Abstract
Background
Although right middle lobe (RML)-atelectasis of the lungs is a common complication of asthma, the relevant data is limited. The aim of this study is to define the characteristics of RML atelectasis in asthma during childhood.
Methods
Children with asthma who had recently developed RML atelectasis were included; anti-inflammatory medications, clarithromycin, and inhaled salbutamol were prescribed, chest-physiotherapy (starting on the sixth day) was applied. Patients were reevaluated on the sixth, fourteenth, thirtieth, and ninetieth days, chest X-rays were taken if the atelectasis had not resolved at the time of the previous visit.
Results
Twenty-seven patients (6.8 (4.8–8.3) years, 48.1% male) with RML atelectasis were included. Symptoms started 15 (7–30) days before admission. The thickness of the atelectasis was 11.8 ± 5.8 mm; FEV1% was 75.9 ± 14.2 and Childhood Asthma Control Test scores were 11.8 ± 5.6 at the time of admission. The atelectasis had been resolved by the sixth (n = 3), fourteenth (n = 9), thirtieth (n = 10), and ninetieth days (n = 3). The treatment response of the patients whose atelectasis resolved in fourteen days was better on the sixth-day (atelectasis thickness: 4.7 ± 1.7 vs. 11.9 ± 7.3 mm, p = 0.021) compared to those whose atelectasis resolved later. Nearly half (54.5%) of the patients whose atelectasis had resolved by fourteen days were using controller medications at the time of admission. However, only two patients (13.3%) were on controller treatment in the latter group (p = 0.032). Regression analysis didn't reveal any prognostic factors for the early resolution of atelectasis.
Conclusions
Early diagnosis and treatment of RML atelectasis prevents complications. Patients who had early resolution of atelectasis had already been on anti-inflammatory medications, and responded better to aggressive treatment within the first week.
Keywords
Asthma control; Atelectasis; Complication; Pediatrics; Prevention
Abbreviations
RML, right middle lobe; FEV1, forced expiratory volume in 1 s; C-ACT, Childhood Asthma Control Test; OR, odds ratio; CI, confidence interval; ICS, inhaled corticosteroids; LABA, long-acting beta agonists
Introduction
Atelectasis is defined as the incomplete expansion of the lung parenchyma, leading to alveolar hypoxia and pulmonary vasoconstriction to prevent ventilation-perfusion mismatching. Children, especially younger children, are more prone to develop atelectasis due to smaller and more collapsible airways, more pliant chest walls, and inefficient collateral ventilation through intra-alveolar and bronchiole-alveolar pores.1 This predisposition is especially notable in patients not only with lung diseases2 and 3 (e.g., pneumonia, asthma, or cystic fibrosis) but also with systemic diseases4 (e.g. neuromuscular diseases).
Although segmental or lobar collapse of the lungs is a common complication of asthma, data about its exact incidence is limited. The incidence has been recorded in 1.62 percent of the general population of children with asthma2 and 11 percent and 36 percent of populations of children with non-hypoxemic and hypoxemic asthma exacerbations, respectively.5 While right middle lobe (RML) atelectasis is typically seen, sometimes the lingula is involved.6 The presence of bronchial inflammation that produces cellular debris, mucus plugs, and edema contributes to the development of atelectasis in asthma.
RML atelectasis can resolve spontaneously, but in some cases the region undergoes atelectasis after acute exacerbation prevents the lobe from re-expanding; in these cases the lobe remains collapsed. Recurrent infections/inflammations and obstruction of the involved lobe may lead to parenchymal damage and bronchiectasis.7 Prompt diagnosis and treatment is mandatory for a better pulmonary outcome. The term ‘RML syndrome’ is used if the RML atelectasis is persistent (with a duration longer than one month) and/or recurrent (≥2 episodes).7 and 8 Although there have been some studies on RML syndrome, little is known about RML atelectasis regarding its response to treatment and the recovery from RML syndrome in childhood asthma. Our aim in this follow-up study was to identify the characteristics and prognostic factors of RML atelectasis in asthma during childhood.
