Abstract
Introduction
Renal cell tumors comprise both benign – oncocytoma – and malignant – clear cell renal cell carcinoma, papillary renal cell carcinoma, and chromophobe renal cell carcinoma – entities. Since the differential diagnosis among renal cell tumors is sometimes difficult on clinical, imaging and pathological grounds, and prognosis is quite dissimilar, epigenetic-based diagnostic biomarkers, specially promoter methylation, might be useful for accurate diagnosis and therapeutic planning.
Materials and methods
EpiTect Methyl II PCR Array was used to screen methylation status of 22 genes, involved in epithelial to mesenchymal transition. Quantitative real-time methylation specific polymerase chain reaction was performed for candidate gene validation, and methylation levels of renal cell tumors subtypes and normal kidney were determined and compared.
Results
MST1R promoter methylation level was significantly higher in clear cell renal cell carcinoma (median: 5367) compared to other renal cell tumors (median: papillary renal cell carcinoma – 1084, chromophobe renal cell carcinoma – 1023, oncocytoma – 1337) and normal kidney (median: 1125), allowing for accurate discrimination from other renal cell tumors with high sensitivity (>96.7%) and specificity (86.7%).
Conclusion
Quantitative MST1R promoter methylation may be useful as biomarker for accurate diagnosis of clear cell renal cell carcinoma in problematic cases.
Resumo
Introdução
Os tumores de células renais englobam neoplasias benignas – oncocitoma – e malignas – carcinoma de células renais de tipo célula clara, papilar e de células cromófobas. Uma vez que o diagnóstico diferencial entre eles não é linear do ponto de vista clínico, imagiológico e patológico, e que o prognóstico de cada subtipo é distinto, o desenvolvimento de biomarcadores de diagnóstico com base em alterações epigenéticas, nomeadamente a metilação do promotor de genes, poderá ser útil para o diagnóstico e planeamento do tratamento.
Material e métodos
A presença de metilação do promotor de 22 genes envolvidos na transição epitélio-mesênquima foi avaliada através da plataforma comercial EpiTectMethylIIPCR Array. Os resultados foram validados, no gene candidato, por “polymerase chain reaction” quantitativo em tempo real específico para metilação. Os níveis de metilação de cada tipo histológico de tumores de células renais e de tecido renal normal foram calculados e comparados.
Resultados
O nível de metilação do promotor do gene MST1R foi significativamente mais elevado nos carcinomas de células renais de tipo célula clara (mediana: 5.367) comparativamente com carcinomas de células renais papilares (mediana: 1.084), carcinomas de células renais de células cromófobas (mediana: 1.023), oncocitomas (mediana: 1.337) e rim normal (mediana: 1.125), permitindo identificar carcinomas de células renais de tipo célula clara com elevada sensibilidade (> 96,7%) e especificidade (86,7%).
Conclusão
O nível de metilação do promotor do gene MST1R poderá constituir um biomarcador útil para o diagnóstico de carcinomas de células renais de tipo célula clara em casos problemáticos.
Keywords
Kidney neoplasms ; Epigenetics ; Biomarkers
Palavras-chave
Neoplasias renais ; Epigenética ; Biomarcadores
Introduction
Renal cancer has an estimated age-standardized incidence and mortality rate respectively of 4.4/100,000 and 1.8/100,000 worldwide.1 In Europe, renal cancer incidence ranks seventh and eighth among all non-cutaneous malignant neoplasms, and in North America kidney and renal pelvis cancers account for 65,150 estimated new cases and 13,680 deaths in both genders in 2013, with men being more affected than women.2 Besides the classical presentation as metastatic disease, mostly because kidney cancer is asymptomatic at its earliest stages, an increasingly higher number of tumors are being incidentally discovered as small masses, frequently posing diagnostic challenges in what concerns the distinction between benign and malignant tumors.3
Renal cell tumors are the most common neoplasms of the kidney (80–85%), followed by urothelial carcinoma of the renal pelvis (15–20%).4 Renal cell tumors (RCTs) are heterogeneous at the genetic, morphological and clinical levels, comprising both benign and malignant entities.5 ; 6 Underscoring the high heterogeneity of RCT, malignant tumors – renal cell carcinomas (RCC) – might have different responses to novel targeted therapies.7 The most frequent malignant RCT is clear cell renal cell carcinoma (ccRCC), followed by papillary renal cell carcinoma (pRCC) and chromophobe renal cell carcinoma (chRCC), accounting respectively for 70%, 10–15% and 5% of all RCT.4 Oncocytoma, the most common benign tumor, accounts for approximately 5% of all RCT.5
Histological subtyping provides relevant prognostic information, independent from tumor pathological stage and grade: ccRCC is the most aggressive subtype, displaying lower cancer-specific survival than pRCC and chRCC, whereas no differences in cancer specific survival are apparent between pRCC and chRCC.8 ; 9
Usually, information on histological subtype is available only after pathological evaluation of the nephrectomy specimen, and thus this valuable diagnostic and prognostic data cannot be taken into account for planning the best time for surgery, especially in cases of small renal tumors (<4 cm) that should benefit from nephron sparing therapy.3 ; 10 For these small tumors, limitations of imagiological techniques and core biopsy histopathological examination preclude accurate characterization of some lesions, seriously limiting the pre-operative information about the biological behavior of the tumor available for the urologist.3 ; 10 Moreover, in the setting of active surveillance protocols,11 ; 12 the development of novel diagnostic biomarkers, preferentially minimally- or non-invasive, is of paramount importance.
