Highlights
- CD4/CD8 ratio in bronchoalveolar lavage fluid can be a useful supplement to other diagnostic methods for sarcoidosis.
- Results suggest that the ratio can be a useful supplement to other diagnostic methods.
Sarcoidosis is a granulomatous inflammatory disease that can be challenging to diagnose. Based on meta-analysis of an extensive evidence base, our study concludes that measuring the CD4/CD8 ratio in bronchoalveolar lavage fluid can be useful for diagnosing sarcoidosis, when used in parallel with other diagnostic tools. We recommend measuring this ratio in patients strongly suspected of having sarcoidosis who cannot tolerate more invasive biopsy.
Abstract
Background
The usefulness of bronchoalveolar lavage fluid (BALF) CD4/CD8 ratio for diagnosing sarcoidosis has been reported in many studies with variable results. Therefore, we performed a meta-analysis to estimate the overall diagnostic accuracy of BALF CD4/CD8 ratio based on the bulk of published evidence.
Methods
Studies published prior to June 2015 and indexed in PubMed, OVID, Web of Science, Scopus and other databases were evaluated for inclusion. Data on sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR), and diagnostic odds ratio (DOR) were pooled from included studies. Summary receiver operating characteristic (SROC) curves were used to summarize overall test performance. Deekss funnel plot was used to detect publication bias.
Results
Sixteen publications with 1885 subjects met our inclusion criteria and were included in this meta-analysis. Summary estimates of the diagnostic performance of the BALF CD4/CD8 ratio were as follows: sensitivity, 0.70 (95%CI 0.64–0.75); specificity, 0.83 (95%CI 0.78–0.86); PLR, 4.04 (95%CI 3.13–5.20); NLR, 0.36 (95%CI 0.30–0.44); and DOR, 11.17 (95%CI 7.31–17.07). The area under the SROC curve was 0.84 (95%CI 0.81–0.87). There was no evidence of publication bias.
Conclusion
Measuring the BALF CD4/CD8 ratio may assist in the diagnosis of sarcoidosis when interpreted in parallel with other diagnostic factors.
Keywords
Bronchoalveolar lavage fluid ; CD4/CD8 ratio ; Sarcoidosis ; Meta-analysis
1. Introduction
Sarcoidosis, a chronic inflammatory disorder of unknown cause, is the most frequently observed interstitial lung disease of unknown origin in Europe (Baughman and Grutters, 2015 ). It usually affects the lung and lymphatic system, but its clinical features are varied and non-specific, and it shows variable radiographic presentation, all of which makes accurate diagnosis a challenge. Typically, sarcoidosis is diagnosed when clinical and/or radiographic findings are supported by histological evidence of non-caseating granulomatous inflammation, and when other causes of granulomas and local reactions can be reasonably excluded (Iannuzzi et al., 2007 , Costabel et al., 2008 and American Thoracic Society: European Respiratory Society Worl ). Another problem with diagnosing sarcoidosis is that, unless patients show typical manifestations of Löfgren syndrome, biopsy is recommended, making diagnosis invasive (Iannuzzi et al., 2007 , Costabel et al., 2008 and American Thoracic Society: European Respiratory Society Worl ). As a result, investigators continue to search for reliable, less invasive methods to diagnose sarcoidosis.
Growing evidence points to the possibility of analyzing the bronchoalveolar lavage fluid (BALF) to aid in diagnosis. In sarcoidosis, TH 1 hyperimmune response to an unknown agent causes CD4 + T lymphocytes to accumulate in affected tissues and leads to the formation of non-caseating granulomas (Baughman et al., 2003 ). As a result, many patients with sarcoidosis show elevated lymphocytosis and CD4/CD8 ratio in BALF, and this elevated ratio has been associated with a diagnosis of sarcoidosis (Baughman et al., 2003 and Costabel, 1997 ). Indeed, clinicians may opt not to perform diagnostic biopsy in patients who present both a clinical picture typical of sarcoidosis and an elevated BALF CD4/CD8 ratio (Kvale, 2003 and Kantrow et al., 1997 ). Several studies have suggested that the BALF CD4/CD8 ratio can supplement the results of other tests when diagnosing sarcoidosis (Wells and Hirani, 2008 , Chretien et al., 1985 and Stoller et al., 1987 ).
However, whether the BALF CD4/CD8 ratio can reliably perform as a diagnostic tool remains controversial. The ratio shows high variability (Kantrow et al., 1997 ), and studies of its diagnostic performance suggest variable sensitivity and specificity. To gain a clearer picture of the diagnostic usefulness of this ratio, we performed a meta-analysis to summarize its overall diagnostic performance based on the available literature.
2. Methods
This study was performed according to the guidelines of the Preferred Reporting Items for Systematic Reviews, as well as the Meta-analysis Statement and methods recommended by the Cochrane Diagnostic Test Accuracy Working Group (Leeflang et al., 2008 ). Institutional review board approval was not required for this retrospective meta-analysis.
2.1. Search Strategy
PubMed, OVID, Web of Science, Scopus, Wanfang, Weipu and CNKI databases were searched for original articles that examined the diagnostic performance of BALF CD4/CD8 for sarcoidosis and that were published up to October 2015. In PubMed, the search string was (((Bronchoalveolar lavage OR Bronchoalveolar'lavage fluid OR BAL OR BALF ) AND sarcoidosis ) AND CD4 /CD8 ratio ). In OVID, references in EMBASE from 1974 to June 2015 and in Medline from 1946 to October 2015 were searched using the following string: “Bronchoalveolar lavage ” OR “Bronchoalveolar lavage fluid ” OR “BAL ” OR “BALF ” AND “CD4 /CD8 ratio ” AND “sarcoidosis ” AND “sensitivity OR specificity OR accuracy ”. Search results were limited to human and clinical trials. In Wanfang, Weipu and CNKI databases, the following search string was used: “Bronchoalveolar lavage fluid ” AND “sarcoidosis ” AND “CD4 /CD8 ratio ”. The “remove duplicates” function was applied during searches in OVID and the Chinese databases. Additional articles were also searched using the “related articles” function in PubMed. References within identified articles were searched manually to find more articles.