Methods
Study population
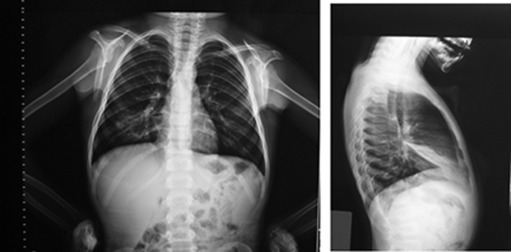
In this prospective study, children between the ages of 3–18 years with an initial diagnosis of asthma who had developed RML atelectasis during follow-up (Fig. 1) were enrolled from the outpatient allergy clinic of the Ankara Education and Research Hospital. The initial diagnosis of asthma was established if there had been a history of respiratory symptoms, such as intermittent wheezing, shortness of breath, coughing, and/or reversible expiratory airflow limitation, defined by at least a twelve percent improvement in forced expiratory volume in 1 s (FEV1) following bronchodilator administration.9 As part of a routine clinical practice, before making an initial diagnosis of asthma the patients who participated in this study had been evaluated for the other etiologies of recurrent respiratory symptoms with other diagnostic tools including chest X-rays and none of them had been found to have any structural pulmonary abnormalities at that time. In this study the chest X-rays revealing atelectasis during follow-up had been required either due to symptoms where we suspected uncontrolled asthma or due to the existence of an exacerbation. In that case, the diagnosis of RML atelectasis was based on radiologic findings, more apparent in the lateral view of a chest X-ray as a wedge-shaped, increased density between the minor and major fissures, with the apex at the hilum and the base towards the pleura10 (Fig. 1). Patients were excluded from the study if they had severe, predefined chronic illnesses other than asthma, including cystic fibrosis, primary ciliary dyskinesia, chest wall defects.
|
|
|
Fig. 1. Posteroanterior and lateral radiographs of five-year-old child with atelectasis of right middle lobe. |
A survey was administered to each participant regarding demographic characteristics (age, gender, family history for atopic diseases, and smoke exposure), asthma control parameters (asthma exacerbations, package of salbutamol consumption, and hospitalizations due to asthma within the last year), and symptomatology of the most recent episode (the onset of symptoms before admission, and the frequency of daytime and nighttime symptoms within the last month).
The children and their parents also filled in the Turkish-language version of the Childhood Asthma Control Test (C-ACT).11 The C-ACT is a self-administered questionnaire with seven items that assesses asthma symptoms during the day and night, the effects of asthma on daily life, and the use of rescue medications in the preceding four weeks.12 A C-ACT score of 19 or lower indicated inadequately controlled asthma.11 Asthma severity was assessed according to the type of controller medications used by the patients, such as inhaled corticosteroids and/or montelukast at the enrollment visit.9
Study measurements
Spirometry using the ZAN100 spirometry system (nSpire Health, Longmont, Colorado, USA), complete blood cell count, and immunoglobulin E measurements (Uni-Cap; Pharmacia, Kalamazoo, Michigan, USA) were done with standard procedures. Skin prick testing (Stallergenes; Antony, France) with twenty-four aeroallergens, including house dust mites; cockroaches; grass, weed, and tree pollens; molds; and cat and dog dander was performed with positive and negative controls. Reactions with an induration of 3 mm or greater than that of the negative control was considered positive, and children with at least one positive test were diagnosed as atopic.
In cases where RML atelectasis was diagnosed during the initial visit, therapy was planned as systemic methylprednisolone (2 mg/kg/day, for five days), clarithromycin for probable underlying infection (20 mg/kg/day, for ten days) and inhaled salbutamol as needed. On the sixth day of treatment, chest physiotherapy13 was started and continued until the improvement of atelectasis. For the purpose of chest physiotherapy13 the patients and their parents were advised to perform percussion and vibration techniques and the exercises of deep breathing and coughing about which they were educated. The medications of the ones who had been already on regular controller treatment was stepped up according to the allowed medications and doses implicated according to age in current GINA guidelines.9 For the ones newly starting to the regular treatment (either montelukast or low dose inhaled corticosteroids) the controller medication was chosen according to the patients age and using capability and started after discussion with the parents of the patients and the patient him/herself. The prescriptions were also given age appropriately in accordance with the GINA guidelines.9
Patients were reevaluated on the sixth, fourteenth, thirtieth, and ninetieth days following admittance, and chest X-rays were taken if the atelectasis had not resolved in the previous visit. Chest X-rays were performed in two projections: posteroanterior and right lateral.