The association between both genetic and epigenetic alterations and renal cancer development is widely acknowleged.4 ; 13 ; 14 Additionally, specific genetic and/or epigenetic alterations were found to be characteristic of certain tumor types, and therefore may act as specific diagnostic biomarkers.13 ; 14 Epigenetic biomarkers, such as methylation status of a gene promoter (promoter hipermethylation), which is associated with repression of gene expression, may be detected in the patients serum or urine.15 ; 16 Thus, it may constitute important pre-operative non-invasive diagnostic tools and/or useful ancillary test for the diagnosis of renal masses through fine needle aspiration cytology or tissue biopsy, allowing for the discrimination between benign and malignant RCT and/or for the identification of more aggressive malignant tumors.
Tumor aggressiveness in the form of local invasion and acquisition of metastatic potential has been associated with epithelial to mesenchymal transition (EMT).17 Key EMT effectors were described to be regulated by epigenetic mechanisms,18 ; 19 and EMT-related genes were found to be deregulated in renal cell tumors.14 Thus, epigenetic deregulation of cellular pathways involved in EMT might account for differences in RCTs aggressiveness, and provide useful diagnostic and prognostic biomarkers.
Therefore, we aimed to systematically evaluate the methylation status of EMT-related genes in the four most frequent RCT subtypes and investigate their potential use as clinically relevant diagnostic and prognostic tools.
Methods
Patients, sample collection and DNA extraction
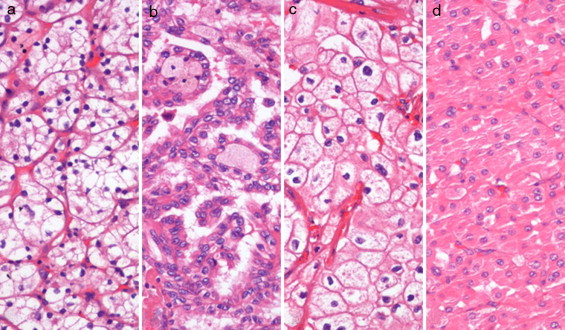
Tumor tissue from 120 total or partial nephrectomy specimens performed between 2003 and 2007 (30 of each ccRCC, pRCC, chRCC and oncocytoma) (Fig. 1 ), and morphologically normal kidney (cortical) tissue from 9 nephrectomy specimens for upper urinary tract neoplasia and 12 renal biopsies performed in the diagnostic work-up of renal dysfunction (without neoplasia or inflammation), were included in this study. All procedures were performed at the Portuguese Oncology Institute – Porto (Portugal), after informed consent was obtained.
|
|
|
Figure 1. Renal cell tumors (RCTs) morphology: (a) clear cell renal cell carcinoma (ccRCC), (b) papillary renal cell carcinoma (pRCC), (c) chromophome renal cell carcinoma (chRCC) and (d) oncocytoma (hematoxylin and eosin staining 200×). |
Tumor tissue samples were obtained immediately after surgery and snap-frozen. Renal biopsies were frozen as part of the routine protocol. All samples were stored at −80 °C and subsequently cut in a cryostat. The presence and extent of tumor were evaluated by H&E stains, to ensure at least 70% of tumor in the samples.
