Abstract
The immobilization of redox enzymes on the surface of electrodes is a typical practice for enzymatic fuel cells (EFCs) but enzyme immobilization usually results in low retaining enzymatic activities and low power densities. Here, we investigated an alternative solution – the use of nonimmobilized thermostable enzymes (e.g., NAD-based glucose 6-phosphate dehydrogenase and flavin-containing diaphorase) and a nonimmobilized mediator (i.e., benzyl viologen or 9,10-anthraquinone-2,7-disulphonic acid, AQDS) for achieving high power densities from glucose 6-phosphate in closed EFCs (called biobatteries). At the same enzyme loading, power densities of biobatteries increased in an order from the case of immobilized enzymes and immobilized vitamin K3 to the case of nonimmobilized enzymes and immobilized vitamin K3 to the case of nonimmobilized enzymes and nonimmobilized AQDS. The maximum power density of the biobattery based on nonimmobilized enzymes and AQDS was 1.1 mW cm−2 of anode at room temperature, 34-times of the biobattery based on immobilized enzymes and immobilized vitamin K3. When enzyme loading was increased by another 10-fold, the highest power density of biobattery was increased to 2.4 mW cm−2 at 37°C. The biobattery based on nonimmobilized enzymes and AQDS retained 60% of its initial current density after running for 2 h. When repeatedly being used for 6 rounds, the biobattery had 35% of its initial current density after 12 days. Our results suggest that closed biobatteries equipped with nonimmobilized thermostable enzymes and mediators feature simple system configuration and have high power densities.
Introduction
Enzymatic fuel cells (EFCs) can convert a number of fuels to electricity directly by using enzymes as low-cost catalysts instead of platinum, suitable for powering portable electronics [1]. When gaseous (low-energy in terms of volumetric density) hydrogen is used, fuel cells usually run under a continuous flow-through regime and the catalyst hydrogenase or platinum must be immobilized on the surface of electrode [2]. However, when high-energy density organic compounds (e.g., sugar, methanol) are used in biological fuel cells, these fuel cells can be operated in a “closed” batch or fed-batch mode [3-5].
Enzyme immobilization becomes a common practice in biosensors and most EFCs because it not only increase the stability of mesophilic enzymes but also facilitate electron transfer from immobilized enzymes to electrodes. Also, immobilized enzymes on the surface of electrodes enable easy separation of biocatalysts from the fuel and reuse of the electrode, especially important when fuel cells are run in a flow-through mode. However, enzyme immobilization has several inherent disadvantages, including decreased enzyme activities, poor mass diffusion, electrode fouling due to enzyme leakage, and possibly bad reproducibility due to complicated enzyme immobilization procedures [5-8].
Electron transfer from redox enzymes to electrodes in EFCs is classified by two mechanisms: direct electron transfer (DET) and mediated electron transfer (MET) [9]. In DET, redox enzymes (e.g., glucose oxidase and laccase) must be immobilized on the surface of electrodes to ensure efficient electron transfer [10, 11]. In MET, one or several small molecular weight, redox-active compounds, are introduced to shuttle electrons between the enzyme active site and electron, such as, NADH, vitamin K3 and benzyl viologen (BV) [4, 5, 12]. In order to retain the mediators in flow-through bioelectrochemical systems, costly mediators are usually immobilized on electrodes. Redox mediators can be immobilized through a variety of means, such as adsorption [13-15], polymerization [16-19], entrapment [20, 21], or covalent linking [22]. Another advantage of immobilizing mediators is a decrease in the electron shuttling distance of the mediator to the electrode, resulting in fast electron transfer rates [23]. For example, low-water solubility vitamin K3 (2-methyl-1,4-naphthoquinone) adsorbed on the surface of anode is a widely used mediator for NAD-dependent redox enzymes thanks to its fast kinetics and small thermodynamic loss [5, 12, 24-27]. In contrast, highly water-soluble mediators, such as BV 9,10-anthraquinone-2,7-disulphonic acid (AQDS), can be used as a nonimmobilized mediator in closed fuel cells [3, 4]. Although AQDS has be used as an electron mediator in microbial fuel cells [28, 29] and metal-free flow-through battery [30], it has not been tested in EFCs, to our limited knowledge.
