels and chemicals reinvent the importance of developing optimal hydrotreating (hydrodeoxygenation, HDO) methods for reducing the amount of oxygenates from complex bio-derived feeds before the thermochemical conversion process into fuels or chemicals.
The amount of oxygenates in bio-derived oils vary depending on the origin of raw materials. Bio-oils produced by pyrolysis of lignocellulosic biomass are rich in oxygenates (35–40%). Therefore, deep hydrotreatment conditions are required to achieve complete deoxygenation from such bio-oils, inducing high processing cost [5]. Plant-based oils or vegetable oils are another class of bio-derived feedstock. Vegetable oils have been extensively studied as a feedstock for the production of bio-chemicals and biofuels through a catalytic HDO route [6-9]. The patented bio-synfining process produces naphtha range hydrocarbons for conventional steam cracking process from vegetable oils and fats [10, 11]. However, most of the widely studied vegetable oils are edible and remain as less favorable feedstocks as their selection to a refinery may create several environmental, economic, and societal issues. Therefore, a proper selection of a sustainable feedstock is vital. Tall oil, a by-product of the Kraft-pulping process, is nonfood chain affecting, economically feasible and low oxygen content feedstock [12]. Tall oil upgrading has already been received substantial attention as a sustainable bio-refinery method for producing biofuels and refinery feeds [12-14]. Tall oil comprises fatty acids, resin acids, and sterols. Different tall oil feeds such as crude tall oil (CTO), distilled tall oil (DTO), and tall oil fatty acid (TOFA) are commercially available, and can be used as valuable raw materials for chemical production. CTO is produced by means of the acidulation of tall oil soap skimmings from the black liquor which is obtained from pulping. DTO and TOFA are obtained from CTO distillation along with other fractions. Among the aforementioned tall oil feeds, CTO is regarded as the most cost-competitive raw material [15].
Sulfided molybdenum catalysts on alumina supports with nickel or cobalt as promoter metals are the most widely employed industrial catalysts for hydrotreating [16]. Supported CoMo and NiMo catalysts are more active in sulfided forms. There have been many studies reported on the origin of catalytic synergy between two main group elements in hydrotreating catalysts [17]. Different theories on the origin of catalytic activity and the nature of active sites can be found in literature [17, 18]. Kubicka et al. [19] reports that during hydroprocessing of vegetable oils over a sulfided NiMo catalyst, nickel sulfide phases are more active for decarboxylation than molybdenum sulfide phases. Therefore, the extent of HDO (hydrogenation/dehydration) versus decarboxylation reactions is highly dependent on factors such as promotor metal (Ni) concentrations and the dispersion of sulfidic phases. It is already learnt from previous studies that in comparison with CoMo catalysts, NiMo catalysts are more active for the HDO of aliphatic oxygenates [20]. Moreover, NiMo catalysts are found to be effective for hydrodeoxygenating cyclic oxygenates to cycloalkanes and also for causing ring opening reactions from cyclics at high temperatures, which produces valuable hydrocarbons for further refinery processes [12, 21]. Based on these observations, it can be suggested that NiMo catalyst is more applicable than a CoMo catalyst for hydrodeoxygenating bio-derived oils such as tall oil which contains aliphatic oxygenates as well as cyclic oxygenates. During catalytic hydrotreating of tall oil, oxygenates are removed by various deoxygenation routes which produces a wide range of hydrocarbons mainly paraffin range hydrocarbons [22, 23].
Among tall oil fractions, catalytic deoxygenation of TOFA has been studied with considerable interest for producing a hydrocarbon fraction suitable as a diesel fuel [24]. Egeberg et al. [2] reports that hydroconversion of TOFA can be slightly different than that of triglycerides (vegetable oils) as the former produces more methane by means of methanation reaction, whereas the latter produce more propane by the scission of glycerol backbone. Catalytic deoxygenation of TOFA investigated by other researchers reveal that a significant yield of n-heptadecane (C17) with high selectivity can be obtained from TOFA using a palladium mesoporous carbon catalyst [24]. Low temperatures are found to be more favorable for the formation of n-octadecane from fatty acids through a HDO route. Hydrotreating of CTO has been discussed in the literature through several patents which mainly focuses the application of hydrotreated products as a diesel fuel [25-27]. Previously, we successfully reported on the use of hydrotreated DTO and CTO as a steam cracker feed [22, 23]. A high degree of deoxygenation was obtained at low temperatures (325–400°C) and space velocity of weight hourly space velocity (WHSV) = 1 h−1, yielding a maximum in paraffins. Our group has also studied the hydrotreating chemistry of sterols in CTO and proposed a reaction scheme for the HDO of sterols at low temperatures in line with studies on the HDO of phenolic compounds and saturated alicyclic alcohols over sulfided catalysts [23]. Importantly, in our study, a high conversion of sterols was obtained at the tested conditions irrespective of space time and temperature.
In this study, a comparative study is presented using different tall oil feedstocks (TOFA, DTO, and CTO) The effect of space time and temperature on the hydrotreating of DTO and CTO is already been reported [22, 23], therefore, they are not discussed further in this study. More specifically, this study presents the results of TOFA hydrotreating under the applied conditions, which are then compared with the results of DTO and CTO hydrotreating. This comparison has carried out on the basis of achieved product distribution, composition of organic phase samples and the conversion of acid fractions at the most favorable space time (WHSV = 1 h−1). The influence of feedstock type and reaction severity (temperature) during hydrotreating of tall oils (6 h of time on stream) over a sulfided NiMo catalyst is thus studied in this article.
Materials and Methods
Commercially available TOFA (SYLFAT® 2) and DTO (SYLVATAL® 25/30 S); and CTO obtained from Stora Enso pulping facilities in Finland were used as feeds for hydrotreating experiments. Detailed chemical and elemental composition and total acid number of the employed feeds are represented in Tables 1 and 2. A commercial alumina (Al2O3) supported NiMo catalyst in sulfided form was employed for hydrotreating studies. The presulfidation was carried out by using a H2S/H2 mixture for 5 h at 400°C (H2S/H2 = 5 vol. %). The test runs (6 h) with each tall oil feed were performed in a continuous down flow fixed bed reactor (stainless steel tube, length: 450 mm long, internal diameter: 15 mm) at different process conditions; temperature = 325–450°C, H2 partial pressure = 5 MPa, weight hourly space velocity (whsv) = 1–3 h−1 and H2/feedstock molar ratio = 17.4. Duplicate experiments were carried out for certain test runs so as to check the reproducibility of yield. In a typical experiment, a known amount of the catalyst (6, 3, and 2 g corresponds to WHSV 1 h−1, WHSV 2 h−1, and WHSV 3 h−1) was loaded in the reactor which was then placed in an oven (Oy Meyer Vastus, Monninkylä, Finland). The catalyst bed temperature was monitored by a temperature controller (TTM-339 series). A set amount of feedstock was fed to the reactor along with hydrogen (62 mol/kg of CTO). TOFA and DTO were used as received for hydrotreating experiments, whereas CTO was preheated in order to reduce the viscosity, and thus to enhance the feeding of the material to the reactor. Gas chromatographic tools such as such as GC-MS (HP-5 MS column), GC×GC-MS (ZB-5 HT inferno and ZB-35 HT inferno columns)and GC×GC-FID/(TOF-MS) (Rtx-1 PONA and BPX-50 columns) were used for quantitatively (using external calibration method) and qualitatively analyzing the organic phase samples obtained from hydrotreating of tall oils [22, 23]. GC-FID (DB-23 and Zebron ZB-1 columns) analyses were carried out to calculate the residual amounts of fatty acids, resin acids, and sterols from organic phase samples. The amount (wt%) of water in aqueous phase samples was determined by Karl Fischer (KF) titration (method: ASTM E 203). Analysis of gas phase components was carried out by FT-IR and micro-GC instruments. The residual oxygen composition of organic phase samples from hydrotreated tall oils was determined by elemental analysis using CHN equipment based on ASTM D 5291 method. Sulfur content was determined by means of ASTM D 4239 method.
