Abstract
Objective
The aim of this study was to compare the performance of multimodal intraoperative neurophysiological monitoring (MIONM) in children below and over 6 years of age.
Methods
43 children, diagnosed with spinal pathologies were divided into two cohorts according to their age and enrolled in the study. Those under the age of 6 consisted group A, whereas those between the age of 6 and 11 consisted group B. All patients underwent spinal surgical procedures according to their diagnosis. A standard anesthesia protocol was given to both groups. Baseline somatosensory evoked potentials (SSEPs) and transcranial electrical motor evoked potentials (tcMEPs) were recorded and evaluated at specific time points for each patient.
Results
Except for the SSEPs in three cases, tcMEPs and SSEPs were recorded for all patients. There was no false-negative whereas 9 false positive recordings due to physiological conditions that all recovered intraoperatively. In 10 patients, MIOMN recorded more than %50 decrement, in which 8 had the kyphosis component. The tcMEPs fully recovered by the end of the operation except for the patient with post-tuberculosis kyphosis. There was no statistically significant difference in the mean threshold values with regard to transcranial stimulus intensity for the tcMEPs between the two groups.
Conclusion
Compared to school aged children, both SSEPs, tcMEPs recordings are feasible and MIONM is effective for early childhood patients undergoing spinal surgery.
Level of evidence
Level III, Diagnostic Study.
Keywords
Intraoperative monitoring ; Motor evoked potentials ; Somatosensory evoked potentials ; Children ; Spine surgery
Introduction
Spinal surgery is a complex procedure that is associated with various risks. The Scoliosis Research Society (SRS) Morbidity and Mortality Committee reported that the annual rate of new neurological complications was 0.95% between 2004 and 2007.1
Multimodal intraoperative neurophysiological monitoring (MIONM) has emerged for the real-time evaluation of neurological status in order to prevent these complications. The ascending sensory tracts can be assessed via somatosensory evoked potentials (SSEP) whereas transcranial motor evoked potentials (tcMEP) primarily evaluate the descending motor tracts.2 Both techniques are combined in MIONM so as to maximize the advantages.3 Success in obtaining reliable MEP is known to be influenced by lesion location, type and duration of anesthetic agent used, existing spinal chord, cranial anomalies, preoperative motor deficit, patients physiological status and age. Among these variables, one of the most important determinant is concluded to be the age of the child.4 ; 5 Although advances in electrophysical monitoring techniques and anesthetic agents have led to an improvement in the reliability of MEP monitoring, the reliability of MEP signals in children younger than 11 years of age is still a concern. Studies conclude that success rates decline in children under the age of 6 compared to older ones probably due to the incomplete maturation of the corticospinal tract, which leads to changes in the morphology of the waveforms and differences in latencies.6 ; 7 ; 8 ; 9
The current literature lacks detailed comparison between early childhood and school aged children in terms of intraoperative neurophysiological characteristics, threshold levels, the elicitability of tcMEPs from different myotomes, and the frequency of absence or decrement in amplitude of more than 50% of the baseline values. We consider 6 years to be the critical age for MIONM, thus SSEPs and tcMEPs recordings may not have the desired efficacy. The aim of this study is to elucidate the difference and compare the efficacy of MIONM in early childhood and school-aged children that have underwent surgical procedures due to spinal pathologies.
Materials and methods
Subjects
43 children younger than 11 years of age that underwent spinal surgery between May 2008 and November 2011 were included in the study. Vast majority of all patients were comprised of idiopathic scoliosis (Table 1 ). MIONM was attempted in all surgical procedures. Children with cardiac pacemakers, cranial anomalies, history of epilepsy and revision cases were excluded. None of the patients had any neurological deficits prior to the surgery. The subjects were divided into two cohorts according to their age. 16 patients mean age 2.7 under the age of six consisted group A and 27 patients mean age 8.2 between 6 and 11 years consisted group B.