EFC-based biobatteries that the high-density fuel is consumed in a closed system without fuel flow-through emerge as next-generation, green, portable micropower sources [9, 31, 32]. However, biobatteries usually suffer from low power densities that hinder their wide implementation [33]. Breakthroughs of the complete oxidation of fuels in closed biobatteries [4, 5, 34] make it possible to the use of nonimmobilized enzymes and mediators because the fuels are completely oxidized via synthetic enzymatic pathways and the final gaseous product CO2 can be released from the devices. In these closed devices equipped with complete oxidation of fuels catalyzed by enzyme cascades, refilling the fuel could not cause wash-out of nonimmobilized enzymes or mediators.
Increasing power density of biobatteries is one of the most important R&D priorities for the development of practical biobatteries powering numerous portable electronic devices. In this study, we re-investigated the feasibility of the use of nonimmobilized enzymes and mediators in biobatteries for retaining the maximum enzyme activity and increasing mass transfer in systems, featuring drastic increases in power outputs and simple system configuration. To address the stability of the nonimmobilized enzymes, we used home-made thermostable enzymes, instead of commercially available mesophilic enzymes, as well as a stable electron mediator.
Experimental
Reagents
All chemicals including glucose 6-phosphate, vitamin K3 (VK3), BV, AQDS, nicotinamide adenine dinucleotide (NAD+), poly-l-lysine (PLL, MW ~70–150 kDa), and 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDC), and N-hydroxysulfosuccinimide (NHS) were purchased from Sigma-Aldrich (St. Louis, MO). Carbon paper (AvCarb MGL200) as the anode supporter was purchased from Fuel Cell Earth (Stoneham, MA). Membrane electrode assembly was purchased from Fuel Cell Store (San Diego, CA). COOH-functionalized multiwall carbon nanotubes (MWCNTs) with an outer diameter of <8 nm were purchased from CheapTubes.com (Brattleboro, VT).
Preparation of enzymes
Geobacillus stearothermophilus 10 diaphorase (DI), G. stearothermophilus 10 glucose 6-phosphate dehydrogenase (G6PDH) were produced and their activities were measured as described previously [27].
Preparation of bioanodes
Enzymes were immobilized on the carbon paper electrode as previously described, through successively casting of PLL, MWCNTs, EDC, NHS, enzymes, and VK3 [12]. For nonimmobilized-enzyme anodes, the anodes without enzymes casted were submerged in the electrolyte containing the same enzyme loading as used for the immobilized system. For nonimmobilized-enzyme-and-mediators EFCs, the same anodes were used without casted by VK3. Instead, soluble BV or AQDS was used as the mediator.
Electrochemical measurements
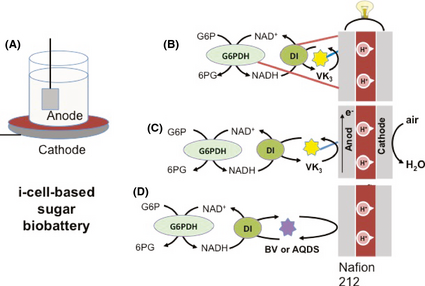
The “i-cell” set-up of EFCs was shown in Figure 1A. Nafion 212 membrane was used to separate an anode and a cathode. An air-breathing carbon cathode was coated with 0.5 mg cm−2 Pt. All electrochemical measurements were conducted using a 1000B Multi-Potentiostat (CH Instruments Inc., Austin, TX) interfaced to a PC. Linear sweep voltammetry was performed at a scan rate of 1 mV sec−1.
|
|
|
Figure 1. Scheme of i-cell configuration biobattery (A), the anode compartment based on two immobilized enzymes (i.e., G6PDH and DI) and the immobilized mediator – VK3 (B), the anode compartment based on two nonimmobilized enzymes and the immobilized mediator – VK3 (C), and the anode compartment based on two nonimmobilized enzymes and the non-immobilized mediator (i.e., benzyl viologen, BV; or 9,10-anthraquinone-2,7-disulfonic acid, AQDS) (D). G6P, glucose 6-phosphate; 6PG, 6-phosphogluconate; NAD+/NADH, nicotinamide adenine dinucleotide; G6PDH, glucose 6-phosphate dehydrogenase; DI, diaphorase. |
To compare immobilized and nonimmobilized enzymes in the EFC, the same amount of enzyme was either immobilized on anodes or soluble in the solution. The electrolyte contained 10 mmol/L G6P, 2 mmol/L NAD+, 100 mmol/L HEPES (pH 7.3), 10 mmol/L MgCl2, 0.5 mmol/L MnCl2, and 100 mmol/L NaCl. Six mL of the electrolyte was stirred at 600 rpm during measurement at the room temperature.