2.2. Selection of Publications
We screened titles and abstracts of identified publications, and those studies that could not be immediately excluded were retrieved as full text. Publications were included in our meta-analysis if they fulfilled the following criteria: (1) they used BALF CD4/CD8 ratio for diagnosing sarcoidosis; (2) they reported sufficient data to calculate true positive (TP), false positive (FP), false negative (FN), and true negative (TN) of the BALF CD4/CD8 ratio for diagnosing sarcoidosis; and (3) they constituted original research published in English or Chinese. To avoid selection bias, we excluded studies involving fewer than 20 subjects. Conference abstracts, reviews, editorials, and case reports were also excluded.
2.3. Data Extraction and Quality Assessment
Two reviewers (YCS and CSP) independently judged the eligibility of publications and extracted the following data: first author, year of publication, country, number of cases and controls, diagnostic standard, sample, method, cut-off values, TP, FP, FN, TN, and study design. Discrepancies in data extraction were resolved by consensus. Efforts were made to contact authors when information was not reported in the article. For studies in which several different cut-off values were tested, only the data associated with the best diagnostic performance was included in this meta-analysis.
The methodological quality of each study was assessed using the Quality Assessment of Diagnostic Accuracy Studies (QUADAS)-2 (Whiting et al., 2011 ). This tool consists of four domains: patient selection, index test, reference standard, as well as flow and timing. Risk of bias was assessed in four domains, the first three of which concern applicability.
2.4. Statistical analysis
Standard methods recommended for diagnostic accuracy meta-analysis were used (Devillé et al., 2002 and Nguyen et al., 2015 ). We analyzed the test accuracy of each study by calculating sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR), diagnostic odds ratio (DOR), together with the corresponding 95%CIs. Summary receiver operating characteristic (SROC) curves and the area under the curve (AUC) were also calculated (Moses et al., 1993 ).
Heterogeneity between studies was evaluated using the χ2 test and Fishers exact tests. If significant heterogeneity existed among studies, meta-regression analysis was performed using covariates reported in most included studies: cut-off values, sample size (< 100 subjects vs. ≥ 100 subjects), study design (prospective vs. retrospective), publication year (before 2005 vs. after 2005), sampling method (consecutive vs. not reported), risk of bias (low vs. high), income in study country (high vs. low or middle, based on World Bank ranking of national economies), and ethnicity (Asian vs. Caucasian). Sensitivity analysis was conducted by subgroups based on the meta-regression results.
Deekss funnel plot was used to detect publication bias (Deeks et al., 2005 ). Post-test probability (PTP) was calculated using the overall prevalence of 20% with Fagan nomograms. Three statistical software programs were used in this meta-analysis: STATA 12.0 (Stata Corp., College Station, TX), Meta-DiSc 1.4 (XI, Cochrane Colloquium, Barcelona, Spain), and RevMan 5.2 (Cochrane Collaboration, Oxford, UK). All statistical tests were two-sided, and P < 0.05 was considered statistically significant.
2.5. Role of the Funding Source
The funders had no role in the study design, collection, analysis or interpretation of the data, or writing of the report. All authors had access to the raw data. The corresponding author had full access to all the data and assumed responsibility for submitting for publication.
3. Results
3.1. Characteristics and Quality of the Included Studies
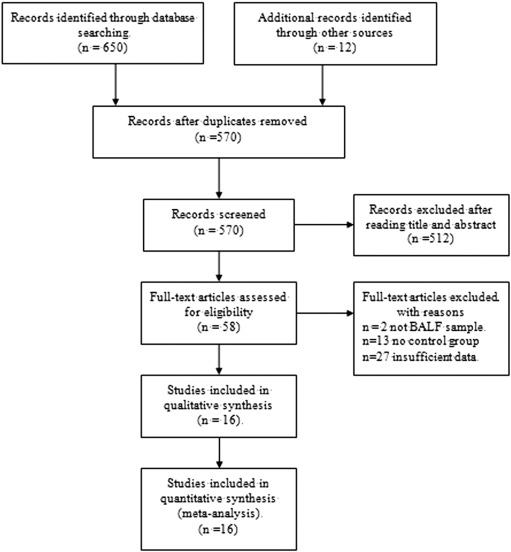
Fig. 1 outlines the study selection, which led to the inclusion of 16 publications in this meta-analysis (Lee et al., 2015 , Suchankova et al., 2013 , von Bartheld et al., 2013 , Hyldgaard et al., 2012 , De Smet et al., 2010 , Korosec et al., 2010 , Danila et al., 2009a , Yao et al., 2008 , Heron et al., 2008 , Fireman et al., 2006 , Smith et al., 2006a , Greco et al., 2005 , Marruchella and Tondini, 2002 , Fireman et al., 1999 , He et al., 1994 and Winterbauer et al., 1993 ). In the studies by Heron et al, BALF CD4/CD8 ratio was analyzed in an analysis cohort and a validation cohort; each was treated as an independent study in our meta-analysis (Heron et al., 2008 ). Consequently, 17 studies were meta-analyzed, 12 of which were prospective and 5 retrospective.