Chest X-rays were reviewed by the radiologist (CS), who was blind to clinical symptoms and findings. Since no standardized validated methods to measure the degree of atelectasis on chest X-rays has been reported until now,14 the radiologist preferred to measure the maximum thickness of atelectasis on lateral chest X-rays that was noted in millimeters which is a frequently used method in order to follow-up the resolution of atelectasis in our tertiary hospital.
Our study was completed in accordance with the ethical standards specified in the Declaration of Helsinki, and was approved by the Medical Ethics Committee of Ankara Education and Research Hospital. All parents and older children provided informed consent.
Statistical analyses
Statistical analyses were performed by using the SPSS 15 package programme (SPSS, Inc., Chicago, Illinois, USA). The Kolmogorov–Smirnov test was used to categorize continuous variables as being normally or non-normally distributed. Categorical variables were expressed as frequencies and numerical variables, as mean (±SD) or median (interquartile range) according to normal or non-normal distribution, respectively. Differences between the groups were compared by chi-square, Students t, or Kruskal–Wallis tests according to the distribution of data. Variables with a p value of less than 0.25 in univariate analysis were examined in the multivariate logistic regression models. Multivariate logistic regression to model the odds of ‘resolution of atelectasis’ versus ‘persistence of atelectasis’ on day 14 was used with a backward likelihood ratio modeling strategy. The size of the effect of each of the risk factors was measured using the odds ratios (OR) and 95 percent confidence intervals (CI). A p value < 0.05 was considered statistically significant.
Results
Twenty-seven children with asthma and RML atelectasis (6.8 (4.8–8.3) years, male 48.1 percent) were included in the study. None of our patients were older than twelve years of age. Atopy was determined in twelve (44.4 percent) patients. Over the course of the last year, patients had experienced frequent asthma attacks [n = 3 (2–6)] and nineteen children had been hospitalized at least once due to asthma exacerbation. Eight (28.6 percent) of the patients were already using controller medications. The characteristics of the subjects are summarized in Table 1.
| Age (year) | 6.8 (4.8–8.3)† |
| Gender (male) (%) | 48.1 |
| Age at diagnosis (year) | 6 (4–8)† |
| Family history for atopic diseases (%) | 48.1 |
| Smoke exposure (%) | 25.9 |
| Allergic rhinitis (%) | 11.1 |
| Atopy (%) | 44.4 |
| Asthma exacerbations/last year | 3 (2–6)† |
| Hospitalization/last year (≥1) (%) | 70.4 |
| Salbutamol consumption, box/last year (n = 14) | 3 (1.8–4.5)† |
| Childhood Asthma Control Test score | 11.8 ± 5.6‡ |
| Asthma severity (%) | |
|---|---|
| Mild intermittent | – |
| Mild persistent | 37 |
| Moderate persistent | 51.9 |
| Severe persistent | 11.1 |
| Symptom onset before admission (day) | 15 (7–30)† |
| Daytime symptom/last month (day) | 18.1 ± 11.5‡ |
| Nighttime symptom/last month (day) | 15.9 ± 11.7‡ |
| Controller medications | |
| Inhaled corticosteroids | 2 |
| Montelukast | 3 |
| Inhaled corticosteroids + montelukast | 2 |
| Inhaled corticosteroids + long-acting beta agonist | 1 |
†. Median (interquartile range).
‡. Mean ± standard deviation.
