Summary
Background
In this study, we have tried to demonstrate the effects of coating style used in oxygenators on various hematologic and clinical parameters.
Materials and methods
Twenty-seven patients were included in the study, who had undergone operations because of elective coronary artery disease. Albumin-coated oxygenator was used in Group I. In Group II, a synthetic polypeptide-coated oxygenator was used. C1-inhib (complement), C3c, C4, interleukins (IL-1β, IL2, IL-6, IL-10), and tumor necrosis factor alpha (TNF-α) levels were examined at four different time intervals. Hemoglobin, hematocrit, leukocyte and platelet counts, drainage, and transfused blood volumes were analyzed.
Results
Albumin levels were significantly lower in Group I than those in Group II 5 minutes after the removal of the cross-clamp. Twenty-four hours after the surgery, Group I patients also had a significantly higher white blood cell count compared to Group II patients. TNF-α levels in Group I were always expressed in considerably higher amounts than those in Group II. IL-6 levels were significantly higher in Group I, but IL-10 levels were observed to be higher in Group II (p < 0.05).
Conclusion
Synthetic polypeptide-coated advanced technology, which employed oxygenators, had an important attenuator effect on acute phase reactants and also on the inflammatory response.
Keywords
complement;interleukin;oxygenator;systemic inflammatory response syndrome
1. Introduction
Systemic Inflammatory Response Syndrome (SIRS) can be the result of coagulation system activation and inflammatory reactions triggered by blood getting into contact with nonbiological membranes during cardiopulmonary bypass.1 ; 2 SIRS is characterized by hypotension, coagulopathy, free oxygen radicals production, and platelet production, which are significantly associated with morbidity and mortality. It is thought that endothelial damage and increased microvascular permeability may play a role in the physiopathological mechanism of this syndrome.3
One of the most important immunological structures affecting the inflammatory reactions that develop after cardiopulmonary bypass (CPB) is the complement system. The severity of the complement response alters depending on the length of CPB4 and the circulation system used.5 During CPB, not only the existence of C3a, C5a, and membrane attack complex (MAC), but also the consumption of the complement components indicate the activation of the complement system in the circulation.6 Considerable complement activations occur within the first 3 days following the CPB.7 During the bypass, the complement system is activated by standard and alternative methods; while the standard activation method leads to an increase both in C3a and C4a, the alternative activation method leads to an increase only in C3a.8 C3a begins to increase with the start of CPB and reaches its peak at the end of the bypass. C3a level depends on the length of the bypass and reverts back to its standard level after 48 hours. C5a is related to instant reperfusion. C5a triggers activated leukocytes, therefore, increases superoxide production and enables them to attach to the coronary artery endothelium.9
There exist important peptides that function in inflammatory and repair roles in the regulation of an organisms immunity system. These peptides are generally called cytokines. Cytokines play a role in both systemic and local inflammation by regulating cell-to-cell communication. These cytokines are functionally proinflammatory (IL-1, IL8) and anti-inflammatory [IL-1RA (IL-1 receptor antagonist), IL-10]. IL-6 shows both characteristics. Proinflammatory cytokines such as IL-1α, IL-1β, and IL-6 may turn out to be harmful mediators when plasma levels are high in cases such as major trauma, sepsis, and shock.10 In cardiac surgery, according to some observations, both pro and anti-inflammatory cytokines expression increase in order to provide an equilibrium between them.11
IL-1 has effects on the emergence of local neutrophil infiltration, delayed type cell sensitivity, fibroplasias, and angiogenesis. After IL-1 injection, local inflammatory response begins. This reaction starts 1 hour after the injection, reaching the peak value in 3-4 hours time. This stimulates IL-6 expression. IL-1 injection induces tumor necrosis factor (TNF) expression in macrophages and IL-6 expression in fibroblasts.
IL-2 is mostly secreted by T4 cells but, it can also be secreted by T-8 cells medullary thymocytes and giant granule lymphocytes. It leads to endothelial permeability increase and leak syndrome due to the secretion of various cytokines.
