Summary
Gas-forming pyogenic liver abscess (GFPLA) is considered to be a very severe form of PLA and carries a high mortality. Klebsiella pneumoniae is the most common pathogen responsible for the disease, whereas cases where Salmonella is cited as the cause are very uncommon. We report the first case of a 53-year-old lady suffering from GFPLA due to Salmonella, who was successfully treated with surgical drainage. To the best of our knowledge, this is the first case of GFPLA caused by Salmonella enteritidis to be reported in the English literature.
Keywords
acute abdomen;free gas;gas forming;liver abscess;Salmonella enteritidis
1. Introduction
Pyogenic liver abscess (PLA) is the most common form of liver abscess. Its incidence varies from 1.1 per 100,000 individuals in Europe to 17.6 per 100,000 in Asia. A rising prevalence rate in liver abscess is also reported.1 The incidence of gas-forming PLAs (GFPLAs) accounts for 10–20% of all PLAs.
Common presentations of PLA include abdominal pain, fever, chills, nausea and vomiting, and a general feeling of illness. However, the classical triad of abdominal pain, fever, and chills may only account for a third of patients.1 Overall, 32.5% of GFPLA patients may present with shock, compared to 11.7% in the nongas-forming group.2
Because the mortality of patients suffering from GFPLA can be as high as 27.7% as compared with the 14.4% mortality in the nongas-forming group,2 prompt recognition of the condition upon presentation is crucial. Any delay in treatment may result in hospital mortality.
2. Case report
A 53-year-old woman, who works as a waitress in a restaurant, was admitted through the emergency department for fever and epigastric pain radiating to the back. Apart from osteoarthritis of the knees, which was relieved by simple analgesia, she has enjoyed good health in the past.
An examination on admission revealed epigastric tenderness, dyspnea, and decreased breath sound on the right. Her white blood cell count was 23 × 109/L on admission, and the serum bilirubin level was 45 μmol/L. Other laboratory results were as follows: serum alkaline phosphatase level, 132 U/L; serum aspartate aminotransferase level, 328 U/L; serum alanine aminotransferase, 376 U/L; international normalized ratio, 1.8; serum glucose level, 1.7 mmol/L.
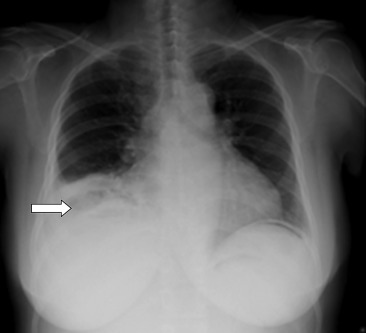
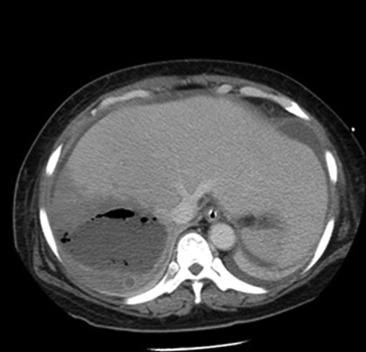
Radiography of the chest was performed (Fig. 1). The chest X-ray showed mild haziness over the right lower zone of the lung. In addition, there was a lucency occupying the right subphrenic area. In view of the patients clinical condition and abnormal chest X-ray result, an urgent computed tomography (CT) scan of the abdomen with contrast was performed that showed a 16.5-cm gas-forming liver abscess involving segments 6–8 (Fig. 2).
|
|
|
Figure 1. Erect chest X-ray on admission: lucency below the right diaphragm. |
|
|
|
Figure 2. Contrast computed tomography: faintly rim-enhancing gas-forming abscess in the right lobe of the liver. |
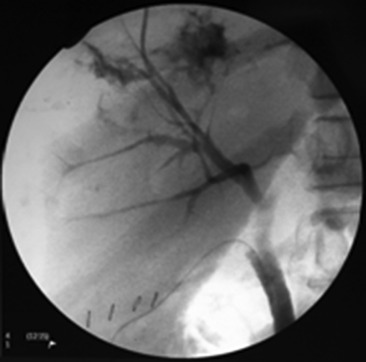
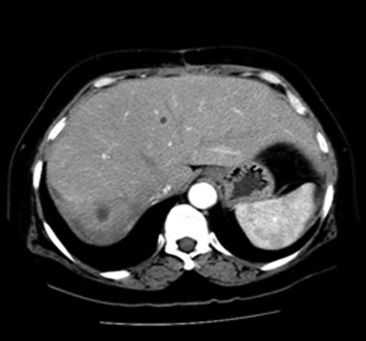
CT-guided drainage of the liver abscess was performed, but only a minimal amount of pus was obtained. The patient continued to run a high fever with hypotension. A decision was made to perform laparotomy and open drainage of the liver abscess. During the laparotomy, a large amount of turbid fluid was noted in the peritoneal cavity. The liver was grossly edematous and the large abscess was seen at the dome of the liver with pus and gas discharge. Open drainage was then performed. Operative cholangiogram showed contrast leakage into the abscess cavity, indicating the communication of the bile duct to the abscess. The common bile duct was explored, and a T-tube was placed to facilitate biliary drainage in a controlled manner (Fig. 3). Abdominal drains were placed in the abscess cavity and the subhepatic area. The patient was supported in the intensive care unit after the operation. Both abscess tissue and pus only yielded Salmonella enteritidis. The bacteria were sensitive to piperacillin. A histology section of the abscess did not yield any evidence of malignancy. The abdominal drains were removed 3 weeks after the operation. The patient was given 3 weeks of intravenous piperacillin and 3 weeks of oral amoxicillin/cavulanate, and was discharged home 49 days after the procedure. The T tube was removed 6 weeks after the operation. A reassessment contrast CT scan showed resolution of the liver abscess at 3 months after the operation ( Fig. 4).
