Abstract
Introduction
Intestinal mucositis is a frequent limiting factor in anticancer therapy and there is currently no broadly effective treatment targeted to cure this side effect.
Objective
This study aimed to evaluate the effects of a mucoadhesive formulation containing curcuminoids (MFC) from Curcuma longa L. on the pathogenesis of 5-fluorouracil (5-FU)-induced intestinal mucositis.
Methods
Three intraperitoneal 5-FU injections (200 mg/kg) were used to induce intestinal mucositis in adult Swiss male mice. Treatment was provided orally (MFC 3.75, 7.5 and 15 mg/kg), thirty minutes before 5-FU injections, daily until euthanasia. Duodenal samples were collected to perform morphometric and histopathological analysis, to investigate the expression of Ki-67, p53, Bax and Bcl-2 by immunohistochemistry, to evaluate neutrophil activity myeloperoxidase (MPO)-mediated and oxidative stress by malondialdehyde (MDA) determination. Mice body weight was assessed as well.
Results
As expected, 5-FU induced a significant weight loss (∼17%, P < 0.001), shortening in villi height (∼55.4%) and crypts depth (∼47%), and increased (∼64%) the histological severity score when compared to other groups (P < 0.05). These pathological changes were markedly alleviated by the three MFC treatment doses (P < 0.05), in special with the dose MFC 15 mg/kg. This dose also stimulated cell proliferation by ∼90% in the epithelial cells lining from villi and crypts (P < 0.05), reduced MPO levels and MDA formation by 60% and 44%, respectively (P < 0.05).
Conclusions
Our data suggest the therapeutic potential of the formulation for treating intestinal mucositis in mice. Supplementary studies are underway searching for the elucidation of mechanisms involved in the protective effects of MFC in order to make this formulation a clinical tool for mucositis treatment.
Keywords
Mucoadhesive formulation ; Curcuminoids ; Curcuma longa L ; Intestinal mucositis ; 5-Fluorouracil
1. I ntroduction
The clinical term mucositis describes a syndrome characterized by ulceration in the entire gastrointestinal tract mucosa and associated symptoms [1] . The frequency of mucositis cases varies and is influenced by factors such as patient’s diagnosis, age, health status, and the type, dose and drug administration frequency [2] .
It is very difficult to thoroughly survey the occurrence of oral and, in particular, intestinal mucositis induced by chemotherapy and/or radiotherapy in humans. This is due to the fact that it is not a locally mediated process but rather, a systemic one, with concomitant oral lesions, emesis, anorexia, adynamia, abdominal pain and, in particular, diarrhea [3] . 5-Fluorouracil, methotrexate and other antimetabolite agents are the most established causes of chemotherapy-induced mucositis. These agents attack tumor cells and constantly dividing normal cell types, which result in destruction of the basal cells and damage to connective tissue in the gastrointestinal tract [1] , [2] and [4] .
Even though scientists have been studying mucositis for over thirty years [5] , there is still no truly effective treatment targeted to cure this side effect [2] and [6] . Current palliative treatments for intestinal mucositis include anti-inflammatories, systemic antibiotics, antidiarrheal medication and nonspecific painkillers [4] , [6] and [7] . While no approach has yet standard gold proven efficacy, numerous agents are in used. Compounds such as growth factors (palifermin), free radical scavengers (amifostine), probiotics (Lactobacillus spp.), zinc sulphate, antioxidants (n-acetyl cysteine, l -glutamine), benzydamine hydrochloride and coating agents (CAM2028, Gelclair, MuGard) have been tested with proved efficacy [4] , [5] , [6] , [7] , [8] and [9] .
Herbal products, particularly those with anti-inflammatory and antioxidant activities such as acteoside [10] , iberogast [11] ,Chimonanthus nitens var. salicifolius[12] , Bidens pilosa L. [13] and Rhodiola algida[14] are promising for the treatment of the mucositis. Curcuma longa L. (also known as turmeric), belongs to the ginger family (Zingiberaceae) and is traditionally used in cooking for flavoring purposes [15] . The anti-inflammatory, antioxidant and antitumor properties of C. longa L. constituents spurred the investigation of the molecular mechanisms associated with these biological activities [15] and [16] . This is a chemically diverse herb and up until 2011, approximately 235 chemical compounds, mainly phenolic and terpenoid, have been identified [17] .