Genomic DNA was extracted as previously described.20 Briefly, DNA was digested overnight with proteinase K (20 mg/mL) in the presence of 10% SDS at 55 °C, extracted with phenolchloroform and precipitated with 100% ethanol.
Tumor classification (WHO), grading (Fuhrman) and staging (TNM) were routinely assessed for all tumor cases in formalin-fixed paraffin-embedded tissue.5 ; 21
Relevant clinical data were collected from clinical charts. This study was approved by the Institutional Review Board (Comissão de Ética para a Saúde) of Portuguese Oncology Institute – Porto, Portugal (CES518/2010).
EpiTect Methyl II qPCR array
The EpiTect Methyl II PCR Array (SABiosciences, Qiagen, Frederick, MD, USA) was used as a screening method to evaluate the promoter methylation status of EMT-related genes in 20 samples (4 ccRCC; 4 pRCC; 6 chRCC; 6 oncocytoma). The EMT commercial assay (cat. no. 524EAHS-901ZA-24) includes 22 genes: CDH1 , CTNNAL1 , DSC2 , DSP , EPCAM , GAB1 , KRT19 , KRT7 , MAP3K5 , MST1R , NID2 , OCLN , PLEK2 , PLSCR1 , PPPDE2 , PTP4A1 , RGS2 , SEH1L , SMAD4 , TGIF1 , TSPAN13 and YES1 .
Assays were performed according to the manufacturers instructions. Briefly, equal amounts of genomic DNA were incubated overnight at 37 °C with a DNA methylation-sensitive restriction enzyme, which digests unmethylated DNA; with a DNA methylation-dependent restriction enzyme that digests methylated DNA; with both enzymes; and without enzyme added/“mock” (Methyl-Profiler DNA Methylation Enzyme Kit, SABiosciences). The enzyme was inactivated at 65 °C for 20 min and, after digestion, the remaining DNA was quantified by real-time PCR using pre-designed primers to the promoter region. DNA amplification was carried on a 7000 Sequence Detection System (Applied Biosystems, Foster City, CA, USA), at 95 °C for 10 min followed by 40 cycles of 97 °C for 15 s and 72 °C for 1 min. PCR product was marked with SYBR® Green and Ct values were obtained. The analysis was performed using a SABiosciences Excel-Based Data Analysis Template, and the percent of hypermethylated DNA was obtained by comparing the amount of DNA in each digest with that of a mock digest, representing the fraction of input DNA containing at least two methylated CpG sites in the targeted gene region.
Bisulfite treatment
Sodium bisulfite converts unmethylated cytosine residues to uracil, whereas methylated cytosine residues remained as such. It was performed with EZ DNA Methylation-Gold Kit (Zymo Research), according to the manufacturers instructions, in a total of 120 tumor samples and 21 morphologically normal kidney tissues, as mentioned above.
Quantitative MSP
Quantitative real-time polymerase chain reaction (qMSP) was performed for all the tested samples after DNA bisulfite treatment.
Primers were designed to amplify methylated bisulfite converted complementary sequences of MST1R promoter using Methyl Primer Express v 1.0 (Applied Biosystems, Foster City, CA, USA). Primer sequences used for Macrophage-stimulating-1 receptor (MST1R ) [GenBank: NM_002447 ] were 5′GCGAGGATTGGTAGTGTTC3′ (forward) and 5′TTCTATCGCCCTCGTAAATC3′ (reverse). A reference gene (β-actin ) was used to normalize for DNA input in each sample.
QMSP analysis was performed using an 7500 Real-time PCR system (Applied Biosystems, Foster City, CA, USA), in a reaction volume of 20 μL consisting of 10 μL of SYBR® Green PCR Master Mix (Applied Biosystems, Foster City, CA, USA), 7 μL of H2 O, 0.5 μL of forward primer, 0.5 μL of reverse primer and 2 μL of bisulfate-modified DNA. Each sample was run in triplicate and “no template controls” were included in each plate as a control for contamination. A calibration curve to quantify the amount of fully methylated alleles in each reaction was constructed with serial dilutions (1:5) of bisulfite converted universally methylated DNA at all CpGs (CpGenome Universal Methylated DNA; Millipore, Billerica, MA). The amplification reaction was carried out at 95 °C for 2 min followed by 50 cycles of 95 °C for 15 s and annealing temperature (60 °C) for 1 min, followed by melting curve analysis.