| Composition (wt.%) | TOFA | DTO | CTO | |
|---|---|---|---|---|
| Free and bonded Fatty Acids | 92.6 | 71.3 | 48.1 | |
| Free | Bonded | |||
| (16:0) Palmitic acid | 0.40 | 0.20 | 2.1 | 0.04 |
| (17:0) Margaric acid | 0.80 | 0.30 | 0.4 | 0.01 |
| (18:0) Stearic acid | 0.90 | 0.70 | 0.8 | 0.01 |
| (18:1) Oleic acid | 27.4 | 15.3 | 8.6 | 0.16 |
| (18:1) 11-octadecenoic acid | 0.70 | 0.50 | 0.3 | 0.01 |
| (18:2) 5.9-octadecadienoic acid | 0.50 | 0.30 | 0.2 | 0.02 |
| (18:2) conj. octadecadienoic acid | 6.00 | 8.30 | 5.5 | 0.36 |
| (18:2) Linoleic acid | 23.1 | 24.3 | 19.1 | 2.4 |
| (18:3) Pinolenic acid | 8.00 | 4.4 | 1.6 | 0.5 |
| (18:3) Linolenic acid | 4.30 | 0.60 | 0.6 | 0.02 |
| (18:3) conj. octadecatrienoic acid | 0.20 | 1.80 | 0.4 | 0 |
| (20:0) Arachidic acid | 0.80 | 0.40 | 0.7 | 0.01 |
| (20:3) 5.11.14-eicosatrienoic acid | 7.90 | 7.60 | 1.4 | 0.08 |
| (20:3) 7.11.14-eicosatrienoic acid | 1.30 | 0.60 | 0.2 | 0.2 |
| Other fatty acids | 10.3 | 6.00 | 1.8 | 0.28 |
| Free and bonded Resin Acids | 1.3 | 23 | 28.5 | |
| Free | Bonded | |||
| 8,15-isopimaradien-18-oic acid | 0.1 | 0.5 | ||
| Pimaric acid | 0.7 | 4.8 | 2.6 | 0.03 |
| Sandaracopimaric acid | – | 0.3 | 0.8 | 0.03 |
| Diabietic acid | – | 0.5 | – | – |
| Palustric acid | – | 2.2 | 3.9 | 0.01 |
| Isopimaric acid | – | 1.1 | 1.3 | 0.01 |
| 7. 9 (11) -abietic acid | – | 0.4 | 0.1 | 0 |
| 13-B-7.9(11)-abietic acid | – | 0.3 | 0.2 | 0.02 |
| Abietic acid | 0.1 | 7.7 | 9.1 | 0.1 |
| Dehydroabietic acid | – | 3.6 | 2.4 | 0.04 |
| Neoabietic acid | – | 0.4 | 3.3 | 0.02 |
| Other resin acids | 0.4 | 1.3 | 2.8 | 0.7 |
| Neutrals | 23 | |||
| Campesterol | – | – | 0.02 | – |
| Campestanol | – | – | 0.17 | – |
| β-Sitosterol | – | – | 2.5 | – |
| β-Sitostanol | – | – | 0.5 | – |
| Lupeol | – | – | 0.5 | – |
| Cycloartenol | – | – | 0.2 | – |
| 24-methylenecycloartenol | – | – | 2.5 | – |
| Prenol-7 | – | – | 0.07 | – |
| α-Sitosterol | – | – | 1.1 | – |
| Methyl betulinate | – | – | 0.02 | – |
| Betulin | – | – | 0.3 | – |
| Betulinic acid | – | – | 0.5 | – |
| Prenol-8 | – | – | 0.9 | – |
| Other | – | – | 12.6 | – |
| Composition | TOFA | DTO | CTO |
|---|---|---|---|
| Elemental composition | |||
| C (wt%) | 76.8 | 77.4 | 78.6 |
| H (wt%) | 11.6 | 11.1 | 11.2 |
| O (wt%) | 11.6 | 11.4 | 9.9 |
| N (ppm) | 12 | 30 | 100 |
| S (ppm) | 53 | 161 | 1800 |
| P (ppm) | – | – | 36 |
| Alkali metals and alkaline earth metals (ppm) | – | – | 56 |
| Total acid number (TAN) mg/kg | 326 | 300 | 129.5 |
Terminology and calculations
For the sake of clarity, the term paraffins in this study correspond to n-alkanes and i-alkanes. Nonaromatics denote cycloalkanes or naphthenes (-mono, -di, or -tri) and other cyclic oxygenates as well as fatty alcohols and unsaturated hydrocarbons. The reactions which mention in this study under hydrotreating conditions are hydrogenation of double bonds, decarboxylation, decarbonylation, HDO, isomerization, hydrocracking of alkane and cyclic structures, and thermal cracking reactions. HDO is represented as a reaction resulted from complete hydrogenation with saturation of double bonds as the first step. Selective deoxygenation denotes the catalytic deoxygenation reactions at low temperatures (Mainly HDO, removal of oxygen as water). Nonselective deoxygenation represents the deoxygenation reactions which occur at high temperatures (thermal or catalytic cracking, removal of oxygen as CO2).
The mass based yield (%) of various product streams in this study is calculated as a relative yield based on the amount (g/h) of feedstock entering to the reactor. Conversion of fatty acids and resin acids was calculated by assuming that residual acid fractions are mostly present in the organic phase. The equation used for calculating the conversion is
|
|
nA, Feed, is the total mol of acids (fatty acid/resin acid) present in the feed, nA, O.P., is the total mol of acids (fatty acid/resin acid) present in the organic phase.