|
|
|
Fig. 1. Study selection. BALF = Bronchoalveolar lavage fluid. |
The mean sample size of eligible studies was 111 (range 30–503), involving 999 patients with sarcoidosis and 886 non-sarcoidosis controls. In all studies, BALF samples were analyzed using flow cytometry. One of the 17 studies blinded diagnosis of patients (von Bartheld et al., 2013 ), while the others did not report blinding. In 10 studies (nine publications) (Hyldgaard et al., 2012 , De Smet et al., 2010 , Korosec et al., 2010 , Yao et al., 2008 , Heron et al., 2008 , Greco et al., 2005 , Marruchella and Tondini, 2002 , Fireman et al., 1999 and He et al., 1994 ), all patients in the case group had biopsy-confirmed sarcoidosis. In seven studies (Lee et al., 2015 , Suchankova et al., 2013 , von Bartheld et al., 2013 , Danila et al., 2009a , Fireman et al., 2006 , Smith et al., 2006a and Winterbauer et al., 1993 ), sarcoidosis was diagnosed based on the combination of clinical, radiological and pathological evidence: diagnosis was based on biopsy showing non-caseating granulomas, after exclusion of other known causes of granulomatosis. Two studies were done in middle-income countries; the others, in high-income countries. Key characteristics of included studies are shown in Table 1 . Patient demographic information is listed in Supplementary Table 1 .
| Author/year [ref] | Country | Ethnicity | Cases/controls | Method | Cut-off value | TP | FP | FN | TN | Study design | Sampling method | Risk of bias | Income |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lee et al. (2015) | South Korea | Asian | 12/57 | FCM | 2.16 | 11 | 9 | 1 | 48 | P | Consecutive | Low | High |
| Suchankova et al. (2013) | Slovakia | Caucasian | 26/27 | FCM | 3.5 | 18 | 1 | 8 | 26 | P | Consecutive | Low | High |
| von Bartheld et al. (2013) | Netherlands | Caucasian | 136/13 | FCM | 3.5 | 73 | 1 | 63 | 12 | P | Consecutive | Low | High |
| Hyldgaard et al. (2012) | Denmark | Caucasian | 19/83 | FCM | 3.8 | 13 | 22 | 6 | 61 | P | Consecutive | High | High |
| De Smet et al. (2010) | Belgium | Caucasian | 36/117 | FCM | 2.62 | 24 | 21 | 12 | 96 | R | Consecutive | Low | High |
| Korosec et al. (2010) | Slovenia | Caucasian | 47/8 | FCM | 3.3 | 33 | 1 | 14 | 7 | P | Consecutive | Low | High |
| Danila et al. (2009a) | Lithuania | Caucasian | 318/185 | FCM | 3.5 | 254 | 18 | 64 | 167 | P | Consecutive | Low | High |
| Yao et al., (2008) | China | Asian | 41/10 | FCM | 4 | 28 | 3 | 13 | 7 | R | Consecutive | High | Middle |
| Heron et al., (2008) | Netherlands | Caucasian | 56/63 | FCM | 3 | 38 | 17 | 18 | 46 | P | Unknown | Low | High |
| Heron et al. (2008) | Netherlands | Caucasian | 26/13 | FCM | 3 | 16 | 2 | 10 | 11 | P | Unknown | Low | High |
| Fireman et al. (2006) | Israel | Caucasian | 67/53 | FCM | 2.5 | 51 | 15 | 16 | 38 | R | Unknown | Low | High |
| Smith et al. (2006a) | USA | Caucasian | 14/12 | FCM | 2.3 | 10 | 2 | 4 | 10 | P | Unknown | Low | High |
| Greco et al. (2005) | Italy | Caucasian | 88/76 | FCM | 3.5 | 48 | 18 | 40 | 58 | R | Consecutive | High | High |
| Marruchella and Tondini (2002) | Italy | Caucasian | 51/38 | FCM | 3.5 | 30 | 5 | 21 | 33 | R | Consecutive | Low | High |
| Fireman et al. (1999) | Israel | Caucasian | 14/16 | FCM | 2.5 | 14 | 3 | 0 | 13 | P | Unknown | Low | High |
| He et al., (1994) | China | Asian | 21/14 | FCM | 3.5 | 18 | 0 | 3 | 14 | P | Consecutive | Low | Middle |
| Winterbauer et al. (1993) | USA | Caucasian | 27/101 | FCM | 4 | 20 | 17 | 7 | 84 | P | Consecutive | Low | High |
BALF = bronchoalveolar lavage fluid; FCM = flow cytometry; FN = false negative; FP = false positive; P = prospective; R = retrospective; TN = true negative; TP = true positive.
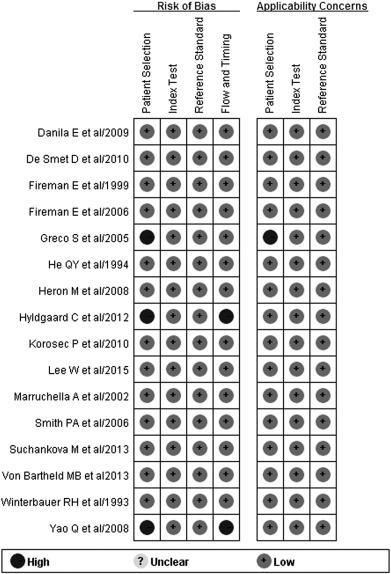
The methodological quality of each study was assessed using QUADAS-2. When a criterion was fulfilled, an answer of Yes was given, Unclear if a criterion was unclear or not reported, and No if a criterion was not achieved. The quality of studies was generally good ( Fig. 2 ), but three studies were judged to have a high risk of bias in the patient selection domain ( Hyldgaard et al., 2012 , Yao et al., 2008 and Greco et al., 2005 ). Two of these studies also showed high risk of bias related to flow and timing ( Hyldgaard et al., 2012 and Yao et al., 2008 ).
