Summary
Solitary benign fibrous mesothelioma (BFM) is uncommon and is termed as solitary fibrous mesothelioma or localized fibrous mesothelioma. Although the most common site for its development is the pleural region (65%), it has also been reported in the peritoneum. They are mostly seen in adults (average age: 54 years). Herein, we present a case of BFM in a 2-year-old male child, who presented to our hospital with abdominal pain and a lump in the abdomen. Differential diagnosis included solitary fibrous tumor, gastrointestinal stromal tumor, and benign fibrous lesions of mesentery. Establishing a preoperative diagnosis is difficult on the basis of clinical parameters or imaging studies and final diagnosis can only be assessed based on the results of histopathological and immunohistochemical examination.
Keywords
benign fibrous mesothelioma;child;peritoneum
1. Introduction
Mesotheliomas can arise from epithelial or mesenchymal elements and can be classified as either diffuse or benign fibrous type. The diffuse types are highly malignant, infiltrating the surrounding tissue, whereas the benign variants are very uncommon and are termed as benign fibrous mesotheliomas (BFMs). BFM has now been termed as solitary fibrous mesothelioma or solitary fibrous tumor (SFT).1 ; 2 It is an exceptionally rare clinical entity and mostly develops in the pleural region (65%); however, in recent years, a few cases have also been reported in the extrathoracic sites including the peritoneum, mesentery, pericardium, and extraperitoneal spaces.3 When mesotheliomas arise at the extrapleural site, it may be difficult to distinguish them from other soft tissue tumors. Therefore, immunohistochemical (IHC) analysis plays a diagnostic role. They are mostly seen in adults between 27 and 64 years of age and their natural history is unknown.4 The etiology and pathophysiology remain elusive amid speculation on their complex genomic imbalances.
2. Case report
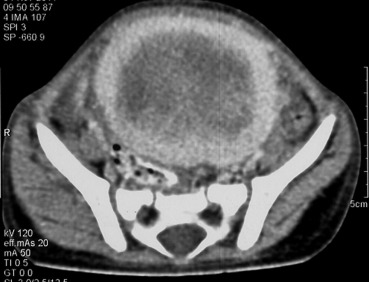
A 2-year-old boy was admitted to the outpatient ward of the pediatrics unit with complaints of excessive crying, episodic diarrhea, and a lump in the abdomen. There was no history of any surgery or trauma. Family history was unremarkable. Clinical examinations revealed that the abdomen was a bit tense. There was a palpable solid mass in the left lower quadrant of the abdomen measuring approximately 10 cm in size. The mass was nonpulsatile with restricted movement and tender on deep palpation. An abdominal ultrasound sonography revealed a large hypoechoic mass arising from the pelvis displacing the bowel loops. Left kidney showed mild grade 1 hydronephrosis. A contrast-enhanced computed tomography revealed a hypodense lesion showing homogeneous contrast enhancement in the pelvic region with focal areas of necrosis (Fig. 1). Radiological findings were highly suggestive of gastrointestinal stromal tumor (GIST). On the basis of clinical presentation and radiological appearances, a tentative diagnosis of GIST was made and an exploratory laparotomy was performed. Peroperatively, the tumor was found to be highly vascular with prominent vessels.
|
|
|
Figure 1. Contrast-enhanced computed tomography scan shows a hypodense lesion with heterogeneous contrast enhancement with areas of necrosis. |
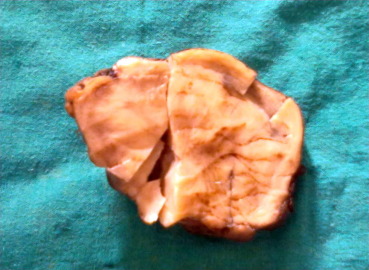
The excised specimen labeled resected tumor of descending colon was submitted for histopathological analysis. A gross examination revealed that the specimen was a well-encapsulated solid, firm to slightly hard, measuring 9 × 6.5 × 2 cm3, and present at the serosal aspect of intestine. The outer surface of the specimen was lobulated. The attached intestine measured 6 cm in length and the cut surface was gray white to pale yellow (Fig. 2). Microsections of the specimen examined showed arrays of bland spindle-shaped cells resembling fibroblasts with variable cellularity and areas of collagenization. Blood vessels showed a hemangiopericytomatous pattern. Mitosis and necrosis were inconspicuous. The histopathological examination included differential diagnosis of BFM or SFT, GIST, mesenteric fibromatosis, and myofibroblastic tumor.
|
|
|
Figure 2. Cut surface of gross specimen, which is pale yellow and homogeneous. |
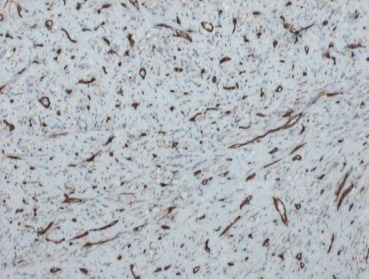
A panel of IHC markers was used to confirm the diagnosis. The tumor was positive for CD34 and vimentin, but was negative for CD117, SMA, S100, and CD99. The final diagnosis of BFM was thus made by analyzing the IHC staining, which played a critical role in diagnosing this entity.