Product yield of paraffins (n-alkanes + i-alkanes) was calculated by assuming that paraffins are solely formed from fatty acid fraction in tall oils. Product yield was calculated by using the following equation.
|
|
The reaction steps discussed in this study in association with the hydrogenation and dexoygenation of fatty acids (linoleic acid and oleic acid) in tall oils over a sulfided NiMo catalyst are shown below by Equations (1), (2), (3), (4), (5), and (6)
Hydrogenation:
|
|
(1) |
|
|
(2) |
|
|
(3) |
Deoxygenation:
|
|
(4) |
|
|
(5) |
|
|
(6) |
Experimental Results
Hydrotreating of TOFA
Hydrotreating experiments with TOFA on a NiMo catalyst resulted in liquid product streams which differ in color and yield depending on the conditions in the reactor. Importantly, the samples from TOFA hydrotreating at low temperatures (<350°C) appeared as oily samples with the tendency of solidification at room temperature. It was also observed that the temperature, 300°C, tested with TOFA resulted the excessive deposition of wax; thus, reactor plugging. In line with earlier studies [22, 23], mass balance error (±5%) was higher in experiments at elevated temperatures (>400°C) in comparison with low-temperature experiments. Liquid products obtained from TOFA hydrotreating were separated into organic and aqueous phases. Table 3 shows the detailed product distribution in organic phases obtained from TOFA hydrotreating at various process conditions. As hydrotreating of TOFA produces more paraffins than other tall oil feeds, the product distribution of major paraffinic hydrocarbons such as n-octadecane and n-heptadecane obtained in TOFA hydrotreating at different process conditions is presented in this study as a separate section.
| Temp (°C) | 325 | 350 | 400 | 450 | 325 | 350 | 400 | 450 | 325 | 350 | 400 | 450 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WHSV (h−1) | 1 | 1 | 1 | 1 | 2 | 2 | 2 | 2 | 3 | 3 | 3 | 3 |
| Composition (wt%) | ||||||||||||
| Paraffins | ||||||||||||
| nC7-C9 | 0.4 | 1.2 | 6.8 | 10.1 | 0.9 | 1.8 | 4.4 | 8.3 | 0.2 | 0.8 | 3.1 | 9.2 |
| nC10-C16 | 1.4 | 2.2 | 4.3 | 8.6 | 4.2 | 2.9 | 3 | 14.1 | 2.8 | 1.9 | 4.4 | 8.3 |
| nC17 | 27.7 | 36 | 36 | 27 | 24 | 31 | 31 | 20 | 21.5 | 29 | 30 | 23 |
| nC18 | 53 | 44 | 32 | 22 | 49.8 | 49 | 35 | 29 | 43 | 41 | 33 | 33 |
| nC19 | 1.2 | 1 | 1.3 | 2.1 | 3.8 | 2.4 | 0.9 | 1.1 | 0.8 | 0.7 | 0.6 | 0.4 |
| nC20 | 2 | 0.8 | 1.4 | 1.5 | 2.4 | 2.1 | 0.2 | 0.9 | 2.9 | 1.5 | 1.4 | 0.2 |
| ialkanes | 2.1 | 2.5 | 4.1 | 6 | 1.4 | 1.7 | 3.4 | 3 | 1.4 | 2.1 | 2.7 | 2.9 |
| Total | 87.6 | 87.7 | 86 | 78.1 | 86.5 | 90.9 | 77.9 | 76.4 | 72.6 | 77 | 75.2 | 77 |
| Nonaromatics | ||||||||||||
| Cycloalkanes | 0.08 | 0.2 | 1.4 | 3 | 0.9 | 0.4 | 0.4 | 2.5 | 0 | 0.03 | 0.9 | 0.3 |
| Olefins | 0.07 | 0.4 | 0 | 0 | 0.08 | 0.03 | 0.01 | 0.6 | 0.1 | 0.2 | 0.5 | 0.6 |
| Alcohols | 2.3 | 3.5 | 2.7 | 0.3 | 0.4 | 0.4 | 1.8 | 1 | 0.3 | 1 | 1 | 0.9 |
| Other | 9.5 | 7.9 | 4.9 | 15.8 | 5.1 | 7 | 9 | 9.1 | 13.9 | 5.9 | 6.8 | 6 |
| Total | 11.9 | 12 | 13 | 19.1 | 6.4 | 7.83 | 11.2 | 13.2 | 14.3 | 7.13 | 9.2 | 7.8 |
| Aromatics | ||||||||||||
| Monoaromatics | 0 | 0 | 1.7 | 4.1 | 0.07 | 0.04 | 0.9 | 1.4 | 0 | 0.04 | 0.8 | 1.2 |
| Overall wt% | 99.5 | 99.7 | 96.7 | 91.3 | 93 | 98.7 | 90 | 90 | 86.9 | 84 | 85.2 | 86 |
| Residual oxygen | 0.8 | 0.8 | 0.46 | 0.4 | 1.3 | 0.9 | 0.7 | 0.9 | 1.6 | 1.9 | 1.1 | 1 |
Product distribution of C17 and C18 hydrocarbons in TOFA hydrotreating
The amount of n-octadecane and n-heptadecane obtained in TOFA hydrotreating on a sulfided NiMo catalyst is shown in Table 3. It is clear from Table 3 that a significant drop in the production n-octadecane occurred with TOFA from 325 to 450°C especially at the longest space time tested (WHSV = 1 h−1), whereas n-heptadecane production steadily increased until 400°C and then sharply decreased at 450°C. As already noted from earlier studies [22, 23], low temperatures (<400°C) are more favorable for the formation of n-octadecane through a HDO route. As the temperature increases, routes consuming less hydrogen such as decarboxylation and decarbonylation are prominent, producing a significant amount of n-heptadecane at these temperatures. Higher concentration of n-heptadecane over n-octadecane was observed beyond 350°C at the longest space time tested (WHSV = 1 h−1). This observation is in agreement with the earlier report on the promotion of decarboxylation route over HDO as a function of increasing temperature on a sulfided NiMo catalyst [28]. It may also imply that decarboxylation + decarbonylation reactions predominate over HDO reaction solely at longer space times, that is, WHSV = 1 h−1 with TOFA. Importantly, in comparison with hydroprocessing of vegetable oils such as rapeseed oil and sunflower oil over a NiMo catalyst [29], a similar yield of n-alkanes from TOFA hydrotreating at lower temperature (350°C) was achieved, especially for the n-C17 + n-C18 fraction. It is noteworthy that the amount of oleic acid (C18:1) and linoleic acid (C18:2) is considerably lower in TOFA compared to the aforementioned vegetable oils. Interestingly, in TOFA hydrotreating, irrespective of space time a similar product distribution of n-heptadecane and n-octadecane was observed at 400°C.
