Abstract
Ultrasound-assisted transesterification of refined palm oil (RPO) and crude palm oil (CPO) to produce biodiesel using a palm oil mill fly ash supported calcium oxide (CaO) catalyst is studied in this work. The reaction time is significantly reduced from a maximum of 360 min under conventional mixing to just 30 min with the use of ultrasound. Under ultrasonic cavitation, the required catalyst loading and methanol to oil molar ratio to produce comparable yields and fatty acid methyl esters (FAME) conversions as conventional mixing are lower. For RPO, the ultrasound-assisted transesterification conditions of 60% ultrasonic amplitude, 30 min reaction time, 4 wt.% catalyst loading, and 9:1 methanol to oil molar ratio result in maximum biodiesel yield and FAME conversion of 85.23% and 97.02%, respectively. As for CPO, maximum biodiesel yield of 73.23% and FAME conversion of 97.04% are obtained under the same conditions with the exception of a methanol to oil molar ratio of 12:1. Key physicochemical properties of the produced biodiesels are found to be within the limits set by EN 14214 and ASTM D 6751. Catalyst reusability tests indicate that the catalyst can be used up to three consecutive cycles after regeneration using methanol washing followed by recalcination at 850°C for 2 h.
Introduction
Heterogeneous catalyzed transesterification has emerged as an alternative biodiesel processing route to homogeneous catalyzed transesterification in light of its potential industrial applicability [1]. The heterogeneous route potentially lowers the overall biodiesel production cost and minimizes environmental impacts due to its simpler production and purification steps [2]. Heterogeneous catalysts can be easily separated from the reaction mixture and regenerated for subsequent use. With increasing catalysis research, they can also be designed to give higher activity, stability, and selectivity, as well as longer lifetime [3, 4]. Nonetheless, a major drawback of heterogeneous catalyzed transesterification is its low reaction rate due to weak interactions between the oil, alcohol, and catalyst because of their mutual immiscibility. To improve mass transfer between the oil and alcohol reactants, the use of ultrasound which results in conditions of intense local turbulence and liquid circulation has been recently proposed.
High power ultrasound generates ultrasonic cavitation, a phenomenon that is defined as the formation, growth, and collapse of microscopic bubbles periodically in the ultrasound irradiated liquid. The implosive collapse of the cavitation bubbles during adiabatic compression results in formation of a high local temperature and pressure spot, called “hotspot.” Furthermore, the collapse and/or formation of cavitation bubbles cause the formation of high-speed microjets, microstreaming, and generation of shockwaves that increase heat and mass transfer in the liquid, as well as velocity gradients that result in shear stresses. These physical effects contribute to effective mixing of solution, disruption of immiscible liquid layers, and promotion of mass transfer at the liquid–solid interface [5]. For instance, Deshmane and Adewuyi [6] showed that the highest soy biodiesel conversion in excess of 90% was achieved within 90 min with the use of ultrasound. In another study using Jatropha curcas oil [7], a conversion of 91% was achieved in just 40 min at a moderate ultrasonic amplitude of 60%. Meanwhile, [8] reported that a high biodiesel conversion of approximately 98% using J. curcas oil was obtained at an optimum condition of 3 wt.% catalyst concentration and 15 min reaction time. Nonetheless, while improvements in reaction rates may be addressed by the use of ultrasound, its effects on biodiesel quality remains relatively unknown. Due to the high local temperature and pressure, species that are present within the cavitation bubbles may undergo pyrolysis, which adversely affects biodiesel quality.
To date, the majority of heterogeneous ultrasound-assisted transesterification studies utilize refined oils as their feedstock [1, 2, 6, 9-11]. Salamatinia et al. [12] have recently reported that biodiesel could be produced from palm, corn, canola, and sunflower oils using ultrasound. They linked the free fatty acids (FFA) content to the biodiesel conversions obtained. In the transesterification of soybean oil, it was observed that ultrasound frequency could enhance mass transfer as 611 kHz resulted in significantly improved mass transfer coefficient (89%) as compared to 20 kHz (48%)[6]. Notably, Badday et al. [7] and Kumar et al. [8] have used nonedible oil J. curcas oil as the raw feedstock for biodiesel production using ultrasound-assisted transesterification.
Both heterogeneous acid and base catalysts have reportedly been used in ultrasound-assisted transesterification processes. Heterogeneous acid catalysts used include carbon, cesium, and alumina doped heteropolyacid catalysts [13-15], 3-(NN,N-triethylamino)-1-propanesulfonic hydrogen sulfate ionic liquid [16], and mixed sulfate zirconia/titania [17]. Meanwhile, heterogeneous base catalysts used include zeolite [1], silicon dioxide supported sodium (Na/SiO2) [8], potassium phosphate (K3PO4) [9], barium oxide (BaO) [11], strontium oxide (SrO) [12], and calcium oxide (CaO) [18].
The main aim of the present work is to investigate the use of ultrasound in heterogeneous catalyzed transesterification using palm oil mill fly ash supported CaO. Palm oil mill fly ash supported CaO is a recently developed heterogeneous catalyst which has been reported as an effective transesterification catalyst [19]. Two different feedstock, that is, refined palm oil (RPO) and crude palm oil (CPO) are studied in order to compare the differences in terms of biodiesel yield and fatty acid methyl esters (FAME) conversion. To the best knowledge of the authors, no attempts have been carried out so far to convert RPO and CPO using ultrasound-assisted transesterification with this heterogeneous catalyst. Parametric experimental tests are carried out to elucidate the effects of ultrasonic amplitude, reaction time, catalyst loading, and methanol to oil molar ratio on yield and FAME conversion. In light of the relatively unknown ultrasound effects on biodiesel quality, the physicochemical properties and glycerides content are analyzed. Lastly, catalyst reusability is examined to determine possible leaching effects caused by ultrasonic cavitation.
Experimental
Materials
Commercial palm cooking oil (Buruh, Malaysia) was purchased from a local supermarket and CPO was provided by Havys Oil Mill Sdn. Bhd. The accompanying CPO sample analysis report recorded a FFA content of 3.7%, a deterioration of bleachability index (DOBI) value of 2.6% and a moisture content of 0.2%. Palm oil mill fly ash was collected from Seri Ulu Langat Palm Oil Mill Sdn. Bhd., Malaysia. Methanol (CH3OH; >99% purity) and calcium carbonate (CaCO3; 99.95% purity) were purchased from Merck (Bandar Sunway, Selangor, Malaysia) and Sigma Aldrich (Subang Jaya, Selangor, Malaysia), respectively. Meanwhile, internal standard methyl heptadecanoate (C17:0) was purchased from Sigma Aldrich. The reference standards for FAME (FAME mix RM-6), glycerol and glycerides contents were also obtained from Sigma Aldrich as follows: N-methyl-N-trimethysilyltrifluoroacetamide (MSTFA), n-heptane, 1,2,4-butanetriol (internal standard No. 1), tricaprin (internal standard No. 2), glycerol, 1-monooleoylglycerol (monoolein), 1,3-dioleoylglycerol (diolein), and 1,2,3-trioleoylglycerol (triolein).