Cyclic voltammetry was performed at different scan rates (1–100 mV sec−1) to compare immobilized VK3 and nonimmobilized BV or AQDS using a 3-electrode system with a Ag/AgCl reference electrode and a Pt wire counter electrode. The anolyte contained 5 mmol/L mediator, 20 mmol/L G6P, 8 mmol/L NAD+, 100 mmol/L HEPES (pH 7.3), 10 mmol/L MgCl2, 0.5 mmol/L MnCl2, and 100 mmol/L NaCl. Twelve units of G6PDH and 16 units of DI were soluble in the 6 mL solution.
Various concentrations (0.2–5 mmol/L) of mediators (VK3, BV or AQDS) were used to study the impact of the mediator concentration on the performance of the EFCs. Other components of the electrolyte were the same as above. Linear sweep voltammetry at the scan rate of 1 mV sec−1 was performed.
Various temperatures (23, 37, or 50°C) were optimized when AQDS was chosen as the optimal mediator in the EFCs, where concentrations of several reaction elements were increased such as 40 mmol/L G6P, 20 U mL−1 G6PDH and 27 U mL−1 DI to achieve high a power density.
To test the long-time performance of the biobatteries, an external load of 150 Ω was applied and the current density was recorded for 2 h. The electrolyte contained 5 mmol/L mediator, 20 mmol/L G6P, 8 mmol/L NAD+, 100 mmol/L HEPES (pH 7.3), 10 mmol/L MgCl2, 0.5 mmol/L MnCl2, 100 mmol/L NaCl, 2 U mL−1 G6PDH, and 2.7 U mL−1 DI. After every 3 days when all the G6P in the batch was consumed, a 20 mmol/L of fresh G6P substrate was supplemented and the current density was recorded for another 2 h to find out it maximum value. This experiment was repeated for 6 rounds and 12 days.
Results and Discussion
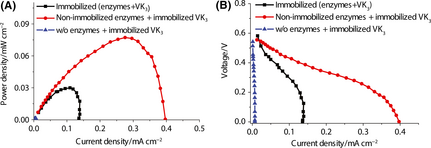
To increase power density of biobatteries, we studied the performance of the glucose 6-phosphate-powered biobatteries equipped with glucose 6-phosphate dehydrogenase (G6PDH) and diaphorase (DI), which represent the most important part of the sugar-oxidation and electron-transfer chain in the complete sugar-oxidation biobattery [5]. The biobattery based on two immobilized enzymes through covalent binding along with immobilized vitamin K3 (VK3) was tested (Fig. 1B) in comparison with the one based on the same amount of non-immobilized enzymes along with immobilized VK3 (Fig. 1C). VK3 was immobilized on the carbon nanotube-casted electrode via simple physical adsorption. The power curves showed that the immobilized-enzyme system presented a maximum power density of 0.032 mW cm−2, approximately a third of the one based on the nonimmobilized enzymes (Fig. 2A). This difference was due to that the covalent binding decreased the apparent activity of immobilized enzymes, resulting in decreased power densities [8]. The mass transport region of the nonimmobilized-enzyme biobattery exhibited a higher current density than that of the immobilized-enzyme one (Fig. 2B). The negative control reaction that the biobattery contained no enzyme generated a power density of 0.0013 mW cm−2 only.
|
|
|
Figure 2. Comparison of the voltage-current (A) and power-current (B) curves of the biobatteries based on two immobilized or non-immobilized enzymes and the immobilized VK3. The electrolyte contained 10 mmol/L G6P, 2 mmol/L NAD+, 100 mmol/L HEPES (pH 7.3), 10 mmol/L MgCl2, 0.5 mmol/L MnCl2, and 100 mmol/L NaCl. Enzymes of 0.4 unit of G6PDH, 16 units of DI were either immobilized on anodes or soluble in the solution. The anode was casted by 10 μL of 300 mmol/L VK3 and the overall reaction volume was 6 mL. |
The use of nonimmobilized enzymes in EFCs were not new [4, 35-38]. However, these studies were often overlooked because of rapid deactivation of nonimmobilized mesophilic enzymes and enzyme wash-out concern. With the discovery of more and more thermostable enzymes from thermophiles and use of engineered enzymes with increased stability, the lifetime of enzymes can be extended greatly even without enzyme immobilization [39]. The advantages of the use of nonimmobilized thermoenzymes in EFCs were the maximum retaining-enzyme activities and great homogeneous mass transfer between nonimmobilized enzymes and soluble substrates. When organic fuels were completely oxidized to CO2 [4, 5, 34], the closed biobatteries enabled to retain nonimmobilized enzymes in the devices without wash-out concerns.