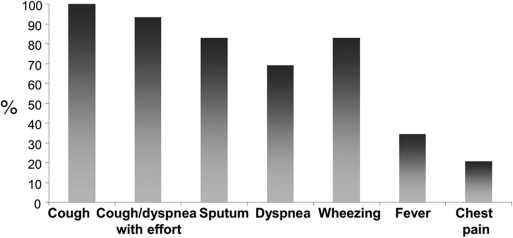
In the current visit, the onset of asthma symptoms was 15 (7–30) days before admission. The most common presenting symptoms were coughing (100 percent), sputum production (85.2 percent), dyspnea (70.4 percent) (Fig. 2). Additionally nine patients (33.3%) had fever at the time of recognition of RML atelectasis. Physical examination revealed pathologic findings in 26 (92.6 percent) children with asthma, including rales (11.1 percent), rhonchi (88.9 percent), wheezing (40.7 percent), and retraction (25.9 percent). Half of the patients were able to perform lung function tests, and FEV1 were 75.9 ± 14.2 percent of the predicted (Table 2). On the chest X-rays, the mean (±SD) of RML atelectasis was 11.8 ± 5.8 mm at maximum width. As a result, at first control visit; the treatment plans were as follows: inhaled corticosteroids (ICS) (n = 10), montelukast (n = 1), ICS + montelukast (n = 6), ICS + long-acting beta agonists (LABA) (n = 8), and ICS + LABA + montelukast (n = 2). Statistically, it is impossible to compare any difference in prognosis between ICS and montelukast since there was only one patient who used montelukast alone. However, when we compare patients using inhaled corticosteroids versus inhaled corticosteroids and montelukast or long acting beta agonists, there is no difference in terms of prognosis.
|
|
|
Fig. 2. Symptoms at the time of admission. |
| FEV1% (n = 14) | 75.9 ± 14.2† |
| FVC % (n = 14) | 78.1 ± 13.4† |
| FEF 25–75% (n = 14) | 72.8 ± 22.2† |
| White blood cells | 8350 (7325–12,350)‡ |
| Eosinophils (%) | 1.3 (0.18–3.3)‡ |
| Polymorphonuclear leucocytes (%) | 62.9 (53.9–81.3)‡ |
| Sedimentation | 18 (12–27)‡ |
| Atelectasis thickness (mm) | 11.8 ± 5.8 |
| Ig E (IU/L) | 71.0 (31.3–351.8)‡ |
†. Mean ± standard deviation.
‡. Median (interquartile range).
As a result of evaluation during the follow-up visits, the resolution of atelectasis had occurred on the sixth, fourteenth, thirtieth, and ninetieth days in three, nine, ten, and three patients, respectively. Likewise, their C-ACT scores increased (admission: 12.2 ± 5.9; first month: 18.9 ± 5.7; second month: 21.5 ± 4.6; third month: 23 ± 2.4). It is important to note that the median (interquartile range) follow-up period (the period between the initial diagnosis of asthma and the appearance of atelectasis) of the study population was 0.66 (0.16–1.3) years and when the follow-up period of the patients whose atelectasis resolved in 14 days were compared with the patients whose atelectasis did not resolve in 14 days, no difference was found [0.9 (0.3–1.9) years vs. 0.3 (0.08–0.9) years, p > 0.05].
The atelectasis did not resolve in two patients, however, they still had linear atelectasis on the ninetieth day. One of these children remained asymptomatic. The other child had undergone high resolution computerized tomography, which revealed RML atelectasis. His immunological assessment, sweat chloride test, nasal biopsy for primary ciliary dyskinesia, and tuberculin test were within normal limits. He was referred to the pediatric pulmonology department, where he had been treated conservatively. By the end of one year, he was still symptomatic despite adequate anti-asthma treatment and developed bronchiectasis.
