Highlights
- Small studies support the use of FFR in non-culprit vessels during ACS.
- FFR demonstrates diagnostic accuracy and reproducibility in AMI, particularly in intermediate lesions.
- Microvascular dysfunction observed from earlier work does not appear to hinder FFR calculation, but further studies are required for validation.
- Vasodilator independent assessment of coronary stenoses has some preliminary data supporting its use in non culprit arteries during ACS, but ongoing trial results are awaited regarding clinical outcomes.
Abstract
Fractional flow reserve (FFR) assessment provides anatomical and physiological information that is often used to tailor treatment strategies in coronary artery disease. Whilst robust data validates FFR use in stable ischaemic heart disease, its use in acute coronary syndromes (ACS) is less well investigated. We critically review the current data surrounding FFR use across the spectrum of ACS including culprit and non-culprit artery analysis. With adenosine being conventionally used to induce maximal hyperaemia during FFR assessment, co-existent clinical conditions may preclude its use during acute myocardial infarction. Therefore, we include a current review of instantaneous wave free ratio as a novel vasodilator independent method of assessing lesion severity as an alternative strategy to guide revascularisation in ACS.
Keywords
Fractional flow reserve;Coronary physiology;Acute coronary syndromes
1. Fractional flow reserve assessment in acute coronary syndromes
Acute coronary syndromes (ACS) are the only clinical condition in which there is a proven mortality benefit with percutaneous coronary intervention (PCI) [1]. In the case of ST-elevation and acute coronary occlusion, the culprit vessel is usually identifiable. However, bystander coronary artery disease or multi-vessel disease occurs in approximately one half of ACS presentations, where diagnosing the culprit lesion can be less straight-forward with an associated poorer prognosis [2]; [3] ; [4]. Whilst in stable coronary artery disease, physiological assessment using techniques such as fractional flow reserve (FFR) assessment is often used to aid operators in deciding which lesion to treat [5]; [6]; [7] ; [8], its use in ACS is less well investigated. (See Table 1.)
| Study/Ref. | N | Exclusions | Study outline | Significant results |
|---|---|---|---|---|
| De Bruyne et al. [24] Circulation 2001 | 57 | Myocardial akinesia, LVSD in non-culprit territories, diameter of target vessel < 2.5 mm | FFRa vs SPECT > 6 days post ACS | Sensitivity/specificity: 82%/87% p < 0.001) |
| Samady et al. [25] J Am Coll Cardiol 2006 | 48 | CTO, ongoing ischaemia, haemodynamic instability, prior MI in index territory, LMS disease, three vessel disease | FFRa vs SPECT and contrast echo < 6 days post ACS | Sensitivity/specificities: SPECT: 83%/93% Contrast echo: 90%/100% (p < 0.001) |
| Ntalianis et al. [27] JACC Cardiovasc Interv. 2010 | 101 | Haemodynamic instability | FFR measured acutely and 35 +/− 4 days in non-culprit arteries post ACS | Acute and follow-up FFR: both 0.77 +/− 0.13 (p = NS) |
| Sels et al. [28] JACC Cardiovasc Interv. 2011 | 328 | LMS disease, previous CABG, STEMI < 5 days prior | Outcomes of using FFR to guide revascularisation in FAME population with ACS vs stable angina (SA) | Absolute risk reduction of ACS vs SA :5.1% vs 3.7% (p = 0.92) |
| Lopez-Palop et al. [29] Rev Esp Cardiol 2012 | 107 | ISR, patients pre-scheduled for angiography | Outcomes of using FFRa to guide revascularisation in non-culprit arteries in ACS | MACE of non-treated vs treated group: 7.4% vs 7.7% (p = 0.52) |
MACE — major adverse cardiovascular events (cardiovascular related death, non-fatal MI, urgent revascularisation), CTO — chronic total occlusion, LMS — left main stem, ISR — in-stent re-stenosis, LVSD — left ventricular systolic dysfunction, CABG — coronary artery bypass graft.
a. FFR cut off value used < 0.75.
Identification of the culprit artery in ST elevation MI is usually straight-forward by utilising information from the surface electrocardiogram and coronary angiography. It is recognised that FFR values in the culprit vessel are higher during acute episodes when compared to measurements made after the microcirculation has had some time to recover [9] ; [10]. It is postulated that this is due to a reduction in the level of attainable hyperaemia in the culprit vessel due to embolisation of thrombus and plaque, ischaemic microvascular dysfunction and myocardial stunning [11]. These states are perhaps most marked in acute ST elevation myocardial infarction. Therefore, the physiological community remain cautious about the application of FFR in culprit artery disease [12]. The use of FFR in assessing the haemodynamic significance of non-culprit lesions in AMI and unstable angina has only been assessed in small studies with differing conclusions. Some studies suggest that transitory microvascular damage in myocardial territories remote from the culprit lesion and the dynamic nature of the injuries limit the reliability of measured indices. Whilst others suggest that FFR can be reliably measured to guide management in this population [13] ; [14].