Comparison of TOFA, DTO, and CTO hydrotreating
Overall product distribution
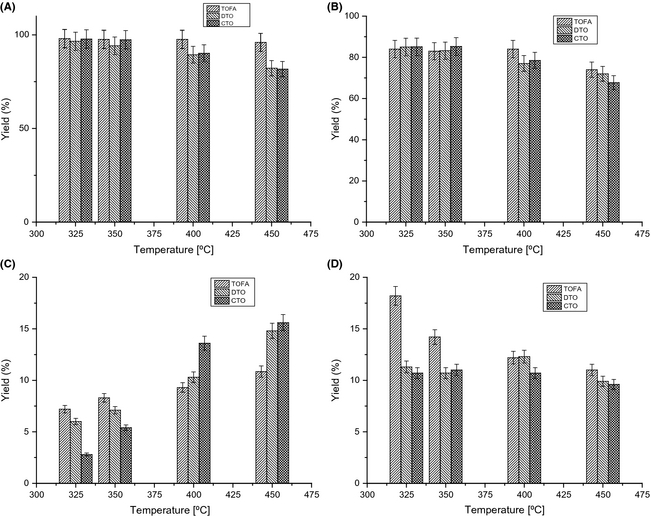
Figure 1 shows the yield of various product streams obtained as a function of temperature at WHSV = 1 h−1 in hydrotreating of tall oils. As noted from Figure 1, among the three tall oil feeds tested, TOFA was found to give the highest yield of liquid products and organic products at different temperatures. It was also observed with the employed tall oil feedstocks that the lowest temperature tested (T = 325°C) resulted to a maximum yield of liquid products. As expected, maximum gas product yield was obtained at 450°C with each tall oil feeds. At this temperature, the highest yield of gas products was obtained from CTO in comparison with DTO and TOFA. The FT-IR detected gas phase components were CO2, CO, methane, ethane, ethylene, propane, propylene, and butane. Importantly, lighter alkanes and alkenes were obtained solely at high temperatures especially from CTO and DTO. As can be seen from Figure 1, among three tall oil feedstocks the highest yield of water was obtained from TOFA. The trend with the production of water from tall oils followed the decreasing order as TOFA–DTO–CTO. Water is obtained as a by-product of HDO (hydrogenation/dehydration) reaction during hydrotreating.
|
|
|
Figure 1. Yield (%) of various product streams obtained in hydrotreating of tall oils over a NiMo catalyst, WHSV = 1 h−1, T = 325–450°C (A) liquid product yield, (B) organic phase product yield, (C) gas product yield, and (D) water yield. |
Composition of organic phase
Figure 2 shows a comparison of the product composition of organic phase samples as a function of temperature obtained in TOFA, DTO, and CTO hydrotreating at WHSV 1 h−1. As can be seen from Figure 2, maximum amount (wt%) of paraffins was obtained from TOFA among three tall oil feeds. This observation appear as an obvious result in relation to the higher amount (wt%) of fatty acids in TOFA.
|
|
|
Figure 2. Product composition (wt%) of organic phase samples in tall oil hydrotreating (A) paraffins,(B) nonaromatics, (C) aromatics, and (D) residual fractions; WHSV =1 h−1, T = 325–450°C. |
Product distribution of aromatic and nonaromatic hydrocarbons
As can be seen from Figure 2, nonaromatics such as cyclic structures (monocyclic and polycyclic hydrocarbons), alcohols, unsaturated hydrocarbons, and sterol derivatives were obtained in higher amount with CTO in comparison with other tall oil feeds. Among the three tall oil feeds, maximum production of aromatics was obtained from DTO. It has been reported previously that the aromatics from DTO hydrotreating comprised primarily of monoaromatics such as substituted benzenes in addition to polyaromatics [22]. Organic phases obtained from three feedstocks especially from CTO also comprised some sulfur (100–500 ppm) and nitrogen (10–20 ppm) [23].
Fatty acid and resin acid conversion
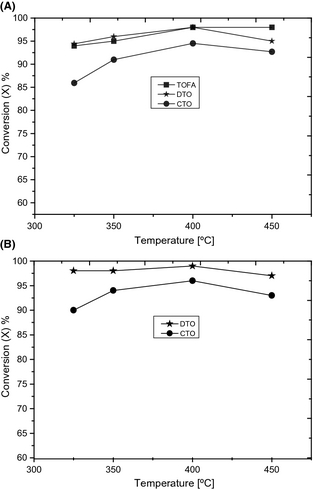
Fatty and resin acid conversions were evaluated based on GC-FID analysis. Figure 3 shows the fatty and resin acid conversions obtained with different tall oil feedstocks at the most favorable space time (WHSV = 1 h−1) as a function of temperatures. Figure 3A shows that fatty acid conversion achieved in a high extent with TOFA and DTO. A similar conversion level achieved with fatty acids in TOFA and DTO in low-temperature hydrotreating experiments (325–400°C). In comparison with TOFA and DTO, a drop in conversion of fatty acids was obtained in CTO hydrotreating under the tested conditions. Figure 3B shows that elevated conversion level was reached for resin acids as well in DTO hydrotreating. In CTO hydrotreating, a low conversion level of resin acids was obtained in line with the conversion of fatty acids from the same feedstock.
|
|
|
Figure 3. Conversion of fatty acids (A) and resin acids (B) obtained in hydrotreating of TOFA, DTO, and CTO at WHSV = 1 h−1, T = 325–350°C. |
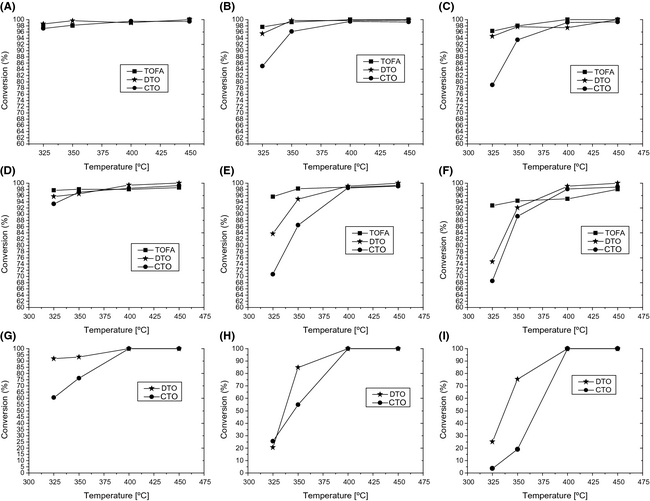
Furthermore, in order to get more insight into the behavior of fatty acids and resin acids during hydrotreating of tall oils, the conversion of individual fatty acids and resin acids in each tall oil feedstock was assessed. Figure 4A–I shows the conversion of linoleic acid, oleic acid, and dehydroabietic acid obtained during the hydrotreating of TOFA, DTO, and CTO at different space times (WHSV = 1–3 h−1) as a function of temperature. It should be stated that all fatty acids and resin acids other than the aforementioned ones have achieved more or less full conversion levels during the hydrotreating of tall oils under the tested conditions; therefore, they are not included in the results.