Catalyst preparation
The wet impregnation method was used to prepare the catalyst at a fixed composition of 45 wt.% calcined CaCO3 loaded onto palm oil mill fly ash [20]. The palm oil mill fly ash was first sieved and dried at 105°C for 24 h to eradicate all moisture. Prior to use, CaCO3 was calcined at 800°C for 90 min to remove CO2 and generate CaO in which the active salt precursor Ca(OH)2 will be subsequently obtained. Typically, to obtain 45 wt.% calcined CaCO3 loaded catalyst, 13.5 g of calcined CaCO3 was added to 200 mL of pure deionized H2O in a three-neck flask under magnetic stirring at 700 rpm to prepare an aqueous solution of Ca(OH)2. The solution was heated in a closed system at 70°C. 16.5 g of fly ash was then slowly added to the solution and mixed vigorously for another 4 h until a homogenous mixture was obtained. The sample was then aged for 18 h to ensure that the Ca(OH)2 precipitated on the fly ash carrier. The removal of adhering H2O in the mixture was accomplished using a hot air oven at 105°C for 24 h. The dried catalyst was then calcined at temperatures of 850°C in a muffle furnace for 2 h. Sufficient catalyst was produced following the methodology described above for the entire set of single factor transesterification parametric study and characterized according to [20]. Since the parametric study was repeated thrice, three batches were made and characterization of each batch of catalyst was carried out to ensure that the quality of the prepared catalyst was consistent and up to the expected requirements.
Experimental setup
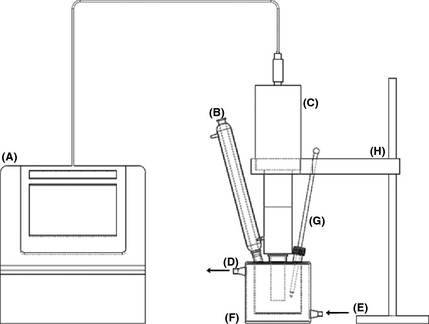
Ultrasound-assisted transesterification reactions were carried out in a customized three-neck jacketed reactor equipped with an ultrasound transducer and a probe (Qsonica Q700 (Newtown, CT, USA); 20 kHz max. power 700 W) as shown in Figure 1. A condenser and a thermometer were inserted into the side joints. Cooling water was allowed to flow through the jacket to control the reaction temperature, which was fixed at 45 ± 2°C. Ultrasonication was introduced in a pulse mode with 10 sec on and 7 sec off to avoid instantaneously temperature spikes and to generate sudden impacts for better cavitation. Conventional mixing transesterification experiments were also carried out to serve as a benchmark for the comparison of the presence and absence of ultrasound. For these experiments, a 250 mL heated three-neck round bottom flask equipped with a reflux condenser was used [20].
|
|
|
Figure 1. Schematic diagram of ultrasonic reactor (A) sonicator; (B) condenser; (C) transducer; (D) cooling water outlet; (E) cooling water inlet; (F) ultrasonic reactor; (G) thermometer and (H) retort stand. |
Ultrasound-assisted transesterification reactions
For all transesterification tests, 100 mL (87 g) of RPO or CPO was measured. The desired amount of fly ash supported CaO catalyst was added to a fixed amount of methanol depending on the methanol to oil molar ratio, and was heated and premixed at 40°C and 500 rpm for 15 min. The catalyst–methanol mixture was then introduced into the reactor and ultrasonication was applied. The effects of ultrasonic amplitude, reaction time, catalyst loading, and methanol to oil molar ratio were investigated in this work. Table 1 shows the nominal and tested parametric settings for the single factor transesterification parametric study. All experiments were repeated thrice.
| Reaction parameters | Tested settings |
|---|---|
| |
| Ultrasonic amplitude (%) | 20, 40, (60)a, 80, 100 |
| Reaction time (min) | 15, (30)a, 45, 60 |
| Catalyst loading (wt.%) | 2, (4)a, 6, 8 |
| Methanol to oil molar ratio (mol/mol) | 6:1, 9:1, (12:1)a, 15:1, 18:1 |
After the reaction, the solid catalyst was filtered out from the mixture with the aid of a vacuum pump. The filtered sample was then separated by centrifugation at 4000 rpm for 15 min. Two phases would be formed, namely an ester-rich phase (top layer) and a glycerol-rich phase (bottom layer). The glycerol was removed using a separating funnel while the excess methanol in the ester-rich phase was removed via a rotary evaporator. The sample from the ester-rich phase underwent further treatment including diluted acetic acid washing (90°C) to remove all dissolved residues and finally hot air oven drying (65°C) to remove all H2O content. The mass of the final product, that is, biodiesel was measured for the determination of yield according to Equation (1) and is presented in this work as the average of three repeats.
|
|
(1) |
Conventional mixing transesterification experiments were additionally performed as a benchmark to compare the presence and absence of ultrasound on biodiesel yield and FAME conversion. The nominal reaction time, catalyst loading, methanol to oil molar ratio, and temperature were set at 180 min, 4 wt.%, 12:1 and 45°C, respectively.
Gas chromatography (GC) analyses
Fatty acid methyl esters analysis
The FAME in the final biodiesel products were qualitatively and quantitatively analyzed three times by gas chromatography (GC). The GC used was a Perkin Elmer (Petaling Jaya, Selangor, Malaysia) Clarus 500 GC equipped with a flame ionization detector (FID). The capillary column used was an Agilent (Petaling Jaya, Selangor, Malaysia) J&W HP-88 column with dimensions of 0.25 mm I.D. × 60 m length × 0.2 μm film thickness. Helium (He) was the carrier gas and its pressure was set at 32 psi. The oven temperature was isothermally held at 185°C for 15 min. Meanwhile, the injector and detector temperatures were set at 240 and 250°C, respectively. For qualitative analysis, 0.1 mL of biodiesel sample was dissolved into 1.9 mL of hexane. 1 μL injections with a split injection ratio of 100:1 were carried out. The identification of all fatty acids composition in the sample was compared to the reference FAME mix RM-6. Meanwhile, quantitative FAME conversion was determined according to EN 14103 [21]. For this, 50 mg of biodiesel sample was mixed with 1 mL of internal standard methyl heptadecanoate (C17) solution (10 mg/mL). Similarly, 1 μL of the mixed sample was injected into the GC. The quantitative analysis provides verification that the FAME conversion in biodiesel is greater than 96.5% (mol/mol) in accordance to the specifications reported in EN 14214 [22]. As detailed in EN 14103, the FAME conversion was calculated using Equation (2).
|
|
(2) |
where is the sum of areas of all peaks ranging from C14:0 and C24:1; AIS is the internal standard (methyl heptadecanoate) peak area; CIS is the concentration of the internal standard solution (mg/mL); VIS is the volume of the internal standard solution used (mL); and m is the mass of the sample (mg).