By demonstrating territories of inducible ischaemia in the context of stable coronary artery disease, lesion selection for coronary intervention can be made easier particularly in multi-vessel disease [15]. Physiological assessment offers an invasive pressure based index of the haemodynamic significance of coronary stenoses and its use has been validated in several clinical trials to guide appropriateness of PCI in stable coronary disease [5]; [6] ; [7].
Data suggests that complete revascularisation of patients with significant multi-vessel disease within a month of primary PCI is associated with an improved survival benefit [2] ; [3]. More recent data from the randomised PRAMI (Preventative Angioplasty in Acute Myocardial Infarction) trial showed that immediate preventative revascularisation of non-culprit arteries with an angiographically significant stenosis at the time of primary PCI for an ST segment elevation myocardial infarction may have prognostic advantages over culprit PCI alone [4] ; [16]. It is important to note, however, that the PRAMI trial compared non-culprit PCI to abstinence from further PCI even where significant or high-grade stenosis was left untreated. This meant that physiologically significant lesions would have been treated in the same manner as intermediate and mild lesions (> 50% stenosis). By deferring high grade stenosis, PRAMI deviated significantly from routine practice where high grade stenoses are known to be associated with high event rates [7]. However, treating all patients with a greater than 50% stenosis means that a significant number of patients will receive stents which may be considered physiologically inappropriate. This strategy of deferral of all lesions greater than 50% stenosis with physiologically negative lesions to conservative management in the control arm may also explain why a staged approach to complete revascularisation of significant stenoses confers a prognostic advantage to immediate PCI of non-culprit vessels [17] ; [18]. It is now recognised from the French Registry, earlier studies and the RIPCORD study that angiography guided revascularisation can be misleading when compared to a physiological guided strategy [19] ; [20].
2. Myocardial infarction and micro-vascular disease
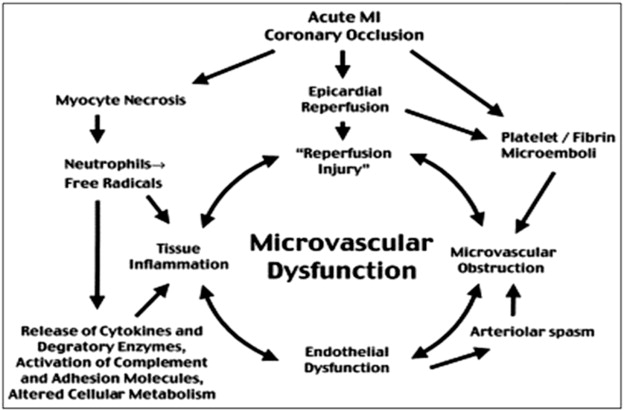
There are various theories describing the pathophysiological mechanisms underlying micro-vascular dysfunction in the peri-infarct period. It is hypothesised that endothelial cell integrity is jeopardised in ischaemic reperfusion injury, with a subsequent reduction in endothelial derived vasodilators such as nitric oxide and an increase in potent vasoconstrictors such as endothelin and oxygen free radicals. This culminates in an overall reduction in myocardial flow. Endovascular injury results in a pro-coagulant and pro-inflammatory state caused by a cascade of activated platelets, neutrophils and adhesion molecules known to mediate myocardial damage [21] (See Fig. 1).
|
|
|
Fig. 1. Pathophysiology of microvascular dysfunction post AMI. |
When accompanied by interstitial oedema and cell contraction, this can result in micro-capillary occlusion. Oxidative stress by the generation of oxygen free-radicals can directly cause further myocardial injury to an ischaemic focus. The release of increased intracellular calcium can alter sarcolemmal calcium regulation which can promote myofibrillar damage in addition to generating a pro-arrhythmic state [22].
Micro-vascular dysfunction in AMI patients is accepted as occurring in territories supplied by culprit arteries and forms the mechanism behind the ‘no re-flow’ phenomenon post intervention. Although regional micro-vascular dysfunction in territories remote from an acutely infarcted area has been described [23], small studies have attempted to address whether this would impact on FFR measurements of non-culprit lesions [12]. This can also aid in the identification of culprit lesions in multi-vessel disease.
3. FFR assessment in AMI
FFR is described as the ratio of maximal hyperaemic myocardial blood flow in the presence of a coronary stenosis to the normal hyperaemic blood flow in the same vessel if it were normal. In other words, the extent to which maximal myocardial blood flow is impeded by an epicardial stenosis. In order to achieve maximal myocardial flow and to minimise intracoronary resistance which is essential in the determination of the FFR, both the epicardial and microvasculature are vasodilated. The commonest agent used to ensure vasodilatation is adenosine [12]. Based on large clinical outcome trials ESC/AHA/ACC guidelines recommend a cut-off value of less than or equal to 0.80 as a guide to perform revascularisation [8].