The ratio of the patients with or without fever at the time of recognition of atelectasis did not differ in terms of the time period passed for resolution of atelectasis. However, the treatment response of the patients whose atelectasis resolved in fourteen days (n = 12) was better on the sixth day (atelectasis thickness: 4.7 ± 1.7 and 11.9 ± 7.3 mm, p = 0.021) compared to the patients whose atelectasis resolved later in terms of thickness of the atelectasis. Nearly half of the patients (54.5%) whose atelectasis had resolved in fourteen days were using controller medications for asthma at the time of admission. But only two patients (13.3 percent), however, were on controller treatment in the group of patients whose atelectasis had resolved later than fourteen days (p = 0.032). The C-ACT scores of the patients whose atelectasis resolved in fourteen days were higher on the thirtieth day compared to the latter group [22.2 ± 3.2 vs. 16.8 ± 6.3, p = 0.019 ] ( Table 3). The time to resolution of atelectasis did not differ in between the patient groups who were evaluated either due to uncontrolled asthma or existence of an exacerbation. Logistic regression analysis for recovery of atelectasis later than fourteen days did not reveal any prognostic factors, including asthma control parameters, level of asthma control (‘uncontrolled’ versus ‘partially controlled’), atopy, serum IgE levels, or thickness of atelectasis upon admission.
| ≤14 days (n = 12) | >14 days (n = 15) | p | |
|---|---|---|---|
| Age† (years) | 7 (4.5–9.9) | 6.8 (5.9–8.1) | >0.05 |
| Gender (male) (%) | 58.3 | 42.9 | >0.05 |
| Smoke exposure (%) | 25 | 28.6 | >0.05 |
| Atopy (%) | 41.7 | 50 | >0.05 |
| Exacerbations/last year† | 3.5 (2–4.8) | 3 (2–6) | >0.05 |
| Hospitalization/last year (%) | 66.7 | 71.4 | >0.05 |
| Onset of symptoms† (day) | 20 (7–30) | 11 (3.5–37.5) | >0.05 |
| FEV1%† | 83.5 (75–89.5) | 76.5 (66.5–79.5) | >0.05 |
| Controller medication use, admission (%) | 54.5 | 13.3 | 0.032 |
| C-ACT, admission‡ | 11.5 ± 4.8 | 12.0 ± 6.5 | >0.05 |
| C-ACT, month 1‡ | 22.2 ± 3.2 | 16.8 ± 6.3 | 0.019 |
| Atelectasis thickness, admission‡ | 10 ± 1 | 12.3 ± 7.5 | >0.05 |
| Atelectasis thickness, (day 6)‡ | 4.7 ± 1.7 | 11.9 ± 7.3 | 0.021 |
†. Median (interquartile range).
‡. Mean ± standard deviation.
Discussion
In this study, we addressed an issue that for a long time has been related to complications of uncontrolled asthma. We showed that most of the study participants' atelectasis (nearly 90 percent) had resolved at the end of three months by appropriate treatment. Most of the patients who demonstrated early resolution of atelectasis had already been on anti-inflammatory medications, and they responded better to aggressive treatment within the first week.
The right middle lobe is the most commonly involved part of the lung to develop atelectasis in children with asthma. The unique anatomic features of the RML (which has a narrow and long lobar bronchus that starts after a sharp angle from the bronchus intermedius) and the relative anatomic confinement of the RML (which results in poor collateral ventilation) both contribute to the obstruction of the RML.15 and 16 Recurrent exacerbations of asthma due to a worsening of the underlying inflammation and/or repeated episodes of infection further contribute to inadequate clearance of secretions and the formation of mucus plugs.17 In this study, the patients represented a population composed of individuals with uncontrolled asthma who had experienced frequent asthma symptoms and therefore used bronchodilators regularly. Furthermore, 70 percent of them had been hospitalized due to an asthma exacerbation within the last year, pointing to the severity of their symptoms.
For the last decade, the treatment of asthma has principally focused on the control of parameters, including daytime or nighttime symptoms, the need for bronchodilator use for relief of symptoms, and activity limitation due to asthma.9 and 18 Risk factors for poor asthma outcomes have been published recently, including excessive bronchodilator use, inadequate ICS use, smoke exposure, low baseline FEV1, sputum or blood eosinophilia, intensive care admission, or the occurrence of one or more severe exacerbations in the previous twelve months.9 Interestingly, complications of asthma such as pneumothorax, pneumomediastinum, or lobar atelectasis have not drawn attention and have not been referred to in the literature. If these complications are not diagnosed and treated properly, the consequences can be hazardous.