Relative levels of methylated promoter DNA in each sample were determined by dividing the mean quantity obtained by qMSP analysis for each gene for the respective value of the internal reference gene (ACTB). This value was multiplied by 1000 for easy tabulation (methylation level = target gene/reference gene × 1000).
Statistical analysis
The frequency of methylated samples was determined for the all four RCT types, considering the highest value determined in the normal kidney tissue as the cutoff. Both median and interquartile ranges of methylation levels were also determined. Differences in methylation levels among RCT subtypes were analyzed using Kruskal–Wallis non-parametric ANOVA followed by Mann–Whitney U test, when appropriate. p values in multiple comparisons were adjusted according to the Bonferroni method. Statistical significance level was set at p < 0.05 (two-sided). Disease specific survival and disease free survival curves (Kaplan–Meier with log-rank test) were computed for histological subtype and MST1R promoter methylation levels. Statistical analysis was performed using IBM® SPSS® Statistics for Windows, version 22.0 (SPSS, Chicago, IL, USA).
Results
Promoter methylation screening with EpiTect Methyl II PCR Array
Five out of the 22 EMT-related genes tested – CDH1 , DSC2 , KRT7 , MST1R and TSPAN13 – displayed hypermethylation at the respective promoter regions. Among these, MST1R depicted the highest percent of hypermethylated DNA – >90% – meaning that at least two CpG sites in the targeted gene region were methylated in more than 90% of input DNA sample. Thus, MST1R promoter metylation was chosen for subsequent validation.
MST1R promoter methylation analysis by qMSP
Relevant clinical and pathological data of the 120 tumors included in this study are depicted in Table 1 . No statistically significant associations between MST1R promoter methylation levels and pathological stage were found (p = 0.06). However, MST1R methylation levels of Fuhrman grade 3 tumors (median: 1451; range: 401–12,367) were higher than those with Fuhrman grade 4 (median: 938, range: 718–7085), and this difference attained statistical significance (p = 0.011).
| Renal cell tumors | Normal kidney | |
|---|---|---|
| Number of patients, n | 120 | 21 |
| Age at diagnosis, median (range) | 60 (29–83) | 60 (19–83) |
| Gender, n (%) | ||
| Male | 70 (58.3) | 11 (52.4) |
| Female | 50 (41.7) | 10 (47.6) |
| Pathological stage, n (%) | n.a. | |
| pT1 | 46 (38.4) | |
| pT2 | 19 (15.8) | |
| pT3 | 25 (20.8) | |
| pT4 | – | |
| n.a. (oncocytoma) | 30 (25.0) | |
| Fuhrman grade, n (%) | n.a. | |
| 1 | 3 (2.5) | |
| 2 | 28 (23.3) | |
| 3 | 43 (35.9) | |
| 4 | 16 (13.3) | |
| n.a. (oncocytoma) | 30 (25.0) | |
n.a., not applicable.
To categorize samples as methylated or unmethylated, a cutoff value was chosen based on the highest methylation ratio value obtained for the respective normal/control samples, ensuring perfect specificity of the assay. We found that in ccRCC the methylation frequency was 60% and, in the remaining histological subtypes, methylation frequency was very low (1%) in pRCC, and absent in chRCC and oncocytoma. The most aggressive RCC subtype, ccRCC, displayed higher MST1R methylation levels compared to pRCC, chRCC and oncocytomas ( Table 2 ), and this was statistically significant (Table 3 ). MST1R methylation levels of chRCC also significantly differed from those of oncocytoma (p = 0.008, Table 3 ).
| Methylation levela median (range) | Methylation frequencyb (%) | |
|---|---|---|
| ccRCC | 5367 (530–12368) | 18/30 (60%) |
| pRCC | 1084 (0–7085) | 1/30 (3%) |
| chRCC | 1023 (524–1701) | 0/30 (0%) |
| Oncocytoma | 1337 (805–2139) | 0/30 (0%) |
| Normal kidney | 1125 (627–4917) | 0/21 (0%) |
ccRCC, clear cell renal cell carcinoma; pRCC, papillary renal cell carcinoma; chRCC, chromophobe renal cell carcinoma.
a. Methylation level = target gene (MST1R ) quantity mean/reference gene (ACTB ) quantity mean × 1000.
b. A sample is considered methylated when its methylation level is higher than the highest methylation level in normal/control samples (4917). Methylation frequency = number of samples methylated/total of samples, for each histological subtype and normal kidney.