Glyceride analysis
Triplicate analyses of free glycerol, monoglycerides (MG), diglycerides (DG), triglycerides (TG), and total glycerol were performed on a Perkin Elmer Clarus 680 GC equipped with an on-column injector, a FID, and a Supelco MET-biodiesel capillary column (14 m × 0.53 mm ID) coated with 0.16 μm film with an integrated 2 m × 0.53 mm I.D. guard. The GC operating conditions were as follows: detector temperature 380°C; programmed-temperature split/splitless injector 60°C hold for 1 min, heat at 15°C/min to 300°C; then heat at 30°C/min up to 380°C; oven temperature program 2 min at 50°C, heat at 15°C/min to 180°C, then heat at 7°C/min to 230°C and finally heat at 10°C/min up to 370°C and hold for 5 min. The carrier gas (He) flow was set at 3 mL/min.
100 mg of homogenized biodiesel sample was accurately measured in a vial. 80 μL of 1,2,4-butanetriol, 100 μL of tricaprin, and 100 μL of MSTFA were added into the vial and mixed vigorously. The sample solution was silylated for 15 min before adding 8 mL of heptane [23]. The silylation is intended to increase the volatility and stability of the hydroxyl compounds injected which therefore improves their detection. 1 μL injections in splitless mode were carried out on the GC.
Determination of the percentage of free glycerol and glycerides
Four calibration solutions were prepared according to EN 14105 [23] for the determination of the percentage of free glycerol and glycerides. The calibration solutions were analyzed thrice under the same conditions as the sample to allow peak identification by comparison of retention times. The glycerol calibration function is given by Equation (3), while the glycerides calibration functions are given by Equation ((4)-(6)).
|
|
(3) |
where Mg is the mass of glycerol (mg); Mei1 is the mass of internal standard 1 (1,2,4-butanetriol) (mg); Ag is the peak area of glycerol; Aei1 is the peak area of internal standard 1 (1,2,4-butanetriol) (mg); ag and bg are the constants from regression method for glycerol
|
|
(4) |
|
|
(5) |
|
|
(6) |
where Mm, Md, Mt are the masses of monoolein, diolein, and triolein, respectively (mg); Mei2 is the mass of internal standard 2 (tricaprin) (mg); Am, Ad, At are the peak areas of monoolein, diolein, and triolein, respectively; Aei2 is the peak area of internal standard 2 (tricaprin); am and bm are the constants from regression method for monoglycerol; ad and bd are the constants from regression method for diglycerol; at and bt are the constants from regression method for triglycerol.
Equation (7)-(11) were used to determine the percentages (m/m) of free glycerol, mono-,di-, and triglycerides, and total glycerol in the biodiesel sample, respectively.
|
|
(7) |
|
|
(8) |
|
|
(9) |
|
|
(10) |
|
|
(11) |
where G is the percentage (m/m) of free glycerol in the sample (%); m is the mass of sample (mg); M, D, T are the percentages (m/m) of mono-, di-, and triglycerides in the sample, respectively (%); GT is the total glycerol (%).
Biodiesel physicochemical properties analysis
The quality of the produced biodiesel under the optimum ultrasonic conditions was evaluated according to the American Society for Testing and Materials (ASTM) standards. The following key physicochemical properties were determined three times and the average of the repeats is presented in this work: (i) kinematic viscosity, determined at 40°C using glass capillary viscometers according to ASTM D 445; (ii) density, determined using a hydrometer according to ASTM D 1298; (iii) flash point, using the rapid equilibrium closed cup method according to ASTM D 93; (iv) acid value, by volumetric titration according to ASTM D 664; (v) higher heating value (HHV), estimated using a bomb calorimeter according to ASTM D 4868 and; (vi) cetane number (CN), calculated from Equation (12) obtained from Artificial Neural Networks (ANN) [24].
|
|
(12) |
where CN indicates the CN while (C18:0), (C18:1), (C18:2), and (C22:1) are the mass concentrations of stearic, oleic, linoleic, and erucic acids, respectively.
Catalyst reusability study
A total of five ultrasound-assisted transesterification cycles were carried out using 100 mL (87 g) of RPO or CPO under the optimum conditions of 60% ultrasonic amplitude, 4 wt.% catalyst loading, 9:1 methanol to oil molar ratio 9:1, 45°C temperature, and 30 min reaction time. After the first cycle of ultrasound-assisted transesterification, the spent solid catalysts were separated from the mixture by filtration and washed with methanol to remove polar compounds on its surface such as glycerol. Then, they were dried in the oven (65°C for 3 h) before recalcining at 850°C for 2 h [20]. To check catalyst deactivation and leaching, determination of CaO and Ca concentrations in the catalyst after each cycle was carried out using XRD and EDS, respectively. EDS was used to semiquantify Ca concentration due to the unavailability of more accurate quantification techniques such as inductively coupled plasma-atomic emission spectroscopy (ICP-AES). Additionally, the surface morphology of the recycled catalysts was characterized by using SEM, while the surface areas were measured using BET surface area analysis.