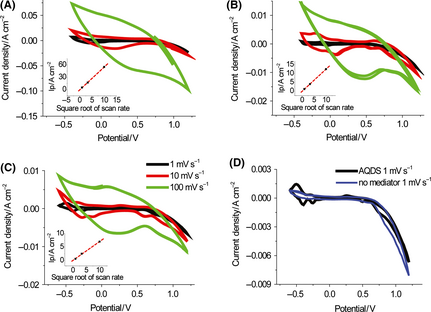
To further increase the power density, we examined whether a nonimmobilized mediator can be used in biobatteries based on nonimmobilized enzymes (Fig. 1D) because adsorbed VK3 was easily desorbed on the surface of the electrode [5]. Two water-soluble mediators –BV and AQDS– were compared to immobilized VK3 for nonimmobilized-enzyme biobatteries. The redox wave could be ascribed to the redox reaction of VK3 (Fig. 3A), BV (Fig. 3B), and AQDS (Fig. 3C) in the anolyte, based on cyclic voltammetry with three different scan rates from 1 to 100 mV sec−1 applied. Based on the average of the peak potentials of the redox couple at the scan rate of 1 mV sec−1, the midpoint potentials were estimated to be −0.10 V, −0.15 V, and −0.25 V versus Ag/AgCl in a 100 mmol/L HEPES buffer (pH 7.3) for VK3, BV, and AQDS respectively. The linear dependence of the peak current on the square root of the scan rate (R2 = 0.99 for all three mediators) as shown in insets of Figure 3A, B, and C indicated that all three mediators regardless of nonimmobilized or immobilized could shuttle the electrons in biobatteries and they were all diffusion-control processes. The immobilized VK3 was actually diffused in the immobilized layer, in accordance with Sonys previous finding [40]. A background scan using the electrolyte in the absence of mediators did not have any redox peak (Fig. 3D).
|
|
|
Figure 3. Comparison of the biobatteries based on the two nonimmobilized enzymes and the immobilized VK3 or nonimmobilized mediators (BV and AQDS): cyclic voltammetry for VK3 (A), BV (B), AQDS (C), and the negative control without mediator (D). The electrolyte included 20 mmol/L G6P, 8 mmol/L NAD+, 100 mmol/L HEPES (pH 7.3), 10 mmol/L MgCl2, 0.5 mmol/L MnCl2, 100 mmol/L NaCl, 2 U mL−1 G6PDH, and 2.7 U mL−1 DI. Scan rates of 1, 10, or 100 mV sec−1 were used. The insets represent the relationship between the peak current density versus the square root of the scan rate. |
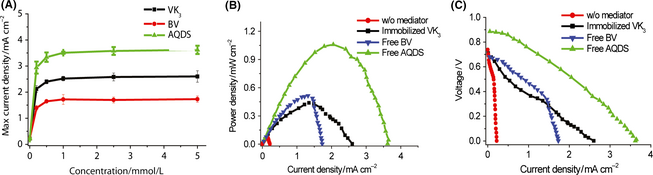
The effect of mediator loading was investigated on maximum current densities obtained from polarization curve scanning (Fig. 4A). Increasing the mediator concentration from 0 to 0.2 mmol/L drastically increased current densities. A further increase of mediator concentration from 0.2 to 1 mmol/L slightly increased in the maximum current densities by 10–20% and then levelled off when the mediator concentrations were more than 1 mmol/L. Nonimmobilized AQDS exhibited the highest maximum current densities, followed by immobilized VK3 and nonimmobilized BV.