|
|
|
Fig. 2. Quality assessment of individual studies in terms of risk of bias and applicability concerns based on the Quality Assessment of Diagnostic Accuracy Studies-2. |
3.2. Diagnostic Accuracy
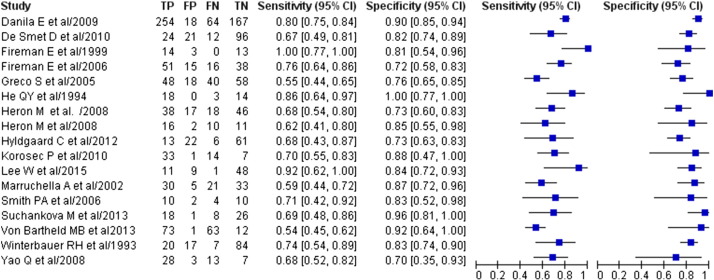
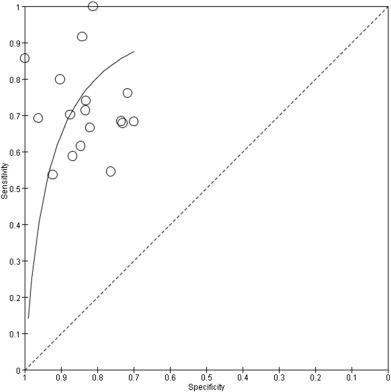
Fig. 3 shows individual study data and provides the Forest plots of sensitivity and specificity for using the BALF CD4/CD8 ratio to diagnose sarcoidosis. Table 2 shows summary estimates of diagnostic performance. Sensitivity ranged from 0.54 to 1.00, and pooled sensitivity was 0.70 (95%CI, 0.64–0.75). Specificity ranged from 0.70 to 1.00, and pooled specificity was 0.83 (95% CI, 0.78–0.86). Pooled PLR and NLR were 4.04 (95%CI, 3.13–5.20) and 0.36 (95%CI, 0.30–0.44), respectively. DOR was 11.17 (95%CI, 7.31–17.07), and the AUC was 0.84 (95%CI, 0.81–0.87) (Fig. 4 ).
|
|
|
Fig. 3. Forest plot of the summary sensitivity and specificity of BALF CD4/CD8 ratio for the diagnosis of sarcoidosis. The sensitivity/specificity of each study is represented as a circle, and the 95%CI is shown as a horizontal line running through the circle. TP = true positive. FP = false positive. FN = false negative. TN = true negative. BALF = bronchoalveolar lavage fluid. |
| Total | Study design | Risk of bias (QUADAS-2) | |||
|---|---|---|---|---|---|
| Prospective | Retrospective | Low | High | ||
| Number of studies | 17 | 12 | 5 | 14 | 3 |
| Sensitivity (95%CI) | 0.70 (0.64–0.75) | 0.72 (0.69–0.76) | 0.64 (0.58–0.70) | 0.72 (0.69–0.75) | 0.60 (0.52–0.68) |
| Heterogeneitya (P) | 58.53 (< 0.001) | 47.43(< 0.001) | 8.89 (0.06) | 52.46 (< 0.001) | 2.68 (0.24) |
| Specificity (95%CI) | 0.83 (0.78–0.86) | 0.84 (0.81–0.87) | 0.79 (0.74–0.83) | 0.84 (0.82–0.87) | 0.75 (0.67–0.81) |
| Heterogeneity (P) | 31.83 (0.01) | 26.97 (0.005) | 4.6 (0.33) | 26.66 (0.01) | 0.28 (0.87) |
| PLR (95%CI) | 4.04 (3.13–5.20) | 4.72 (3.2–6.97) | 2.9 (2.28–3.69) | 4.48 (3.33–6.04) | 2.41 (1.78–3.28) |
| Heterogeneity (P) | 33.98 | 29.23 (0.002) | 3.53 (0.47) | 27.17 (0.01) | 0.15 (0.93) |
| NLR (95%CI) | 0.36 (0.30–0.44) | 0.33 (0.24–0.44) | 0.47 (0.38–0.58) | 0.35 (0.27–0.44) | 0.55 (0.44–0.69) |
| Heterogeneity (P) | 50.55 | 40.61(< 0.001) | 5.81 (0.21) | 42.55 (< 0.001) | 1.31 (0.52) |
| DOR (95%CI) | 11.17 (7.31–17.07) | 17.43 (9.23–32.56) | 6.36 (4.28–9.46) | 15.1 (9.12–24.99) | 4.45 (2.61–7.60) |
| Heterogeneity (P) | 1.50E + 08 | 25.16 (0.01) | 3.73 (0.44) | 27.51 (0.01) | 0.49 (0.78) |
| AUC | 0.84 | 0.87 | 0.78 | 0.86 | 0.77 |
a. Q-value; AUC: Area under the curve; DOR: diagnostic odds ratio; NLR: negative likelihood ratio; PLR: positive likelihood ratio.
|
|
|
Fig. 4. Summary receiver operating characteristic (SROC) curve of BALF CD4/CD8 ratio for the diagnosis of sarcoidosis. Each open circle is the result of a single study. BALF = bronchoalveolar lavage fluid. |
3.3. Meta-regression Analysis
Chi-squared values for pooled diagnostic performance parameters were high: sensitivity, 58.53; specificity, 31.83; PLR, 33.98; NLR, 50.55; and DOR, 1.5 × 108 (all P < 0.05). This indicates significant heterogeneity among the included studies (Table 2 ). In order to identify possible reasons for this heterogeneity, meta-regression was conducted to assess the effect of study quality on the relative DOR (RDOR) of BALF CD4/CD8 ratio for diagnosis of sarcoidosis. The characteristics of these covariates are listed in Table 1 . Diagnostic accuracy was not significantly affected by sample size (P = 0.24), publication year (P = 0.34), sampling method (P = 0.56), income (P = 0.93), or cut-off value (P = 0.57). Ethnicity (Asian vs. Caucasian) did not significantly affect RDOR (P = 0.93) Two factors may affect the diagnostic accuracy: study design (P = 0.048) and risk of bias (P = 0.03). The meta-regression results are shown in detail in Table 3 .