|
|
|
Figure 4. Conversion of individual fatty acids and resin acids; linoleic acid: (A) WHSV =1 h−1, (B) WHSV =2 h−1, (C) WHSV = 3 h−1; oleic acid: (D) WHSV = 1 h−1, (E) WHSV = 2 h−1, (F) WHSV = 3 h−1;dehydroabietic acid: (G) WHSV = 1 h−1, (H) WHSV = 2 h−1, and (I) WHSV = 3 h−1. |
Discussion
Based on distribution of gaseous products obtained from tall oil hydrotreating experiments, it can be assumed that in TOFA and DTO hydrotreating at high temperatures (>400°C), mainly nonselective deoxygenation occurs with a sulfided NiMo catalyst by means of cracking (thermal or catalytic) [12, 28] which requires no hydrogen and produces short-chain oxygenates and CO2 as well as hydrocarbons (cycloalkanes and aromatics) [30]. With CTO, it is suggested that nonselective deoxygenation can be lower at high temperatures (>400°C) from sterols [23] although sterol hydrotreating chemistry at high temperature has not been elucidated yet. The gas product composition with high concentration (~60% yield of total gaseous products) of lighter alkanes and alkenes from CTO hydrotreating may imply that sterols in CTO are mainly converted by thermal decomposition (cracking) of side chains as well as the ring opening at high temperatures (>400°C). The thermal decomposition and ring opening reactions from sterols produce gas phase light hydrocarbons along with oxygenates and unsaturated cyclic structures [31]. A ring opening and thermal decomposition route is also expected from resin acids in CTO and DTO at high temperatures, producing lighter alkanes and alkenes [32] On the other hand, the increased yield of gaseous products in low temperature (<400°C) TOFA and DTO hydrotreating in comparison with the CTO hydrotreating at similar conditions is attributed to the increased formation of CO2 (~40%), CO (~20%), and methane (~20%) through selective deoxygenation routes. Interestingly, in contrast with methane, propane was obtained only in minor yields in low temperature (<400°C) tall oil hydrotreating.
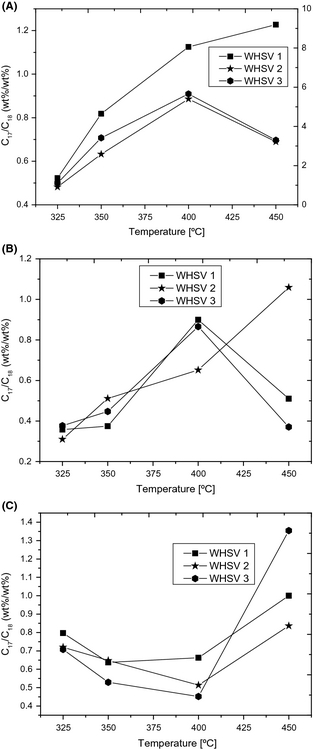
n-Octadecane and n-heptadecane were obtained as the major paraffinic hydrocarbons from tall oil hydrotreating through HDO, decarboxylation and decarbonylation reactions. In order to further evaluate the extent of decarboxylation + decarbonylation reactions versus HDO reaction in tall oil hydrotreating, the C17/C18 ratio of the liquid products was calculated. Figure 5 shows the comparison of the C17/C18 ratio obtained from the hydrotreating of TOFA, DTO, and CTO at different process conditions (space time and temperature). It is interesting to note from Figure 5 that in agreement with TOFA hydrotreating, DTO hydrotreating at different WHSVs also results an increase in C17/C18 ratio as a function of increasing temperature until 400°C. Surprisingly with CTO, a decrease in C17/C18 ratio was observed from 325 to 400°C in all tested cases. The higher C17/C18 ratio obtained in low temperature (325°C and 350°C) experiments irrespective of space time is attributed to the decrease in formation of n-octadecane. This result suggests that the complex nature of CTO feedstock plays a significant role during its hydrotreating over a NiMo catalyst. The selective deoxygenation reactions may be reduced with CTO at lower temperatures (<400°C) due to a number of reasons, which is related to the reactivity of fatty acids in CTO and discussed in a separate section in this study. At 450°C, it appears that a comparison of C17/C18 ratio as a function of WHSV is challenging for the tested tall oil feeds. Presumably, the formation n-octadecane and n-heptadecane can be significant at shorter space times (WHSV = 2 h−1 and WHSV = 3 h−1), whereas cracking reactions which produce shorter chain alkanes from C17 and C18 hydrocarbons can also be significant as a function of increasing space time.
|
|
|
Figure 5. C17/C18 (wt%/wt%) ratio obtained from tall oil hydrotreating (A) TOFA (B) DTO (C) CTO; WHSV = 1–3 h−1, T = 325–450°C. |
Regarding the other fractions in organic phase, monoaromatics are mainly formed from fatty acid fractions through intermediate n-alkanes by means of cyclization and aromatization reaction routes [30]. In addition to this, a direct route for the formation of cycloalkanes and aromatics prior to deoxygenation has also been proposed in the literature [33]. Monoaromatics can also be resulted from resin acids and sterols in tall oil by selective ring opening and dehydrogenation reactions which are valid on the employed catalyst at high temperatures. As fatty acids act as the major precursor for monoaromatics formation at high temperatures, it is obvious that lower amount of fatty acids in CTO in comparison with DTO causes a low yield of monoaromatics [30]. Interestingly, the ratio of polyaromatics to monoaromatics was higher with CTO compared to DTO, and it can be credited to the supplement formation of polyaromatics from the sterol fraction in CTO, either prior to or after deoxygenation.
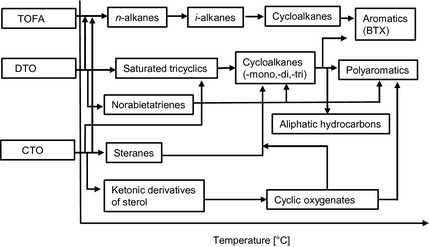
Based on the obtained composition of reaction products in organic phase samples, a reactivity scale is proposed for the formation of major products from each tall oil feeds as a function of temperature (325–450−°C), and represented as Figure 6. It should be noted that the proposed reactivity scale excludes the possible thermal or catalytic cracking reactions which occur directly from tall oil fractions in high-temperature experimental runs.
|
|
|
Figure 6. Proposed reactivity scale for the formation of major products from tall oil feeds in hydrotreating (T = 325–450°C). |
Conversion and reactivity of acid fractions in tall oil
Fatty acids
As noted from Figure 3A, a decrease in conversion of fatty acids was obtained in CTO hydrotreating. Therefore, it is important to assess the effect of other fractions on the conversion of fatty acids in CTO. It is already learnt from DTO hydrotreating that resin acids are not inducing any significant effect on fatty acids in terms of inhibiting its reactivity. In addition to resin acids CTO also contains a considerable amount of neutrals (sterols). In view of the earlier study on the HDO of phenolic compounds and alicyclic alcohols [20], it can be proposed that the sulfur anion vacancy (CUS) site which is responsible for the HDO of acid fractions are also responsible for the HDO of sterols (direct C–O hydrogenolysis). Interestingly, in our previous study on CTO hydrotreating [23], a high conversion range of sterols occurred at the tested conditions irrespective of space time and temperature. Therefore, based on these observations we may only conclude that a high conversion of sterols is achievable in spite of the competitive adsorption on CUS between acid fractions and sterols in CTO.