| Diagnosis | Age <6 yr | Age 6–11 yr | N |
|---|---|---|---|
| Congenital scoliosis | 5 | 2 | 7 |
| Idiophatic scoliosis | 13 | 13 | |
| Kyphosis (Traumatic, post-tuberculosis, idiopathic) | 3 | 3 | |
| Tethered cord | 4 | 3 | 7 |
| Tethered cord + Diastematomyelia | 4 | 2 | 6 |
| Diastematomyelia | 3 | 4 | 7 |
Anesthesia
A propofol bolus of 2 mg/kg was administered after the initiation of a remifentanil infusion of 0.2 μg/kg/min, and 100% oxygen (O2 ) was delivered via a facemask or laryngeal mask. Following the baseline recordings, 0.5 mg/kg of atracurium besilate, a short-acting myorelaxant, was administered and the patient was intubated. The anesthesia was continued with total intravenous anesthesia (TIVA) with respect to the cardiovascular parameters (remifentanil 0.1–0.2 μg/kg/min and propofol 6–8 mg/kg/h), and the induction of bolus doses of remifentanil and propofol was avoided. Mechanical ventilation with air-oxygen mixtures was performed with respect to the end-tidal CO2 level, which was maintained between 30 and 35 mmHg. Normotensive anesthesia was performed to maintain the systolic blood pressure between 100 and 130 mmHg. In case of hypotension, which would inhibit the eliciting potentials, the doses of propofol and remifentanil were decreased to 2–4 mg/kg/h and 0.05–0.1 mg/kg/h, respectively. An infusion of ketamine hydrochloride (0.025 mg/kg/h) was also added for the maintenance of the anesthesia.
Monitoring equipment
The Nicolet Endeavor CR™ (Viasys Healthcare, Nicolet Biomedical, Madison, Wisconsin, USA) system was used for MIONM, and the tcMEPs and SSEPs were recorded. In addition, pedicle screw/hole stimulation, free-run electromyography, direct nerve root stimulation and F-waves were also monitored.10
Evoked potentials
Intraoperative monitoring was accomplished in multimodal manner including SSEPs, tcMEPs, direct nerve root stimulation.
SSEPs
The SSEPs were recorded from the scalp on C3′-FPz and C4′-FPz montages via corkscrew electrodes or subdermal needle electrodes by stimulating the median or ulnar nerves bilaterally with self-adhesive surface electrodes. In the lower extremities, the bilateral tibial nerves were stimulated by surface electrodes, and the SSEPs were recorded from inion-FPz and FZ′-FPz via corkscrew electrodes. The filter settings were adjusted to between 100 and 300 Hz. The stimulus frequency was 1.7 Hz and the duration was 500 μs for the tibial nerve and 300 μs for the median nerve. Furthermore, the stimulus strength ranged from 25 to 60 mA for the tibial and median nerves.11
tcMEPs
The tcMEPs were recorded by subdermal needle electrodes from either the abductor pollicis brevis or more commonly from the bilateral abductor digiti quinti due to the intra-arterial interventions to the radial artery at the wrist. In the lower extremities, the tcMEPs were recorded from bilateral iliopsoas (IP), adductor magnus (AddM), vastus lateralis (VL), tibialis anterior (TA), and abductor hallucis muscles (AHL) that involved the L1-S2 myotomes. In addition, the bilateral external anal sphincter muscles were monitored as needed to prevent injury to the lower spinal cord, especially for tethered cord syndrome. Corkscrew electrodes from M3-Mz6 and M4-Mz6 for the left and right motor cortex were respectively applied to the stimuli. The duration of the stimulus was 1000 μs, and the maximum intensity was 400 V. The frequency of the stimuli, including the five consecutive train stimuli, was 250 Hz and the filters were set to 30 and 500 Hz. The number of train stimuli was increased to 10 when five consecutive trains of 400 V elicited no tcMEPs. A threshold level of stimulus intensity was determined when satisfying tcMEP recordings were obtained from at least the muscles that were essential for the relevant surgical procedure.10
Direct nerve root stimulation
The tissues suspected to be of nervous structures were stimulated by a bipolar stimulator, and the motor responses were recorded. The stimulus duration was 0.1 ms, and the maximum stimulus intensity was 20 mA.
Assessment of the patients
All cases were evaluated the day before the surgery by the anesthesia and neurophysiological monitoring staff, and written informed consent was obtained from the parents of all patients for both the surgical procedure and MIONM application.
Recording of tcMEPs and SSEPs
Recording was repeated regularly four times during the surgery The regular time points were as follows: after the induction of anesthesia (first baseline), immediately after the beginning of the intervention (second baseline), at the middle of the surgical intervention (third baseline), and at the end of the surgical procedure (fourth baseline). Additional recordings were obtained after osteotomies, correction maneuvers according to the surgeons claim. After excluding hypotension, hypothermia, anemia, regulation problems of anesthesia dose, the MIONM team informed the surgeon whenever tcMEP values decreased by more than 50% of the second baseline. Regarding the SSEPs, at least a 50% decrease in amplitude and/or a 10% prolongation of latency was recorded to reflect a serious event during surgery.
