Dear Editor,
Entecavir is a nucleoside analog, which is used for the treatment of chronic hepatitis B. It is a potent and highly selective inhibitor of DNA polymerase, with a high antiviral efficacy and high genetic barrier to viral resistance.1 and 2 Several common side effects of entecavir such as headache, stomachache, and upper respiratory tract infection have been reported, but cutaneous side effects are extremely rare and have been reported in only 3 cases in the English literature.3, 4 and 5 Here we report a very unusual case of drug eruption caused by entecavir.
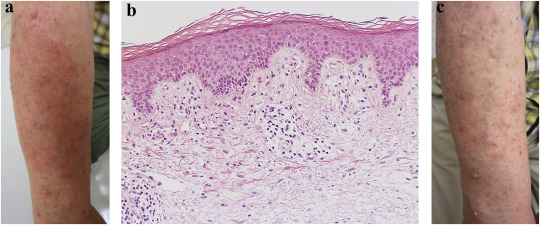
A 65-year-old man was referred to us for consultation regarding a bilateral eruption on his arms. He had a history of chronic renal failure spanning more than 40 years. He had undergone a renal transplantation at the age of 24 years, and received treatment with azathioprine (100 mg daily), prednisolone (5 mg daily), and nifedipine. He had developed a hepatitis B virus (HBV) infection and had been taking 0.5 mg entecavir daily. Six months after taking entecavir, he developed bilateral erythematous plaques on his arms (Fig. 1a). Multiple papillomatous plaques on the arms were verruca vulgaris often seen in the patients after organ transplantation. Laboratory examination revealed increased white blood cell count, 8600/μL (normal range, 3400–7300/μL) without eosinophilia; elevated cholinesterase level, 531 U/L (normal range, 206–476 U/L); and decreased estimated glomerular filtration rate, 45.86 mL/min/1.73 m2 (normal range, >90 mL/min/1.73 m2). Serological test results for the HBV surface antigen and its antibodies were both positive. Histological examination of a punch biopsy specimen from the left forearm revealed superficial perivascular lymphocytic infiltrates with mild vacuolar alteration, which was consistent with a drug eruption (Fig. 1b). Initially, we suspected drug-induced photosensitivity due to nifedipine. However, lesions did not improve even after discontinuation of nifedipine. Minimal erythema doses for UVA and UVB were within normal limits. A drug lymphocyte stimulation test (DLST) results with nifedipine was negative. Based on these results, we suspected that the drug eruption was due to entecavir. DLST with entecavir was performed and the result was positive with a stimulation index of 4.0. Based on the clinical course and the result of DLST, we diagnosed drug eruption caused by entecavir. As the eruption was not severe and entecavir seemed to be effective against HBV, entecavir was not discontinued but reduced to 0.5 mg every second day, and topical corticosteroid treatment was initiated. The eruption gradually improved but did not completely disappear (Fig. 1c).
|
|
|
Fig. 1.. (a) Clinical findings. Pruritic erythematous plaques on the right arm. (b) Histopathological findings of erythematous plaque, which show superficial perivascular lymphocytic infiltration with mild vacuolar alteration (hematoxylin and eosin staining, original magnification ×200). (c) Clinical findings 1 month after reduction of entecavir. |
Only 3 cases of drug eruption caused by entecavir have been reported previously in the literature (Table 1). We reviewed the cases of 3 men and 1 woman, including our patient. The median age of onset was 63.5 years. The types of eruptions included immediate allergy,3 maculopapular type,4 granulomatous type5 and erythematous type (our patient). The interval between the start of entecavir administration and the onset of symptoms varied in range from 2 days to 6 months.
| Ref. | Case no | Year | Age | Sex | Clinical manifestations | Time interval† | Treatment | Patch test | DLST | Scratch test | Recurrrence |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 3 | 1 | 2009 | 30 | M | Immediate allergy | 2 d | Discontinuation, Topical steroid, Oral anti-histamine | – | nw | + | nw |
| 4 | 2 | 2011 | 62 | M | Maculopapular | 7 d | Discontinuation, Topical steroid | nw | + | ND | nw |
| 5 | 3 | 2013 | 65 | F | Granulomatous | 2 m | Discontinuation, Topical metronidazol, Oral minocycline | – | nw | nw | +(3 months after retreatment) |
| Our case | 4 | 2014 | 65 | M | Erythematous plaque | 6 m | Reduce,Topical steroid | ND | + | ND | + |
Ref, reference; DLST, drug lymphocyte stimulation test; M, male; F, female; d, day; m, month; nw, not written; ND, not done.