It has been hypothesised that microvascular congestion may attenuate hyperaemic blood flow, following AMI, leading to a reduced trans-stenotic pressure gradient. The resulting effect could theoretically lead to an underestimation of lesion severity producing an artificially high FFR reading. Tamita et al. reported that post-interventional FFR was higher in AMI patients than in the stable angina patients following PCI with no significant differences in IVUS parameters (mean luminal areas) [9]. Tani et al. described an exploratory finding in patients post AMI who had related wall ischaemia on myocardial perfusion single-photon emission computed tomography (SPECT), but had non-physiologically significant FFR measurements of 0.87 and 0.89 in the left anterior descending and right coronary arteries respectively. The mismatch between the diagnostic modalities led the authors to suggest applying caution when interpreting FFR in culprit lesions post MI [10]. It is important to note, however, that both of these small studies measured FFR in the peri-infarct period in culprit arteries only and no assessments were performed in non-culprit bystander lesions.
De Bruyne et al. examined 57 patients at least six days post AMI, the sensitivity and specificity of FFR with a cut-off level of less than 0.75 to detect a perfusion defect on SPECT was 82% and 87%, respectively. When only true positive and negative SPECT imaging was considered, the corresponding values were 87% and 100% (p < 0.001) [24]. Accordingly, the authors concluded that when measured greater than six days post AMI, FFR accurately reflects the haemodynamic lesion severity and its impact on myocardial perfusion despite the damaged infarct zone micro-vasculature.
Samady et al. went on to study 48 patients earlier post AMI (3.7 +/− 1.3 days) than De Bruyne et al. and compared the relationship of FFR with SPECT and myocardial contrast echo. To identify true reversibility, follow-up SPECT was performed 11 weeks after PCI. The sensitivity, specificity, and concordance of FFR ≤ 0.75 for detecting true reversibility on SPECT were 88%, 93%, and 91% (chi-square p < 0.001) and for detecting reversibility on myocardial contrast echo were 90%, 100%, and 93% (chi-square p < 0.001), respectively. The optimal FFR value for discriminating inducible ischaemia on non-invasive imaging was demonstrated as 0.78, similar to findings from De Bruyne et al. Thus FFR of the infarct related artery accurately identified reversibility on non-invasive imaging, supporting its use early post AMI.
Whilst these studies evolved the physiological field towards a growing population of unstable patients, many of these studies were performed in the era of thrombolysis, when coronary angiography was performed 3 days after the acute event. Thus it may be difficult to translate some of the findings directly to contemporary clinical practice. Additionally, in accordance with study design at the time, patients with occluded infarct-related arteries, haemodynamic instability, prior MI in the index vessel territory, complex three vessel disease, left main stenosis or ongoing ischaemia were excluded from the study, whereas nowadays such patients reflect a substantial proportion of the patient population encountered in real world practice. The authors also stated that the reduction in post MI vasodilator reserve may result in FFR and non-invasive imaging underestimating the haemodynamic significance of residual lesions in culprit arteries [25].
Ntalianis et al. sought to assess reliability of FFR of non-culprit coronary stenoses during PCI in AMI. 101 patients undergoing PCI for AMI were prospectively recruited, approximately three quarters of which were ST elevation MI and one quarter non-ST-elevation MI. FFR measurements in 112 non-culprit stenoses, in which PCI was contemplated on the basis of the angiogram (greater than 50% stenosis), were obtained immediately post PCI of the culprit vessel and repeated 35 +/− 4 days later. No difference was found between the FFR value of the non-culprit stenoses between the acute and follow-up phase of the study (0.77 +/− 0.13 vs. 0.77 +/− 0.13, respectively, P = NS). Only 2 patients had FFR values higher than 0.80 acutely and lower than 0.75 at follow-up but TIMI flow, corrected TIMI frame count, percentage diameter stenosis, minimum luminal diameter and index of microcirculatory resistance (IMR) did not change between the acute and followup phases. Although IMR did not change, the authors caution that this may have been due to the relatively short duration between the two measurements [26]. Additionally, most cases included in the study had inferior and lateral wall ST changes on ECG or non-ST-elevation MI. Haemodynamically unstable patients were also excluded on account of extensive necrosis and cardiogenic shock precluding hyperaemic assessment of non-culprit stenoses, in line with accepted medical practice. The study confirmed that non-culprit stenoses with an FFR less than 0.80 underwent PCI at follow-up angiography, and not in the acute setting in line with current guidelines at the time of publication. However, it was not sufficiently powered to assess differences in clinical outcome, but focused on FFR utility as a diagnostic tool in the setting of AMI [27].
Outcome data from a subset analysis from the FAME study may help to further support the application of FFR in AMI when the culprit artery is unknown. Of the original 1005 patients included in FAME, 328 had unstable angina or non-ST-elevation MI and were included if troponin titres were positive with creatinine kinase levels less than 1000 units per litre. Although the analysis was not powered to detect differences in subgroups, or to detect superiority of one treatment modality over another, using FFR to guide PCI resulted in similar risk reductions of major adverse cardiovascular events in patients with unstable angina and non-ST-elevation MI as those found with stable angina. The number of stents used in this population was reduced without a concomitant increase in hospital stay or procedure time. Similar to some previous studies, extensive ST-elevation MI was excluded on account of presumed micro-vascular obstruction within an infarcted area causing persistent myocardial stunning [28].