| Covariate | No. of studies | Coefficient | RDOR (95%CI) | P |
|---|---|---|---|---|
| Sample size | ||||
| ≥ 100 | 8 | − 0.61 | 0.54 (0.19–1.58) | 0.24 |
| < 100 | 9 | |||
| Study design | ||||
| Prospective | 12 | − 0.91 | 0.40 (0.16–0.99) | 0.048 |
| Retrospective | 5 | |||
| Publication year | ||||
| After 2005 | 13 | − 0.59 | 0.55 (0.15–2.02) | 0.34 |
| Before 2005 | 4 | |||
| Sampling method | ||||
| Consecutive | 12 | 0.34 | 1.41 (0.42–4.75) | 0.56 |
| Unknown | 5 | |||
| Risk of Bias | ||||
| Low | 14 | 1.14 | 3.12 (1.12–8.7) | 0.03 |
| High | 3 | |||
| Income | ||||
| High | 15 | 0.08 | 1.08 (0.15–7.68) | 0.93 |
| Low/middle | 2 | |||
| Cut-off value | ||||
| 3–4 | 13 | − 0.35 | 0.70 (0.19–2.59) | 0.57 |
| < 3 or > 4 | 4 | |||
| Ethnicity | 0.08 | 1.08 (0.15–7.68) | 0.93 | |
| Asian | 2 | |||
| Caucasian | 15 |
BALF: bronchoalveolar lavage fluid. RDOR: relative diagnostic odds ratio
Based on these meta-regression results, we performed sensitivity analysis by stratifying study participants based on study design and risk of bias. These two analyses suggested that prospective studies and high-quality studies (with low risk of bias) reported much better diagnostic performance than retrospective studies or studies with high or uncertain risk of bias (Table 2 ).
3.4. Publication Bias Evaluation
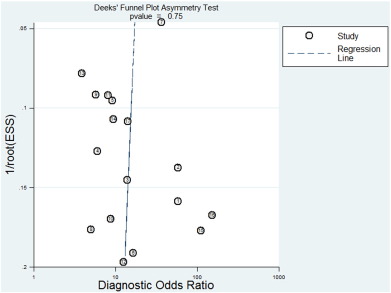
Deekss funnel plot asymmetry test was used to assess likelihood of publication bias in the final set of studies. The slope coefficient was associated with P = 0.75, and the shape of the funnel plot of the pooled DOR of the CD4/CD8 ratio was not obviously asymmetrical (Fig. 5 ).
|
|
|
Fig. 5. Funnel plots for assessing the risk of publication bias. The funnel graph shows the log of the diagnostic odds ratio (DOR) plotted against the standard error of the log of the DOR (an indicator of sample size). Solid circles represent each study in the meta-analysis. The line indicates the regression result. |
4. Discussion
Numerous studies have focused on the potential value of BALF CD4/CD8 ratio for diagnosis of sarcoidosis. This is based on the fact that CD4 + T cells interacting with antigen-presenting cells appear to trigger formation of sarcoid granulomas and help maintain them (Iannuzzi et al., 2007 and Baughman et al., 2003 ). In addition, activated alveolar macrophages and CD4 + T cells participate in the influx of mononuclear cells into the alveoli that often precedes sarcoid granuloma formation in the lung (Baughman et al., 2011 and Jones, 2002 ). Diagnostic studies have reported highly variable sensitivity and specificity when using the CD4/CD8 ratio, prompting us to perform what we believe to be the first meta-analysis to assess the available evidence on the diagnostic usefulness of this ratio in sarcoidosis.
The pooled results indicate relatively low diagnostic sensitivity of 0.70, and specificity of 0.83, suggesting a relatively high rate of missed diagnoses (30%) and misdiagnoses (17%). Likelihood ratios > 10 and < 0.1 are considered as strong indicators to rule in or rule out a diagnosis, respectively (Deeks and Altman, 2004 ). In the present meta-analysis, PLR was 4.04 and NLR was 0.36, suggesting relatively low ability to discriminate sarcoidosis from non-sarcoidosis, although the AUC in SROC analysis was relatively high (0.84). While it appears the BALF CD4/CD8 ratio is not robust enough on its own to diagnose sarcoidosis, pooled DOR was moderate (11.17), suggesting that it may be a helpful ancillary tool that, when interpreted together with other diagnostic factors, can improve sarcoidosis diagnosis.
QUADAS-2 was used to assess methodological quality of the studies included in our meta-analysis. QUADAS-2 provides more detailed and rigorous assessment than the earlier QUADAS, such as in the explanation of indeterminate results. Although this instrument can be used to assign quality scores (Whiting et al., 2003 ), it is fundamentally a qualitative tool, which we used to characterize risk of bias along four domains as low, high, or unclear. This analysis suggested quality differences among the included studies, and meta-regression suggested that study quality may have affected the reported diagnostic performance of the BALF CD4/CD8 ratio. In addition, diagnostic performance of the BALF CD4/CD8 ratio was variable even among studies at low risk of bias. These findings highlight the need for better-designed diagnostic studies, particularly prospective studies and studies with low risk of bias. These studies should carefully address the issue of variability of the BALF CD4/CD8 ratio as a diagnostic tool.
The studies included in this meta-analysis varied in their cut-off values for the CD4/CD8 ratio, which usually fell between 2 and 4. No international standards exist about what cut-off value to use, and this value is likely to vary with clinical context, depending on country, ethnicity, examination equipment, disease severity, and history of corticosteroid treatment. Our meta-regression of cut-off values suggests that different cut-off values did not substantially affect the diagnostic accuracy of the BALF CD4/CD8 ratio (P = 0.57, Table 2 ). This contrasts with previous studies showing that a CD4/CD8 ratio ≥ 3.5 strongly suggests sarcoidosis, but is not specific enough on its own to diagnose the disease ( Marruchella and Tondini, 2002 and Costabel et al., 2010 ). Further work should aim to identify the cut-off value that provides optimal diagnostic accuracy, and researchers should be open to the possibility that different cut-offs are needed for different types of patients or clinical contexts. In addition, all included studies utilized flow cytometry to determine the BALF CD4/CD8 ratio based on a protocol developed for peripheral blood samples. Several studies have reported that flow cytometric typing of lymphocytes from BALF correlates well with results from conventional immunocytochemistry ( Brandt et al., 1996 , Ma et al., 2001 , Smith et al., 2006b and Szpechcinski et al., 2011 ), though the results may depend on combinations of antibodies and gating strategies. Therefore future studies are needed to optimize these parameters for typing lymphocytes in BALF.