IL-6 is released by T and B lymphocytes, monocytes, endothelial cells, epithelial cells and fibroblasts. It is an indicator of an acute inflammatory response. IL-6 level in plasma rises proportionally to the tissue damage caused by the surgeon and the inflammatory response induced by CPB.
IL-10 inhibits TH1 (T helper lymphocytes) and natural cells' cytokine production. It increases B cell proliferation and antibody production and inhibits cellular immunity. By inhibiting TNF-α and nitrous oxide (NO) production, endogen IL-10 has a role in the protection of myocardium during ischemic reperfusion.
TNF is composed of two peptides called TNF-α and TNF-β. TNF-α is an endogenous pyrogen and an acute phase reactant secreted by activated macrophages. TNF-α has role in local neutrophilic infiltration, tumor necrosis, cachexia, and neutrophilia, and it has radioprotective effects at the same time. TNF-α, like proinflammatory cytokines, may individually turn into potential harmful mediators, when their plasma levels increase substantially.10
Oxygenators are accused of causing hemolysis because of abrupt pressure changes and increased surface tension. Perfusion through an oxygenator increases platelet membrane fragility, leading to increased possibility of hemolysis, which then decreases after a 24-hour period. Further trauma to blood components can be caused by the aspiration system.2 One of the accused tools leading to coagulation cascade activation and inflammatory responses is the “oxygenator” and numerous attempts have been made to change its surface with the hope of preventing these catastrophic events. Therefore, we investigated the effects of coating style (albumin or synthetic peptide) used in oxygenators within various hematologic and clinical parameters.
2. Materials and methods
Twenty-seven patients undergoing elective coronary artery bypass grafting (CABG) were included in the study. Our exclusion criteria among the patients was as follows: any anticoagulant, steroid or anti-inflammatory drug usage within the last 7 days prior to enrollment, previous bypass surgery and anyone in need of additional surgical procedures. The patients were randomly allocated into two groups based on oxygenators used. In Group I, albumin-coated (Jostra REF VKMO 4230, Hirrlingen, Germany) oxygenator was used (n = 14, male:female ratio = 10:4, mean age = 58 ± 8 years), and in Group II, a synthetic peptide-coated oxygenator (Jostra REF VKMO 4200, Hirrlingen, Germany) was used (n = 13, male:female = 10:3, mean age = 58 ± 10 years).
A radial artery and a central vein were cannulated in all patients. Pentothal sodium 3–4 mg/kg, cisatracurium (Nimbex) 0.15–0.2 mg/kg and fentanyl were used for anesthesia induction. Thereafter, fentanyl (100 μg) and Nimbex (0.02 mg/kg with 30-minute intervals) were used for anesthesia. Intravenous heparin (5 mg/kg) was given for systemic heparinization, which was neutralized with protamine sulfate following the cessation of extracorporeal circulation. We preferred isothermic blood cardioplegia and abstained from steroid drugs during the surgery. In the case of a low preoperative hemoglobin level, erythrocyte suspension was added to the individual patients prime volume (Isolyte S 1000 mL, Gelofusine 500 mL, mannitol 100 mL, heparin 50 mg, 3 ampule NaHCO3, antibiotic). Blood samples were taken from radial artery at the following time points:
- T1: Following anesthesia induction, just before surgical incision.
- T2: 5 minutes after the removal of cross-clamp.
- T3: At 6th hour from the start of CPB surgery.
- T4: At 24th hour from the start of CPB surgery.
The blood taken in flat test tubes was allowed to stand at +4°C until it became decomposed with a 10-minute centrifugation in 3000 rotation. The decomposed sera were kept at −70°C in a deep freeze. C1inhib, C3c, and C4 were studied using the nephelometric method with a Behring Nephelometer Analyzer device, and the results were indicated in terms of g/dL.
IL-1β, IL2, IL-6, and IL-10 were studied by the ELISA method using immunoassay kits (BioSource International, Inc USA). The results were obtained by using a Biotek ELISA reader at 450 nm and interleukin results were calculated as picograms (pg)/mL.