It has been shown that formulations containing mucoadhesive polymers, such as poloxamer 407, have the potential to treat mucositis [4] , [6] and [13] . Mucoadhesive systems interact with mucin that covers mucosa, thereby promoting prolonged and localized contact between the pharmaceutical dosage form and absorptive tissue [18] .
Given the above, a mucoadhesive liquid formulation based on poloxamer 407 and curcuminoids from C. longa L. was developed to improve curcuminoids solubility and to promote intimate contact between the herbal active ingredients and gastrointestinal mucosa. This formulation is hypothesized to be effective in treating mice bearing 5-FU-induced intestinal mucositis.
2. Material and methods
2.1. Mucoadhesive formulation containing curcuminoids (MFC)
MFC was prepared by mixing polyethylene glycol 400 and propilenglycol (1:1, v/v) in a heated reactor (65–70 °C). Then, 9.0% (w/v) poloxamer 407 (Basf, Ludwigshafen, Germany) was added to the mixture under mechanical stirring until complete dispersion. After that, curcuminoids from C. longa L. extract (>95% curcuminoid content, Gamma Comércio Importação & Exportação LTDA, São Paulo, Brazil) was added to the organic phase and pH was adjusted by using 0.1 M citric acid (pH 6.5). The final concentration of curcuminoids was 25 mg/mL. An aqueous fraction was prepared by dispersing 6% (w/v) of poloxamer 407 in purified water, under constant stirring. After dispersion, Soluplus® (Basf, Ludwigshafen, Germany), sodium metabisulphite and sodium bisulphite were added under stirring. The aqueous fraction was then heated up to 65 °C and poured into organic fraction. The mixture was stirred for 30 min. MFC was then bottled in ambar flask and stored at room temperature until use. A blank mucoadhesive formulation (control) was prepared as described above, but without adding curcuminoids extract.
2.2. Mice
Swiss (8–10-week-old) male albino mice provided by our Institute’s Central Bioterium were kept under controlled temperature (25 ± 2 °C) and light conditions (12h-light–dark cycle), with free access to water and standard chow pellets (Presence nutrição animal, São Paulo, Brazil). Mice were acclimatized for a week before beginning the experiments. All animal treatment and surgical procedures were carried out in accordance with the norms of the Brazilian College of Animal Experimentation (COBEA) and approved by the local Research Ethics Committee (Federal University of Goiás 036/2012).
2.3. Experimental design
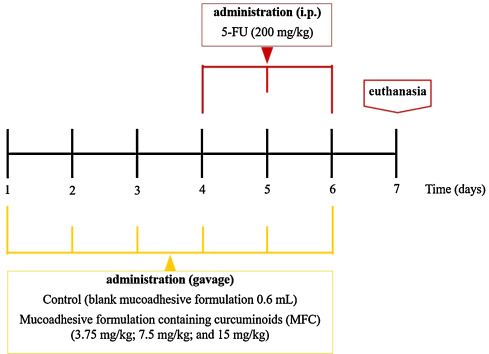
Considering that the 5-FU regime was able to induce mucositis homogeneously in the mice, as well as ethical aspects in the use of animals, three independent essays were performed with mice randomly allocated into five experimental groups (Fig. 1 ) of 5 animals each. Groups were as follow: negative control (blank mucoadhesive formulation 0.6 mL/animal, via gavage, from day 1–6); positive control (5-FU 200 mg/kg, i.p., days 4–6); and treated (MFC 3.75 mg/kg; 7.5 mg/kg and 15 mg/kg, via gavage, from day 1–6). 5-FU (Sigma–Aldrich, St. Louis, USA) was diluted in sterile water and injected intraperitoneally (i.p.), daily, for 3 consecutive (days 4, 5 and 6) days into mice to induce mucositis. During the induction of mucositis, MFC treatment was given thirty minutes before 5-FU injections, daily throughout the trial to the respective groups via gavage. On the 7th day of the experiment, animals were anesthetized by 10 mg/kg of xylazine and 100 mg/kg of ketamine hydrochloride administered i.p. and euthanized by cervical dislocation [19] . Duodenal samples, 10 cm after pyloric sphincter were removed to perform the assays.
|
|
|
Fig. 1. Schematic representation of MFC treatment and intestinal mucositis induction by 5-FU. |
2.4. Body weight assessment
Mice were daily weighed throughout the entire experimental period (7 days). The values were expressed as weight variation (%) in relation to the weight at the beginning of the experimental period [13] .
