Summary
Objective
To explore the effect of retroperitoneal laparoscopic debridement and drainage on infected necrosis in severe acute pancreatitis.
Materials and methods
This retrospective study included 18 patients with severe acute pancreatitis (SAP) undergoing retroperitoneal laparoscopic debridement and drainage from May 2006 to April 2012 in our hospital. All patients had infected retroperitoneal necrosis and single or multiple peritoneal abscesses. Eleven patients transferred to our hospital were treated with the retroperitoneal laparoscopic debridement and drainage within 24–72 hours after admission. Conservative treatments were given to eight patients. Retroperitoneal laparoscopic debridement and drainage were applied 3–11 days after admission.
Results
All patients had infection of necrotic pancreas or peripancreatic tissues. Twelve patients had organ failure. Three patients underwent secondary surgery. Laparotomy with debridement and drainage were applied to one patient who had a huge lesser sac abscess 7 days after first surgery. The other two patients were given secondary retroperitoneal laparoscopic debridement and drainage. One case was complicated by retroperitoneal hemorrhage, four cases had pancreatic leakage, and no intestinal fistula was found. The patients' heart rate, respiration, temperature, and white blood cell count were significantly improved 48 hours after surgery compared with those prior to surgery (p<0.05). The average length of stay in hospitals was 40.8 days (range, 6–121 days), and the drainage tube indwelling time was 44.4 days (range, 2–182 days).
Conclusion
Retroperitoneal laparoscopic debridement and drainage is an SAP surgical treatment with a minimally invasive procedure and a good effect, and can be applied for infected retroperitoneal necrosis in early SAP.
Keywords
debridement;drainage;laparoscopy;retroperitoneal approach;severe acute pancreatitis
1. Introduction
Severe acute pancreatitis (SAP) is often accompanied by infectious pancreatic necrosis, necrosis and infection of peripancreatic tissue, and pancreatic pseudocyst infection. Most patients have severe systemic inflammatory responses, some of them even accompanied by multiple organ failure (MOF). The mortality rate of SAP is as high as 30% (14–62%).1 Infection of necrotic pancreas or peripancreatic tissues is the main cause of MOF and increased mortality. At present, the consensus of the treatment for infected pancreatic or surrounding tissue necrosis is to apply adequate drainage.2 Because the traditional laparotomy with debridement and drainage has a large traumatic injury and a high mortality rate, the application of minimally invasive techniques is attracting more and more attention.2; 3 ; 4 The minimally invasive techniques being used for infected retroperitoneal necrosis in SAP include percutaneous drainage, peroral transgastric/transduodenal necrosectomy, laparoscopic percutaneous drainage, and hand-assisted laparoscopic debridement and drainage.4; 5; 6; 7 ; 8 As a minimally invasive surgery, retroperitoneal laparoscopic debridement and drainage is rarely reported to be used for infected necrosis of SAP.9 Delayed laparoscopic retroperitoneal debridement and drainage was thought to be carried out only when retroperitoneal liquefaction occurs.10 Here, we retrospectively analyzed 18 patients admitted to our hospital from May 2006 to April 2012, who underwent retroperitoneal laparoscopic debridement and drainage of SAP, to investigate the efficacy and the timing of surgery.
2. Materials and methods
2.1. General information
Eighteen patients (10 males and 8 females, average age 47.3 years, range 32–78 years) were included in this study. All studies were approved by the ethics committee of the First Affiliated Hospital of PLA General Hospital and carried out under the institutional guidelines. Contrast-enhanced abdominal computed tomography (CT) scan indicated infection of necrotic pancreas or peripancreatic tissues, and abscesses in the lesser sac, left anterior pararenal space, retroduodenal space, and paracolic gutters (Table 1). Fifteen patients had family history of biliary stones, one patient had a history of hyperlipidemia, and two patients had drinking history. CT scan showed Grade C, Grade D, or Grade E. CT severity index was 5.4 ± 1.6, Acute Physiology and Chronic Health Evaluation II (APACHE II) score was 13.3 ± 5.4, and Ranson score was 4.6 ± 1.2. Except for infected pancreatic or surrounding tissue necrosis, some patients were complicated with lack of effective circulating blood (9 cases), hypoxemia (6 cases), and acute renal failure (2 cases; Table 2).