There are several factors that should be addressed which may influence the BALF CD4/CD8 ratio. One factor is smoking, which is associated with higher total cell number, higher proportions of CD8 + lymphocytes and CD4 + cells, and lower CD4 +/CD8 + ratio in BALF (Hoser et al., 1999 ). Numbers of CD4 + and CD8 + cells in BALF are also affected in smokers with comorbidities such as chronic obstructive pulmonary disease (Forsslund et al., 2014 ). Several studies included in this meta-analysis reported smoking history, while its effect on BALF CD4/CD8 ratio remains unclear (Korosec et al., 2010 and Fireman et al., 2006 ). Future studies should examine the potential impact of smoking on BALF cell numbers and proportions, and thereby on the potential diagnostic usefulness of the BALF CD4/CD8 ratio. Another factor was the stage of sarcoidosis. As pulmonary sarcoidosis advances from stage I to stage III, the number of CD8 + cells increases and the number of CD4 + cells decreases, leading to a decrease in the CD4/CD8 ratio (Danila et al., 2008 ). Indeed, previous work has shown that the diagnostic sensitivity of the BALF CD4/CD8 ratio decreases with increasing stage of sarcoidosis (Danila et al., 2009a ); on the other hand, the ratio may be less clinically useful in stage I disease, for which clinical and radiographic features on their own show high diagnostic reliability. A third factor that should be addressed is the recovery rate of BALF, which has been reported to range widely from 23.3% to 91.3% (Fireman et al., 2006 and Winterbauer et al., 1993 ). Most studies included in our meta-analysis did not report the recovery rate, which may bias diagnostic accuracy. A fourth factor was whether or not participants were on corticosteroid treatment, which can modify lymphocyte proportions in BALF and thereby the BALF CD4/CD8 ratio (Danila et al., 2009b ). Indeed, some studies suggest that this ratio shows lower diagnostic sensitivity in patients on such treatment (Danila et al., 2009a and He et al., 1994 ). None of the patients in nine studies (eight publications) received corticosteroids (Suchankova et al., 2013 , De Smet et al., 2010 , Korosec et al., 2010 , Yao et al., 2008 , Heron et al., 2008 , Fireman et al., 2006 , Fireman et al., 1999 and Winterbauer et al., 1993 ), while the other studies did not report corticosteroid status (Lee et al., 2015 , von Bartheld et al., 2013 , Hyldgaard et al., 2012 , Smith et al., 2006a , Greco et al., 2005 and Marruchella and Tondini, 2002 ). Future studies should take into account possible confounding by smoking status, recovery rate, and corticosteroid treatment when assessing the diagnostic performance of BALF CD4/CD8 ratio. Studies should also look systematically at whether the ratio is more clinically useful for advanced stages of the disease, where clinical and radiographic features on their own can be less informative.
The findings of this meta-analysis should be interpreted with caution because of several limitations. First, we excluded conference abstracts, reviews, editorials, case reports and articles not written in English or Chinese, which may bias our results. In addition, we omitted unpublished studies and studies not indexed in our set of databases. Nevertheless, our funnel plots suggested no significant risk of publication bias. Second, there may have been misclassification bias. The diagnosis of sarcoidosis in the case group was biopsy-confirmed in only 12 studies (11 publications); in the remaining five studies (Lee et al., 2015 , Suchankova et al., 2013 , Danila et al., 2009a , Fireman et al., 2006 and Smith et al., 2006a ), sarcoidosis was diagnosed based on the combination of clinical, radiological, pathological and follow-up observation. Some cases lacked histological evidence of non-caseating granulomas. Third, most studies in our meta-analysis did not report blinding, which increases the risk of analytical bias. Future diagnostic studies should avoid these methodological problems. Future studies should also examine the cost-effectiveness of determining BALF lymphocyte ratios relative to more invasive biopsy, as well as compare the ratio with endobronchial ultrasound transbronchial needle aspiration, which has been shown to be a safe and effective diagnostic procedure (Costabel et al., 2010 and Agarwal et al., 2012 ).
In conclusion, our meta-analysis provides the most comprehensive evidence to date that the determination of the BALF CD4/CD8 ratio can aid in the diagnosis of sarcoidosis, with an elevated ratio associated with greater likelihood of disease in patients who present a typical clinical/radiological picture of the disease. However, the ratio is not specific or selective enough to use on its own; rather, it must always be combined with other established diagnostic factors and tests.
The following is the supplementary data related to this article.
Supplementary Table 1.
Patient demographic information.
Author Contributions
Y.C.S. and C.S.P. conceived the project, performed the systematic review and meta-analysis, and drafted and revised the manuscript. These authors also take full responsibility for the integrity of the data and the accuracy of the data analysis. Y.Q.W., C.W. and D.D.L. contributed to the systematic review and helped draft the manuscript. Z.L.L., T.Y. and L.C. contributed to data acquisition and analysis. F.Q.W. drafted and revised the manuscript.
Declaration of interests
All authors have read the journals policy on disclosure of potential conflicts of interest and have none to declare.
Acknowledgments
This work was supported by grants 81230001 and 81300032 from the National Natural Science Foundation of China .