An observational 2 centre descriptive cohort study was conducted in Spain to further observe the use of FFR in safely deciding on management in angiographically moderate non-culprit lesions in 107 patients undergoing PCI for an acute coronary syndrome. Based on FFR measurements, 81 patients (75.7%) were not revascularised and all lesions studied in the remaining 26 patients (24.3%) were revascularised. There were no significant differences in clinical profile or baseline angiographic characteristics of the two groups studied. At 1 year follow-up, no significant differences were observed between the treated group and not-treated group in terms of cardiac death (p = 0.971), revascularisation of the lesion studied (p = 0.8), non-fatal MI or fixed combined episode of a major adverse cardiovascular event (p = 0.97). Additionally, there were no significant differences in need for urgent coronary angiography, revascularisation or re-hospitalisation due to cardiac causes between the two groups [29]. These results support the data obtained from the FAME subgroup analysis, in that the safety and reliability of FFR to assess the functional significance of angiographically inconclusive lesions can be performed at the same time as culprit lesion revascularisation in AMI. As with FAME and data from Fischer et al. [13], FFR-guided deferral of PCI in inconclusive lesions was also deemed safe. This also corroborated data from the 5 year follow-up of the DEFER study [30]. As the study was observational, comparisons could not be made with alternative non-invasive functional tests, which would have provided an interesting comparison with prior studies [24].
Recent data from a single centre experience of FFR measurement in non-ST-elevation MI demonstrated a reduction in diagnostic variability between cardiologists, with the FFR result triggering a change in the initial treatment decision in 46% of the 100 patients prospectively recruited [31]. The data collected included measurements of FFR in culprit lesions in 42% of cases. FFR may therefore be useful in guiding decisions regarding revascularisation in AMI by reducing the disparity that exists between clinicians in the optimal treatment strategy for intermediate lesions.
4. Practical implications of FFR in AMI
The major advantages associated with FFR measurements include reproducibility, high degree of spatial resolution [13], high specificity when compared with non-invasive modalities such as myocardial SPECT [10]. FFR provides a practical assessment of physiological significance which can be used to provide immediate answers in the cardiac catheterisation laboratory. However, FFR requires maximal microvascular dilatation in order to provide an accurate functional assessment of coronary lesions [9] ; [19].
As previously mentioned, micro-vascular dysfunction of both the culprit and non-culprit areas during the acute phase of ST-elevation MI has been shown in early human studies [26]. Possible pathophysiological mechanisms include local neurohormonal mediated vasoconstriction, elevated left ventricular end diastolic pressures and extensive adjacent territory ischaemia [32] ; [33]. Despite this, small studies have demonstrated that FFR values obtained in non-culprit lesions during AMI may be reliable and reproducible [11]; [27]; [28] ; [29]. Additionally, FFR has been validated and incorporated into current guidelines as a surrogate for ischaemic testing in the cardiac catheterisation laboratory [13]. Despite this, the necessity to induce hyperaemic conditions using agents such as adenosine appears to constitute a major deterrent to the widespread adoption of FFR in clinical practice, particularly in the United States [34].
Adenosine, although commonly administered for arrhythmias, FFR assessment and non-invasive imaging modalities, has contraindications limiting universal use [35] ; [36]. It is not uncommon for patients with AMI to present with concurrent atrio-ventricular block, bradycardia, hypotension, severe airway diseases or cardiogenic shock precluding the use of adenosine to induce hyperaemia in this group [10]. These factors mean that a proportion of patients presenting with AMI would be ineligible for FFR assessment, and other functional or anatomical assessments would be required. Cases of MI and death have been reported following administration of adenosine or adenosine receptor agonists [37] ; [38], prompting the United States Food and Drug Administration to issue a safety announcement in November 2013 advising its avoidance in patients with cardiovascular instability.
Whilst adenosine is the most frequently used agent, there are alternatives with similar efficacy to attain hyperaemia for measurement of FFR. Nitroprusside administered intracoronary (IC) has yielded similar vasodilatation to IC adenosine [39]. Rudzinski et al. found that an intracoronary nitroprusside bolus of 100 μg was at least as effective, faster acting and better tolerated than conventional intravenous (IV) adenosine infusion [40]. The use of IC nicorandil as a bolus was found to be similar in hyperaemic efficacy to that of IV adenosine in 210 patients studied by Jang et al. [41]. There was also a strong linear correlation between the FFR measurements obtained by administration of both agents (r2 = 0.934) with less fluctuation in mean blood pressure, PR interval and chest discomfort following nicorandil administration.
However speed of revascularisation is paramount in AMI and may preclude the use of FFR. Leesar et al. studied the impact of FFR on costs, procedural time and clinical outcome of managing AMI. Seventy patients with recent non-ST-elevation MI or unstable angina with an intermediate single vessel lesion were randomised to either angiography followed by SPECT, or acute FFR-guided revascularisation. When compared with the SPECT strategy, the FFR-guided approach reduced cost (US$2113 +/− $120 vs. $1,329 +/− $44, p < 0.05) and hospital duration, with no significant increase in procedure time, radiation exposure or clinical event rates at 1 year follow-up [42].