TNF-α was studied by the ELISA method using immunoassay kits (BioSource International, Inc USA). The results were obtained by using Biotek ELISA reader at 450 nm. Hemoglobin, hematocrit, leukocyte, and platelet counts were analyzed with a Coulter Max M mode device. Moreover, we also used an Olympus AU 5200 machine for determining total protein and albumin levels.
Drainage and transfused blood volumes were recorded at 4-hour intervals at 0, 8, and 12 hours postoperatively.
2.1. Statistical analysis
Variables were expressed as a mean for standard deviation. To compare groups, Student t test and Mann-Whitney U test were used according to the normality test results assessed by Kolmogorov-Smirnov test. A p value less than 0.05 was considered as significant.
3. Results
There were no statistically significant differences between the groups in terms of age, gender, body surface area, cross-clamp time, body temperature during bypass, hematocrit, and drainage volumes corrected by time and transfusion requirements (Table 1).
| Group I | Group II | p | |
|---|---|---|---|
| Age (y) | 58 ± 8 | 58 ± 10 | NS |
| Sex | 4 females, 10 males | 3 females, 10 males | NS |
| BSA (m²) | 1.84 ± 0.14 | 1.91 ± 0.15 | NS |
| Duration of pump (min) | 69.28 ± 35.37 | 78.38 ± 23.8 | NS |
| Duration of cross clamping (min) | 39.4 ± 21.8 | 43.3 ± 13.6 | NS |
| Temperature (°C) | 29.28 ± 2.19 | 29.23 ± 1.3 | NS |
| HTC in CPB (%) | 21.5 ± 3.34 | 23.76 ± 3.72 | NS |
| 0–4 h drainage (mL) | 338.8 ± 244 | 266.9 ± 167 | NS |
| 4–8 h drainage (mL) | 155.7 ± 75.7 | 132.6 ± 104.2 | NS |
| 8–12 h drainage (mL) | 91.15 ± 104.16 | 105.7 ± 109.5 | NS |
| Post-operative blood transfusion (unit) | 1 ± 1.22 | 0.61 ± 1.19 | NS |
BSA = body surface area; CPB = cardiopulmonary bypass; HTC = hematocrit; NS = not significant.
Total protein, hematocrit, and platelet count values were similar in both of the groups at the same intervals. Albumin levels were significantly lower in Group I at time 2 (Table 2). Group I patients had also significantly higher white blood cell count 24 hours after surgery.
| Group I | Group II | p | |
|---|---|---|---|
| Albumin (mg/dL) | |||
| T1 | 3.60 ± 0.28 | 3.76 ± 0.29 | NS |
| T2 | 2.65 ± 0.23 | 2.97 ± 0.41 | <0.05 |
| Total protein (mg/dL) | |||
| T1 | 7.4 ± 0.50 | 6.93 ± 0.60 | NS |
| T2 | 4.73 ± 0.42 | 4.81 ± 1.09 | NS |
| HTC (%) | |||
| T1 | 40.7 ± 4.46 | 40.69 ± 5.21 | NS |
| T2 | 29.2 ± 4.86 | 32.53 ± 3.62 | NS |
| Platelet count/mL | |||
| T1 | 272,642 ± 69,540 | 231,461 ± 79,234 | NS |
| T2 | 122,714 ± 41,244 | 146,000 ± 50,146 | NS |
| WBC/mL | |||
| T1 | 8240 ± 3035 | 8018 ± 3244 | NS |
| T2 | 12,164 ± 2837 | 11,528 ± 2684 | <0.05 |
HTC = hematocrit; NS = not significant; T1 = following anesthesia induction but just before surgical incision; T2 = at 24th hour from starting point of cardiopulmonary bypass surgery; WBC = white blood cell.
Postoperative total protein, albumin, hematocrit, and platelet counts were significantly lower, and white blood cell counts were significantly higher in both of the groups.