Chest radiography is not routinely recommended for the diagnosis of asthma; instead it helps to recognize complications or to exclude chronic infections and anatomic abnormalities, especially during childhood.19 and 20 Typical findings of an asthmatic lung on chest X-rays are hyperinflation due to intrinsic positive end-expiratory pressure, paucity of vascular markings in the upper zones of the lung, and thickened bronchial walls and peribronchial tissues.20 The threshold for ordering a chest X-ray is high for physicians today because of a fear of side effects from radiation, and the difficulty of convincing parents (even those whose children have persistent respiratory symptoms) of the need for such X-rays. With the invention of newer techniques such as positron emission tomography or magnetic resonance imaging, however, the role of imaging in the diagnosis and management of asthma has increased.21
A precise diagnosis of the underlying pathology is essential for proper treatment of symptoms, especially in cases of atelectasis, where no single therapy is likely to be successful in all forms. Evidence-based studies on the management of lobar atelectasis are lacking.22 We treated our patients with systemic corticosteroids not only for persistent or recurrent asthma symptoms, but also for atelectasis. Corticosteroids are the cornerstones of asthma treatment, as they reduce the number of inflammatory cells in the airways, block neutrophil adherence, and decrease microvascular leakage; they are also highly effective in inhibiting mucus hypersecretion by suppressing the underlying inflammation. In addition, they inhibit the synthesis of arachidonic acid-derived inflammatory mediators like montelukast, which leads to a further decrease in hyperreactivity.23, 24, 25 and 26 On the sixth day of the treatment, atelectasis improved or had undergone resolution in thirteen patients in this study, which may be due to the rapid and potent anti-inflammatory actions of corticosteroids and montelukast.
We used clarithromycin (used for probable underlying infection) for the management of atelectasis, whether or not the patient had fever. There is an association of RML collapse with bacterial infections in children with asthma27 and bronchoalveolar lavage cultures of patients with RML atelectasis revealed the presence of Haemophilus influenzae, Streptococcus pneumonia, and Staphylococcus aureus. 27 and 28 Macrolides exhibit not only antimicrobial features29 but also immunomodulatory actions, such as the inhibition of pro-inflammatory transcription factors, goblet cell hyperplasia and the reduction of airway interleukin-8 and neutrophil numbers. 30, 31 and 32 Nevertheless, placebo controlled trials are needed to demonstrate the effectiveness of antibiotics in RML atelectasis.
The basic therapeutic options for asthma rely on the use of controller and reliever medications. A lack of ICS treatment as a controller medication is a risk factor for both developing fixed airflow limitation and future exacerbation.9 Additionally one of our studys important findings was the rapid improvement of atelectasis in children who had been using controller medications. There were 8 patients who had been already on controller treatment such as montelukast and/or ICS with or without LABA at the time of recognition of RML atelectasis (Table 1). The constant reduction of inflammation of the airways by regular ICS and/or montelukast intake might contribute to the rapid response and resolution of atelectasis. Furthermore, glucocorticoid receptor α expression is down-regulated in vitro by glucocorticoids, but in vivo, down-regulation of the receptor by glucocorticoids treatment does not appear to occur. 33
A limitation of our study was that we did not determine the frequency of RML syndrome in children with asthma who admitted to our allergy department and who had taken chest X-rays during the study period; this was beyond our aim. A case–control study may demonstrate the efficacy of different management protocols. Another limitation would be the sample size. Further studies regarding right middle lobe atelectasis with more number of participants would be informative since studies on this topic are relatively scarce in pediatric age group.
In conclusion, treatment with controller medications in known asthmatics might decrease the severity and speed the resolution of RML atelectasis, and that in most children the problem might resolve within 90 days with treatment. Control chest X-rays may be required after a one-month follow-up to ensure that the atelectasis is resolved. If there is no recovery, cases should be followed closely to prevent the development of complications and the need for surgical interventions. Baseline treatment of asthma with anti-inflammatory medications not only controls asthma symptoms but also accelerates the resolution of atelectasis.
Conflict of interest
The authors have no conflict of interest to declare.