2.5. Morphometric and histopathological analysis
Duodenal samples were fixed in 10% phosphate buffered formalin, embedded in paraffin, sectioned at 5 μm thickness (Leica RM 2155, Heidelberg, Germany) and stained by hematoxylin and eosin (H&E). For intestinal morphometric examination, villus height (estimated from top of the villus to crypt-villus junction) and crypt depth (defined as adjacent intussusception) were determined using the AxioVision 40 software (Carl Zeiss, Jena, Germany) and a light microscope (Axio Scope A1Carl Zeiss, Jena, Germany) at 20X and 40X objectives. In this triple-blinded trial, ten villi and crypts from each animal duodenal slide were measured and averaged for each group.
Histopathological analysis evaluated the severity of intestinal tissue damage caused by 5-FU using ten different histological parameters, which consisted in villi and crypts size reduction, villi and crypts disruptions and abscess formation, muscularis thickening, epithelium and muscular layer integrity, inflammatory cells infiltration, vacuolization and mucosal edema. For each score, a value of 0–3 was given and combined for an overall score [10] and [13] .
2.6. Immunohistochemistry for Ki-67, p53, Bax and Bcl-2
Duodenal samples were embedded in paraffin, sectioned at 3 μm thickness (Leica RM 2155, Heidelberg, Germany) and mounted on silane-coated slides. The slides were then deparaffinized, rehydrated in graded ethanol and washed with tris-buffered saline (TBS) (pH 7.4). This was followed by incubation in a citric acid buffer solution (pH 6.0) at 96 °C for 20 min. Afterwards, slides were gradually cooled to room temperature and incubated in a 3% H2 O2 solution for 30 min to block endogenous peroxidase activity. They were then incubated in bovine serum albumin (BSA 1%) for 20 min. The next step was overnight incubation with one of the following primary antibodies (Santa Cruz Biotechnology, Santa Cruz, USA) at 4 °C: Ki-67 mouse monoclonal IgG1 (1:100); p53 mouse monoclonal IgG1 (1:50); Bax rabbit polyclonal IgG (1:50) and Bcl-2 mouse monoclonal IgG1 (1:50). After that, the streptavidin biotin peroxidase technique was performed. Slides were washed with TBS, incubated with secondary antibodies (ImmunoCruz™ mouse ABC staining systems sc-2017 or sc-2018, Santa Cruz Biotechnology, Santa Cruz, USA) for 30 min, washed again to eliminate excess free antibodies, incubated with reagent enzyme (avidin + biotin HRP) and washed once more. Another incubation was performed with streptavidin-peroxidase complex for 30 min followed by a TBS wash. Finally, slides were incubated in DAB developing solution (3.3'-diaminobenzidine) and counterstained with hematoxylin. The assays were performed in triplicate. Negative controls were obtained by replacing primary antibodies per 1% PBS–BSA.
Each slide was analyzed and photographed using a light microscopy (Axio Scope A1Carl Zeiss, Jena, Germany) at 40X objective. Also for each slide, a hundred duodenal epithelial cells were counted in ten consecutive fields to determine the percentage of positively stained cells expressing Ki-67, p53, Bax and Bcl-2.
2.7. Duodenal tissue homogenate preparation
Tissue homogenate procedure was carried out as previously described by Ávila et al. [13] . Briefly, 250 mg of duodenal tissue from participants of each group were collected and homogenized with hexadecyltrimethylammonium bromide buffer 0.5% by a dispersing machine (T25-digital Ultra-Turrax® -IKA, Germany) at 10.000 rpm, 25 °C for 5 min. Homogenates were centrifuged at 2000 rpm, 4 °C for 15 min, and supernatants obtained were used for MPO and MDA analysis.