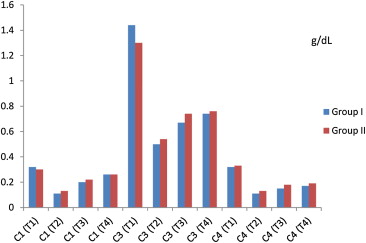
C1, C3, and C4 checked at four different intervals had no statistical difference in both groups (p > 0.05). While C1, C3, and C4 prominently decreased in surgery, a slight increase was observed following CPB ( Fig. 1).
|
|
|
Figure 1. The levels of complement in Groups I and II (g/dL). C = complement; CPB = cardiopulmonary bypass; dL = deciliter; g = gram; T1 = just after the anesthesia induction before surgical incision; T2 = 5 minutes after the removal of cross-clamp; T3 = 6 hours after the start of CPB; T4 = 24 hours after the start of CPB. |
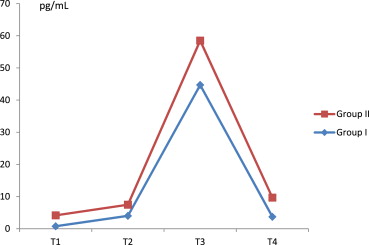
Statistical comparisons and levels of cytokines between Group I and Group II are shown in Table 3. Regarding Group II, the increase of TNF-α in T1–T3 was significant (p < 0.05), while the decrease in T3–T4 was not significant (p > 0.05). In Group I, the increase of TNF-α in T1–T3 was very significant (p < 0.01), while the increase in T1–T4 was less significant (p < 0.05) ( Fig. 2). There was an important difference of TNF-α levels in T4 time between the two groups.
| Cytokines (pg/mL) | Group I | Group II | p |
|---|---|---|---|
| IL-1 (T1) | 0 | 0.0615 ± 0.22 | NS |
| IL-1 (T2) | 0 | 0 | NS |
| IL-1 (T3) | 0 | 0.146 ± 0.36 | NS |
| IL-1 (T4) | 0 | 0.53 ± 1.91 | NS |
| IL-2 (T1) | 0 | 0.348 ± 0.643 | NS |
| IL-2 (T2) | 0 | 0.361 ± 0.83 | NS |
| IL-2 (T3) | 0 | 0.216 ± 0.475 | NS |
| IL-2 (T4) | 0.107 ± 0.305 | 0.103 ± 0.202 | NS |
| IL-6 (T1) | 7.857 ± 25.01 | 1.461 ± 3.619 | NS |
| IL-6 (T2) | 92.642 ± 106.9 | 17.61 ± 24.07 | <0.05 |
| IL-6 (T3) | 608.28 ± 82.62 | 255.38 ± 69.55 | <0.05 |
| IL-6 (T4) | 486.78 ± 153.86 | 285.69 ± 88.2 | <0.05 |
| IL-10 (T1) | 0.528 ± 1.58 | 0.476 ± 1.71 | NS |
| IL-10 (T2) | 35.52 ± 41.01 | 51.64 ± 62.58 | NS |
| IL-10 (T3) | 16.14 ± 33.64 | 26.97 ± 30.03 | <0.05 |
| IL-10 (T4) | 14.028 ± 37.36 | 4.061 ± 5.27 | NS |
| TNF-α (T1) | 0.776 ± 1.67 | 3.39 ± 5.64 | NS |
| TNF-α (T2) | 4.014 ± 11.56 | 3.47 ± 5.36 | NS |
| TNF-α (T3) | 44.71 ± 115.76 | 13.81 ± 17.43 | NS |
| TNF-α (T4) | 3.69 ± 5.79 | 5.97 ± 9.11 | NS |
CPB = cardiopulmonary bypass; mL = milliliter; NS = not significant; pg = picogram; T1 = just after the anesthesia induction before surgical incision; T2 = 5 minutes after the removal of cross clamp; T3 = 6 hours after the start of CPB; T4 = 24 hours after the start of CPB.
|
|
|
Figure 2. TNF-α exchange ratio. CPB = cardiopulmonary bypass; T1 = just after the anesthesia induction before surgical incision; T2 = 5 minutes after the removal of cross-clamp; T3 = 6 hours after the start of CPB; T4 = 24 hours after the start of CPB; TNF-α = tumor necrosis factor alpha. |
Neither IL-1 nor IL-2 was found in the plasma of both the groups. This shows that there was no pattern difference among them.