|
|
|
Figure 3. Operative cholangiogram: contrast extravasation via right anterior bile duct into the abscess. |
|
|
|
Figure 4. Computed tomography scan after 3 months showed regression of the liver abscess without gas content. |
3. Discussion
GFPLA is a rare condition that carries a high mortality. The usual cause of PLA is mainly due to portal seeding from appendicitis and diverticulitis. With the improvement of antibiotics, biliary tract diseases have become the most common identifiable cause, although most of them are cryptogenic in origin.3 Patients with compromised immunology may be more often affected. Diabetes mellitus is not regarded as a cause of PLA; however, a high prevalence of the disease is noted among patients suffering from PLA in Asia. Reports suggested that about 43% of patients suffering from PLA are known to have diabetes.1 ; 4 An even higher proportion of diabetic patients are noted in gas-forming entities associated with higher mortality rates.1; 2 ; 4
In the management of this patient, a prompt decision to operate was made. Apart from drainage of the liver abscess, cholecystectomy was performed as cholecystitis was the usual source for liver abscess formation. Operative cholangiogram was performed in the same operation to identify the presence of a possible biliary stone or sludge that could have led to liver abscess formation. Because the liver abscess contained a large portion of solid debris, open drainage and debridement of the abscess had been performed. The displaced intrahepatic duct was unavoidably broken down together with the abscess. Because the operative cholangiogram had successfully identified the leakage, exploration of the common bile duct and T-tube placement for external drainage was performed. The T-tube placement aimed to ensure adequate drainage of the bile and enhanced the healing of the biliary system. Alternatively, Endoscopic retrograde cholangiopancreatography (ERCP) could be performed after the operation if persistent leakage of the bile is noted in the drainage tube. Because the patient was in shock during the operation, the surgeon decided to advocate a more definitive approach for biliary drainage. In our institute, T tubes are usually removed 6 weeks after the operation in order to allow a complete maturation of the T-tube track. (We had an unpleasant experience of leakage from the tract when the T tube was removed prematurely.)
The usual organisms found in the liver abscess were Gram-negative bacterium. Mezhir et al3 reported that multiple microorganisms were usually cultured in Western countries, and the most common species include Streptococcus, Enterococcus, and Escherichia coli. By contrast, studies in Asia showed that Klebsiella pneumoniae were the most predominant bacteria (63–79%), followed by Escherichia coli. Single microorganisms were responsible in the majority of cases in Asia. 1; 2; 5 ; 6 In the case of GFPLAs, K. pneumoniae is the most frequently cultured organism, accounting for 87% of cases. 2Salmonella is a rare pathogen for PLAs. Since 2000, only two reports of Salmonella-related PLA have appeared in the English literature. Both reports found that patients with immunocompromised status were at risk for this condition. 7 ; 8 This study represents the first report of a GFPLA due to Salmonella species without an underlying pathology.