2.8. Myeloperoxidase (MPO) activity
MPO activity assay was performed by adding 10 μL of samples previously prepared to 96-well plastic plates (TPP, Trasadingen, Switzerland), containing 100 μL of phosphate buffered saline, 100 μL of H2 O2 0.1% and 50 μL of orto -dianisidine 1.3%. The absorbance reading was performed at 450 nm wavelength. Results were expressed as active MPO unit per milligram of tissue [20] .
2.9. Malondialdehyde (MDA) determination
Tissue MDA determination was carried out according to a modified method developed by Ohkawa et al. [21] . Briefly, 250 μL of each previously prepared supernatant sample was homogenized with 25 μL of butylated hydroxytoluene 4%. Immediately afterwards, 1 mL of trichloroacetic acid solution 12%, 1 mL of thiobarbituric acid solution 0.73% and 750 μL of tris–HCl buffer were added, and the tubes were incubated at 95 °C water bath for 50 min. This mixture was later homogenized with 1.5 mL of n-butanol, centrifuged at 5000 rpm, 25 °C for 10 min, and supernatants were collected to determine the absorbance at 532 nm wavelength.
2.10. Statistical analysis
Data are reported as means ± standard error of the mean. The inter-group variation was evaluated by One- or Two-way Analysis of Variance (ANOVA) followed by Bonferroni post-tests using the Windows version of GraphPad Prism 5.01 software (San Diego, CA, USA). P– < 0.05 was considered statistically significant.
3. Results
3.1. Effect of MFC on animal body weight variation
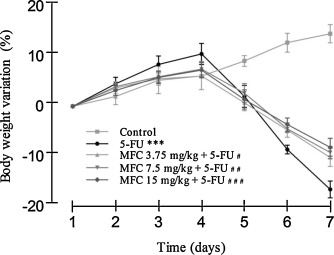
Intestinal mucositis usually leads to decrease in food intake and absorption capacity, and body weight analysis is an objective criterion for determining its severity [12] and [13] . As shown in Fig. 2 , all groups gained body weight until the 4th day, with the exception of the control group that gained weight throughout the entire experimental period, reaching a peak of 14.5 ± 0.33%. From day 4, animals only exposed to 5-FU doses (200 mg/kg) showed significantly body weight loss (P < 0.001), reaching ∼17% reduction on day 7. In these animals, MFC treatment attenuated 5-FU-induced body weight loss at all tested doses. Animals exposed to 5-FU and treated with 3.75; 7.5 or 15 mg/kg of MFC showed body weight of 10.2 ± 0.34%, 9.2 ± 0.28%, and 8.1 ± 0.3%, respectively.
|
|
|
Fig. 2. Body weight variation in mice subjected to intestinal mucositis and treated with MFC in relation to initial mass. Experiments performed in triplicate with 5 animals per group. Mice were daily weighed throughout the experimental period (7 days). Data expressed as mean ± SEM. ***P < 0.001 comparing 5-FU vs Control; #P < 0.05 comparing 5-FU vs MFC 3.75 mg/kg + 5-FU; ##P < 0.001 comparing 5-FU vs MFC 7.5 mg/kg + 5-FU; and ###P < 0.001 comparing 5-FU vs MFC 15 mg/kg + 5-FU. For statistical analysis, one-way ANOVA and Bonferroni post-tests were applied. |
3.2. Protective effect of MFC on 5-FU-induced intestinal mucositis
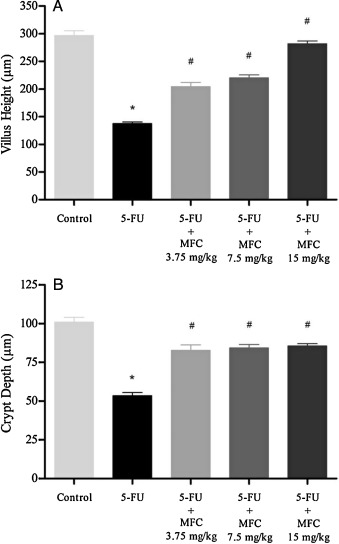
It is know that the reduction of intestinal length is a direct indicator of intestinal damage induced by chemotherapy drugs such as 5-FU and methotrexate [4] , [5] , [6] , [10] , [11] , [12] and [13] . Therefore, lengths of duodenal villi and crypts with different doses of MFC treatment were determined. As shown in Fig. 3 A and B, 100% of animals exposed to 5-FU showed a significant shortening in villi height (∼55.4%) and crypts depth (∼47%) when compared to control (P < 0.05). This reduction was markedly alleviated by the three MFC treatment doses (P < 0.05), with special emphasis on the MFC 15 mg/kg. These results suggested that the 5-FU-induced duodenal mucositis could be prevented and/ or reduced by MFC.