Authors' contributions
OS had primary responsibility for the development of the protocol, analytic framework of the study and data analysis, and had primary responsibility for patient screening with CO and prepared the manuscript. CO participated in the development of the protocol, contributed to patient screening and preparation of the manuscript. OC and CS contributed to the design, data analysis and manuscript preparation. YD contributed to the design and data analysis of the study. All authors read and approved the final manuscript.
References
- 1 D.G. Peroni, A.L. Boner; Atelectasis: mechanisms, diagnosis and management; Paediatr Respir Rev, 1 (2000), pp. 274–278
- 2 B.E. Sekerel, F. Nakipoglu; Middle lobe syndrome in children with asthma: review of 56 cases; J Asthma, 41 (2004), pp. 411–417
- 3 J.M. Marchant, J.P. Masel, F.L. Dickinson, I.B. Masters, A.B. Chang; Application of chest high-resolution computer tomography in young children with cystic fibrosis; Pediatr Pulmonol, 31 (2001), pp. 24–29
- 4 J.D. Finder; Airway clearance modalities in neuromuscular disease; Paediatr Respir Rev, 11 (2010), pp. 31–34
- 5 S.L. Tsai, E.F. Crain, E.J. Silver, H.S. Goldman; What can we learn from chest radiographs in hypoxemic asthmatics?; Pediatr Radiol, 32 (2002), pp. 498–504
- 6 A.K. Ayed; Resection of the right middle lobe and lingula in children for middle lobe/lingula syndrome; Chest, 125 (2004), pp. 38–42
- 7 K.N. Priftis, M.B. Anthracopoulos, D. Mermiri, A. Papadopoulou, P. Xepapadaki, C. Tsakanika, et al.; Bronchial hyperresponsiveness, atopy, and bronchoalveolar lavage eosinophils in persistent middle lobe syndrome; Pediatr Pulmonol, 41 (2006), pp. 805–811
- 8 K. De Boeck, T. Willems, D. Van Gysel, L. Corbeel, R. Eeckels; Outcome after right middle lobe syndrome; Chest, 108 (1995), pp. 150–152
- 9 Global Strategy for Asthma Management and Prevention. Updated 2014. Available from: http://www.ginasthma.org/ [accessed 30.06.14].
- 10 T. Gudbjartsson, G. Gudmundsson; Middle lobe syndrome: a review of clinicopathological features, diagnosis and treatment; Respiration, 84 (2012), pp. 80–86
- 11 B.E. Sekerel, O.U. Soyer, O. Keskin, N. Uzuner, M. Yazicioglu, M. Kilic, et al.; The reliability and validity of Turkish version of Childhood Asthma Control Test; Qual Life Res, 21 (2012), pp. 685–690
- 12 A.H. Liu, R. Zeiger, C. Sorkness, T. Mahr, N. Ostrom, S. Burgess, et al.; Development and cross-sectional validation of the Childhood Asthma Control Test; J Allergy Clin Immunol, 119 (2007), pp. 817–825
- 13 M. Duggan, B.P. Kavanagh; Atelectasis; R.W. Wilmott, T.F. Boat, A. Bush, V. Chernick, R.R. Deterding, F. Ratjen (Eds.), Kendig’s and Chernick’s Disorders of Respiratory Tract in Children, Elsevier Saunders, Philadelphia (2012), pp. 564–569
- 14 T. Hendriks, M. de Hoog, M.H. Lequin, A.H. Devos, P.J.F.M. Merkus; DNase and atelectasis in non-cystic fibrosis pediatric patients; Crit Care, 9 (2005), pp. R351–R356
- 15 S. Bertelsen, E. Struve-Christensen, A. Aasted, J. Sparup; Isolated middle lobe atelectasis: aetiology, pathogenesis, and treatment of the so-called middle lobe syndrome; Thorax, 35 (1980), pp. 449–452
- 16 J.T. Einarsson, J.G. Einarsson, H. Isaksson, T. Gudbjartsson, G. Gudmundsson; Middle lobe syndrome: a nationwide study on clinicopathological features and surgical treatment; Clin Respir J, 3 (2009), pp. 77–81