Results and Discussion
Effects of ultrasonic amplitude
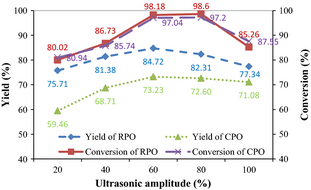
Figure 2 shows the effects of ultrasonic amplitude on biodiesel yield and FAME conversion for both RPO and CPO. As the amplitude increases from 20% to 60%, the yield of RPO biodiesel increases from 75.71% to 84.72%, while the yield of CPO biodiesel increases from 59.46% to 73.23%. As the amplitude increases, the size of cavitation bubbles increases leading to more intense collapse of bubbles which form very fine and stable emulsions [25]. This rapid emulsification accelerates the transport of reactants to and from the boundary between the two phases of oil and methanol resulting in higher interfacial surface area for mass transfer and hence higher biodiesel yield [26]. Wu et al. [27] have proven that very fine and stable emulsions produced in the presence of ultrasound were approximately 2.4 times smaller in droplets size than those produced in the absence of ultrasound. In this work, a maximum yield is recorded at 60% ultrasonic amplitude and any increase beyond that causes a drop in yields for both RPO and CPO. This is because a large number of cavitation bubbles is generated in the mixture with high ultrasonic amplitude. These bubbles will coalesce forming larger bubbles which will then act as a barrier to the transfer of acoustic energy through the liquid phase [8]. As observed in Figure 2, FAME conversion reaches a maximum above 80% in all cases. The highest FAME conversions of 98.18% and 97.04% are achieved with 60% ultrasonic amplitude for RPO and CPO biodiesels, respectively. Thus, the recommended amplitude for transesterification of both oils using palm oil mill fly ash supported CaO catalyst is 60% for the best yield and conversion.
|
|
|
Figure 2. Effects of ultrasonic amplitude on biodiesel yield and FAME conversion under nominal parametric settings (catalyst loading 4 wt.%, methanol to oil molar ratio 12:1, temperature 45°C, time 30 min). |
Effects of reaction time
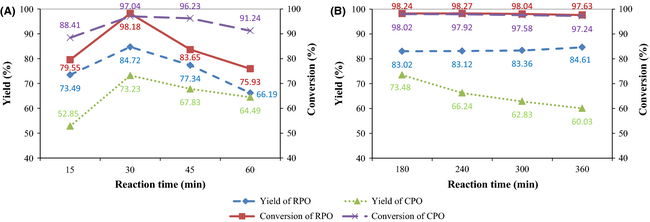
Figure 3A shows the effects of ultrasonic reaction time on biodiesel yield and FAME conversion for both RPO and CPO. An increase from 73.49% to 84.72% in RPO yield and a high FAME conversion of more than 98% are observed in the first 30 min. Meanwhile, for CPO, an increase from 52.85% to 73.23% in yield and a high FAME conversion of approximately 97% are attained in the first 30 min. The observed trends can be attributed to the sonochemical effect induced by acoustic cavitation stimulated by ultrasonic irradiation [28]. The speed of ultrasound in a liquid medium varies with the properties of the medium. The speed of ultrasound is 1140 m/sec in methanol, 1410 m/sec in biodiesel and 1453 m/sec in palm oil [25]. The change in velocity of ultrasound in adjacent mediums may enhance mixing between two phases because of the changes in acoustic streaming patterns, which further reduces the mass transfer resistance and helps to speed up the reaction [25]. After 30 min, the yield drops and the FAME conversion is lower due to promotion of the reverse reaction. The yield and conversion of RPO biodiesel drop to 66.19% and 75.93%, respectively, while the yield and conversion of CPO biodiesel drop to 64.19% and 91.24%, respectively, at 60 min. These results are in line with that reported by [11] whereby only 75% conversion was detected at 60 min in CaO catalyzed ultrasound-assisted transesterification of palm oil.
|
|
|
Figure 3. Effects of reaction time on biodiesel yield and FAME conversion under nominal parametric settings of (A) ultrasonic amplitude 60%, catalyst loading 4 wt.%, methanol to oil molar ratio 12:1, temperature 45°C; (B) stirring speed 700 rpm, catalyst loading 4 wt.%, methanol to oil molar ratio 12:1, temperature 45°C. |
The results for the conventional mixing transesterification experiments are shown in Figure 3B. For both RPO and CPO, the ultrasound-assisted transesterification reactions are very fast (15–60 min) as compared to the conventional mixing ones (180–360 min). The underlying mechanism behind the fast reactions is the collapse of cavitational bubbles near the methanol/oil boundaries leading to ultrasonic jets of methanol impinging into oil and resulting in excellent mixing. These results are in line with [29-31] who reported that the conversion of biodiesel was complete at approximately 60 min regardless of the type of oil. Figure 3B also shows that the highest RPO yield of 84.61% is obtained with 360 min of conventional mixing. In contrast, a similar RPO yield of 84.72% is achieved in just 30 min with the use of ultrasonic cavitation. For CPO, a shorter reaction time of 180 min is required with conventional mixing to achieve the highest yield of 73.48%. Compared to RPO, this represents a 50% reduction in reaction time. Nevertheless, significant reductions in reaction time of above 90% can only be achieved by using ultrasonic cavitation. In general, ultrasound reduces the reaction times of conventional mixing from the range of 180 to 360 min to 30 min with similar yields and conversions.
Effects of catalyst loading
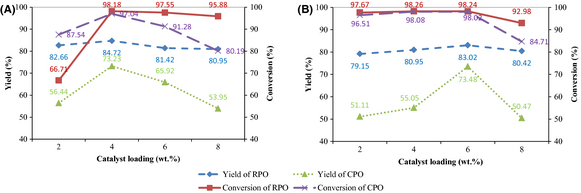
The effects of catalyst loading on biodiesel yield and FAME conversion in ultrasound-assisted transesterification for RPO and CPO can be seen in Figure 4A. The RPO yields for all catalyst loadings are comparable within the range of 81–84%. These results are supported by [11] in which the yields were reportedly similar with increasing amount of heterogeneous catalysts (CaO and SrO) in the ultrasonic reactor. It was suggested that catalyst loading may not be the dominant factor controlling the rate of reaction in ultrasonic systems. Conversely, the CPO yield increases from 56.44% to 73.23% when the catalyst loading increases from 2 wt.% to 4 wt.%. Further increase in catalyst loading reduces the yield to 53.95%. The presence of excess catalyst leads to soap formation and difficulty in esters separation [32]. In these cases, soap formation was visually observed as a milky third layer which formed between the top biodiesel layer and the bottom glycerol layer. In this work, a concentration of 4 wt.% gave the best biodiesel yields. As for RPO FAME conversion, when the catalyst loading increases from 2 wt.% to 4 wt.%, the conversion increases from 66.71% to 98.18% and reaches a constant beyond 4 wt.%. Maximum FAME conversion of 97.04% for CPO is also obtained at a catalyst loading of 4 wt.%, but the conversion decreases to 80.19% when 8 wt.% catalyst loading is used.