Data analysis
All statistical analyses were performed using SPSS (Version 16.0. Chicago). Descriptive statistics are expressed as percentages and mean ± SD. Power was found to be ≥ 0.80 for specified sample size using a 2 sided test. Parametric data was compared using ANOVA between groups whereas nonparametric data was evaluated using Chi-Square and Mann Whitney U tests. A p value of less than 0.05 was accepted as being statistically significant.
Results
tcMEPs
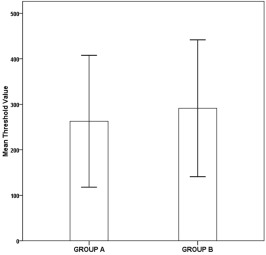
The mean threshold value of the transcranial stimulus intensity for tcMEP recordings of all patients (n = 43) was 281 V (min: 176 V, max: 400 V), and the mean threshold values were 263 V and 291 V for groups A and B, respectively. The difference in these threshold values was not statistically significant (p = 0.232) (Fig. 1 ).
|
|
|
Fig. 1. A bar graph demonstrating the mean threshold values (volt) of both age groups. The error bars represent ±2 standard deviation (p = 0.232). |
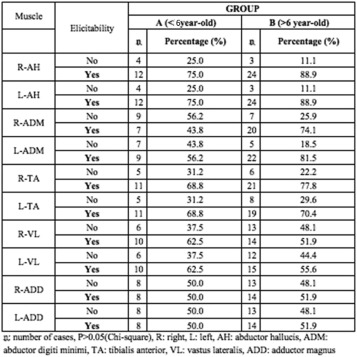
The rate of successful tcMEP recordings of the relevant muscles (IP, AddM, VL, TA, AH) was evaluated using a chi-square test for each muscle, and there were no statistically significant differences between the two groups (p > 0.05) (Fig. 2 ). In addition, no statistically significant differences were found regarding the mean amplitude values of the tcMEPs recorded from all muscles at different time points throughout the operation (p > 0.05).
|
|
|
Fig. 2. The elicitability rate of tcMEPs from various muscles for both age groups. |
There was also no statistically significant difference between the groups regarding a decrease in amplitude of tcMEPs (p = 0.719). Ten patients showed a decrement in tcMEP amplitude of 50% or more. Eight patients out of the ten were diagnosed with a kyphosis component. In two patients the decrement of tcMEP amplitude was observed immediately after the correction maneuver. All tcMEPs fully recovered by the end of the operation and patients were neurologically intact postoperatively except for the patient with post-tuberculosis kyphosis.
A total of nine false positive tcMEP values (5 in group A, 4 in group B) were recorded but all recovered within a maximum of 1 h after appropriate management of physiologic conditions (e.g, hypothermia, hypovolemia). There were no false-negative outcomes in either group.
All patients remained intact neurologically except for one patient in group B. A pedicle subtraction osteotomy at the T11 level was performed. A 75% loss of tcMEPs in the right AddM and VL muscles was detected just after the pedicle screw insertion at the L1 level. The amplitude of tibial and ulnar SSEPs and tcMEPs recorded from the bilateral ADM, TA, and AH muscles were similar when compared with the baseline values. Pedicle screw stimulation could not be performed due to technical reasons. Motor weakness in right hip flexion and knee extension was detected during the immediate post-operative period. An urgent CAT scan confirmed misplacement of the pedicle screw at the L1 level. The pedicle screw was revised and the patient fully recovered over the next four weeks.
SSEPs
Transient changes in the amplitude of the tibial SSEPs were observed in two patients in-group A and three patients in-group B. These changes were related to hypotension, and the patients completely recovered by the end of the surgery.
Discussion
Since clinical examination is unreliable to assess the integrity of central motor pathways in the children younger than 6 years of age, electromagnetic stimulation of the motor cortex provides the opportunity to assess the integrity of corticospinal tracts. Physiology based studies that describe critical age-related changes, that appear mainly to be based on the development of cortical synaptic efficacy generate important data in achieving successful MIONM.12 ; 13
A study conducted by Koh et al provide corresponding neurophysiological evidence that muscle action potential onset latency apparently decreases starting from the age of 6. Although children aged less than 8 years required the facilitation of an isometric muscle contraction, there was a significant increase in the incidence of obtaining a muscle action potential in relaxed muscles between the ages of 8 and 11 year. Maturation of central motor pathways continues up until the age of 11 years and by the age of 11, adult values (79 m/s) of V1 (crown to heel length/latency to the onset of the evoked muscle action potential) are reached.14 In recent years, age has also become a prominent criteria and has been implicated in the classification and treatment algorithm for children with spinal deformities. Although still under discussion, under the age of six is defined as early onset and those over the age of six are defined as late onset in spinal deformities, in which the overall success rate for lower extremity MEP was reported 66% in spinal surgery.