References
- Agarwal et al., 2012 R. Agarwal, A. Srinivasan, A.N. Aggarwal, D. Gupta; Efficacy and safety of convex probe EBUS-TBNA in sarcoidosis: a systematic review and meta-analysis; Respir. Med., 106 (2012), pp. 883–892
- American Thoracic Society: European Respiratory Society Worl American Thoracic Society: European Respiratory Society World association of sarcoidosis and other granulomatous disorders; Statement on sarcoidosis; Am. J. Respir. Crit. Care Med., 160 (1999), pp. 736–755
- Baughman and Grutters, 2015 R.P. Baughman, J.C. Grutters; New treatment strategies for pulmonary sarcoidosis: antimetabolites, biological drugs, and other treatment approaches; Lancet Respir. Med., 3 (2015), pp. 813–822
- Baughman et al., 2003 R.P. Baughman, E.E. Lower, R.M. du Bois; Sarcoidosis; Lancet, 361 (2003), pp. 1111–1118
- Baughman et al., 2011 R.P. Baughman, D.A. Culver, M.A. Judson; A concise review of pulmonary sarcoidosis; Am. J. Respir. Crit. Care Med., 183 (2011), pp. 573–581
- Brandt et al., 1996 B. Brandt, M. Thomas, M. von Eiff, G. Assmann; Immunophenotyping of lymphocytes obtained by bronchoalveolar lavage: description of an all-purpose tricolor flow cytometric application; J. Immunol. Methods, 194 (1996), pp. 95–102
- Chretien et al., 1985 J. Chretien, A. Venet, C. Danel, D. Israel-Biet, D. Sandron, A. Arnoux; Bronchoalveolar lavage in sarcoidosis; Respiration, 48 (1985), pp. 220–223
- Costabel, 1997 U. Costabel; CD4/CD8 ratios in bronchoalveolar lavage fluid: of value for diagnosing sarcoidosis?; Eur. Respir. J., 10 (1997), pp. 2699–2700
- Costabel et al., 2008 U. Costabel, S. Ohshimo, J. Guzman; Diagnosis of sarcoidosis; Curr. Opin. Pulm. Med., 14 (2008), pp. 455–461
- Costabel et al., 2010 U. Costabel, F. Bonella, S. Ohshimo, J. Guzman; Diagnostic modalities in sarcoidosis: BAL, EBUS, and PET; Semin. Respir. Crit. Care Med., 31 (2010), pp. 404–408
- Danila et al., 2008 E. Danila, L. Jurgauskiene, R. Malickaite; BAL fluid cells and pulmonary function in different radiographic stages of newly diagnosed sarcoidosis; Adv. Med. Sci., 53 (2008), pp. 228–233
- Danila et al., 2009a E. Danila, J. Norkūniene, L. Jurgauskiene, R. Malickaite; Diagnostic role of BAL fluid CD4/CD8 ratio in different radiographic and clinical forms of pulmonary sarcoidosis; Clin. Respir. J., 3 (2009), pp. 214–221
- Danila et al., 2009b E. Danila, L. Jurgauskiene, J. Norkuniene, R. Malickaite; BAL fluid cells in newly diagnosed pulmonary sarcoidosis with different clinical activity; Ups. J. Med. Sci., 114 (2009), pp. 26–31
- De Smet et al., 2010 D. De Smet, G.A. Martens, B.V. Berghe, et al.; Use of likelihood ratios improves interpretation of laboratory testing for pulmonary sarcoidosis; Am. J. Clin. Pathol., 134 (2010), pp. 939–947
- Deeks and Altman, 2004 J.J. Deeks, D.G. Altman; Diagnostic tests 4: likelihood ratios; BMJ, 329 (2004), pp. 168–169
- Deeks et al., 2005 J.J. Deeks, P. Macaskill, L. Irwig; The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed; J. Clin. Epidemiol., 58 (2005), pp. 882–893
- Devillé et al., 2002 W.L. Devillé, F. Buntinx, L.M. Bouter, et al.; Conducting systematic reviews of diagnostic studies: didactic guidelines; BMC Med. Res. Methodol., 2 (2002), p. 9
- Fireman et al., 1999 E. Fireman, I. Topilsky, J. Greif, et al.; Induced sputum compared to bronchoalveolar lavage for evaluating patients with sarcoidosis and non-granulomatous interstitial lung disease; Respir. Med., 93 (1999), pp. 827–834
- Fireman et al., 2006 E. Fireman, T. Boikaner, I.E. Priel; Combined CD4/CD8 ratio in induced sputum and pulmonary function testing for non-invasive identification of sarcoidosis; Transl. Res., 148 (2006), pp. 87–95
- Forsslund et al., 2014 H. Forsslund, M. Mikko, R. Karimi, et al.; Distribution of T-cell subsets in BAL fluid of patients with mild to moderate COPD depends on current smoking status and not airway obstruction; Chest, 145 (2014), pp. 711–722
- Greco et al., 2005 S. Greco, A. Marruchella, M. Massari, C. Saltini; Predictive value of BAL cellular analysis in differentiating pulmonary tuberculosis and sarcoidosis; Eur. Respir. J., 26 (2005), pp. 360–361 (author reply 362)
- He et al., 1994 Q.Y. He, S. Kitamura, Y. Ishii; Measurement of T cell subsets in bronchoalveolar lavage fluid for diagnosis of pulmonary sarcoidosis; Zhonghua Yi Xue Za Zhi, 74 (1994), pp. 87–89 (126-27)
- Heron et al., 2008 M. Heron, W.A. Slieker, P. Zanen, et al.; Evaluation of CD103 as a cellular marker for the diagnosis of pulmonary sarcoidosis; Clin. Immunol., 126 (2008), pp. 338–344
- Hoser et al., 1999 G. Hoser, J. Kawiak, J. Domagała-Kulawik, P. Kopiński, W. Droszcz; Flow cytometric evaluation of lymphocyte subpopulations in BALF of healthy smokers and nonsmokers; Folia Histochem. Cytobiol., 37 (1999), pp. 25–30