GFPLA is a condition where vigorous metabolism and growth of bacteria are seen. Bacteria obtain their energy through fermentation of glucose, which leads to an accumulation of acids and introduction of an acidic environment. Salmonella, a gas-forming microorganism, will produce formic hydrogen lyase, which converts formic acid to CO2 and H2. The accumulation of gas involves the increased production of gas, impaired transportation of gas, and equilibrium between the gas in the local tissue and that in the abscess.9Salmonella-related GFPLA is rare. Our case is the first report since Lee et al 7 had reported a spontaneous GFPLA by the bacteria within a hepatocellular carcinoma. The gas-forming ability of Salmonella is also reported in other organs. 10
Empirical antibiotics are mandatory for the treatment of PLA. Systemic antibiotics alone have been shown to be effective for small-sized abscesses (<3 cm) in well-selected patients.11 Drainage of the abscess, either percutaneous or operative, facilitates recovery in larger size abscesses. There is no consensus on the mode of drainage with regard to the characteristics of the abscess. Hope et al11 reported that 83% of uniloculated abscesses larger than 3 cm can be drained percutaneously, whereas only 33% of the complex multilocular abscess can be drained percutaneously. Tan et al6 also suggested that surgical drainage for abscesses larger than 5 cm provides better clinical outcome in terms of reduced need for reintervention and shorter hospital stays, with comparable morbidity and mortality rates. However, others reported conflicting results.3 In general, percutaneous drainage is regarded as the priority choice in view of the improvements in imaging and avoidance of general anesthesia. Surgical drainage is reserved for those who had been unresponsive to medical treatment and for whom the percutaneous option failed, and those who had ruptured abscess with peritonitis, anatomical difficulty in performing percutaneous drainage, or primary intra-abdominal pathology.1; 2 ; 5 Nonetheless, if biliary communication is evident, concomitant biliary drainage should be considered.
The prognosis of the PLA can be predicted by the severity of the disease at presentation. A retrospective study had reviewed 298 patients suffering from PLA. A multivariate analysis identified GFPLA, anaerobic infection, Acute Physiology and Chronic Health Evaluation II (APACHE) II score, and Simplified Acute Physiology II (SAP II) score as independent variables and to be statistically significantly affecting the mortality rate of PLA. In patients affected by PLA, an APACHE II score of more than 14 or a SAP II score of more than 27 resulted in a markedly increased risk of mortality, whereas an APACHE II score lower than 6 or a SAP II score below 22 indicated a zero mortality rate.5 Our patient is classified as a high-risk patient because she has an APACHE II score of 6 and a SAP II score of 31.
Immediate diagnosis of the condition, decisive surgical management, and dedicated intensive care during the postoperative period are the key to surviving this critical disease.
References
- 1 L.T. Tian, K. Yao, X.Y. Zhang, et al.; Liver abscesses in adult patients with and without diabetes mellitus: an analysis of clinical characteristics, features of the causative pathogens, outcomes and predictors of fatality: a report based on a large population, retrospective study in China; Clin Microbiol Infect, 18 (2012), pp. E314–E330
- 2 F.F. Chou, S.M. Sheen-Chen, Y.S. Chen, T.Y. Lee; The comparison of clinical course and results of treatment between gas-forming and non-gas-forming pyogenic liver abscess; Arch Surg, 130 (1995), pp. 401–405
- 3 J.J. Mezhir, Y. Fong, L.M. Jacks, et al.; Current management of pyogenic liver abscess: surgery is now second-line treatment; J Am Coll Surg, 210 (2010), pp. 975–983
- 4 C.C. Yang, C.Y. Chen, X.Z. Lin, et al.; Pyogenic liver abscess in Taiwan: emphasis on gas-forming liver abscess in diabetes; Am J Gastroenterol, 88 (1993), pp. 1911–1915
- 5 S.C. Chen, C.C. Huang, S.J. Tsai, et al.; Severity of disease as main predictor for mortality in patients with pyogenic liver abscess; Am J Surg, 198 (2009), pp. 164–172
- 6 Y.M. Tan, A.Y.F. Chung, P.K.H. Chow, et al.; An appraisal of surgical and percutaneous drainage for pyogenic liver abscesses larger than 5 cm; Ann Surg, 241 (2005), pp. 485–490
- 7 C.C. Lee, S.K. Poon, G.H. Chen; Spontaneous gas-forming liver abscess caused by Salmonella within hepatocellular carcinoma: a case report and review of the literature; Dig Dis Sci, 47 (2002), pp. 586–589
- 8 Y.P. Chou, C.S. Changchien, K.W. Chiu, et al.; Salmonellosis with liver abscess mimicking hepatocellular carcinoma in a diabetic and cirrhosis patient: a case report and review of the literature; Liver Int, 26 (2006), pp. 498–501
- 9 H.L. Lee, H.C. Lee, H.R. Guo, et al.; Clinical significance and mechanism of gas formation of pyogenic liver abscess due to Klebsiella pneumoniae; J Clin Microbiol, 42 (2004), pp. 2783–2785
- 10 P.H. Hung, Y.L. Chiu, P.R. Hsueh; Gas-forming splenic abscess due to Salmonella enterica serotype enteritidis in a chronically hemodialyzed patient; J Microbiol Immunol Infect, 40 (2007), pp. 276–278
- 11 W.W. Hope, D.V. Vrochides, W.L. Newcombe, et al.; Optimal treatment of hepatic abscess; Am Surg, 74 (2008), pp. 178–182
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?