|
|
|
Figure 4. Effects of catalyst loading on biodiesel yield and FAME conversion under nominal parametric settings of (A) ultrasonic amplitude 60%, methanol to oil molar ratio 12:1, temperature 45°C, time 30 min; (B) stirring speed 700 rpm, methanol to oil molar ratio 12:1, temperature 45°C, time 180 min. |
Figure 4B illustrates the RPO and CPO biodiesel yields and FAME conversions for conventional mixing transesterification at different catalyst loadings. It can be observed that the biodiesel yields increase with increase in catalyst loading, but they drop once the optimum loading of 6 wt.% is reached. The results indicate that lesser catalyst loading is required to produce comparable maximum yields with the use of ultrasound as compared to conventional mixing regardless of the type of oil. In ultrasonication, for both RPO and CPO, 4 wt.% catalyst loading is required to achieve the highest yields while 2 wt.% lesser concentration is necessary to produce similar yields to 6 wt.% under conventional mixing. Hence, this clearly shows that ultrasound reduces not only the reaction time in biodiesel production, but also the catalyst loading for comparable yields. Additionally, both RPO and CPO achieve highest FAME conversions at 4 wt.% catalyst loading similar to the ultrasound-assisted transesterification. Maximum FAME conversions for conventional mixing are 98.26% and 98.08% for RPO and CPO, respectively. These are comparable to the maximum FAME conversions for ultrasonication, that is, 98.18% and 97.04% for RPO and CPO, respectively.
Effects of methanol to oil molar ratio
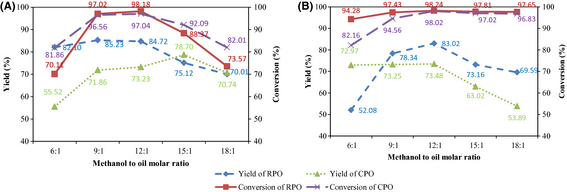
Figure 5A depicts the effects of methanol to oil molar ratio on RPO and CPO biodiesel yields and FAME conversions for ultrasound-assisted transesterification. Specifically for RPO, by increasing the molar ratio from 6:1 to 9:1, the yield increases from 82.10% to 85.23%. Further increase till 15:1 causes the yield to drop to 75.12%. As for CPO, the yield increases from 55.52% to 78.70% when the molar ratio increases from 6:1 to 15:1. The yield subsequently drops to 70.74% at 18:1. Since transesterification is reversible, when the molarity of the reactants decreases over time till a point at which the molarity is below equilibrium state, the reaction tends to shift backward [33]. Additionally, excess methanol promotes emulsification of products that increases the difficulty in separation and leads to reduction in yield. Meanwhile, the FAME conversions for both oils exceed the minimum requirement of 96.5% only at molar ratios of 9:1 and 12:1 whereby the conversions are 97.02% and 98.18%, respectively, for RPO, and 96.56% and 97.04%, respectively, for CPO. At these molar ratios, the RPO yields are comparable at 85.23% and 84.72% for 9:1 and 12:1, respectively. Similarly, CPO yields are 71.86% and 73.23% for 9:1 and 12:1, respectively. Thus, from an economic viewpoint, a molar ratio of 9:1 is preferred as less methanol is used to give above required conversions and comparable yields. This optimum molar ratio of 9:1 agrees well with those reported by [34] and [35].
|
|
|
Figure 5. Effects of methanol to oil molar ratio on biodiesel yield and FAME conversion under nominal parametric settings of (A) ultrasonic amplitude 60%, catalyst loading 4 wt.%, temperature 45°C, time 30 min; (B) stirring speed 700 rpm, catalyst loading 4 wt.%, temperature 45°C, time 180 min. |
The effects of methanol to oil molar ratio on RPO and CPO biodiesel yields and FAME conversions for conventional mixing can be seen in Figure 5B. A molar ratio of 12:1 is required to attain maximum RPO yield as compared to a molar ratio of 9:1 under ultrasonic cavitation. Nonetheless, a different trend is observed for CPO in which a higher molar ratio of 15:1 is necessary for ultrasound-assisted transesterification. This can be attributed to the fact that CPO has a higher viscosity than RPO hence more methanol is required during transesterification [36]. By comparing Figure 5A and B, it can be seen that maximum yields under ultrasonic cavitation are higher. The highest RPO yield increases from 83.02% to 85.23%, while the highest CPO yield improves from 73.48% to 78.70%. On the other hand, the average FAME conversion using conventional mixing is higher than ultrasonication. For RPO biodiesel, only a molar ratio of 9:1 or above can produce conversions which exceed the minimum requirement of 96.5%, whereas for CPO biodiesel, a molar ratio of 12:1 or above is required. Maximum conversions for both RPO and CPO are attained at a molar ratio of 12:1, that is, 98.24% and 98.02%, respectively. At the same molar ratio of 12:1 under ultrasonication, 98.18% and 97.04% conversions are obtained for RPO and CPO, respectively. The conversions are lower, but they exceed the minimum requirement.
To summarize, the optimum transesterification conditions for RPO under ultrasonic cavitation are 60% ultrasonic amplitude, 30 min reaction time, 4 wt.% catalyst loading, and 9:1 methanol to oil molar ratio. This results in biodiesel yield and FAME conversion of 85.23% and 97.02%, respectively. Meanwhile, for the ultrasound-assisted transesterification of CPO, the optimum transesterification conditions are the same except for methanol to oil molar ratio, which is at 12:1. Under these conditions, a yield of 73.23% and an FAME conversion of 97.04% are obtained.
Biodiesel properties analysis
Table 2 summarizes the tested physicochemical properties of the biodiesels produced under optimum conditions. The biodiesels produced from RPO and CPO using ultrasonication have slightly lower viscosities (5.02 and 5.79 mm2/sec, respectively) as compared to those produced using conventional mixing (5.03 and 5.98 mm2/sec, respectively). All these viscosities remain within the standard range of 1.9 to 6.0 mm2/sec at 40°C specified in ASTM D 6751 biodiesel standard. High viscosity results in poor fuel atomization, incomplete combustion, and carbon deposition on the injector. Additionally, the densities of RPO and CPO biodiesels from ultrasonication (862.3 and 863.9 kg/m3, respectively) are slightly lower than the RPO and CPO biodiesels from conventional mixing (863.1 and 865.3 kg/m3), respectively. Likewise to viscosity, these densities fully conform to the limits given by ASTM D 6751, that is, between 860 and 900 kg/m3 at 15°C. A higher biodiesel density means that there is a delivery of a greater mass of fuel for a fixed volumetric flowrate. Hence, CPO biodiesel has this added advantage over RPO biodiesel since its density is higher in both production methods. Its higher density can be linked to the fact that the density of CPO at 950 kg/m3 is higher than that of RPO at 900 kg/m3 [4]. Variations in biodiesel density are mostly dependent on the feedstock used rather than the production process.