The effect of sulfur on fatty acid conversion
It is widely accepted that metal impurities present in the feed influences the hydroprocessing reactions on a sulfided NiMo catalyst [3]. However, the effects of metal impurities on the conversion of fatty acids are ruled out in this research study as the test runs were carried out only for 6 h of time-on-stream. The drop in conversion of fatty acids in CTO hydrotreating is considered in this study mainly based on the sulfur content in the feedstock. It has been reported that the sulfur content in the feed, for instance, elemental sulfur, preferably in a range 2000–5000 w ppm is able to enhance the extent of decarboxylation reactions over a sulfided NiMo catalyst [26]. This increase in the magnitude of decarboxylation reactions is attributed to the increased catalyst acidity by means of adsorption of sulfur species onto the catalyst surface. Senol et al. [34] and Ryymin et al. [35] studied the effects of sulfur additives (H2S and dimethyl disulfide (DMDS)) in the HDO of aliphatic oxygenates and published that in the presence of sulfur additives, reductive reactions such as hydrogenation (saturation of double bonds), and HDO (addition of hydrogen adjacent to carbonyl carbon, which results the step-wise formation of alkanes and water through intermediate alcohol) are suppressed on a sulfided NiMo catalyst. On the basis of this aspect, we can conclude that the unexpected higher C17/C18 ratio obtained in CTO hydrotreating at low temperature (<350°C) (Fig. 5) is presumably due to the higher concentration (1800 ppm) of sulfur in CTO compared to DTO and TOFA, and their influence on selective deoxygenation routes from fatty acids. However, a significant increase in the decarboxylation route (C17 formation) in comparison with HDO route (C18 formation) is not observed in CTO hydrotreating by virtue of the sulfur content in the feed, but rather it seemed like both HDO and decarboxylation are occurred in a similar extent in CTO hydrotreating. It may presume that in comparison with TOFA and DTO hydrotreating, decarboxylation route was more prominent in CTO hydrotreating in low-temperature experiments apparently due to the effects of sulfur content. The drop in HDO of fatty acids in CTO at low temperatures compared to acids in other feedstocks can be attributed to the decreased reductive reactions due to the effect of sulfur. However, no conclusive comments are drawn here associating the structure–activity relationship of the employed catalyst during hydrotreating of tall oils.
Reaction mechanism assessment of fatty acids
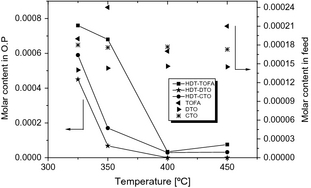
The reaction mechanism of fatty acids is discussed here based on the conversion of individual fatty acids presented in Figure 4. Figure 4A–C shows that linoleic acid, one of the main fatty acids present in tall oil (present in similar concentrations in TOFA, DTO, and CTO), was converted in similar extent with all feedstocks at longer space time (WHSV = 1 h−1). However, at shorter space times (WHSV = 2 and 3 h−1) a prominent drop in reactivity of linoleic acid especially at low temperatures (325–350°C) is observed with CTO compared to other feedstocks. It is also observed from Figure 4D–F that other major fatty acid, oleic acid, converted in a slightly lesser extent in CTO hydrotreating even at WHSV 1 h−1 compared to the hydrotreating of TOFA and DTO. At shorter space times (WHSV = 2 and 3 h−1), a significant drop in the conversion of oleic acid was also observed in DTO hydrotreating. Oleic acid can be formed from linoleic acid by partial hydrogenation [36], therefore, we perceive the possibility of oleic acid formation from linoleic acid at least favorable space times such as WHSV = 2 and 3 h−1. The formation of oleic acid at these conditions indirectly results a drop in the conversion of oleic acid. Furthermore, Figure 7 is plotted to show the molar amount (moles) of stearic acid (a saturated fatty acid (C18) which is reported to form as an intermediate acid during the hydrotreating of C18 unsaturated fatty acids) in feedstock and hydrotreated products, respectively, at WHSV=1 h−1. It is evident from Figure 7 that high concentrations of stearic acid is obtained in low temperature (<400°C) hydrotreating experiments. This observation confirms that fatty acids (linoleic acid and oleic acid) undergo deoxygenation through an initial fatty acid chain double-bond hydrogenation as proposed by other researchers [8, 36, 37]. The saturated fatty acid formed; stearic acid in this case, can then undergo deoxygenation via different routes.
|
|
|
Figure 7. Concentration (molar content) of stearic acid in feeds and hydrotreated products at WHSV = 1 h−1. |
It can be suggested that hydrogenation is prominent at elevated temperatures as well, and produce saturated fatty acids. However, as shown by reactions (4), (5), and (6), n-alkanes are formed (up to an optimum temperature level) at the expense of intermediate fatty acids, which in turn results a drop in the concentration of saturated fatty acids (stearic acid) in the product stream from high temperatures (>400°C); as evident from Figure 7. Interestingly for all feeds short-chain fatty acids as well as partially hydrogenated fatty acids were detected in product streams, where for CTO the concentration of partially hydrogenated fatty acids especially at low temperatures was markedly higher than for DTO and TOFA. With TOFA and DTO, a high yield (wt%) of short-chain fatty acids at high temperatures are resulted by means of cracking reactions.