3. Discussion
BFM is now called SFT and is a rare entity. It was first described in 1950, and since then only few cases have been reported. SFT is usually a localized benign neoplasm arising from submesothelial layer. Approximately 65% of SFTs are pleural in origin, but can also be found at other sites such as mediastinum, pericardium, mesothelium, peritoneum, mesentery, retroperitoneal spaces, and paranasal sinuses. SFTs arising in peritoneal or mesenteric regions are called extrapleural fibrous tumors.5 However, cases of SFT that are of extrapleural (peritoneum) origin are extremely rare (2.8/100,000).2 They are mostly seen in adults between 27 and 64 years of age, with the average age being 54 years. Clinical features vary depending on the site and size of tumor. The etiology and pathophysiology remain elusive. On radiological images, they appear as a well-defined solid tumor, which may contain cystic areas with focal areas of calcification. Radiological appearances are not diagnostic and reveal nonspecific overlapping benign fibrous lesions of mesentery (extrapleural SFT, mesenteric fibromatosis, sclerosing mesenteritis, and inflammatory pseudotumor), GISTs, lymphoma, metastasis, carcinoid, and soft tissue sarcomas.6 Establishing preoperative diagnosis is difficult by analyzing clinical parameters or imaging findings alone, and therefore, complete diagnosis totally depends on pathological findings.
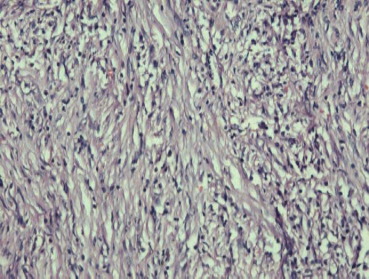
Histologically, extrapleural SFT are well-circumscribed firm masses with a pale cut surface. Microscopic analysis revealed that they were composed of spindle-shaped cells with a variable number of hyalinized collagen matrixes and with a wide range of histological features regarding cellular form and arrangement (Fig. 3). A hemangiopericytomatous pattern was common.7 Mitotic activity may be variable with rare necrosis. IHC analysis revealed that the tumor cells were diffusely positive for CD34 and vimentin, but negative for CD117, SMA, CD99, and S100, which ruled out the differential diagnoses of GIST, mesenteric fibromatosis, and myofibroblastic tumors.8 Therefore, the final diagnosis rested on IHC analysis (Fig. 4). However, SFT should be considered in the differential diagnosis of any mesenchymal lesion arising from gastrointestinal tract. They are usually benign but have high predilection of recurrences and aggressive behavior. Therefore, long-term follow-up is recommended.9
|
|
|
Figure 3. Histological findings (200×) show proliferation of spindle-shaped cells with areas of collagenization and branching blood vessels. |
|
|
|
Figure 4. Immunohistochemical image showing CD34-positive spindle cells and vessels. |
Our case is unique in its presentation because it is seen the peritoneal region of a 2-year-old child, and both the age and site of origin are rare. Available studies report the pleura as the most common site of SFT with incidental reporting in other mesothelial layers. SFT is an under-recognized entity owing to lack of IHC in most of the centers. However, with the induction of IHC and increased applications of markers, cases are now being reported in relation to the abdominal cavity and other extrapleural sites.
References
- 1 T. Adachi, Y. Sugiyama, S. Saji; Solitary fibrous benign mesothelioma of the peritoneum: report of a case; Surg Today, 29 (1999), pp. 915–918
- 2 N. Okike, P.E. Bernatz, L.B. Woolner; Localized mesothelioma of the pleura: benign and malignant variants; J Thorac Cardiovasc Surg, 75 (1978), pp. 363–372
- 3 V.D. Srinivasan, J.D. Wayne, M.S. Rao, D.L. Zynger; Solitary fibrous tumor of the pancreas: case report with cytologic and surgical pathology correlation and review of the literature; JOP, 9 (2008), pp. 526–530
- 4 J.S. Gold, C.R. Antonescu, C. Hajdu, et al.; Clinicopathologic correlates of solitary fibrous tumors; Cancer, 94 (2002), pp. 1057–1068
- 5 R.H. Young, P.B. Clement, W.T. McCaughey; Solitary fibrous tumors (‘fibrous mesotheliomas’) of the peritoneum. A report of three cases and a review of the literature; Arch Pathol Lab Med, 114 (1990), pp. 493–495
- 6 A.D. Levy, J. Rimola, A.K. Mehrotra, L.H. Sobin; From the archives of the AFIP: benign fibrous tumors and tumorlike lesions of the mesentery: radiologic-pathologic correlation; Radiographics, 26 (2006), pp. 245–264
- 7 C. Gengler, L. Guillou; Solitary fibrous tumour and haemangiopericytoma: evolution of a concept; Histopathology, 48 (2006), pp. 63–74
- 8 M.I. Lau, F.J. Foo, M.C. Sissons, P. Kiruparan; Solitary fibrous tumor of small bowel mesentery: a case report and review of the literature; Tumori, 96 (2010), pp. 1035–1039
- 9 Y. Ekıcı, S. Uysal, G. Güven, G. Moray; Solitary fibrous tumor of the lesser omentum: report of a rare case; Turk J Gastroenterol, 21 (2010), pp. 464–466
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?