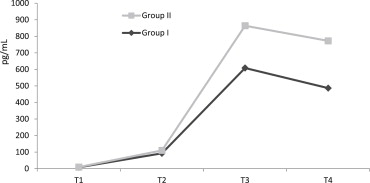
In Group I, there had been a significant increase in IL-6 level (7.857 ± 25.01 pg/mL) from the start of the operation (p < 0.001). In IL-6, we could see a decrease in T3 and T4 (608.28 ± 82.62 and 486.78 ± 153.86 pg/mL) (p < 0.05), but over the T1 value, there was a level of T4. There was a significant increase in IL-10 (p < 0.001). We could see an insignificant decrease in T10 between T3 and T4 (16.14 ± 33.64 and 14.028 ± 37.36 pg/mL) (p > 0.05), but the T4 level was over T1.
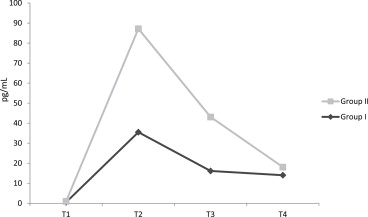
When we look at the cytokines in Group II, we see a significant increase in IL-6 level as the operation is started up (p < 0.05). IL-6 showed an increase in T3–T4 but it was not significant (p < 0.05). The increase of IL-10 in T3–T4 (26.97 ± 30.03 to 4.061 ± 5.27 pg/mL) was significant (p < 0.01).
There was a significant difference between IL-6 in T2, T3, and T4 levels (Fig. 3) and IL-10 in the T3 level (p < 0.001) ( Fig. 4). IL-10 was significantly high in Group II (T3) (p < 0.05). In Group I, IL-6 in T2, T3, and T4 levels were found to be significantly high (p < 0.001).
|
|
|
Figure 3. The levels of IL-6 in Groups I and II (pg/mL). CPB = cardiopulmonary bypass; IL = interleukin; mL = milliliter; pg = picogram; T1 = just after the anesthesia induction before surgical incision; T2 = 5 minutes after the removal of cross-clamp; T3 = 6 hours after the start of CPB; T4 = 24 hours after the start of CPB. |
|
|
|
Figure 4. The levels of IL-10 in Groups I and II (pg/mL). CPB = cardiopulmonary bypass; IL = interleukin; mL = milliliter; pg = picogram; T1 = just after the anesthesia induction before surgical incision; T2 = 5 minutes after the removal of cross-clamp; T3 = 6 hours after the start of CPB; T4 = 24 hours after the start of CPB. |
4. Discussion
Previous studies have suggested that the oxygenator coating type may affect protein absorption and platelet adhesion-aggregation. Moreover, the postoperative clinical course may also be better depending on the oxygenator coating type. The use of biocompatible membranes in the extracorporeal circulation can decrease the side-effects related to blood element-membrane interactions.1 The protein coating provides a hydrophilic surface via electrostatics and van der Waals forces. This electrically inactive surface creates a milieu that prevents fibrinogen binding, leading to platelet activation.
Polypeptide impregnation is a safe and an effective option for oxygenator coating.12 Albumin coating may prevent platelet activation and consumption during CPB only for some time.13
Blood-synthetic membrane interaction in the extracorporeal circulation leads to thrombocytopenia, discharge of platelet granule contents and thromboxane synthesis. Subsequently, subcellular degeneration and a decrease in thrombocyte sensitivity may ensue. The changes in thrombocyte count and functions lengthen the bleeding time and result in significant blood loss. Factor XII is also activated by blood-nonbiological membrane interaction. This activation triggers intrinsic coagulation pathway. Mechanical trauma caused by the oxygenator itself may jeopardize both proteins and platelets in the extracorporeal circulation system. Thrombocyte function and count are further adversely affected by this kind of trauma.14 ; 15 Denaturized proteins increase red blood cell grouping and red cell vulnerability to shear stress.1; 2; 14; 16; 17 ; 18 Thrombocyte and neutrophil activations have an important role in reperfusion injury. White blood cells are very important to define acute inflammation during the first 6-24 hours.19 In our study, we found a decrease in postoperative platelet and hematocrit levels in both groups. The difference between the groups was not significant (p > 0.05).