| p value* | ||||
|---|---|---|---|---|
| ccRCC | pRCC | chRCC | Oncocytoma | |
| Normal kidney | <0.001 | 0.108 | 0.066 | 0.716 |
| Oncocytoma | <0.001 | 0.128 | 0.001 | – |
| chRCC | <0.001 | 0.535 | – | – |
| pRCC | <0.001 | – | – | – |
- . Mann–Whitney test, Bonferronis correction (statistically significant when p < 0.01). ccRCC, clear cell renal cell carcinoma; pRCC, papillary renal cell carcinoma; chRCC, chromophobe renal cell carcinoma; n.s., not significant.
MST1R methylation levels allowed for the discrimination between ccRCC and the remainder RCT – pRCC, chRCC and oncocytoma – with high sensitivity (96.7%, 100%, 100%, respectively) and specificity (86.7% for the three comparisons), as well as between chRCC and oncocytoma, with 96.7% sensitivity and 53.3% specificity (Table 4 ).
| % | ccRCC vs. | chRCC vs. | ||
|---|---|---|---|---|
| pRCC | chRCC | Oncocytoma | Oncocytoma | |
| Sensitivity | 96.7 | 100 | 100 | 96.7 |
| Specificity | 86.7 | 86.7 | 86.7 | 53.3 |
| PPV | 87.9 | 88.2 | 88.2 | 67.4 |
| NPV | 96.3 | 100 | 100 | 94.1 |
| Accuracy | 91.7 | 93.3 | 93.3 | 75 |
PPV, positive predictive value; NPV, negative predictive value; ccRCC, clear cell renal cell carcinoma; pRCC, papillary renal cell carcinoma; chRCC, chromophobe renal cell carcinoma.
MST1R promoter methylation levels did not associate with cancer-specific survival (p = 0.21) nor disease-free survival (p = 0.091).
Discussion
Macrophage-stimulating-1 receptor (MST1R ), also known as Ron (Recepteur d’Origine Nantais) is a receptor tyrosine kinase, member of the MET proto-oncogene family. MST1R binds macrophage stimulating protein (MSP) and is found in macrophages, epithelial cells, osteoclasts and hematopoietic cells. 22 ; 23 ; 24 In normal epithelial cells, MST1R is involved not only in cell proliferation and survival (inhibition of apoptosis) but also in integrin-dependent cell adhesion and motility. 22 ; 23 ; 24 MSP/MST1R signaling deregulation was found in cancer cells, 25 ; 26 ; 27 and MST1R altered expression was reported in several human neoplasms, including hepatocellular, 28 breast,29 colon,30 lung,31 ovarian,32 nasopharyngeal33 and bladder34 cancer, but not in renal cancer. Several oncogenic MST1R variants have been described, 25 ; 26 ; 27 and hypermethylation of a specific region of MST1R promoter was reported to be associated with the transcription of an oncogenic MST1R variant, 35 but MST1R promoter methylation had not been previously described in RCTs.
In this study, we showed that quantitative MST1R promoter methylation is a common feature of ccRCC, and its levels are significantly higher than those observed in pRCC, chRCC, oncocytoma and normal renal tissue, suggesting not only a role in renal tumorigenesis but also a specific implication in the genesis of the clear cell type. Furthermore, the distribution of MST1R promoter methylation levels allowed for accurate discrimination of ccRCC from other RCT, a finding that is of particular diagnostic relevance. It had been previously reported that the promoter methylation level of a panel of three genes – CDH1 , PTGS2 and RASSF1A – could discriminate the four main histological types of RCT, although with limited sensitivity. 36 Thus, either alone or in conjunction with that gene panel, MST1R promoter methylation might provide a useful ancillary tool for the histopathological or cytopathological evaluation of suspicious renal masses.