| Physicochemical property | Unit | Test method | ASTM D 6751 and EN 14214 limits | Conventional mixing | Ultrasonic cavitation | ||
|---|---|---|---|---|---|---|---|
| RPO | CPO | RPO | CPO | ||||
| |||||||
| Kinematic viscosity | mm2/sec; 40°C | ASTM D 445 | 1.9-6.0a | 5.03 | 5.98 | 5.02 | 5.79 |
| Density | kg/m3; 15°C | ASTM D 1298 | 860-900a | 863.1 | 865.3 | 862.3 | 863.9 |
| Flash point | °C | ASTM D 93 | 130 minimuma | 168 | 144 | 170 | 164 |
| Acid value | mg KOH/g | ASTM D 664 | 0.5 maximuma | 0.32 | 0.42 | 0.46 | 0.49 |
| High heating value | MJ/kg | ASTM D 4868 | 35 minimuma | 39.0 | 38.1 | 38.1 | 37.5 |
| Cetane numberc | – | ASTM D 613 | 47 minimuma | 50.4 | 48.1 | 49.8 | 49.0 |
| FAME conversion | % (mol/mol) | EN 14103 | 96.5 minimumb | 96.8 | 97.0 | 98.2 | 97.1 |
| Free glycerol | % | EN 14105 | 0.02 maximumb | 0.0069 | 0.0059 | 0.0068 | 0.0039 |
| Total glycerol | % | EN 14105 | 0.25 maximumb | 0.0604 | 0.0610 | 0.0163 | 0.0347 |
| Monoglyceride | % | EN 14105 | 0.8 maximumb | 0.1245 | 0.1722 | 0.0297 | 0.0976 |
| Diglyceride | % | EN 14105 | 0.2 maximumb | 0.1702 | 0.1730 | 0.0790 | 0.1660 |
| Triglyceride | % | EN 14105 | 0.2 maximumb | 0.1068 | 0.084 | 0.077 | 0.0344 |
The flash points for biodiesels produced using conventional mixing is 168°C for RPO and 144°C for CPO. Meanwhile, for biodiesels produced using ultrasonication, the flash points are 170°C for RPO and 164°C for CPO. All the flash points are well above the specified minimum of 130°C in ASTM D 6751, but the biodiesels produced using ultrasonication have higher flash points especially for CPO. Since the flash point is an indicator of the absence/presence of volatile alcohol, the high flash points of biodiesels as compared to diesel indicate that the biodiesels are safer fuels [4]. From Table 2, it can be observed that acid values for both biodiesels produced using ultrasonication and conventional mixing range between 0.32 and 0.49 mg KOH/g, which satisfy the maximum limit of 0.5 mg KOH/g in ASTM D 6751. It is important to meet this requirement to avoid problems associated with deposit and corrosion of fuel injection systems. For HHV, the ASTM D 6751 imposes a HHV higher than 35 MJ/kg for biodiesel. The HHV of biodiesels produced are 39.0 and 38.1 MJ/kg for RPO and CPO, respectively, using conventional mixing, and 38.1 and 37.5 MJ/kg for RPO and CPO, respectively, using ultrasonication. The HHV values are slightly less with ultrasonication, but the results are above the acceptable minimum. These HHV are similar to those reported by [37] which ranged from 37.8 to 39.4 MJ/kg for used frying palm oil biodiesel. The final measured physicochemical property is CN which is an important indicator of the quality of the fuel and is usually measured using the standard engine test, that is, ASTM D 613. However, CN is relatively difficult to measure and is not always determined owing to the lack of measurement facility [38]. In this work, the CN is predicted using Equation (12), which has been demonstrated to produce predicted CN with a level of confidence of greater than 90% [24]. All the predicted CN ranging from 48.1 to 50.4 exceed the minimum value of 47 required by ASTM D 6751, which indicates good quality combustion in compression ignition engines.
Overall, the measured biodiesel physicochemical properties indicate that there are no significant variations in viscosity, density, flash point, acid value, HHV, and CN with the use of ultrasonication as compared to conventional mixing. Additionally, all biodiesels produced meet the requirements stipulated in ASTM D 6751. The results reveal that not only ultrasonication is effective in achieving significantly shorter reaction times for transesterification, but it does not adversely affect biodiesel quality.
FAME conversion and glycerides analysis
Table 2 also lists the FAME conversions and glycerides analysis for both RPO and CPO biodiesels produced under conventional mixing and ultrasonication. The FAME conversions of all biodiesels exceed the minimum limit of 96.5% specified in EN 14214. In the presence of ultrasound, it can be observed that the FAME conversion improves from 96.8% to 98.2% for RPO, and slightly improves from 97.0% to 97.1% for CPO.
Irrespective of the mixing method, the CPO biodiesel has lower free glycerol, but higher total glycerol concentrations as compared to the RPO biodiesel. In addition, higher MG and DG, but lower TG are measured in the CPO biodiesel as compared to the RPO biodiesel. For all biodiesels, both free and total glycerol concentrations are lowered with the use of ultrasonic cavitation. This is especially so for CPO whereby the free glycerol is reduced from 0.0059% to 0.0039% while the total glycerol is reduced from 0.0610% to 0.0347%. Both the free and total glycerol concentrations are below the maximums stipulated in EN 14105. For other by-products including MG, DG and TG, their concentrations are reduced under the influence of ultrasonic cavitation. The observed reductions in these by-products can be linked to the good mixing of ultrasonic cavitation which promote mass transfer at the liquid–solid interfaces. Due to the enhanced mass transfer, lesser by-products are formed as more are converted into biodiesel. In short, it can be concluded that ultrasonic cavitation during transesterification produces biodiesel which meets EN standards in terms of purity.
Catalyst reusability study
Catalyst reusability cycles
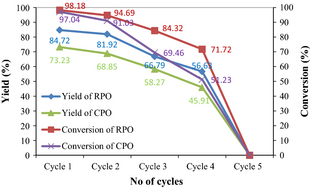
Figure 6 shows the RPO and CPO biodiesel yields and FAME conversions for five consecutive cycles of catalyst reuse. Both yield and conversion decrease after each subsequent cycle. For the first cycle, the yields of RPO and CPO are 84.72% and 73.23%, respectively. Meanwhile, the conversions of RPO and CPO are 98.18% and 97.04%, respectively. Significantly, both yield and conversion from the fourth cycle reduce to 56.63% and 71.72%, respectively, for RPO; and 45.91 and 51.23%, respectively, for CPO. Under ultrasonic cavitation, the microbubbles of reaction products especially glycerol have more potential to adsorb onto the CaO surface and generate glyceroxides species as the alternative active sites [9]. Kouzu et al. [39] reported that glyceroxides species are more active than CaO itself in transesterification. However, the CaO surface does not only adsorb glycerol, but also other reaction products which lead to the deactivation of CaO within the catalyst. Deactivation of catalyst becomes more dominant than generation of glyceroxides species after each subsequent cycle.