Postoperative drainage can be affected by several non-surgical factors, such as: platelet dysfunction and inflammatory response. The previous studies for albumin coating showed contradictions on postoperative drainage and transfusion requirements.20 We did not find any significant difference about drainage volume and postoperative transfusion needs in our study.
It was detected that the levels of C3 and C4 decreased during CPB and the levels of C3a and C4a increased because of activation.12 C3a increased with the start of CPB and reached its maximum level at the end of the bypass. C3a level depends on the length of the bypass and reverts back to its standard level after 48 hours. The complements increase with the contact activation, but is devastated by the pump. Therefore, the detection of the initial increase and decrease in the complement levels come into prominence. It is identified that, in the studies carried out using oxygenators coated with protein, the level of C5b-9 (membrane attack complex) is less high compared to the others.21
TNF-α is a potent proinflammatory cytokine. This may turn into harmful mediators when plasma levels are high in pathologic cases such as major trauma, sepsis, and shock.10 It is mostly associated with ischemic–reperfusion damage.17; 22; 23; 24 ; 25 It can lead to hemorrhagic necrosis by its synergistic effect with complement. The increase in TNF-α starts with the beginning of CPB and reaches its maximum value 1 hour after the end of CPB.26 There is some research in the literature that observe that there is no systemic TNF-α secretion in normothermic CPB.27 In some cases, levels of TNF-α remain normal during CPB.27 ; 28 The significant increase in TNF-α is seen at the end of this technique, but in other research, it is shown to occur after the aortic cross-clamp removal. The endogen mediators such as TNF-α, IL-1, and IL-6 are important adhesion molecules during the acute inflammatory phase.29
In Group I of our study, we saw a significant increase in TNF-α after cross-clamp removal, but that showed no relation to the start of CPB. We also got the same result in Group II of the study.
When we looked at TNF-α levels that increased after the cross-clamp removal in both groups, we got results that are in agreement with those in literature. Even though the increase in Group I TNF-α level in T1 and T3 is quite significant, there was no statistical difference between the groups (p > 0.05).
It has been shown that cytokine secretion after cardiac surgery is increased and proinflammatory cytokine response level is proportional to ischemic time.30 Pro-inflammatory cytokines can cause damage to myocardial contractility. Recently, it was observed that there was a relation between the increase in cytokine levels, left ventricular wall damage, and myocardial ischemia.
There was no observed secretion of IL-1β during normothermic CPB and there has been no measurement of IL-1 level in any patient.27 Even after hypothermic bypass, IL-1 was not found in plasma. In both groups of our study, we could not detect any IL-1 and IL-2 levels in plasma. This has made us think that there is not any free IL-1 and IL-2 in plasma. However, it has been considered that during CPB, the levels of these two interleukins increase within the cell and affects the inflammatory response.
In both the groups, we saw a significant increase in IL-6 and IL-10 levels after the start of CPB. Six hours after the start of CPB, there was a significant increase in IL-6 level, while IL-10 levels were observed to be insignificantly decreased. However, in Group II, there was a significant increase in IL-10 levels (p < 0.05), but IL-6 levels were significantly low (p < 0.01). Here we observed the anti-inflammatory IL-10 effect which decreased the IL-6 level. According to these results, the synthetic peptide-coated oxygenator used during CPB seems to have specifically induced the systemic inflammatory response. As a result, we might say that in Group II patients, the IL system seems to be activated toward the induction of the anti-inflammatory response.
The synthetic peptide-coated oxygenator showed an inflammatory effect by increasing the IL-levels in the acute phase. The IL-6 level in Group II seemed to be less increased when compared to Group I. In albumin-coated group, the increase in IL-6 and IL-10 levels in the first phase seems to be statistically significant. Although IL-10 levels were higher at T2 and T3 time points, it was only significantly higher at T3 in Group II. Albumin-coated oxygenators induce, mostly, the inflammatory response, while synthetic peptide-coated oxygenators after the initial phase of CPB induce a significant anti-inflammatory response.