Although previous studies on RCT identified promoter hypermethylation of several genes37 ; 38 using genome-wide promoter methylation analysis, data validation in an independent clinical series of samples is mostly lacking, impairing the evaluation of its usefulness as diagnostic biomarkers. On the other hand, most studies are focused in ccRCC, the most common RCT subtype, but increasing attention must be paid to diagnostic biomarkers which allow for the discrimination among RCTs, especially chRCC and oncocytoma, as diagnosis carries quite different prognosis and, eventually, therapeutic options. Global methylation profiles of chRCC and oncocytoma have been published,39 but again without validation in independent set of clinical samples. Although MST1R promoter methylation surfaced in the array as promising biomarker, we further extended our analysis to primary RCT and normal kidney tissues, to assess its potential clinical usefulness. Furthermore, this analysis allowed for the estimation of the sensitivity and specificity of this novel epigenetic-based biomarker, which depicted high diagnostic performance. However, because it does not allow for the coverage of all relevant differential diagnostic problems in RCT, the integration with other biomarkers, such as the microRNA that we recently demonstrated to distinguish among RCT subtypes,40 might be relevant, especially in the setting of patient selection for surveillance protocols. Importantly, these molecular techniques might be performed in clinical samples obtained by non- or minimally-invasive procedures, such as urine or serum,16 or serve as ancillary tools for fine needle aspiration cytology (FNAC), as previously shown.40
This study was performed in tumor tissue after nephrectomy, which limits the scope of this new diagnostic biomarker. Further validation of MST1R promoter methylation in urine and/or serum samples is warranted. Additionally, at present, the precise role of MST1R promoter methylation in ccRCC carcinogenesis is not known, nor is its relevance for the development of non-ccRCC. Elucidation of the biological role of MST1R in renal carcinogenesis might allow for the development of novel therapeutic strategies, as tyrosine kinase inhibitors that act on MST1R might modulate its effects on neoplastic cells. 41 ; 42
Conclusions
MST1R methylation levels might be a useful diagnostic biomarker, allowing for the discrimination of ccRCC from other RCT and normal renal tissue, with high sensitivity and specificity. Further studies are warranted to independently assess MST1R promoter methylation level as a new diagnostic biomarker for RCTs, and also to illuminate the role of MST1R promoter methylation in renal carcinogenesis.
Ethical disclosures
Protection of human and animal subjects
The authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of data
The authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consent
The authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
Conflicts of interest
The authors have no conflicts of interest to declare.
Funding
Bolsa de Investigação da Associação Portuguesa de Urologia (2010) & CIPOP 4 – 2008. ASP-L is supported by FCT - Fundação para a Ciência e Tecnologia-SFRH/SINTD/94217/2013.
References
- 1 J. Ferlay, I. Soerjomataram, M. Ervik, R. Dikshit, S. Eser, C. Mathers, et al.; GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 [Internet]; International Agency for Research on Cancer, Lyon, France (2013) Available from: http://globocan.iarc.fr [accessed 07.09.14]
- 2 R. Siegel, D. Naishadham, A. Jemal; Cancer statistics, 2013; CA Cancer J Clin, 63 (2013), pp. 11–30
- 3 I.S. Gill, M. Aron, D.A. Gervais, M.A. Jewett; Clinical practice. Small renal mass; New Engl J Med, 362 (2010), pp. 624–634
- 4 M.M. Baldewijns, I.J. van Vlodrop, L.J. Schouten, P.M. Soetekouw, A.P. de Bruine, M. van Engeland; Genetics and epigenetics of renal cell cancer; Biochim Biophys Acta, 1785 (2008), pp. 133–155