|
|
|
Figure 4. The effect of mediator concentration (A), and voltage-current curve (B) and power-current curve (C) at 5 mmol/L mediator loading. The electrolyte included 20 mmol/L G6P, 8 mmol/L NAD+, 100 mmol/L HEPES (pH 7.3), 10 mmol/L MgCl2, 0.5 mmol/L MnCl2, 100 mmol/L NaCl, 2 U mL−1 G6PDH, and 2.7 U mL−1 DI. Linear sweep voltammetry at the scan rate of 1 mV sec−1 was performed. |
Polarization curves were presented at 5 mmol/L of the mediator loading in order to further compare three mediators (Fig. 4B). The AQDS-mediated biobattery showed the highest open circuit potential due to its higher redox potential as compared to BV and VK3. The highest current density of 3.6 mA cm−2 and power density of 1.1 mW cm−2 were obtained from the AQDS-mediated biobattery (Fig. 4C). Both BV- and VK3-mediated biobatteries had a similar maximum power density of ~0.45 mW cm−2 whereas the maximum current density of the VK3-mediated one was nearly 1.7-fold that based on nonimmobilized BV (Fig. 4C). These data were approximately 10-fold higher than the negative control without any mediators. According to the polarization curve (Fig. 4C), the BV-mediated biobattery had a significant mass transport loss in the high current density region, implying that the electron-transfer via BV to anode became rate-limiting at this region. It was found that DI had the specific activity of 3.0 U mg−1 on VK3, 5.2 U mg−1 on AQDS, and 0.5 U mg−1 on BV, indicating that the same amount of DI was the least efficient to transfer electrons base on BV. Therefore, at the high current density region DI activity may not be high enough to transfer electrons from it to BV, resulting in a dramatic mass transport loss according to its polarization curve. As for the other two nonimmobilized mediator-based biobatteries, the dominating voltage loss was Ohmic loss rather than mass transport loss. More details regarding voltage losses will be investigated through a comprehensive analysis on the biobattery resistance and mathematic modeling in the future.
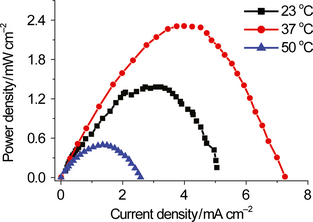
The reaction condition was further optimized to enhance power density of such biobatteries based on nonimmobilized enzymes and mediators. The amount of the substrate and enzymes was increased by 2- or 10-fold and the reaction temperature was further optimized at 23, 37, and 50°C (Fig. 5). At 23°C, the high enzyme-loading biobattery had a power density of 1.4 mW cm−2. At 37°C, the highest power density of 2.4 mW cm−2 was obtained. The power density at 50°C was dramatically decreased to 0.5 mW cm−2, possibly due to the deactivation of the enzymes and the cofactors. This result suggests another threefold increase in the maximum power density compared to our previous result (i.e., 0.8 mW cm−2) [5]. This biobattery were among highest-power-density EFCs reported [41-43].
|
|
|
Figure 5. The power-current curve at different reaction temperatures. The electrolyte included 40 mmol/L G6P, 8 mmol/L NAD+, 5 mmol/L AQDS, 100 mmol/L HEPES (pH 7.3), 10 mmol/L MgCl2, 0.5 mmol/L MnCl2, 100 mmol/L NaCl, 20 U mL−1 G6PDH, and 27 U mL−1 DI. Linear sweep voltammetry at the scan rate of 1 mV sec−1 was performed. |
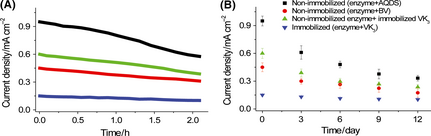
The biobattery equipped with nonimmobilized enzymes and a nonimmobilized mediator AQDS utilized thermoenzymes such as DI and G6PDH to minimize the enzyme deactivation issue because both enzymes had a long lifetime of several weeks at the room temperature [27]. Long time performance based on the current generation was evaluated for up to 12 days or 6 rounds of reusability testing. In all tests, an external load of 150 Ω was applied and the current density was recorded for 2 h. The biobattery based on non-immobilized enzymes and AQDS retained 60% of its initial current density, the one based on VK3 or BV could retain 65–70%, and the one based on immobilized enzymes retained more than 75% (Fig. 6A). When repeatedly being used for six rounds, after 12 days, the biobattery based on AQDS still had 35% of its initial current density, while the one based on immobilized enzymes retained 65% (Fig. 6B). Battery based on nonimmobilized enzymes and mediators produced more power densities that the based on nonimmobilized enzymes and mediators although the latter had better stability.