| Necrotic sites | No. | % |
|---|---|---|
| Lesser sac | 15 | 83 |
| Left perinephric space | 11 | 61 |
| Right perinephric space | 2 | 11 |
| Left paracolic gutter | 6 | 33 |
| Right paracolic gutter | 2 | 11 |
| Retroduodenal | 2 | 11 |
| Characteristic | Value |
|---|---|
| Age,(y) | 51.3 (30–78) |
| Sex, M/F | 10/8 |
| Etiology | |
| Gallstone | 15 |
| Alcohol | 2 |
| Hypercholesterolemia | 1 |
| CT severity index | 5.4 ± 1.6 |
| Ranson score | 4.6 ± 1.2 |
| APACHE II score | 13.3 ± 5.4 |
| Duration of stay (d) | 40.8 (6–121) |
| Hypovolemia | 9 |
| Hypoxemia | 6 |
| ARF | 2 |
APACHE II = Acute Physiology and Chronic Health Evaluation II; ARF = acute renal failure; CT = computed tomography; d = days; M/F = male/female.
2.2. Surgical approaches
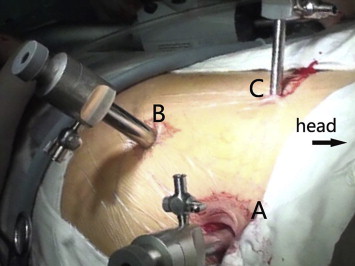
After general anesthesia, the patients were placed in the lateral position (60°) with the affected side up. Right lateral position was used if the abscess was located in the lesser sac, pancreatic tail, left perinephric space, or left paracolic gutter. Left lateral position was used in the cases where the abscess was located in the pancreatic head, retroduodenal space, right perinephric space, or right paracolic gutter. The waist was boosted to allow better exposure. An armpit pad was used, side pads, and front and rear sides of the body were fixed with position frames. The skin was incised about 12 mm on the posterior axillary line and 2 cm under the 12th costal margin. Long curved forceps were used to separate muscles and fascia bluntly. The peritoneum was pushed away to avoid going into peritoneal cavity after injury during finger dissection of the retroperitoneal space. A water capsule filled with 300–400 mL of normal saline for 3–5 minutes was used to expand retroperitoneal space, and form a lacuna. A 12-mm Trocar (Hangzhou Kang Ji medical instrument co., LTD, Hangzhou, China) was placed after removal of normal saline and the water capsule. A 10-mm transverse incision was created 1 cm above the iliac crest in the midaxillary line. Forceps were used to separate muscles and fascia bluntly to the retroperitoneal space. A 10-mm Trocar was placed and inflated until the retroperitoneal space pressure showed 10–14 mmHg. Then, the viewer was used. A 5-mm Trocar was inserted at 2 cm below the anterior axillary line of costal arch under endoscopic direct observation. The perirenal fascia was opened along the front of the psoas muscle from retrocolic space and until the pancreatic tail (Fig. 1). Then, it was opened along the top of the pancreatic tail until the top part of pancreas and the lesser sac was reached. The pus was sucked out and necrotic tissues removed. Attention was paid to avoid injuries to pancreas and splenic arteries and veins during the removal of surrounding necrotic tissues. Two or three drainage tubes were placed in the top part of pancreas and retrocolic space after removal of necrotic tissue and pus. For patients with right retroperitoneal abscess, the perirenal fascia was opened, and the anteromedial dissection was continued until the duodenum was reached. Then, two or three drainage tubes were placed in retroduodenal space, anterior extrarenal space, and retrocolic space.