Fatty acid conversion versus paraffin yield
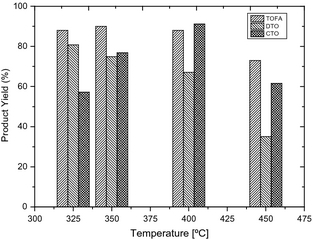
The product yield (%) of paraffins was calculated in this study in order to correlate fatty acid conversion and paraffin formation. It is important to observe that product yield of paraffins corresponds to the amount of fatty acids in feedstocks especially in DTO and CTO hydrotreating experiments. Palanisamy et al. [38] proposes a route for the formation of n-alkanes and i-alkanes from resin acids (abietic acid) through a ring opening mechanism over a sulfided NiMo catalyst. This ring opening mechanism through C–C bond cleavage may be valid in our experimental conditions producing acyclic paraffinic hydrocarbons (n-alkanes and i-alkanes). Figure 8 shows the yield of paraffins as a function of temperature, obtained from TOFA, DTO, and CTO hydrotreating at WHSV = 1 h−1. It should be taken into consideration that product yield of paraffins can be lower at a high temperature (>400°C) due to the successive formation of cycloalkanes and aromatics from paraffins. Considering this, it can be seen from Figure 8 that a high yield (%) of paraffins was obtained from TOFA hydrotreating, which is no surprise as fatty acids were converted in high extent in that case. A high yield of paraffins was obtained from low-temperature experimental runs in DTO hydrotreating. Remarkably, a high yield of paraffins was obtained from CTO hydrotreating at temperatures 350 and 400°C. As already discussed, fatty acids in CTO are converted in a lesser extent than fatty acids in TOFA and DTO. Furthermore, fatty alcohols and fatty acid esters were detected in significant concentrations in samples from low temperature (325–400°C) hydrotreating of CTO. These fatty alcohols and fatty acids are resulted from the incomplete HDO of fatty acids. This observation also signifies that even at low temperatures all converted fatty acids in CTO were not turned into paraffins. Therefore, a possibility of paraffin formation from fractions other than fatty acids is proposed in CTO hydrotreating. It is known that CTO feed contains a considerable fraction of fatty alcohols (6–8 wt%) along with other fractions; consequently, one route can be suggested as paraffin formation from fatty alcohols via hydrogenation. Ring opening activity of sulfided NiMo catalyst with resin acids as proposed by Palanisamy et al. [38] can also be extend to sterols in CTO. Nonetheless, it is suggested that detailed studies are needed in order to further evaluate the possible routes for the formation of paraffins from cyclic structures in tall oils.
|
|
|
Figure 8. Product yield of paraffins (%) from TOFA, DTO, and CTO as a function of hydrotreating temperature (WHSV = 1 h−1). |
Resin acids and their reaction mechanism assessment
From DTO hydrotreating, it is known that fatty acids did not impose any inhibiting effects on resin acid conversions over a sulfided NiMo catalyst, and vice versa [22]. Figure 3 shows that conversion of resin acids in DTO and CTO follows the same trend as of fatty acids. Therefore, it is proposed that resin acid conversion is also altered by the same effects which are related to fatty acid conversion. Figure 4G–I shows that the conversion of resin acid, dehydroabietic acid, was lower in the case of DTO and CTO hydrotreating at low temperatures (<400°C), which implies that a temperature above 350°C may be needed for achieving a complete conversion of dehydroabietic acid. Earlier, it has been reported by our group that 18-norabietane (C19H34) is formed from resin acids [e.g., abietic acid (C19H29COOH)] via complete hydrogenation and deoxygenation [22]. Coll et al. propose that deoxygenation from resin acids can occur by means of complete hydrogenation which produces intermediate aldehydes and alcohols and finally a hydrocarbon structure with same number of carbon atoms as the parent resin acids [12]. However, the formation of 18-norabietane as a major product from resin acids at low-temperature hydrotreating [22] implies that deoxygenation was mainly occurred through a hydrogenation and decarboxylation/decarbonylation step and not through an intermediate aldehyde–alcohol route as proposed by Coll et al. [12] The formation of a small fraction of abietane from resin acids also implies that hydrogenation/dehydration mechanistic route is still valid from resin acids. Furthermore, it was reported [22] that norabietatrienes and monoaromatic tricylic structures are formed even at 350°C from abietic-type resin acids presumably through a parallel dehydrogenation and decarboxylation reaction route as proposed by Dutta et al. [21] The decarboxylation step involved in this reaction route can be catalytic (in the presence of a HDO catalyst under H2 pressure) or noncatalytic (thermal reaction in the absence of a HDO catalyst). Interestingly, as evidence to noncatalytic route, it has been reported that norabietatrienes are formed during the thermal treatment of abietic acid at 350°C [22, 39]. Therefore, different reaction routes from resin acids are proposed for low-temperature hydrotreating (<400°C) as in the case of fatty acids. Moreover, it may infer that during the hydrotreating of DTO at low temperatures (<400°C), most of the resin acids are consumed through a decarboxylation or decarbonylation as well as through the noncatalytic step (direct decarboxylation + dehydrogenation) without inducing much interaction with the active site responsible for HDO (hydrogenation/dehydration) reaction.
Conclusions
The activity of a commercial alumina supported NiMo catalyst was evaluated for hydrotreating of different tall oil feedstocks. In this study, sulfided NiMo catalyst was found to be active for producing wide-range of products from tall oil feedstocks through various reaction routes existing at different temperatures. At lower temperatures (<400°C), selective deoxygenation routes such as, HDO, decarboxylation, and decarbonylation routes were prominent. At higher temperatures reactions such as cracking (thermal or catalytic) and dehydrogenation were prominent, which produced cycloalkanes and aromatics as main products. Importantly, the trend obtained for the formation of n-octadecane and n-heptadecane in this study especially from TOFA is found to be in well agreement with the behavior of sulfided NiMo catalyst reported in the literature for hydrotreating of vegetable oils. Moreover, fatty acids were found to convert in high extent in TOFA and DTO hydrotreating. In CTO hydrotreating, a decrease in conversion of acid fractions was observed at low temperatures, which is attributed to the complex nature of CTO with significant amount of sulfur compounds in it. Sulfur compounds are proposed to alter the deoxygenation reactions particularly decarboxylation reactions from acid fractions at low temperatures in CTO hydrotreating. Furthermore, it is proposed based on the results in this study that deoxygenation reaction from resin acids at low temperatures mainly occurs through a hydrogenation and decarboxylation/decarbonylation step.
Acknowledgments
Jinto Manjaly Anthonykutty acknowledges the financial support from VTT graduate school. The authors also acknowledge Stora Enso for supporting this research and their vision on wood-based olefins.
Conflict of Interest
None declared.
References
- Furimsky, E. 2000. Catalytic hydrodeoxygenation. Appl. Catal. A: Gen.199:147–190.
- Egeberg, R., N. Michaelsen, L. Skyum, and P. Zeuthen. 2010. Hydrotreating in the production of green diesel. PTQ Q2 Available at www.digitalrefining.com/article/1000156 (accessed 31 May 2014).
- Furimsky, E., and F. E. Massoth. 1999. Deactivation of hydroprocessing catalysts. Catal. Today52:381–495.
- Furimsky, E., and F. E. Massoth. 1993. Regeneration of hydroprocessing catalysts. Catal. Today17:537–659.
- Elliott, D. C.2007. Historical developments in hydroprocessing bio-oils. Energy Fuels21:1792–1815.
- Kubička, D., P. Šimáček, and N. Žilková. 2009. Transformation of vegetable oils into hydrocarbons over mesoporous-alumina-supported CoMo catalysts. Top. Catal.52:161–168.