5. Alternative assessments of ischaemia
Whilst FFR is the most widely accepted tool for invasive assessment of coronary lesions, recently novel physiological indices, instant wave-free ratio (iFR), basal stenosis resistance (BSR) and new imaging modalities have been introduced which allow physiological investigation without the need for hyperaemia [43] ; [44]. Whilst the iFR and BSR both provide rapid on table decision making techniques in a similar way to FFR, CT-FFR is perhaps less ideal for non-culprit AMI patients as the imaging and interpretation takes several hours.
6. Instantaneous wave free ratio (iFR)
Davies et al. identified a period of the cardiac cycle during which intracoronary resistance is naturally minimised and stable as observed during hyperaemia, thus obviating the need for pharmacological vasodilatation. Data from the Adenosine Vasodilator Independent Stenosis Evaluation Study (ADVISE) and subsequent ADVISE registry, South Korean Registry, and ADVISE-in Practice studies all showed diagnostic efficiency using iFR to define a functionally significant lesion when compared to conventional FFR [43] ; [45]. More recently the multicentre, prospective, core-lab analysed ADVISE II study demonstrated that using a hybrid iFR/FFR approach delivered an overall classification agreement with FFR of 94%, avoiding the need to administer hyperaemic medication in 70% of patients. The only study which has reported different levels of agreement between iFR and FFR is the VERIFY study [46]. However, when the results of this study were independently analysed in core-lab setting, the reported findings of the VERIFY investigators could not be substantiated, and high levels of classification match between iFR and FFR were identified. The reason for this difference has largely been put down to a poor algorithm applied by the VERIFY investigators [47].
The CLARIFY (Classification Accuracy of Pressure-Only Ratios Against Indices using Flow Study) study demonstrated that further reductions in resistance by administration of adenosine when performing iFR did not improve diagnostic categorisation, when compared to FFR. The hyperaemic stenosis resistance (HSR) was also used as an arbiter to determine the most accurate assessment of the haemodynamic significance of a lesion when discrepancy existed between iFR and FFR (4 cases, 7.7% of the study population). Although only 51 vessels were analysed, iFR, iFR with adenosine administration and FFR had an almost equivalent high level of diagnostic agreement with HSR; with the receiver-operating characteristic (ROC) area under the curve of 0.93, 0.94 and 0.96, respectively [42].
These studies raise an interesting question as to whether hyperaemia is even required, when a vasodilator independent method of coronary analysis can now be performed in the catheter laboratory with a similar level of diagnostic accuracy to FFR. Using such an approach would theoretically remove the obstacles encountered in administering adenosine to achieve hyperaemia in AMI patients where microvascular congestion predominates. FFR in small studies has shown reliability in assessing non-culprit lesions in AMI despite earlier studies demonstrating microvascular dysfunction in both culprit and non-culprit territories during acute ST elevation MI. By removing the issues associated with achieving maximal hyperaemia in microcirculatory dysfunction during ACS, iFR may prove a useful non-vasodilator dependent alternative in some cases to conventional FFR. Encouraging preliminary data from the FORECAST study supports this premise [48]. This promising data provides an interesting platform for further investigation of iFR assessment in AMI, and may circumvent some of the problems with adenosine administration during FFR measurement.
Whilst most of the data to date in AMI has been in small patient populations, the FLAIR and SWEDEHEART studies will be the first studies to test the application of both FFR and iFR in large contemporary populations undergoing routine assessment of intermediate non-culprit lesions. FLAIR is a global, 2500 patient clinical outcomes trial in which patients will be randomised to either receiving iFR or FFR guided therapy using a single cut-off point (FFR ≤ 0.80, and iFR < 0.90).
7. Conclusions
FFR provides a combination of anatomical and physiological information useful in tailoring treatment strategies in coronary artery disease, with robust clinical data validating its use in stable ischaemic heart disease. There are small studies supporting the use of FFR in evaluating the severity of non-culprit lesions during acute coronary syndromes following revascularisation of the culprit vessel. It appears to demonstrate diagnostic accuracy and reproducibility in AMI, particularly in intermediate lesions. The presence of microvascular dysfunction as observed in experimental and clinical studies in non-culprit arterial territories appears to not significantly hinder FFR calculation. However, larger studies such as FLAIR and SWEDEHEART are needed to validate these findings.
Conflict of interest
The authors report no possible conflicts of interest, sources of financial support, corporate involvement and confirm that the contents have not been partially or wholly published elsewhere.