|
|
|
Figure 6. Reusability cycle of RPO and CPO biodiesel yield and FAME conversion (ultrasonic amplitude 60%, catalyst loading 4 wt.%, methanol to oil molar ratio 9:1, temperature 45°C, time 30 min). |
Characterization of recycled catalysts
Figure 7 depicts the SEM images of the catalyst after each cycle while Table 3 lists the BET surface areas of fresh and recycled catalysts, and CaO and Ca concentrations. The catalyst size increases slightly with each subsequent cycle as shown in Figure 7. Accordingly, as listed in Table 3, the BET surface area decreases after each subsequent cycle. This may be caused by adsorption of compounds onto the catalyst surface. In general, a larger particle size is typically associated with a smaller surface area which decreases the rate of reaction. As a result, lower biodiesel yields and FAME conversions occur after each subsequent cycle.
| Catalyst | CaO concentration (wt.%) | Ca concentration (wt.%) | BET surface area (m2/g) |
|---|---|---|---|
| Fresh | 83 | 38 | 3.539 |
| 2nd cycle | 73 | 34 | 3.499 |
| 3rd cycle | 64 | 26 | 3.423 |
| 4th cycle | 58 | 17 | 3.385 |
| 5th cycle | 53 | 9 | 3.359 |
|
|
|
Figure 7. SEM images (2000 × magnification) of catalysts from (A) 1st cycle (B) 2nd cycle (C) 3rd cycle and (D) 4th cycle. |
When ultrasonic cavitation was applied to the transesterification process, the CaO and Ca concentrations decrease after each cycle. Since the obtained Ca concentrations are considered semiquantitative, all the associated discussions are relying on the figures in Table 3 to obtain valid trends when parameters are changed. The actual magnitudes must be treated with a certain degree of conservatism. After the fourth cycle, the CaO concentration decreases from 83 wt.% to 53 wt.% while the Ca concentration decreases from 38 wt.% to only 9 wt.%. This indicates that the palm oil mill fly ash supported CaO catalyst experiences leaching in ultrasound-assisted transesterification. Despite dissolution of CaO and Ca into the reaction mixture, the catalyst can be reasonably reused up to three times.
Conclusions
Ultrasound-assisted transesterification of RPO and CPO to produce biodiesel using a palm oil mill fly ash supported calcium oxide (CaO) catalyst has been investigated. The optimum transesterification conditions for RPO under ultrasonic cavitation are 60% ultrasonic amplitude, 30 min reaction time, 4 wt.% catalyst loading, and 9:1 methanol to oil molar ratio. This results in biodiesel yield and FAME conversion of 85.23% and 97.02%, respectively. Meanwhile, for the ultrasound-assisted transesterification of CPO, the optimum transesterification conditions are the same except for methanol to oil molar ratio, which is at 12:1. Under these conditions, a yield of 73.23% and an FAME conversion of 97.04% are obtained. With the use of ultrasonic cavitation in transesterification, the reaction time is significantly reduced from a maximum of 360 under conventional mixing to just 30 min. Additionally, the required catalyst loading and methanol to oil molar ratio are reduced as compared to conventional mixing for comparable yields and conversions. All produced biodiesels have physicochemical properties such as kinematic viscosity, density, flash point, acid value, HHV, and CN within the limits specified in ASTM D 6751. The FAME and all glycerides contents are also within the limits specified in EN 14214. The catalyst can be reused feasibly up to three consecutive cycles after regeneration. In conclusion, the potential of palm oil mill fly ash supported calcium oxide (CaO) catalyst to be used in conjunction with ultrasound-assisted transesterification to produce RPO and CPO biodiesels has been demonstrated.
Acknowledgments
This work was supported by the Ministry of Higher Education (MOHE), Malaysia as well as the Faculty of Engineering at The University of Nottingham Malaysia Campus. The authors would like to express gratitude to Havys Oil Mill Sdn. Bhd. for providing the crude palm oil and Seri Ulu Langat Palm Oil Mill for the supply of palm oil mill boiler ash.
Conflict of Interest
None declared.
References
- Georgogianni, K. G., A. K. Katsoulidis, P. J. Pomonis, G. Manos, and M. G. Kontominas. 2009. Transesterification of rapeseed oil for the production of biodiesel using homogeneous and heterogeneous catalysis. Fuel Process. Technol.90:1016–1022.
- Salamatinia, B., H. Mootabadi, S. Bhatia, and A. Z. Abdullah. 2010. Optimisation of ultrasonic-assisted heterogeneous biodiesel production from palm oil: a response surface methodology approach. Fuel Process. Technol.91:441–448.
- Puna, J. F., J. F. Gomes, M. J. N. Correia, A. P. S. Dias, and J. C. Bordado. 2010. Advances on the development of novel heterogeneous catalysts for transesterification on triglycerides in biodiesel. Fuel89:3602–3606.
- Salamatinia, B., A. Z. Abdullah, and S. Bhatia. 2012. Quality evaluation of biodiesel produced through ultrasound-assisted heterogeneous catalytic system. Fuel Process. Technol.97:1–8.
- Kojima, Y., H. Imazu, and K. Nishida. 2013. Physical and chemical characteristics of ultrasonically-prepared water-in-diesel fuel: effects of ultrasonic horn position and water content. Ultrason. Sonochem.21:722–728.
- Deshmane, V. G., and Y. G. Adewuyi. 2013. Synthesis and kinetics of biodiesel formation via calcium methoxide base catalysed transesterification reaction in the absence and presence of ultrasound. Fuel107:474–482.
- Badday, A. S., A. Z. Abdullah, and K.-T. Lee. 2013. Optimisation of biodiesel production process from Jatropha oil using supported heteropolyacid catalyst and assisted by ultrasonic energy. Renew. Energy50:427–432.
- Kumar, D., G. Kumar, Poonam, C.P. Singh. 2010. Ultrasonic-assisted transesterification of jatropha curcas oil using solid catalyst, Na/SiO2. Ultrason. Sonochem.17:839–844.
- Choedkiatsakul, I., K. Ngaosuwan, and S. Assabumrungrat. 2013. Application of heterogeneous catalysts for transesterification of refined palm oil in ultrasound-assisted reactor. Fuel Process. Technol.111:22–28.