In the current study, the mean threshold value of the stimulus intensity for tcMEP recordings was slightly lower in group A (263 V) than in group B (p > 0.05). This data reveals that we might achieve recording potentials with similar intensities of the stimulus. Recently, Fulkerson et al reported their results among children younger than three years of age, and their mean threshold value of stimulus intensity of 533 V. This was much more higher than our results.15 The authors explained that this was caused by the relationship between the high threshold value and the immaturity of the motor pathways in children that age. When we calculated the mean threshold intensity of the stimuli of the patients in our study under the age of three, we found that the mean threshold value was as low as 218 V. The big difference between the two series of patients might be due to the different anesthesia protocol that was used. They maintained the anesthesia with 100–250 μg/kg/min of propofol and combined that with intermittent fentanyl boluses of 2 μg/kg if needed. Compared to our patients, they were anesthetized with higher doses of propofol. Yang et al proposed to combine propofol with sevoflurane in order to avoid the side effects of prolonged propofol administration in patients under the age of three.16 However, they did not determine the threshold values of their subjects.
In the current study, one patient presented with a significant decrease in tcMEPs of the right AddM and VL muscles after the insertion of the pedicle screw to the L1 level. Additionally, motor weakness compatible with the neurophysiological monitoring was determined during the early post-operative period, which suggests the importance of recording tcMEPs involving different myotomes. If we would have only recorded them from one or two muscles (TA and AH), we would not have been able to detect the selective impairment of the proximal lumbar roots. The tibial SSEPs corresponding to the lower lumbar roots were normal and proved to be unhelpful with regard to these kinds of selective neural injuries. However, since this occurred in one of our patient, we did not revise the procedure as we could not confirm the misplacement of the pedicle screw by stimulation due to technical reasons nor could we confirm the unaffected tcMEPs recorded from other lower extremity muscles.
The elicitability rate of tcMEPs from different myotomes was almost the same in both groups. The number of cases with successful recordings of tcMEPs numbered more than the unsuccessful recordings when all myotomes were considered, demonstrating that tcMEP recording from different muscles is feasible and effective for both age groups.
The elicitability rates of tcMEPs from various muscles may differ according to the same stimulation conditions. This has been highlighted in previous studies in which muscles such as the AH, APB, and ADM have been presented as the optimal muscles to record tcMEPS because of the relatively wider representation of these muscles on the motor homunculus.17 ; 18 The results in our study were comparable to the previous studies in that they revealed relatively high elicitability rates of tcMEPs from the AH, TA and ADM muscles compared with the ADD and VL in both groups. The highest rate of elicited tcMEPs was obtained from the AH muscle in both groups, with over 75% elicitability. Additionally, the elicitability of tcMEPs from the VL muscle was lower than 60% in the two groups. McIntyre et al compared the availability of tcMEPs and SSEPs in patients younger than 72 months diagnosed with a wide range of pathologies (e.g brain tumor, tethered cord, posterior spinal fusion, cervical decompression) and concluded that tcMEPs can be obtained more easily than SSEPs if a permissive anesthetic technique is used.19 In the current study, there was no difference between SSEPs and tcMEPs recordings in both children younger than 72 months and between 72 and 132 months. Similar to the above mentioned study, lacking of homogeneity between compared groups may be one of the drawbacks of the study. In scientific aspect, such comparison might not be ideal but readers can consider the fact that both groups have no neurological findings and the mean threshold values seems similar at final evaluation. It would also be extremely difficult to homogenize groups in terms of diagnosis, since early and late onset pathologies are strongly related with the age of the child.
This consecutive series of patients revealed that, compared to school aged children, SSEPs, tcMEPs recordings are both feasible and MIONM is effective for early childhood patients undergoing spinal surgery.
Acknowledgement
The authors would like to express their special thanks to Assoc. Prof. Y. Hu for reviewing the manuscript before submission.