References
- [1] S.R. Mehta, C.P. Cannon, K.A. Fox, L. Wallentin, W.E. Boden, R. Spacek, et al.; Routine versus selective invasive strategies in patients with acute coronary syndromes: a collaborative analysis of randomized trials; JAMA, 293 (2005), pp. 2908–2917
- [2] S.J. Brener, S. Milford-Bleland, M.T. Roe, D.L. Bhatt, W.S. Weintraub, R.G. Brindis; Culprit-only or multivessel revascularization in patients with acute coronary syndromes: an American College of Cardiology National Cardiovascular Database Registry report; Am Heart J, 155 (1) (2008 Jan), pp. 140–146
- [3] P. Sorajja, B.J. Gersh, D.A. Cox, M.G. McLaughlin, P. Zimetbaum, C. Costantini, et al.; Impact of multi-vessel disease on reperfusion success and clinical outcomes in patients undergoing primary percutaneous coronary intervention for acute myocardial infarction; Eur Heart J, 28 (2007), pp. 1709–1716
- [4] D.S. Wald, J.K. Morris, N.J. Wald, A.J. Chase, R.J. Edwards, L.O. Hughes, et al.; Randomized trial of preventive angioplasty in myocardial infarction; N Engl J Med, 369 (12) (2013 Sep 19), pp. 1115–1123
- [5] G.J. Bech, B. De Bruyne, N.H.J. de Pijls, E.D. Muinck, J.C. Hoorntje, J. Escaned, et al.; Fractional flow reserve to determine the appropriateness of angioplasty in moderate coronary stenosis: a randomized trial; Circulation, 103 (2001), pp. 2928–2934
- [6] P.A.L. Tonino, B. De Bruyne, N.H.J. Pijls, U. Siebert, F. Ikeno, M. van' t Veer, et al.; Fractional flow reserve versus angioplasty for guiding percutaneous coronary intervention; N Engl J Med, 360 (2009), pp. 213–224
- [7] B. De Bruyne, N.H.J. Pijls, B. Kalesan, E. Barbato, P.A. Tonino, Z. Piroth, et al.; Fractional Flow reserve — guided PCI versus medical therapy in stable coronary disease; N Engl J Med, 367 (2012), pp. 991–1001
- [8] N.H.J. Pijls, W.F. Fearon, P.A. Tonino, U. Siebert, F. Ikeno, B. Bornschein, et al.; Fractional flow reserve versus angiography for guiding percutaneous coronary intervention in patients with multivessel coronary artery disease: 2 year follow-up of the FAME study; J Am Coll Cardiol, 56 (2010), pp. 177–184
- [9] K. Tamita, T. Akasaka, T. Takagi, A. Yamamuro, K. Yamabe, M. Katayama, et al.; Effects of microvascular dysfunction on myocardial fractional flow reserve after percutaneous coronary intervention in patients with acute myocardial infarction; Catheter Cardiovasc Interv, 57 (4) (2002 Dec), pp. 452–459
- [10] S. Tani, I. Watanabe, C. Kobari, M. Matsumoto, T. Miyazawa, Y. Iwamoto, et al.; Mismatch between results of myocardial fractional flow reserve measurements and myocardial perfusion SPECT for identification of the severity of ischaemia; Jpn Heart J, 45 (2004), pp. 867–972
- [11] A.F.C. Yong, W.F. Fearon; Coronary microvascular dysfunction after ST-segment elevation myocardial infarction. Local or global phenomenon?; Circ Cardiovasc Interv, 6 (2013), pp. 201–203
- [12] B. De Bruyne, J. Sarma; Fractional Flow reserve: a review; Heart, 94 (2008), pp. 949–959
- [13] J.J. Fischer, X.Q. Wang, H. Samady, I.J. Sarembock, E.R. Powers, L.W. Gimple, et al.; Outcome of patients with acute coronary syndromes and moderate coronary lesions undergoing deferral of revascularisation based on fractional flow reserve assessment; Catheter Cardiovasc Interv, 68 (2006), pp. 544–548
- [14] J.M. Potvin, J. Rodes-Cabau, O.F. Bertrand, O. Gleeton, C.N. Nguyen, G. Barbeau, et al.; Usefulness of fractional flow reserve measurements to defer revascularisation in patients with stable or unstable angina pectoris, non ST-elevation and ST-elevation myocardial infarction, or atypical chest pain; Am J Cardiol, 98 (2006), pp. 289–297
- [15] W. Wijns, P. Kolh, N. Danchin, C. Di Mario, V. Falk, T. Folliguet, et al.; Guidelines on myocardial revascularization: the Task Force on Myocardial Revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS); Eur Heart J, 31 (2010), pp. 2501–2555
- [16] S.J. Brener, S.A. Murphy, C.M. Gibson, P.M. DiBattiste, L.A. Demopoulos, C.P. Cannon; Efficacy and safety of multi-vessel revascularisation in patients with acute coronary syndromes; Am J Cardiol, 90 (2002), pp. 631–633