|
|
|
Figure 1. Trocar sites. Trocar A was located in the posterior axillary line 2 cm below the 12th rib cartilage margin. Trocar B was located in the midaxillary line 1 cm above the iliac crest. Trocar C was located in the anterior axillary line 2 cm below the costal margin. |
2.3. Timing of surgery
All patients underwent a preoperative CT scan, and infection of necrotic pancreas or peripancreatic tissues was confirmed. Infection or effusion was found in the lesser sac, the perinephric space, the left and right retrocolic space, or the retroduodenal space. Effusions in the lesser sac and left perinephric space were most common (Table 1). Retroperitoneal laparoscopic debridement and drainage was applied to all patients at 1–11 days after admission. Eleven patients were treated in other hospitals for 4–30 days and transferred to our hospital after their conditions deteriorated and organ failure was found. The peritoneal laparoscopic debridement and drainage was applied within 24–72 hours after admission to hospital. Eight patients were admitted to hospital 8–48 hours after the onset of upper abdomen or left upper abdominal pain and were given conservative treatments after admission. However, their conditions either did not alleviate or continued to deteriorate. Persistent abdominal pain, fever, or even organ failure was found. Retroperitoneal laparoscopic debridement and drainage was applied at 3–11 days after admission.
2.4. Statistical analysis
Data are expressed as mean ± standard error of the mean. Data analysis was conducted with SPSS 13.0 software (SPSS Inc., Chicago, IL, USA). Student t test was used for statistic analysis, and p<0.05 was considered statistically significant.
3. Results
All patients had infection of necrotic pancreas or peripancreatic tissues. No intestinal fistula was found. The patients' heart rate, respiration, temperature, and white blood cell count were significantly improved at 48 hours after surgery compared with those prior to surgery (Table 3; p<0.05). The average length of stay in hospital was 42.8 days (range, 17–121 days), and the drainage tube indwelling time was 46.9 days (range, 6–182 days).
| Time of measurement | Heart rate (beats/min) | Breathing rate (breaths/min) | Temperature (°C) | WBC (×109/L) |
|---|---|---|---|---|
| Prior to operation | 125.9 ± 7.6 | 29.7 ± 3.9 | 38.7 ± 0.9 | 19.3 ± 6.7 |
| 48 h after operation | 108.3 ± 7.8* | 21.0 ± 1.9* | 38.2 ± 0.9* | 13.7 ± 2.6* |
Data are presented as mean ± standard error of the mean.
- When compared with rate prior to surgery, p<0.05.
Three patients out of 18 underwent secondary surgery. Among those three patients, one had a fever of 39–40°C after surgery. Abdominal CT scan confirmed that this patient had a huge lesser sac indicating inadequate drainage, so laparotomy with debridement and drainage were conducted on the seventh postoperative day. Secondary retroperitoneal laparoscopic debridement and drainage were applied to the other two patients. One case was complicated by retroperitoneal hemorrhage and was improved after conservative hemostatic treatment. Four patients had pancreatic leakage, and 150–280 mL of pancreatic juice was drained daily for those four patients. Their conditions were gradually improved after a longer period (56–182 days).
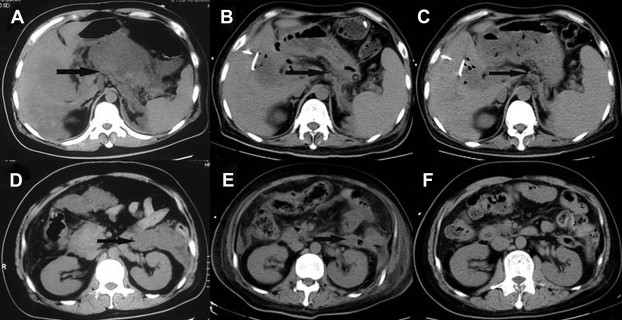
Two patients complicated by acute renal failure were hemodialyzed for 24–48 hours. Three patients showed acute respiratory distress syndrome (ARDS) and noninvasive mechanical ventilation was given to them. Two patients' conditions were improved after noninvasive mechanical ventilation was given. Laparoscopic debridement of necrotic tissue and drainage was conducted immediately, and endotracheal tube was used in the Intensive Care Unit (ICU) for a 78-year-old obese patient (body mass index = 36) with hypercholesterolemia and a history of hypertension and chronic heart failure, admitted to hospital because of 72 hours of left upper abdominal pain and high body temperature (39.8°C). His condition deteriorated sharply 3 days after admission, and ARDS was found. This patient died 2 days after surgery due to acute heart failure and ARDS. The other patients were cured or discharged with drainage tube (Fig. 2).