References
- 1 D.K. Hamilton, J.S. Smith, C.A. Sansur, et al.; Rates of new neurological deficit associated with spine surgery based on 108,419 procedures: a report of the scoliosis research society morbidity and mortality committee; Spine, 36 (2011), pp. 1218–1228
- 2 M.R. Nuwer, E.G. Dawson, L.G. Carlson, L.E. Kanim, J.E. Sherman; Somatosensory evoked potential spinal cord monitoring reduces neurologic deficits after scoliosis surgery: results of a large multicenter survey; Electroencephalogr Clin Neurophysiol, 96 (1995), pp. 6–11
- 3 H. Iwasaki, T. Tamaki, M. Yoshida, et al.; Efficacy and limitations of current methods of intraoperative spinal cord monitoring; J Orthop Sci, 8 (2003), pp. 635–642
- 4 X. Chen, D. Sterio, X. Ming, et al.; Success rate of motor evoked potentials for intraoperative neurophysiologic monitoring: effects of age, lesion location, and preoperative neurologic deficits; J Clin Neurophysiol, 24 (2007), pp. 281–285
- 5 G.B. Russell; Motor evoked potential; G.B. Russell, L.D. Rodichok (Eds.), Primer of Intraoperative Neurophysiologic Monitoring, Butterworth-Heinemann, Boston (1995), p. 159
- 6 F.J. Frei, S.E. Ryhult, E. Duitmann, C.C. Haster, J. Luetschg, T.O. Erb; Intraoperative monitoring of motor-evoked potentials in children undergoing spinal surgery; Spine, 32 (2007), pp. 911–917
- 7 A. Beric; Transcranial electrical and magnetic stimulation; E. Niedermeyer, F. Lopes da Silva (Eds.), Electroencephalography, Williams& Wilkins, Baltimore (1999), pp. 836–850
- 8 A. Nezu, S. Kimura, S. Uehara, T. Kobayashi, M. Tanaka, K. Saito; Magnetic stimulation of motor cortex in children: maturity of corticospinal pathway and problem of clinical application; Brain Dev, 19 (1997), pp. 176–180
- 9 G.H. Thompson, L.E. Lenke, B.A. Akbarnia, R.E. McCarthy, R.M. Campbell; Early onset scoliosis: future directions; J Bone J Surg Am, 89 (2007), pp. 163–166
- 10 M. Beyazova, A. Senkoylu, M. Zinnuroglu; Intraoperative neurophysiologic monitoring in spine surgery: Gazi Medical School experience; J Turk Spinal Surg, 21 (2010), pp. 49–58
- 11 K.D.K. Luk, Y. Hu, Y.W. Wong, J.C. Leong; Variability of somatosensory evoked potentials in different stages of scoliosis surgery; Spine, 24 (1999), pp. 1799–1804
- 12 N. Gogtay, J.N. Giedd, L. Lusk, et al.; Dynamic mapping of human cortical development during childhood through early adulthood; Proc Natl Acad Sci, 101 (2004), pp. 8174–8179
- 13 A.T. Barker, I.L. Freestone, R. Jalinous, J.A. Jarratt; Magnetic stimulation of the human brain and peripheral nervous system: an introduction and the results of an initial clinical evaluation; Neurosurgery, 20 (1987), pp. 100–109
- 14 T.H. Koh, J.A. Eyre; Maturation of corticospinal tracts assessed by electromagnetic stimulation of the motor cortex; Arch Dis Child, 63 (1988), pp. 1347–1352
- 15 D.H. Fulkerson, K.B. Satyan, L.M. Wilder, et al.; Intraoperative monitoring of motor evoked potentials in very young children; J Neurosurg Pediatr, 7 (2011), pp. 311–317
- 16 J. Yang, Z. Huang, H. Shu, et al.; Improving successful rate of transcranial electrical motor-evoked potentials monitoring during spinal surgery in young children; Eur Spine J, 21 (2012), pp. 980–984
- 17 A.R. Moller; Intraoperative Neurophysiologic Monitoring; (2nd ed.)Humana Press Inc, New York (2006), p. 185
- 18 M.V. Simon; Intraoperative Neurophysiology a Comprehensive Guide to Monitoring and Mapping; (1st ed.)Demos Medical Publishing, Totowa (NJ) (2009), p. 18
- 19 I.W. McIntyre, L. Francis, J.J. McAuliffe; Transcranial motor-evoked potentials are more readily acquired than somatosensory-evoked potentials in children younger than 6 years; Anesth Analg, 122 (2016), pp. 212–218
Document information
Published on 31/03/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?