- 5 S.G. Eble JN, J.I. Epstein, I.A. Sesterhenn; Pathology and genetics of tumours of the urinary system and male genital organs; World Health Organization Classification of Tumours, Lyon (2004)
- 6 F. Algaba, H. Akaza, A. Lopez-Beltran, G. Martignoni, H. Moch, R. Montironi, et al.; Current pathology keys of renal cell carcinoma; Eur Urol, 60 (2011), pp. 634–643
- 7 R. Fisher, M. Gore, J. Larkin; Current and future systemic treatments for renal cell carcinoma; Semin Cancer Biol, 23 (2013), pp. 38–45
- 8 J.C. Cheville, C.M. Lohse, H. Zincke, A.L. Weaver, M.L. Blute; Comparisons of outcome and prognostic features among histologic subtypes of renal cell carcinoma; Am J Surg Pathol, 27 (2003), pp. 612–624
- 9 A. Lopez-Beltran, J.C. Carrasco, L. Cheng, M. Scarpelli, Z. Kirkali, R. Montironi; 2009 update on the classification of renal epithelial tumors in adults; Int J Urol, 16 (2009), pp. 432–443
- 10 A. Volpe, J.A. Cadeddu, A. Cestari, I.S. Gill, M.A. Jewett, S. Joniau, et al.; Contemporary management of small renal masses; Eur Urol, 60 (2011), pp. 501–515
- 11 M.C. Smaldone, A. Kutikov, B.L. Egleston, D.J. Canter, R. Viterbo, D.Y. Chen, et al.; Small renal masses progressing to metastases under active surveillance: a systematic review and pooled analysis; Cancer, 118 (2012), pp. 997–1006
- 12 P.L. Crispen, R. Viterbo, S.A. Boorjian, R.E. Greenberg, D.Y. Chen, R.G. Uzzo; Natural history, growth kinetics, and outcomes of untreated clinically localized renal tumors under active surveillance; Cancer, 115 (2009), pp. 2844–2852
- 13 M. Esteller; Epigenetics in cancer; New Engl J Med, 358 (2008), pp. 1148–1159
- 14 R. Henrique, A.S. Luis, C. Jeronimo; The epigenetics of renal cell tumors: from biology to biomarkers; Front Genet, 3 (2012), p. 94
- 15 R. Henrique, V.L. Costa, C. Jeronimo; Methylation-based biomarkers for early detection of urological cancer; Crit Rev Oncogen, 13 (2007), pp. 265–282
- 16 M.O. Hoque, S. Begum, O. Topaloglu, C. Jeronimo, E. Mambo, W.H. Westra, et al.; Quantitative detection of promoter hypermethylation of multiple genes in the tumor, urine, and serum DNA of patients with renal cancer; Cancer Res, 64 (2004), pp. 5511–5517
- 17 R. Kalluri, R.A. Weinberg; The basics of epithelial–mesenchymal transition; J Clin Investig, 119 (2009), pp. 1420–1428
- 18 M. Cieslik, S.A. Hoang, N. Baranova, S. Chodaparambil, M. Kumar, D.F. Allison, et al.; Epigenetic coordination of signaling pathways during the epithelial-mesenchymal transition; Epigenet Chromatin, 6 (2013), p. 28
- 19 L.J. Talbot, S.D. Bhattacharya, P.C. Kuo; Epithelial-mesenchymal transition, the tumor microenvironment, and metastatic behavior of epithelial malignancies; Int J Biochem Mol Biol, 3 (2012), pp. 117–136
- 20 P. Patricio, J. Ramalho-Carvalho, P. Costa-Pinheiro, M. Almeida, J.D. Barros-Silva, J. Vieira, et al.; Deregulation of PAX2 expression in renal cell tumours: mechanisms and potential use in differential diagnosis; J Cell Mol Med, 17 (2013), pp. 1048–1058
- 21 S. Edge, D.R. Byrd, C.C. Compton, A.G. Fritz, F.L. Greene, A. Trotti; AJCC cancer staging manual; (7th ed.)Springer, New York (2010)
- 22 A. Danilkovitch, E.J. Leonard; Kinases involved in MSP/RON signaling; J Leukoc Biol, 65 (1999), pp. 345–348
- 23 A. Danilkovitch, S. Donley, A. Skeel, E.J. Leonard; Two independent signaling pathways mediate the antiapoptotic action of macrophage-stimulating protein on epithelial cells; Mol Cell Biol, 20 (2000), pp. 2218–2227
- 24 D. Angeloni, A. Danilkovitch-Miagkova, S.V. Ivanov, R. Breathnach, B.E. Johnson, E.J. Leonard, et al.; Gene structure of the human receptor tyrosine kinase RON and mutation analysis in lung cancer samples; Genes Chromosomes Cancer, 29 (2000), pp. 147–156
- 25 A. Danilkovitch-Miagkova; Oncogenic signaling pathways activated by RON receptor tyrosine kinase; Curr Cancer Drug Targets, 3 (2003), pp. 31–40