We detected lower postoperative total protein and albumin levels in both groups probably due to protein breakdown and adhesion to the nonbiological membrane during extracorporeal circulation. This decrease reached statistical significance for albumin levels only in the albumin-coated group. This may suggest a less inactive membrane surface in the polypeptide-coated group.
The therapy aiming to equilibrate the inflammatory reactions after the cardiac surgery may decrease the myocardial damage and incidence level of complications. In Group I, IL-6 was found to be high. The substantial increase in the IL-6 level has a more harmful effect on the organism by its pro-inflammatory action when compared to its anti-inflammatory character. In Group II, the significant high level of IL-10 shows that anti-inflammatory character is more effective. Furthermore, the low level of albumin and high level of white blood cells in Group I shows that blood has been exposed to trauma. According to these results, the usage of synthetic polypeptide-coated oxygenator seems to be quite reasonable. We believe that with the improvement of technology, other types of coated oxygenators will be invented, which might lead to a decrease in SIRS in minimal levels.
References
- 1 S. Günaydin; Clinical performance and biocompatibility of novel coating technologies; XVIII Mech Perfus (2003), pp. 15–28
- 2 A.Y. Bozer; Miyokard korunması. Kalp hastalıkları ve Cerrahisi: Hacettepe Üniversitesi Tıp Fakültesi; Toraks ve Kalp Damar Cerrahisi yayını (1985), pp. 125–168
- 3 N.E. Moat, N. Rebuck, D.F. Shore, T.W. Evans, A.H. Finn; Humoral and cellular activation in a simulated extracorporeal circuit; Ann Thorac Surg, 56 (1993), pp. 1509–1514
- 4 Y. Tabardel, J. Duchateau, D. Schmartz, et al.; Corticosteroids increase blood interleukin-10 levels during cardiopulmonary bypass in men; Surgery, 119 (1996), pp. 76–80
- 5 E. Ovrum, T.E. Mollnes, E. Fosse, et al.; Complement and granulocyte activation in two different types of heparinized extracorporeal circuits; J Thorac Cardiovasc Surg, 110 (1995), pp. 1623–1632
- 6 R.A. Moore, E.A. Geller, E.S. Matthews, S.B. Botros, A.B. Jose, D.L. Clark; The effects of hypothermic cardiopulmonary bypass on patients with low-titer, nonspecific cold aglutinin; Ann Thorac Surg, 37 (1984), pp. 233–238
- 7 P. Bruins, H. te Velthuis, A.P. Yazdanbakhsh, et al.; Activation of the complement system during and after cardiopulmonary bypass surgery: Post surgery activation involves C-reactive protein and is associated with postoperative arrhythmia; Circulation, 96 (1997), pp. 3542–3548
- 8 K.A. Shastri, G.L. Logue, M.P. Stern, S. Rehman, S. Raza; Complement activation by heparin-protamine complexes during cardiopulmonary bypass: Effect of C4a null allele; J Thorac Cardiovasc Surg, 114 (1997), pp. 482–488
- 9 R.D. Riley, H. Sato, Z.Q. Zhao, et al.; Recombinant human complement C5a receptor antagonist reduces infarct size following surgical revascularization; Circulation, 96 (1997) I-679
- 10 B. Stephen, B.S. Horton, W. Warwick, et al.; IL-6 and IL-8 levels after cardiopulmonary bypass are not affected by surface coating; Ann Thorac Surg, 68 (1999), pp. 1751–1755
- 11 T. Kawamura, R. Wakusawa, K. Inada; İnterleukin-10 and İnterleukin-1 receptor antagonists increase during cardiac surgery; Can J Anaesth, 44 (1997), pp. 38–42