- [17] R. Kornowski, R. Mehran, G. Dangas, E. Nikolsky, A. Assali, B.E. Claessen, et al.; Prognostic impact of staged versus “one-time” multivessel percutaneous intervention in acute myocardial infarction: analysis from the HORIZONS-AMI (harmonizing outcomes with revascularization and stents in acute myocardial infarction) trial; J Am Coll Cardiol, 58 (7) (2011), pp. 704–711
- [18] P.J. Vlaar, K.D. Mahmoud, D.R. Holmes Jr., G. van Valkenhoef, H.L. Hillege, I.C. van der Horst, et al.; Culprit vessel only versus multivessel and staged percutaneous coronary intervention for multivessel disease in patients presenting with ST-segment elevation myocardial infarction: a pairwise and network meta-analysis; J Am Coll Cardiol, 58 (7) (2011), pp. 692–703
- [19] N. Curzen, O. Rana, Z. Nicholas, P. Golledge, A. Zaman, K. Oldroyd, et al.; Does routine pressure wire assessment influence management strategy at coronary angiography for diagnosis of chest pain?: the RIPCORD study; Circ Cardiovasc Interv, 7 (2) (2014), pp. 248–255
- [20] J.J. Fischer, H. Samady, J.A. McPherson; Comparison between visual assessment and quantitative angiography versus fractional flow reserve for native coronary narrowings of moderate severity; Am J Cardiol, 90 (2002), pp. 210–215
- [21] M.T. Roe, E.M. Ohman, A.C.P. Maas, R.H. Christenson, K.W. Mahaffey, C.B. Granger, et al.; Shifting the open-artery hypothesis downstream: the quest for optimal reperfusion; J Am Coll Cardiol, 37 (1) (2001), pp. 9–18
- [22] A. Prasad, B.J. Gersh; Management of microvascular dysfunction and reperfusion injury; Heart, 91 (2005), pp. 1530–1532
- [23] C.M. Kramer, W.J. Rogers, T.M. Theobald, T.P. Power, S. Petruolo, N. Reichek; Remote non-infarcted region dysfunction soon after first anterior myocardial infarction. A magnetic resonance tagging study; Circulation, 94 (1996), pp. 660–666
- [24] De Bruyne, N.H.J. Pijls, J. Bartunek, K. Kulecki, J.W. Bech, H. De Winter, et al.; Fractional flow reserve in patients with prior myocardial infarction; Circulation, 104 (2001), pp. 157–162
- [25] H. Samady, W. Lepper, E.R. Powers, K. Wei, M. Ragosta, G.G. Bishop, et al.; Fractional flow reserve of infarct-related arteries identifies reversible defects on non-invasive myocardial perfusion imaging early after myocardial infarction; J Am Coll Cardiol, 47 (2006), pp. 2187–2193
- [26] N.G. Uren, T. Crake, Lefroy, R. de Silva, G.J. Davies, A. Maseri; Reduced coronary vasodilator function in infracted and normal myocardium after myocardial infarction; N Engl J Med, 331 (1994), pp. 222–227
- [27] A. Ntalianis, J.W. Sels, G. Davidavicius, N. Tanaka, O. Muller, C. Trana, et al.; Fractional flow reserve for the assessment of non-culprit coronary artery stenoses in patients with acute myocardial infarction; JACC Cardiovasc Interv, 3 (12) (2010 Dec), pp. 1274–1281
- [28] J.W. Sels, P.A. Tonino, U. Siebert, W.F. Fearon, M. Van't Veer, B. De Bruyne, et al.; Fractional flow reserve in unstable angina and non-ST-segment elevation myocardial infarction: experience from the FAME (Fractional flow reserve versus angioplasty for multi-vessel evaluation) study; JACC Cardiovasc Interv, 4 (11) (2011 Nov), pp. 1183–1189
- [29] Carrillo P. Lopez-Palop, F. Torres, I. Lozano, A. Frutos, P. Avanzas, A. Cordero, et al.; Results of fractional flow reserve measurement to evaluate non-culprit coronary artery stenosis in patients with acute coronary syndrome; Rev Esp Cardiol, 65 (2) (2012), pp. 164–170
- [30] N.H. Pijls, P. van Schaardenburgh, G. Manoharan, E. Boersma, J.W. Bech, M. van't Veer, et al.; Percutaneous coronary intervention of functionally nonsignificant stenosis: 5 year follow-up of the DEFER study; J Am Coll Cardiol, 49 (21) (2007), pp. 2105–2111
- [31] D. Carrick, M. Behan, F. Foo, J. Christie, W.S. Hillis, J. Norrie, et al.; Usefulness of fractional flow reserve to improve diagnostic efficiency in patients with non-ST elevation myocardial infarction; Am J Cardiol, 111 (1) (2013 Jan 1), pp. 45–60
- [32] P.L. Van Herck, S.G. Carlier, M.J. Claeys, S.E. Haine, P. Gorissen, H. Miljoen, et al.; Coronary micro-vascular dysfunction after myocardial infarction: increased zero flow pressure both in the infarcted and in the remote myocardium is mainly due to left ventricular filling pressure; Heart, 93 (2007), pp. 1231–1237