- 26 Y. Lu, H.P. Yao, M.H. Wang; Multiple variants of the RON receptor tyrosine kinase: biochemical properties, tumorigenic activities, and potential drug targets; Cancer Lett, 257 (2007), pp. 157–164
- 27 M.H. Wang, R. Zhang, Y.Q. Zhou, H.P. Yao; Pathogenesis of RON receptor tyrosine kinase in cancer cells: activation mechanism, functional crosstalk, and signaling addiction; J Biomed Res, 27 (2013), pp. 345–356
- 28 Q. Chen, D.W. Seol, B. Carr, R. Zarnegar; Co-expression and regulation of Met and Ron proto-oncogenes in human hepatocellular carcinoma tissues and cell lines; Hepatology, 26 (1997), pp. 59–66
- 29 P. Maggiora, S. Marchio, M.C. Stella, M. Giai, A. Belfiore, M. De Bortoli, et al.; Overexpression of the RON gene in human breast carcinoma; Oncogene, 16 (1998), pp. 2927–2933
- 30 W.S. Chen, H.J. Kung, W.K. Yang, W. Lin; Comparative tyrosine-kinase profiles in colorectal cancers: enhanced arg expression in carcinoma as compared with adenoma and normal mucosa; Int J Cancer J, 83 (1999), pp. 579–584
- 31 C.G. Willett, M.H. Wang, R.L. Emanuel, S.A. Graham, D.I. Smith, V. Shridhar, et al.; Macrophage-stimulating protein and its receptor in non-small-cell lung tumors: induction of receptor tyrosine phosphorylation and cell migration; Am J Respir Cell Mol Biol, 18 (1998), pp. 489–496
- 32 P. Maggiora, A. Lorenzato, S. Fracchioli, B. Costa, M. Castagnaro, R. Arisio, et al.; The RON and MET oncogenes are co-expressed in human ovarian carcinomas and cooperate in activating invasiveness; Exp Cell Res, 288 (2003), pp. 382–389
- 33 Y.C. Chou, C.L. Chen, T.H. Yeh, S.J. Lin, M.R. Chen, S.L. Doong, et al.; Involvement of recepteur d’origine nantais receptor tyrosine kinase in Epstein-Barr virus-associated nasopharyngeal carcinoma and its metastasis; Am J Pathol, 181 (2012), pp. 1773–1781
- 34 H.L. Cheng, H.S. Liu, Y.J. Lin, H.H. Chen, P.Y. Hsu, T.Y. Chang, et al.; Co-expression of RON and MET is a prognostic indicator for patients with transitional-cell carcinoma of the bladder; Br J Cancer, 92 (2005), pp. 1906–1914
- 35 D. Angeloni, A. Danilkovitch-Miagkova, T. Ivanova, E. Braga, E. Zabarovsky, M.I. Lerman; Hypermethylation of Ron proximal promoter associates with lack of full-length Ron and transcription of oncogenic short-Ron from an internal promoter; Oncogene, 26 (2007), pp. 4499–4512
- 36 V.L. Costa, R. Henrique, F.R. Ribeiro, M. Pinto, J. Oliveira, F. Lobo, et al.; Quantitative promoter methylation analysis of multiple cancer-related genes in renal cell tumors; BMC Cancer, 7 (2007), p. 133
- 37 C.J. Ricketts, M.R. Morris, D. Gentle, M. Brown, N. Wake, E.R. Woodward, et al.; Genome-wide CpG island methylation analysis implicates novel genes in the pathogenesis of renal cell carcinoma; Epigenetics, 7 (2012), pp. 278–290
- 38 I. Ibragimova, M.J. Slifker, M.E. Maradeo, G. Banumathy, E. Dulaimi, R.G. Uzzo, et al.; Genome-wide promoter methylome of small renal masses; PLOS ONE, 8 (2013), p. e77309
- 39 A.A. Slater, M. Alokail, D. Gentle, M. Yao, G. Kovacs, E.R. Maher, et al.; DNA methylation profiling distinguishes histological subtypes of renal cell carcinoma; Epigenetics, 8 (2013), pp. 252–267
- 40 R.M. Silva-Santos, P. Costa-Pinheiro, A. Luis, L. Antunes, F. Lobo, J. Oliveira, et al.; MicroRNA profile: a promising ancillary tool for accurate renal cell tumour diagnosis; Br J Cancer, 109 (2013), pp. 2646–2653
- 41 I. Dussault, S.F. Bellon; From concept to reality: the long road to c-Met and RON receptor tyrosine kinase inhibitors for the treatment of cancer; Anti-cancer Agents Med Chem, 9 (2009), pp. 221–229
- 42 J.M. O’Toole, K.E. Rabenau, K. Burns, D. Lu, V. Mangalampalli, P. Balderes, et al.; Therapeutic implications of a human neutralizing antibody to the macrophage-stimulating protein receptor tyrosine kinase (RON), a c-MET family member; Cancer Res, 66 (2006), pp. 9162–9170
Document information
Published on 11/04/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?