- Choudhury, H. A., S. Chakma, and V. S. Moholkar. 2013. Mechanistic insight into sonochemical biodiesel synthesis using heterogeneous base catalyst. Ultrason. Sonochem.21:169–181.
- Mootabadi, H., B. Salamatinia, S. Bhatia, and A. Z. Abdullah. 2010. Ultrasonic-assisted biodiesel production process from palm oil using alkaline earth metal oxides as the heterogeneous catalysts. Fuel89:1818–1825.
- Salamatinia, B., H. Mootabadi, I. Hashemizadeh, and A. Z. Abdullah. 2013. Intensification of biodiesel production from vegetable oils using ultrasonic-assisted process: optimisation and kinetic. Chem. Eng. Process.73:135–143.
- Badday, A. S., A. Z. Abdullah, and K. T. Lee. 2013. Ultrasound-assisted transesterification of crude Jatropha oil using alumina-supported heteropolyacid catalyst. Appl. Energy105:380–388.
- Badday, A. S., A. Z. Abdullah, and K. T. Lee. 2013. Ultrasound-assisted transesterification of crude Jatropha oil using cesium doped heteropolyacid catalyst: interactions between process variables. Energy60:283–291.
- Badday, A. S., A. Z. Abdullah, and K. T. Lee. 2014. Transesterification of crude Jatropha oil by activated carbon-supported heteropolyacid catalyst in an ultrasound-assisted reactor system. Renew. Energy62:10–17.
- Guo, W., H. Li, G. Ji, and G. Zhang. 2012. Ultrasound- assisted production of biodiesel from soybean oil using Bronsted acidic ionic liquid as catalyst. Bioresour. Technol.125:332–334.
- Boffito, D. C., V. Crocella, C. Pirola, B. Neppolian, G. Cerrato, M. Ashokkumar, et al. 2013. Ultrasonic enhancement of the acidity, surface area and free fatty acids esterification catalytic activity of sulphated ZrO2- TiO2 systems. J. Catal.297:17–26.
- Verziu, M., S. M. Coman, R. Richards, and V. I. Parvulescu. 2011. Transesterification of vegetable oils over CaO catalysts. Catal. Today167:64–70.
- Ho, W. W. S., H. K. Ng, and S. Gan. 2012. Development and characterisation of novel heterogeneous palm oil mill boiler ash-based catalysts for biodiesel production. Bioresour. Technol.125:158–164.
- Ho, W. W. S., H. K. Ng, S. Gan, and S. H. Tan. 2014. Evaluation of palm oil mill fly ash supported calcium oxide as a heterogeneous base catalyst in biodiesel synthesis from crude palm oil. Energy Convers. Manage.88:1167–1178.
- EN 14103. 2003. Fat and oil derivatives – Fatty Acid Methyl Esters (FAME) – Determination of ester and linolenic acid methyl esters contents.
- EN 14214. 2003. Automotive fuels – Fatty Acid Methyl Esters (FAME) for diesel engines – Requirements and test methods.
- EN 14105. 2003. Fat and oil derivatives – Fatty Acid Methyl Esters (FAME) – Determination of free and total glycerol and mono-, di- and triglyceride contents.
- Cheenkachorn, K.2004. Prediction properties of biodiesels using statistical models and artificial neural network. Sust. Energy Environ.3003:176–179.
- Naresh, N. M., and G. A. Yusuf. 2009. Optimisation of the synthesis of biodiesel via ultrasound-enhanced base- catalysed transesterification of soybean oil using a multifrequency ultrasonic reactor. Energy Fuels23:2757–2766.
- Naresh, N. M., and G. A. Yusuf. 2010. Application of taguchi method to investigate the effects of process parameters on the transesterification of soybean oil using high frequency ultrasound. Energy Fuels24:2120–2126.
- Wu, P., Y. Yang, J. A. Colucci, and E. A. Grulke. 2007. Effect of ultrasonication on droplet size in biodiesel mixtures. J. Am. Oil Chem. Soc.84:877–884.
- Omotola, B., P. Leslie, A. Bamikole, and A. Farouk. 2009. Low-cost feedstock conversion to biodiesel via ultrasound technology. Energies3:1691–1703.
- Stavarache, C., M. Vinatoru, R. Nishimura, and Y. Maeda. 2005. Fatty acids methyl esters from vegetable oil by means of ultrasonic energy. Ultrason. Sonochem.12:367–372.
- Stavarache, C., M. Vinatoru, and Y. Maeda. 2006. Ultrasonic versus silent methylation of vegetable oils. Ultrason. Sonochem.13:401–407.
- Stavarache, C., M. Vinatoru, and Y. Maeda. 2007. Aspects of ultrasonically assisted transesterification of various vegetable oils with methanol. Ultrason. Sonochem.14:380–386.
- Dias, A. P. S., J. Puna, M. J. N. Correia, I. Nogueira, J. Gomes, and J. Bordado. 2013. Effect of the oil acidity on the metholysis performances of lime catalyst biodiesel from waste frying oils (WFO). Fuel Process. Technol.116:94–100.
- Kotwal, M. S., P. S. Niphadkar, S. S. Deshpande, V. V. Bokade, and P. N. Joshi. 2009. Transesterification of sunflower oil catalysed by flyash-based solid catalysts. Fuel88:1773–1778.
- Arzamendi, G., E. Arguinarena, I. Campo, S. Zabala, and L. M. Gandia. 2008. Alkaline and alkaline earth metals compounds as catalysts for the methanolysis of sunflower oil. Catal. Today133–135:305–313.
- Kawashima, A., K. Matsubara, and K. Honda. 2009. Acceleration of catalytic activity of calcium oxide for biodiesel production. Bioresour. Technol.89:1818–1825.
- Georgogianni, K. G., M. G. Kontominas, P. J. Pomonis, D. Avlonitis, and V. Gergis. 2008. Alkaline conventional and in situ transesterification of cottonseed oil for the production of biodiesel. Energy Fuels22:2110–2115.
- Encinar, J. M., J. F. Gonzalez, and A. Rodriguez-Reinares. 2007. Ethanolysis of used frying oil. Biodiesel preparation and characterization. Fuel Process. Technol.88:513–522.
- Encinar, J. M., J. F. Gonzalez, and A. Pardal. 2012. Transesterification of castor oil under ultrasonic irradiation conditions. Preliminary results. Fuel Process. Technol.103:9–15.
- Kouzu, M., S.-Y. Yamanaka, J.-S. Hidaka, and M. Tsunomori. 2009. Heterogeneous catalysis of calcium oxide used for transesterification of soybean oil with refluxing methanol. Appl. Catal. A355:94–99.
Document information
Published on 01/06/17
Submitted on 01/06/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?