- Kikhtyanin, O. V., A. E. Rubanov, A. B. Ayupov, and G. V. Echevsky. 2010. Hydroconversion of sunflower oil on Pd/SAPO-31 catalyst. Fuel89:3085–3092.
- Guzman, A., J. E. Torres, L. P. Prada, and M. L. Nunez. 2010. Hydroprocessing of crude palm oil at pilot plant scale. Catal. Today156:38–43.
- Kovács, S., T. Kaszaa, A. Thernesz, I. W. Horváthb, and J. Hancsóka. 2011. Fuel production by hydrotreating of triglycerides on NiMo/Al2O3/F catalyst. Chem. Eng. J.176–177:237–243.
- Abhari, R., and Havlik P. Z. 2009. Hydrodeoxygenation process. US patent publication 2009/0163744.
- Abhari, R., H. L. Tomlinson, and E. G. Roth. 2009. Biorenewable naphtha. US patent publication 2009/0300971) A1.
- Coll, R., S. Udas, and W. A. Jacoby. 2001. Conversion of the rosin acid fraction of crude tall oil into fuels and chemicals. Energy Fuels15:1166–1172.
- Sharma, R. K., and N. N. Bakhshi. 1991. Catalytic conversion of crude tall oil to fuels and chemicals over HZSM-5: effect of co-feeding steam. Fuel Process. Technol.27:113–130.
- Furrer, R. M., and N. N. Bakhshi. 1988. Catalytic conversion of tall oil to chemicals and gasoline range hydrocarbons. Res. Thermochem. Biomass Convers956.
- Kirshner, M. 2005. Chemical profile: tall oil. Chemical Market Reporter: 34.
- Furimsky, E.1998. Selection of catalysts and reactors for hydroprocessing. Appl. Catal.17:177–206.
- Furimsky, E.1983. Chemistry of catalytic hydrodeoxygenation. Catal. Rev-Sci. Eng.25:421–458.
- Topsøe, H., R. G. Egeberg, and K. G. Knudsen. 2004. Future challenges of hydrotreating catalyst technology. Prepr. Pap-Am. Chem. Soc. Div. Fuel Chem.49: 568–569.
- Kubička, D., and L. Kaluza. 2010. Deoxygenation of vegetable oils over sulfided Ni, Mo, and NiMo catalysts. Appl. Catal. A: Gen.372:199–208.
- Şenol, O. I., E. M. Ryymin, T. R. Viljava, and A. O. I. Krause. 2007. Effect of hydrogen sulphide on the hydrodeoxygenation of aromatic and aliphatic oxygenates on sulphided catalysts. J. Mol. Catal. A: Chem.277:107–112.
- Dutta, R. P., and H. H. Schobert. 1993. Hydrogenation/dehydrogenation reactions of rosin. Fundam. Studies Coal Liquefaction38:1140–1146.
- Anthonykutty, J. M., K. M. Van Geem, R. D. Bruycker, J. Linnekoski, A. Laitinen, J. Räsänen, et al. 2013. Value added hydrocarbons from distilled tall oil via hydrotreating over a commercial NiMo catalyst. Ind. Eng. Chem. Res.52:10114–10125.
- Anthonykutty, J. M., J. Linnekoski, A. Harlin, A. Laitinen, and J. Lehtonen. Catalytic upgrading of crude tall oil into a paraffin-rich liquid. Biomass Conv. Bioref. doi: 10.1007/s13399-014-0132-8
- Rozmysłowicz, B., P. Mäki-Arvela, S. Lestari, O. A. Simakova, K. Eränen, I. L. Simakova, et al. 2010. Catalytic deoxygenation of tall oil fatty acids over a palladium-mesoporous carbon catalyst: a new source of biofuels. Top. Catal.53:1274–1277.
- Knuuttila, P., P. Kukkonen, and Ulf Hotanen. 2010. Method and apparatus for preparing fuel components from crude tall oil. US patent publication WO 2010/097519 A2.
- Stigsson, L., and V Naydenov. 2009. Conversion of crude tall oil to renewable feedstock for diesel range fuel compositions. European patent publication WO 2009/131510.
- Diaz, M. A. F., R. A. Markovits, and S. A. Markovits. 2005. Process for refining a raw material comprising black liquor soap, crude tall oil or tall oil pitch. EP1568 760 A1.
- Kubičková, I., and D. Kubička. 2010. Utilization of triglycerides and related feedstocks for production of clean hydrocarbon fuels and petrochemicals: a review. Waste Biomass Valorization1:293–308.
- Mikulec, J., J. Cvengros, L. Jorı′kova, M. Banic, and A. Kleinova. 2010. Second generation diesel fuel from renewable sources. J. Clean. Prod.18:917–926.
- da Rocha Filho, G. N., D. Brodzki, and G. Djéga-Mariadassou. 1993. Formation of alkanes, alkylcycloalkanes and alkylbenzenes during the catalytic hydrocracking of vegetable oils. Fuel72:543–549.
- Wagner, J. L., V. P. Ting, and C. J. Chuck. 2014. Catalytic cracking of sterol-rich yeast lipid. Fuel130:315–323.
- Severson, R. F., and W. H. Schuller. 1972. The thermal behavior of some resin acids at 400–500°C. Can. J. Chem.50:2224.
- Scharmann, H., W. R. Eckert, and A. Zeman. 1969. Elucidation of structure of the methyl esters of cyclic fatty acids V: mass-spectrometry of isomeric methyl esters of phenyl undecanoic acid. Fette Seifen Anstrichmiftel71:118.
- Senol, O. I., T.-R. Viljava, and A. O. I. Krause. 2007. Effect of sulfiding agents on the hydrodeoxygenation of aliphatic esters on sulphided catalysts. Appl. Catal. A: Gen.326:236–244.
- Ryymin, E.-M., M. L. Honkela, T.-R. Viljava, and A. O. I. Krause. 2010. Competitive reactions and mechanisms in the simultaneous HDO of phenol and methyl heptanoate over sulphided NiMo/γ-Al2O3. Appl. Catal. A: Gen.389:114–121.
- Monnier, J., H. Sulimma, A. Dalai, and G. Caravaggio. 2010. Hydrodeoxygenation of oleic acid and canola oil over alumina-supported metal nitrides. Appl. Catal. A382:176–180.
- Snåre, M., I. Kubicˇkova, P. Ma1ki-Arvela, K. Era1nen, and D. Y. Murzin. 2006. Heterogeneous catalytic deoxygenation of stearic acid for production of biodiesel. Ind. Eng. Chem. Res.45:5708–5715.
- Palanisamy, S. 2013. Co-processing fat-rich material into diesel fuel. Doctoral thesis, Chalmers University
- Bernas, A., T. Salmi, Y. D. Murzin, J. Mikkola, and M. Rintola. 2012. Catalytic transformation of abietic acid to hydrocarbons. Top. Catal.55:673–679.
Document information
Published on 01/06/17
Submitted on 01/06/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?