- 17 A. Sehitogullari, F. Sayir, U. Cobanoglu, S. Bilici; Surgical treatment of right middle lobe syndrome in children; Ann Thorac Med, 7 (2012), pp. 8–11
- 18 National Asthma Education and Prevention Program; Expert Panel Report 3: Guidelines for the Diagnosis and Management of Asthma; National Institute of Health, Bethesda, MD (2007) NIH Publication No. 08-4051. Available from: http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.htm [accessed 20.06.14]
- 19 L. Benayoun, A. Druilhe, M.C. Dombret, M. Aubier, M. Pretolani; Airway structural alterations selectively associated with severe asthma; Am J Respir Crit Care Med, 167 (2003), pp. 1360–1368
- 20 A. Sung, D. Naidich, I. Belinskaya, S. Raoof; The role of chest radiography and computed tomography in the diagnosis and management of asthma; Curr Opin Pulm Med, 13 (2007), pp. 31–36
- 21 M. Castro, S.B. Fain, E.A. Hoffman, D.S. Gierada, S.C. Erzurum, S. Wenzel, National Heart, Lung, and Blood Institutes Severe Asthma Research Program; Lung imaging in asthmatic patients: the picture is clearer; J Allergy Clin Immunol, 128 (2011), pp. 467–478
- 22 M.B. Schindler; Treatment of atelectasis: where is the evidence?; Crit Care, 9 (2005), pp. 341–342
- 23 P. Chanez, A. Bourdin, I. Vachier, P. Godard, J. Bousquet, A.M. Vignola; Effects of inhaled corticosteroids on pathology in asthma and chronic obstructive pulmonary disease; Proc Am Thorac Soc, 1 (2004), pp. 184–190
- 24 P.J. Barnes; Effect of corticosteroids on airway hyperresponsiveness; Am Rev Respir Dis, 141 (2 Pt 2) (1990), pp. S70–S76
- 25 R. Djukanovic, J.W. Wilson, K.M. Britten, S.J. Wilson, A.F. Walls, W.R. Roche, et al.; Effect of an inhaled corticosteroid on airway inflammation and symptoms in asthma; Am Rev Respir Dis, 145 (1992), pp. 669–674
- 26 P.J. Barnes; Current and future therapies for airway mucus hypersecretion; Novartis Found Symp, 248 (2002), pp. 237–249 discussion 249–53, 277–82
- 27 C. Springer, A. Avital, N. Noviski, C. Maayan, I. Ariel, P. Mogel, et al.; Role of infection in the middle lobe syndrome in asthma; Arch Dis Child, 67 (1992), pp. 592–594
- 28 K.N. Priftis, D. Mermiri, A. Papadopoulou, M.B. Anthracopoulos, G. Vaos, P. Nicolaidou; The role of timely intervention in middle lobe syndrome in children; Chest, 128 (2005), pp. 2504–2510
- 29 J.M. Zuckerman; Macrolides and ketolides: azithromycin, clarithromycin, telithromycin; Infect Dis Clin North Am, 18 (2004), pp. 621–649
- 30 E.H. Wong, J.D. Porter, M.R. Edwards, S.L. Johnston; The role of macrolides in asthma: current evidence and future directions; Lancet Respir Med, 2 (2014), pp. 657–670
- 31 T. Tanabe, S. Kanoh, K. Tsushima, Y. Yamazaki, K. Kubo, B.K. Rubin; Clarithromycin inhibits interleukin-13-induced goblet cell hyperplasia in human airway cells; Am J Respir Cell Mol Biol, 45 (2011), pp. 1075–1083
- 32 J.L. Simpson, H. Powell, M.J. Boyle, R.J. Scott, P.G. Gibson; Clarithromycin targets neutrophilic airway inflammation in refractory asthma; Am J Respir Crit Care Med, 177 (2008), pp. 148–155
- 33 L. Pujolsa, J. Mullol, C. Picado; Glucocorticoid receptor in human respiratory epithelial cells; Neuroimmunomodulation, 16 (2009), pp. 290–299
Document information
Published on 05/04/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?