|
|
|
Figure 2. Laparoscopic retroperitoneal debridement and gallbladder puncture and drainage treatment for a 37-year-old male patient. (A) Lesser sac abscess prior to surgery. (B) Lesser sac abscess was significantly improved 20 days after surgery. (C) Lesser sac had no significant effusion 160 days after surgery. (D) Perinephric abscess prior to surgery. (E) Perinephric abscess was markedly improved 20 days after surgery. (F) No perirenal effusion was found 160 days after surgery. Arrow shows position of retroperitoneal infection. |
4. Discussion
Most surgeons consider that the best timing for SAP surgery should be 2–4 weeks after disease onset because they believe the mortality of surgery is high in early SAP.11; 12 ; 13 They also think that, as time goes on, the boundaries between necrotic tissue and normal pancreatic tissues become more clear and this makes debridement much easier. The study by Radenkovic et al showed that the operative mortality of SAP patients was 34%.14 It has been shown that abnormal immune regulation is very important in the development of SAP and further promotes inflammatory response.15; 16; 17 ; 18 The multicenter national survey by Takeda et al showed that the mortality of acute necrotizing pancreatitis was 20.8%, and mortality of acute necrotizing pancreatitis associated with infection was 33.3%.19 No difference was found between the mortality of early surgery and delayed surgery. Acute pancreatitis clinical guidelines indicate that the mortality rate of SAP with aseptic necrosis was 12% (2–44%), and mortality rate of SAP with infected necrosis was as high as 30% (14–62%).1 This shows that the seriousness of the SAP is not a result of pancreatic necrosis, but dictated by the infection caused by systemic inflammatory response syndrome and MOF.20; 21 ; 22 It is very important to do early drainage with a smaller invasion, and alleviate the patients inflammatory response as soon as possible in the treatment of SAP.
SAP was previously treated by routine abdominal debridement and drainage,23 ; 24 open packing and secondary suture,10 ; 25 and closed surgical drainage or lesser omental sac lavage.26 ; 27 All these methods have shortcomings such as large trauma, long recovery time, and high death rate. Minimally invasive techniques have been emphasized more in recent years. Parekh et al performed retroperitoneal debridement and drainage of necrotic foci with hand-assisted laparoscopy.4 Brokelman et al found that laparoscopic surgery will destroy the integrity of the peritoneum, and weaken immunological and other biological characteristics as open surgery does.28 Peng et al showed that, compared with traditional open surgery, laparoscopic retroperitoneal resection may reduce the perioperative immune response.29 Retroperitoneal laparoscopic debridement and drainage of infected necrotic tissue has smaller trauma to the peritoneum and keeps peritoneal immune function, so it could minimize injury to the patients immune system. It can remove the peripancreatic and retroperitoneal infected necrotic tissues at the same time, and therefore reduce systemic inflammatory response.