|
|
|
Figure 6. The current-time curve for the first 2 h (A) and for 12 days with repeatedly adding fresh G6P substrate every 3 days (B). The electrolyte contained 5 mmol/L mediator, 20 mmol/L G6P, 8 mmol/L NAD+, 100 mmol/L HEPES (pH 7.3), 10 mmol/L MgCl2, 0.5 mmol/L MnCl2, 100 mmol/L NaCl, 2 U mL−1 G6PDH, and 2.7 U mL−1 DI. An external load of 150 Ω was applied. |
The use of nonimmobilized enzymes for high-power EFCs is not a new concept but this practice was often overlooked. For example, Whitesides and his coworkers used three nonimmobilized cascade NAD-based redox enzymes in a closed methanol fuel cell achieved a power density of 0.67 mW cm−2 [4]. Fujita et al. demonstrated that a refuelable mediated biofuel cell based on nonimmobilized enzymes that were entrapped in a hierarchical porous carbon electrode exhibited a power density of 1 mW cm−3 through 10 refuelling cycles [44]. Another high-power EFC based on glucose oxidase achieved a power density of 1.30 mW cm−2, where glucose oxidase was entrapped with carbon nanotubes without any chemical covalent linking [43].
Our comparative study between the two nonimmobilized mediators (Fig. 4) suggested that AQDS was better than BV in terms of power outputs. AQDS, which has been used in an organic-inorganic aqueous flow battery [30] and microbial fuel cells [45], is inexpensive, highly soluble, stable, and undergoes extremely rapid two-electron redox reaction. This new biobattery configuration exhibited more than 50 times higher power outputs compared to those based on immobilized VK3, the same amount of immobilized enzymes, and the same electrode material [5, 12, 27]. Our data suggested that the use of nonimmobilized enzymes and mediator could be an alternative solution to achieve high power density and high potential for biobatteries and simplify the configuration of biobatteries.
Conclusions
A biobattery equipped with two nonimmobilized G6PDH and DI as well as nonimmobilized AQDS as a mediator was demonstrated to achieve high power densities as high as 2.4 mW cm−2. Recent advances in the complete oxidation of organic fuels and the development of thermostable enzymes made it possible to use nonimmobilized enzymes and mediators in closed biobatteries. Further advances in thermostable enzymes, high-activity redox enzymes, and high-surface area and high-conductivity 3-D electrodes could lead to high-power, low-cost, and long-lasting biobatteries with simple fuel cell configuration.
Conflict of Interest
None declared.
References
- Bhatnagar, V., and P. Owende. 2015. Energy harvesting for assistive and mobile applications. Energy Sci. Eng.3:153–173.
- Baffert, C., K. Sybirna, P. Ezanno, T. Lautier, V. Hajj, I. Meynial-Salles, et al. 2012. Covalent attachment of FeFe hydrogenases to carbon electrodes for direct electron transfer. Anal. Chem.84:7999–8005.
- Liu, X.H., M. Q. Hao, M. N. Feng, L. Zhang, Y. Zhao, X. W. Du, et al. 2013. A One-compartment direct glucose alkaline fuel cell with methyl viologen as electron mediator. Appl. Energy106:176–183.
- Palmore, G.T.R., H. Bertschy, S. H. Bergens, and G. M. Whitesides. 1998. A methanol/dioxygen biofuel cell that uses NAD+-dependent dehydrogenases as catalysts: application of an electro-enzymatic method to regenerate nicotinamide adenine dinucleotide at low overpotentials. J. Electroanal. Chem.443:155–161.
- Zhu, Z. G., T. K. Tam, F. F. Sun, C. You, and Y. H. P. Zhang. 2014. A high-energy-density sugar biobattery based on a synthetic enzymatic pathway. Nat. Commun.5:3026–3033.
- Gao, F., O. Courjean, and N. Mano. 2009. An improved glucose/O(2) membrane-less biofuel cell through glucose oxidase purification. Biosens. Bioelectron.25:356–361.
- Johnston, W., N. Maynard, B. Y. Liaw, and M. J. Cooney. 2006. In situ measurement of activity and mass transfer effects in enzyme immobilized electrodes. Enzyme Microb. Technol.39:131–140.
- Zhao, X.Y., H. F. Jia, J. Kim, and P. Wang. 2009. Kinetic limitations of a bioelectrochemical electrode using carbon nanotube-attached glucose oxidase for biofuel cells. Biotechnol. Bioengin.104:1068–1074.
- Barton, S. C., J. Gallaway, and P. Atanassov. 2004. Enzymatic biofuel bells for implantable and microscale devices. Chem. Rev.104:4867–4886.