- 12 G. Wimmer-Greinecker, G. Matheis, S. Martens, G. Oremek, U. Abdel-Rahman, A. Moritz; Synthetic protein treated versus heparin coated cardiopulmonary bypass surfaces: similar clinical results and minor biochemical differences; Eur J Cardio Thorac Surg, 15 (1999), pp. 1–7
- 13 K. Adrian, K. Mellgren, M. Skogby, L.G. Friberg, G. Mellgren; The effect of albumin priming solution on platelet activation during experimental long-term perfusion; Perfusion, 13 (1998), pp. 187–191
- 14 Y. Sawa, Y. Shimazaki, K. Kadoba, et al.; Attenuation of cardiopulmonary bypass-derived inflammatory reactions reduces myocardial reperfusion injury in cardiac operations; J Thorac Cardiovasc Surg, 111 (1996), pp. 29–35
- 15 D.R. Jones, R.C. Hill, M.J. Hollingsed, et al.; Use of heparin-coated cardiopulmonary bypass; Ann Thorac Surg, 56 (1993), pp. 566–568
- 16 K.M. Mullane, S. Moncada; The salvage of ischemic myocardium by BW 755 C in anaesthetized dogs; Prostaglandins, 24 (1982), pp. 255–266
- 17 A.P. Metinko, S.L. Kunkel, T.J. Standiford, R.M. Strieter; Anoxia-hiperoxia induces monocyte derived interleukin-8; J Clin Invest, 90 (1992), pp. 791–798
- 18 J. Butler, G.M. Rocker, S. Westaby; Inflammatory response to cardiopulmonary bypass; Ann Thorac Surg, 55 (1993), pp. 552–559
- 19 L.C. Junqueira, C. Carneiro; Histologia básica; (10th ed.)Guanabara Koogan, Rio de Janeiro (2004), pp. 223–237
- 20 O. Moen, E. Fosse, V. Brockmeier, et al.; Disparity in blood activation by two different heparin-coated cardiopulmonary bypass systems; Ann Thorac Surg, 60 (1995), pp. 1317–1323
- 21 R.D. Dowling, M.E. Brown, R.O. Whittington Jr., J.J. Quinlan, J.M. Armitage; Clinical cardiopulmonary bypass without systemic anticoagulation; Ann Thorac Surg, 56 (1993), pp. 1176–1178
- 22 T. Sakai, T.W. Latson, C.W. Whitten, et al.; Perioperative measurements of interleukin-6 and alpha-melanocyte-stimulating hormone in cardiac transplant patients; J Cardiothorac Vasc Anesth, 7 (1993), pp. 17–22
- 23 D.E. Hammerschmidt, D.F. Stroncek, T.K. Bowers, et al.; Complement activation and neutropenia occurring during cardiopulmonary bypass; J Thorac Cardiovasc Surg, 81 (1981), pp. 370–377
- 24 S. Wan, J.L. Leclere, J.L. Vincent; Inflammatory response to cardiopulmonary bypass. mechanisms involved and possible therapeutic strategies; Chest, 112 (1997), pp. 676–692
- 25 O.A. Khair, R.J. Davies, J.L. Devalia; Bacterial-induced release of inflammatory mediators by bronchial epithelial cells; Eur Respir J, 9 (1996), pp. 1913–1922
- 26 C. Larry, M.D. Casey; Role of cytokines in the pathogenesis of cardiopulmonary-induced multisystem organ failure; Ann Thorac Surg, 56 (1993), pp. 92–96
- 27 B. Frering, I. Philip, M. Dehoux, C. Rolland, J.M. Langlois, J.M. Desmonts; Circulating cytokines in patients undergoing normothermic cardiopulmonary bypass; J Thorac Cardiovasc Surg, 108 (1994), pp. 636–641
- 28 A. Finn, S. Naik, N. Klein, R.J. Levinsky, S. Strobel, M. Elliot; İnterleukin-8 release and neutrophil degranulation after pediatric cardiopulmonary bypass; J Thorac Cardiovasc Surg, 105 (1993), pp. 234–241
- 29 F. Ieda, M. José, S. Martin, Y. Winston Bonetti; Leukocytes and the inflammatory response in ischemia-reperfusion injury; Rev Bras Cir Cardiovasc, 25 (2010), pp. 575–584
- 30 S. Wan, J.M. Desmet, L. Barvais, M. Goldstein, J. Vincent, J.L. Leclere; Myocardium is a major source of proinflammatory cytokines in patients undergoing cardiopulmonary bypass; Thorac Cardiovasc Surg, 112 (1996), pp. 806–811
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?