†. Time interval means between start of entecavir and onset of symptoms.
Recently, defective regulatory T cells (Tregs) are thought to be one of the key factors in the development of drug eruption.6 Zhang JY et al.7 reported frequencies of Tregs gradually decreased from the baseline to month 6 in patients undergoing entecavir treatment, and the ratio of Tregs to Th17 cells were the lowest at month 3 and exhibited a reverse “V”-type change. We speculated that long-term duration to onset in this case might be caused by this gradually decreased Tregs and also the immunosuppressive effects of azathioprine and prednisolone.
Another interesting observation is that, although entecavir has been used worldwide, all 4 cases were from East Asian countries. Recently, the relationship between the genetic background and drug eruptions has attracted considerable attention.8, 9 and 10 For example, in the case of carbamazepine-induced Stevens–Johnson syndrome and toxic epidermal necrolysis, a strong association of HLA-B*1502 and HLA-B*1511, both of which are alleles of the HLA-B75 serotype, has been found in Han Chinese and Japanese patients, respectively.8 and 9 However, these HLA alleles are not found in Caucasian populations. In addition, HLA-B*5801 has been reported to be a risk factor for allopurinol-induced Stevens–Johnson syndrome/toxic epidermal necrolysis.10
According to these observations, which suggest that certain HLA alleles could be genetic markers for certain drug eruptions, we could speculate that genetic differences might contribute to a distinctive hypersensitivity rate for entecavir. Further investigation is indispensable to determine whether this speculation is correct.
Conflict of interest
The authors have no conflict of interest to declare.
References
- 1 R.A. de Man, L.M. Wolters, F. Nevens, D. Chua, M. Sherman, C.L. Lai, et al.; Safety and efficacy of oral entecavir given for 28 days in patients with chronic hepatitis B virus infection; Hepatology, 34 (2001), pp. 578–582
- 2 C.M. Lange, J. Bojunga, W.P. Hofmann, K. Wunder, U. Mihm, S. Zeuzem, et al.; Severe lactic acidosis during treatment of chronic hepatitis B with entecavir in patients with impaired liver function; Hepatology, 50 (2009), pp. 2001–2006
- 3 K. Sugiura, M. Sugiura, T. Takashi, H. Naoki, A. Itoh; Immediate allergy, drug-induced eruption, by entecavir; J Eur Acad Dermatol Venereol, 23 (2009), pp. 487–489
- 4 S. Yamada, Y. Sawada, M. Nakamura; Maculopapular-type drug eruption caused by entecavir; Eur J Dermatol, 21 (2011), pp. 635–636
- 5 J. Yoon, D. Park, C. Kim; A granulomatous drug eruption induced by entecavir; Ann Dermatol, 25 (2013), pp. 493–495
- 6 R. Takahashi, Y. Kano, Y. Yamazaki, M. Kimishima, Y. Mizukawa, T. Shiohara; Defective regulatory T cells in patients with severe drug eruptions: timing of the dysfunction is associated with the pathological phenotype and outcome; J Immunol, 182 (2009), pp. 8071–8079
- 7 J.Y. Zhang, C.H. Song, F. Shi, Z. Zhang, J.L. Fu, F.S. Wang; Deceased ratio of Treg cells to Th17 cells correlates with HBV DNA suppression in chronic hepatitis B patients undergoing entecavir treatment; PLoS One, 5 (2010), p. e13869
- 8 W.H. Chung, S.I. Hung, H.S. Hong, M.S. Hsih, L.C. Yang, H.C. Ho, et al.; Medical genetics: a marker for Stevens-Johnson syndrome; Nature, 428 (2004), p. 486
- 9 N. Kaniwa, Y. Saito, M. Aihara, K. Matsunaga, M. Tohkin, K. Kurose, et al.; HLA-B*1511 is a risk factor for carbamazepine-induced Stevens-Johnson syndrome and toxic epidermal necrolysis in Japanese patients; Epilepsia, 51 (2010), pp. 2461–2465
- 10 S.I. Hung, W.H. Chung, L.B. Liou, C.C. Chu, M. Lin, H.P. Huang, et al.; HLA-B*5801 allele as a genetic marker for severe cutaneous adverse reactions caused by allopurinol; Proc Natl Acad Sci U S A, 102 (2005), pp. 4134–4139
Document information
Published on 05/04/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?