- Liu, Y., M. Wang, F. Zhao, Z. Xu, and S. Dong. 2005. The direct electron transfer of glucose oxidase and glucose biosensor based on carbon nanotubes/chitosan matrix. Biosens. Bioelectron.21:984–988.
- Yu, Y., Z. Chen, S. He, B. Zhang, X. Li, and M. Yao. 2014. Direct electron transfer of glucose oxidase and biosensing for glucose based on PDDA-capped gold nanoparticle modified graphene/multi-walled carbon nanotubes electrode. Biosens. Bioelectron.52:147–152.
- Zhu, Z.G., Y. R. Wang, S. D. Minteer, and Y. H. P. Zhang. 2011. Maltodextrin-powered enzymatic fuel cell through a non-natural enzymatic pathway. J. Power Sources196:7505–7509.
- Malinauskas, A., T. Ruzgas, and L. Gorton. 2000. Electrochemical study of the redox dyes Nile Blue and Toluidine Blue adsorbed on graphite and zirconium phosphate modified graphite. J. Electroanal. Chem.484:55–63.
- Persson, B.1990. A chemically modified graphite electrode for electrocatalytic oxidation of reduced nicotinamide adenine-dinucleotide based on a phenothiazine derivative, 3-beta-naphthoyl-toluidine blue-O. J. Electroanal. Chem.287:61–80.
- Liu, J. L., J. H. Li, and S. J. Dong. 1996. Interaction of brilliant cresyl blue and methylene green with DNA studied by spectrophotometric and voltammetric methods. Electroanalysis8:803–807.
- Bala, W., Z. Lukasiak, M. Rebarz, P. Dalasinski, A. Bratkowski, D. Bauman, et al. 2004. Photoluminescence characterization of vacuum deposited PTCDA thin films. Opto-Electron. Rev.12:445–448.
- Bai, J., X. J. Bo, B. Qi, L. P. Guo, et al. 2010. A novel polycatechol/ordered mesoporous carbon composite film modified electrode and its electrocatalytic application. Electroanalysis22:1750–1756.
- Zhou, D.M., J. J. Sun, H. Y. Chen, and H. Q. Fang. 1998. Electrochemical polymerization of toluidine blue and its application for the amperometric determination of beta-D-glucose. Electrochim. Acta43:1803–1809.
- Moore, C.M., N. L. Akers, A. D. Hill, Z. C. Johnson, and S. D. Minteer. 2004. Improving the environment for immobilized dehydrogenase enzymes by modifying Nafion with tetraalkylammonium bromides. Biomacromolecules5:1241–1247.
- Doaga, R., T. McCormac, and E. Dempsey. 2009. Electrochemical sensing of NADH and glutamate based on meldola blue in 1,2-diaminobenzene and 3,4-ethylenedioxythiophene polymer films. Electroanalysis21:2099–2108.
- Prieto-Simon, B., and E. Fabregas. 2004. Comparative study of electron mediators used in the electrochemical oxidation of NADH. Biosens. Bioelectron.19:1131–1138.
- Ju, H.X., Y. Xiao, X. J. Lu, and H. Y. Chen. 2002. Electrooxidative coupling of a toluidine blue O terminated self-assembled monolayer studied by electrochemistry and surface enhanced Raman spectroscopy. J. Electroanal. Chem.518:123–130.
- Kano, K., and T. Ikeda. 2000. Fundamentals and practices of mediated bioelectrocatalysis. Anal. Sci.16:1013–1021.
- Sato, F., M. Togo, M. K. Islam, T. Matsue, J. Kosuge, N. Fukasaku, et al. 2005. Enzyme-based glucose fuel cell using Vitamin K-3-immobilized polymer as an electron mediator. Electrochem. Comm.7:643–647.
- Togo, M., A. Takamura, T. Asai, H. Kaji, and M. Nishizawa. 2007. An enzyme-based microfluidic biofuel cell using vitamin K-3-mediated glucose oxidation. Electrochim. Acta52:4669–4674.
- Sugiyama, T., Y. Goto, R. Matsumoto, H. Sakai, Y. Tokita, and T. Hatazawa. 2010. A mediator-adapted diaphorase variant for a glucose dehydrogenase-diaphorase biocatalytic system. Biosens. Bioelectron.26:452–457.
- Zhu, Z. G., F. F. Sun, X. Z. Zhang, and Y. H. P. Zhang. 2012. Deep oxidation of glucose in enzymatic fuel cells through a synthetic enzymatic pathway containing a cascade of two thermostable dehydrogenases. Biosens. Bioelectron.36:110–115.