In this study, 11 patients were treated in other hospitals for 4–30 days. Infected necrosis in pancreas or pancreatic surrounding tissue, retroperitoneal foci, and multiple abscesses were found after they were admitted to our hospital. Retroperitoneal laparoscopic debridement and drainage was performed at 24–72 hours after admission. Eight patients showed abdominal pain and were admitted to hospital at 8–48 hours later and peritoneal infection were confirmed by CT scan. Laparoscopic debridement drainage was applied at 3–11 days after admission. One 78-year-old obese patient with hypercholesterolemia and a history of chronic heart failure died 2 days after surgery due to acute heart failure and ARDS. In this study, three patients underwent secondary surgery. One patient was complicated by retroperitoneal hemorrhage and was improved after conservative hemostatic treatment. Four cases had pancreatic leakage and their conditions gradually improved after a longer period of drainage. The patients' heart rate, respiration, temperature, and white blood cell count were significantly improved at 48 hours after surgery when compared with those prior to surgery, indicating that retroperitoneal laparoscopic debridement and drainage for SAP peritoneal infection was safe and effective. Although early peritoneal laparoscopic debridement and drainage may result in incomplete removal of necrotic pancreas, it has been reported that complete removal is not necessary as long as enough drainage is provided.3 All the patients in this study had surgery within 11 days after admission and the surgeries were safe and effective. This indicates that retroperitoneal laparoscopic debridement and drainage might suitable for treatment of early-infected retroperitoneal necrosis in severe acute pancreatitis and prevention of secondary organ dysfunction.
Although many people think that delayed surgery for infected retroperitoneal necrosis of SAP may be less risky, it might be a better choice if minimally invasive surgical debridement and drainage is applied prior to organ failure to reduce the systemic inflammatory response. Based on this study, we conclude that retroperitoneal laparoscopic debridement and drainage is a good surgical approach for infected retroperitoneal necrosis of SAP. Due to the small sample number in this study, the advantage of this surgical approach needs to be further confirmed.
References
- 1 P.A. Banks, M.L. Freeman; Practice guidelines in acute pancreatitis; Am J Gastroenterol, 101 (2006), pp. 2379–2400
- 2 E.L. Bradley, N.D. Dexter; Management of severe acute pancreatitis: a surgical odyssey; Ann Surg, 251 (2010), pp. 6–17
- 3 Y.C. Chang, H.M. Tsai, X.Z. Lin, C.H. Chang, J.P. Chuang; No debridement is necessary for symptomatic or infected acute necrotizing pancreatitis: delayed, mini-retroperitoneal drainage for acute necrotizing pancreatitis without debridement and irrigation; Dig Dis Sci, 51 (2006), pp. 1388–1395
- 4 D. Parekh; Laparoscopic-assisted pancreatic necrosectomy: a new surgical option for treatment of severe necrotizing pancreatitis; Arch Surg, 141 (2006), pp. 895–903
- 5 R.E. Brolin, L. Flancbaum, F.R. Ercoli, et al.; Limitations of percutaneous catheter drainage of abdominal abscesses; SurgGynecol Obstet, 173 (1991), pp. 203–210
- 6 K.D. Horvath, L.S. Kao, A. Ali, K.L. Wherry, C.A. Pellegrini, M.N. Sinanan; Laparoscopic assisted percutaneous drainage of infected pancreatic necrosis; SurgEndosc, 15 (2001), pp. 677–682
- 7 J. Escourrou, H. Shehab, L. Buscail, et al.; Peroral transgastric/transduodenal necrosectomy: success in the treatment of infected pancreatic necrosis; Ann Surg, 248 (2008), pp. 1074–1080
- 8 B.I. Babu, A.K. Siriwardena; Current status of minimally invasive necrosectomy for post-inflammatory pancreatic necrosis; HPB (Oxford), 11 (2009), pp. 96–102