- [33] C.M. Gibson, K.A. Ryan, S.A. Murphy, R. Mesley, S.J. Marble, R.P. Giugliano, et al.; Impaired coronary blood flow in non-culprit arteries in the setting of acute myocardial infarction. The TIMI Study Group. Thrombolysis in myocardial infarction; J Am Coll Cardiol, 34 (1999), pp. 974–982
- [34] N.S. Kleimann; Bringing it all together integration of physiology with anatomy during cardiac catheterisation; J Am Coll Cardiol, 58 (2011), pp. 1219–1221
- [35] E.H. Botvinick; Current methods of pharmacologic stress testing and the potential advantages of new agents; J Nucl Med Technol, 37 (1) (2009 March), pp. 14–25
- [36] M.J. Henzlova, M.D. Cerqueira, J.J. Mahmarian; Quality Assurance Committee of the American Society of Nuclear Cardiology. Stress protocols and tracers; J Nucl Cardiol, 13 (6) (2006 Nov), pp. e80–e90
- [37] S. Shah, D. Parra, R.S. Rosenstein; Acute myocardial infarction during regadenoson myocardial perfusion imaging; Pharmacotherapy, 33 (2013), pp. 90–95
- [38] D.H. Hsi, R. Marreddy, M. Moshiyakhov, U. Luft; Regadenoson induced acute ST-segment elevation myocardial infarction and multivessel coronary thrombosis; J Nucl Cardiol, 20 (2013), pp. 481–484
- [39] W.A. Parham, A. Bouhasin, J.P. Ciaramita, S. Khoukaz, S.C. Herrmann, M.J. Kern; Coronary hyperaemic dose responses of intracoronary sodium nitroprusside; Circulation, 109 (2004), pp. 1236–1243
- [40] W. Rudzinski, A.H. Waller, A. Rusovici, A. Dehnee, A. Nasur, M. Benz, et al.; Comparison of efficacy and safety of intracoronary sodium nitroprusside and intravenous adenosine for assessing fractional flow reserve; Catheter Cardiovasc Interv, 81 (3) (2013), pp. 540–544
- [41] H.J. Jang, B.K. Koo, H.S. Lee, J.B. Park, J.H. Kim, M.K. Seo, et al.; Safety and efficacy of a novel hyperaemic agent, nicorandil, for invasive physiological assessment in a catheterisation laboratory: a prospective multicentre study; Eur Heart J, 34 (27) (2013), pp. 2055–2062
- [42] M.A. Leesar, T. Abdul-Baki, N.I. Akkus, A. Sharma, T. Kannan, R. Bolli; Use of fractional flow reserve versus stress perfusion scintigraphy after unstable angina. Effect on duration of hospitalization, cost, procedural characteristics and clinical outcome; J Am Coll Cardiol, 41 (2003), pp. 1115–1121
- [43] S. Sen, J. Escaned, I.S. Mailk, G.W. Mikhail, R.A. Foale, R. Mila, et al.; Development and validation of a new adenosine-independent index of stenosis severity from coronary wave-intensity analysis: results of the ADVISE (ADenosine Vasodilator Independent Stenosis Evaluation Study); J Am Coll Cardiol, 59 (2012), pp. 1392–1402
- [44] S. Sen, K.N. Asrress, S. Nijjer, R. Petraco, I.S. Malik, R.A. Foale, et al.; Diagnostic classification of the instantaneous wave-free ratio is equivalent to fractional flow reserve and is not improved with adenosine administration: results of CLARIFY (Classification Accuracy of Pressure-Only Ratios Against Indices Using Flow Study); J Am Coll Cardiol, 61 (2013), pp. 1409–1420
- [45] J.J. Park, R. Petraco, C.W. Nam, J.H. Doh, J. Davies, J. Escaned, et al.; Clinical validation of the resting pressure parameters in the assessment of functionally significant coronary stenosis; results of an independent, blinded comparison with fractional flow reserve; Int J Cardiol, 168 (4) (2013 Oct), pp. 4070–4075
- [46] C. Berry, M. van 't Veer, N. Witt, P. Kala, O. Bocek, S.A. Pyxaras, et al.; VERIFY (VERification of Instantaneous Wave-Free Ratio and Fractional Flow Reserve for the Assessment of Coronary Artery Stenosis Severity in EverydaY Practice): a multicenter study in consecutive patients; J Am Coll Cardiol, 61 (13) (2013), pp. 1421–1427
- [47] S. Nijjer, R. Petraco, S. Sen, D. Francis, J. Davies; RESOLVE could not verify the VERIFY study. Re: improvement in coronary haemodynamics after percutaneous coronary intervention: assessment using instantaneous wave-free ratio; Heart, 99 (23) (2013), pp. 1740–1748
- [48] C. Indolfi, A. Polimeni, C. Spaccarotella, S. Sorrentino, J. Sabatino, M. Miceli, et al.; Instantaneous wave-Free Ratio (iFR) and Gradient (iFG): new promising adenosine-independent alternative to fractional flow reserve. Preliminary results from the FORECAST Study; J Am Coll Cardiol, 60 (17S) (2012)
Document information
Published on 19/05/17
Submitted on 19/05/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?