- Sund, C., S. McMasters, S. Crittenden, L. Harrell, and J. Sumner. 2007. Effect of electron mediators on current generation and fermentation in a microbial fuel cell. Appl. Microbiol. Biotechnol.76:561–568.
- Logan, B.E., C. Murano, K. Scott, N. D. Gray, and I. M. Head. 2005. Electricity generation from cysteine in a microbial fuel cell. Water Res.39:942–952.
- Huskinson, B., M. P. Marshak, C. Suh, S. Er, M. R. Gerhardt, C. J. Galvin, et al. 2014. A metal-free organic-inorganic aqueous flow battery. Nature505:195–198.
- Luckarift, H., P. Atanassov, and G. Johnson. 2014. Enzymatic fuel cells: from fundamentals to applications. Wiley, New York, NY.
- Zhang, Y.-H. P.2013. Next generation biorefineries will solve the food, biofuels, and environmental trilemma in the energy-food-water nexus. Energy Sci. Eng.1:27–41.
- Cooney, M.J., V. Svoboda, C. Lau, G. Martin, and S. D. Minteer. 2008. Enzyme catalysed biofuel cells. Energy Environ. Sci.1:320–337.
- Xu, S., and S. D. Minteer. 2012. Enzymatic biofuel cell for oxidation of glucose to CO2. ACS Catal.2:91–94.
- Davis, G., H.A.O. Hill, W. J. Aston, I. J. Higgins, and A. P. F. Turner. 1983. Bioelectrochemical fuel-cell and sensor based on a quinoprotein, Alcohol-Dehydrogenase. Enzyme Microb. Technol.5:383–388.
- Yue, P. L., and K. Lowther. 1986. Enzymatic oxidation of C1 compounds in a biochemical fuel-cell. Chem. Eng. J.33:B69–B77.
- Bourourou, M., K. Elouarzaki, M. Holzinger, C. Agnes, A. Le Goff, N. Reverdy-Bruas, et al. 2014. Freestanding redox buckypaper electrodes from multi-wall carbon nanotubes for bioelectrocatalytic oxygen reduction via mediated electron transfer. Chem. Sci.5:2885–2888.
- Palmore, G. T. R., and H. H. Kim. 1999. Electro-enzymatic reduction of dioxygen to water in the cathode compartment of a biofuel cell. J. Electroanal. Chem.464:110–117.
- Zhang, Y.-H.P. 2015. Production of biofuels and biochemicals by in vitro synthetic biosystems: OPPORTUNITIES and challenges. Biotechnol. Adv. (in press). doi: 10.1016/j.biotechadv.2014.10.009.
- Sakai, H., T. Nakagawa, H. Mita, R. Matsumoto, T. Sugiyama, H. Kumita, et al. 2009. A high-power glucose/oxygen biofuel cell operating under quiescent conditions. ECS Trans.16:9–15.
- Sakai, H., T. Nakagawa, Y. Tokita, T. Hatazawa, T. Ikeda, S. Tsujimura, et al. 2009. A high-power glucose/oxygen biofuel cell operating under quiescent conditions. Energy Environ. Sci.2:133–138.
- Reuillard, B., A. Le Goff, C. Agnes, M. Holzinger, A. Zebda, C. Gondran, et al. 2013. High power enzymatic biofuel cell based on naphthoquinone-mediated oxidation of glucose by glucose oxidase in a carbon nanotube 3D matrix. Phys. Chem. Chem. Phys.15:4892–4896.
- Zebda, A., C. Gondran, A. Le Goff, M. Holzinger, P. Cinquin, and S. Cosnier. 2011. Mediatorless high-power glucose biofuel cells based on compressed carbon nanotube-enzyme electrodes. Nat. Commun.2:370.
- Fujita, S., S. Yamanoi, K. Murata, H. Mita, T. Samukawa, T. Nakagawa, et al. 2014. A repeatedly refuelable mediated biofuel cell based on a hierarchical porous carbon electrode. Sci. Rep.4:4937.
- da Silva, M. E. R., P. I. M. Firmino, and A. B. dos Santos. 2012. Impact of the redox mediator sodium anthraquinone-2,6-disulphonate (AQDS) on the reductive decolourisation of the azo dye reactive red 2 (RR2) in one- and two-stage anaerobic systems. Biores. Technol.121:1–7.
Document information
Published on 01/06/17
Submitted on 01/06/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?