- Hyldgaard et al., 2012 C. Hyldgaard, S. Kaae, M. Riddervold, H.J. Hoffmann, O. Hilberg; Value of s-ACE, BAL lymphocytosis, and CD4 +/CD8 + and CD103 + CD4 +/CD4 + T-cell ratios in diagnosis of sarcoidosis; Eur. Respir. J., 39 (2012), pp. 1037–1039
- Iannuzzi et al., 2007 M.C. Iannuzzi, B.A. Rybicki, A.S. Teirstein; Sarcoidosis; N. Engl. J. Med., 357 (2007), pp. 2153–2165
- Jones, 2002 N.P. Jones; Sarcoidosis; Curr. Opin. Ophthalmol., 13 (2002), pp. 393–396
- Kantrow et al., 1997 S.P. Kantrow, K.C. Meyer, P. Kidd, et al.; The CD4/CD8 ratio in BAL fluid is highly variable in sarcoidosis; Eur. Respir. J., 10 (12) (1997), pp. 2716–2721
- Korosec et al., 2010 P. Korosec, M. Rijavec, M. Silar, I. Kern, M. Kosnik, K. Osolnik; Deficiency of pulmonary Vα24 Vβ11 natural killer T cells in corticosteroid-naïve sarcoidosis patients; Respir. Med., 104 (2010), pp. 571–577
- Kvale, 2003 P.A. Kvale; Is it difficult to diagnose sarcoidosis?; Chest, 123 (2003), pp. 330–332
- Lee et al., 2015 W. Lee, W.S. Chung, K.S. Hong, J. Huh; Clinical usefulness of bronchoalveolar lavage cellular analysis and lymphocyte subsets in diffuse interstitial lung diseases; Ann Lab Med., 35 (2015), pp. 220–225
- Leeflang et al., 2008 M.M. Leeflang, J.J. Deeks, C. Gatsonis, P.M. Bossuyt, Cochrane Diagnostic Test Accuracy Working Group; Systematic reviews of diagnostic test accuracy; Ann. Intern. Med., 149 (2008), pp. 889–897
- Ma et al., 2001 W. Ma, W. Cui, Q. Lin; Improved immnunophenotyping of lymphocytes in bronchoalveolar lavage fluid (BALF) by flow cytometry; Clin. Chim. Acta, 313 (2001), pp. 133–138
- Marruchella and Tondini, 2002 A. Marruchella, M. Tondini; Reliability of bronchoalveolar lavage in the routineclinical assessment of patients with sarcoidosis. A retrospective analysis; Panminerva Med., 44 (2002), pp. 257–260
- Moses et al., 1993 L.E. Moses, D. Shapiro, B. Littenberg; Combining independent studies of a diagnostic test into a summary ROC curve: data analytic approaches and some additional considerations; Stat. Med., 12 (1993), pp. 1293–1316
- Nguyen et al., 2015 A.H. Nguyen, E.J. Miller, C.S. Wichman, I.G. Berim, D.K. Agrawal; Diagnostic value of tumor antigens in malignant pleural effusion: a meta-analysis; Transl. Res., 166 (2015), pp. 432–439
- Smith et al., 2006a P.A. Smith, L.M. Kohli, K.L. Wood, C.A. Hage, H.L. Twigg, K.S. Knox; Cytometric analysis of BAL T cells labeled with a standardized antibody cocktail correlates with immunohistochemical staining; Cytometry B Clin. Cytom., 70 (2006), pp. 170–178
- Smith et al., 2006b P.A. Smith, L.M. Kohli, K.L. Wood, C.A. Hage, H.L. Twigg 3rd, K.S. Knox; Cytometric analysis of BAL T cells labeled with a standardized antibody cocktail correlates with immunohistochemical staining; Cytometry B Clin. Cytom., 70 (2006), pp. 170–178
- Stoller et al., 1987 J.K. Stoller, J.A. Rankin, H.Y. Reynolds; The impact of bronchoalveolar cell analysis on clinicians' diagnostic reasoning about interstitial lung disease; Chest, 92 (1987), pp. 839–843
- Suchankova et al., 2013 M. Suchankova, M. Bucova, E. Tibenska, et al.; Triggering receptor expressed on myeloid cells-1 and 2 in bronchoalveolar lavage fluid in pulmonary sarcoidosis; Respirology, 18 (2013), pp. 455–462
- Szpechcinski et al., 2011 A. Szpechcinski, P. Kopinski, D. Giedronowicz, A. Rozy, P. Jagus, M. Szolkowska, J. Chorostowska-Wynimko; Simple flow cytometric protocol of CD4 +/CD8 + lymphocyte ratio assessment in bronchoalveolar lavage fluids from patients with interstitial lung diseases; Anal. Quant. Cytol. Histol., 33 (2011), pp. 289–296
- von Bartheld et al., 2013 M.B. von Bartheld, O.M. Dekkers, A. Szlubowski, et al.; Endosonography vs conventional bronchoscopy for the diagnosis of sarcoidosis: the GRANULOMA randomized clinical trial; JAMA, 309 (2013), pp. 2457–2464
- Wells and Hirani, 2008 A.U. Wells, N. Hirani; Interstitial lung disease guideline; Thorax, 63 (2008), pp. v1–v58
- Whiting et al., 2003 P. Whiting, A.W. Rutjes, J.B. Reitsma, P.M. Bossuyt, J. Kleijnen; The development of QUADAS: a tool for the quality assessment of studies of diagnostic accuracy included in systematic reviews; BMC Med. Res. Methodol., 3 (2003), p. 25
- Whiting et al., 2011 P.F. Whiting, A.W. Rutjes, M.E. Westwood, et al.; QUADAS- 2: a revised tool for the quality assessment of diagnostic accuracy studies; Ann. Intern. Med., 155 (2011), pp. 529–536
- Winterbauer et al., 1993 R.H. Winterbauer, J. Lammert, M. Selland, R. Wu, D. Corley, S.C. Springmeyer; Bronchoalveolar lavage cell populations in the diagnosis of sarcoidosis; Chest, 104 (1993), pp. 352–361
- Yao et al., 2008 Q. Yao, Z.J. Xu, H. Huang, et al.; Predictive value of single and combined indexes in the diagnosis of sarcoidosis; Zhonghua Jie He He Hu Xi Za Zhi, 31 (2008), pp. 488–491
Document information
Published on 06/04/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?