- 9 C. Tang, B. Wang, B. Xie, H. Liu, P. Chen; Treatment of severe acute pancreatitis through retroperitoneal laparoscopic drainage; Front Med, 5 (2011), pp. 302–305
- 10 U. Navaneethan, S.S. Vege, S.T. Chari, T.H. Baron; Minimally invasive techniques in pancreatic necrosis; Pancreas, 38 (2009), pp. 867–875
- 11 E.L. Bradley, J.T. Fulenwider; Open treatment of pancreatic abscess; Surg Gynecol Obstet, 159 (1984), pp. 509–513
- 12 R. Mofidi, A.C. Lee, K.K. Madhavan, O.J. Garden, R.W. Parks; Prognostic factors in patients undergoing surgery for severe necrotizing pancreatitis; World J Surg, 31 (2007), pp. 2002–2007
- 13 G.H. Sakorafas, C. Lappas, A. Mastoraki, S.G. Delis, M. Safioleas; Current trends in the management of infected necrotizing pancreatitis; Infect Disord Drug Targets, 10 (2010), pp. 9–14
- 14 D.V. Radenkovic, D.D. Bajec, G.G. Tsiotos, et al.; Planned staged reoperative necrosectomy using an abdominal zipper in the treatment of necrotizing pancreatitis; Surg Today, 35 (2005), pp. 833–840
- 15 M.W. Wichmann, T.P. Hüttl, H. Winter, et al.; Immunological effects of laparoscopic vs open colorectal surgery: a prospective clinical study; Arch Surg, 140 (2005), pp. 692–697
- 16 Z. Liu, Y. Shen, N. Cui, J. Yang; Clinical observation of immunity for severe acute pancreatitis; Inflammation, 34 (2011), pp. 426–431
- 17 A. Nishio, M. Asada, K. Uchida, T. Fukui, T. Chiba, K. Okazaki; The role of innate immunity in the pathogenesis of experimental autoimmune pancreatitis in mice; Pancreas, 40 (2011), pp. 95–102
- 18 V. Mylona, V. Koussoulas, D. Tzivras, et al.; Changes in adaptive and innate immunity in patients with acute pancreatitis and systemic inflammatory response syndrome; Pancreatology, 11 (2011), pp. 475–481
- 19 K. Takeda, S. Matsuno, M. Sunamura, M. Kobari; Surgical aspects and management of acute necrotizing pancreatitis: recent results of a cooperative national survey in Japan; Pancreas, 16 (1998), pp. 316–322
- 20 M.G. Raraty, S. Connor, D.N. Criddle, R. Sutton, J.P. Neoptolemos; Acute pancreatitis and organ failure: pathophysiology, natural history, and management strategies; Curr Gastroenterol Rep, 6 (2004), pp. 99–103
- 21 C.J. McKay, A. Buter; Natural history of organ failure in acute pancreatitis; Pancreatology, 3 (2003), pp. 111–114
- 22 M.S. Petrov, S. Shanbhag, M. Chakraborty, A.R. Phillips, J.A. Windsor; Organ failure and infection of pancreatic necrosis as determinants of mortality in patients with acute pancreatitis; Gastroenterology, 139 (2010), pp. 813–820
- 23 E.D. Davidson, E.L. Bradley 3rd; Marsupialization" in the treatment of pancreatic abscess; Surgery, 89 (1981), pp. 252–256
- 24 A. Lumsden, E.L. Bradley 3rd; Secondary pancreatic infections; Surg Gynecol Obstet, 170 (1990), pp. 459–467
- 25 H.S. Ho, C.H. Frey; The role of antibiotic prophylaxis in severe acute pancreatitis; Arch Surg, 132 (1997), pp. 487–492
- 26 B. Rau, A. Bothe, H.G. Beger; Surgical treatment of necrotizing pancreatitis by necrosectomy and closed lavage: changing patient characteristics and outcome in a 19-year, single-center series; Surgery, 138 (2005), pp. 28–39
- 27 C. Fernández-del Castillo, D.W. Rattner, M.A. Makary, A. Mostafavi, D. McGrath, A.L. Warshaw; Débridement and closed packing for the treatment of necrotizing pancreatitis; Ann Surg, 228 (1998), pp. 676–684
- 28 W.J. Brokelman, M. Lensvelt, R. Borel, J.H. Klinkenbijl, M.M. Reijnen; Peritoneal changes due to laparoscopic surgery; Surg Endosc, 25 (2011), pp. 1–9
- 29 B. Peng, J.H. Zheng, H. Li; Effect of retroperitoneal laparoscopic radical nephrectomy of renal carcinoma (nephroma) on perioperative cell immunity; J Endourol, 22 (2008), pp. 2161–2164
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?