|
|
|
Fig. 3. Morphometric analysis of mice duodenum subjected to intestinal mucositis and treated with MFC. On day 7, duodenum segments were removed to measure villus height (panel A) and crypt depth (panel B). Data expressed as mean ± SEM. *P < 0.05 comparing 5-FU vs Control and #P < 0.05 comparing 5-FU vs MFC 3.75 mg/kg + 5-FU, MFC 7.5 mg/kg + 5-FU, MFC 15 mg/kg + 5-FU. For statistical analysis, two-way ANOVA and Bonferroni post-tests were applied. |
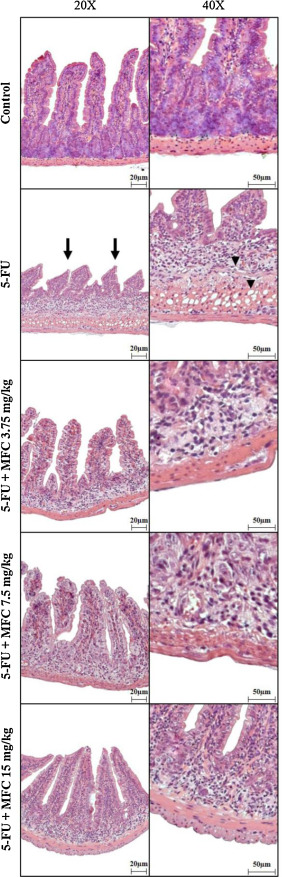
Furthermore, 5-FU group had a significantly increased (∼64%) histological severity score when compared to all other groups, characterized by villi and crypts disruption and shortening, intense inflammatory infiltration, vacuolization and mucosal edema (Fig. 4 ; Table 1 ). Histological severity scores were significantly (P < 0.05) decreased in MFC treated groups, especially MFC 15 mg/kg, except for those related to inflammatory infiltration.
|
|
|
Fig. 4. Histological sections of mice duodenum subjected to intestinal mucositis and treated with MFC. Control (panels A and B) showing normal villi and crypts. 5-FU (panels C and D) showing villi shortening (arrows), crypts deepening, intense inflammatory infiltration, vacuolization and mucosal edema (arrowheads). MFC 3.75 mg/kg + 5-FU (panels E and F), MFC 7.5 mg/kg + 5-FU (panels G and H), MFC 15 mg/kg + 5-FU (panels I and J) showing villi and crypts preserved – both in size and integrity –, however, inflammatory infiltrate was observed. H&E staining (panels A, C, E, G, I; 20X and B, D, F, H, J; 40X). |
| Groups | Scores values |
|---|---|
| Control | 3 (1, 5) |
| 5-FU* | 15 (9, 18) |
| MFC 3.75mg/kg+5-FU** | 7 (3, 10) |
| MFC 7.5mg/kg+5-FU** | 7 (3, 12) |
| MCF 15mg/kg+5-FU** | 5 (2, 9) |
Data expressed as median and range in parenthesis.
- . P < 0.001 comparing 5-FU vs control.
- . P < 0.05 comparing 5-FU vs MFC 3.75 mg/kg + 5-FU, MFC 7.5 mg/kg + 5-FU, and MFC 15 mg/kg + 5-FU. For statistical analysis, one-way ANOVA and Bonferroni post-tests were applied.
In view of these analyses, MFC 15 mg/kg demonstrated the most effective results in treating intestinal mucositis and was then chosen for subsequent investigations.
3.3. Effect of MFC on immunohistochemical detection of Ki-67, p53, Bax and Bcl-2
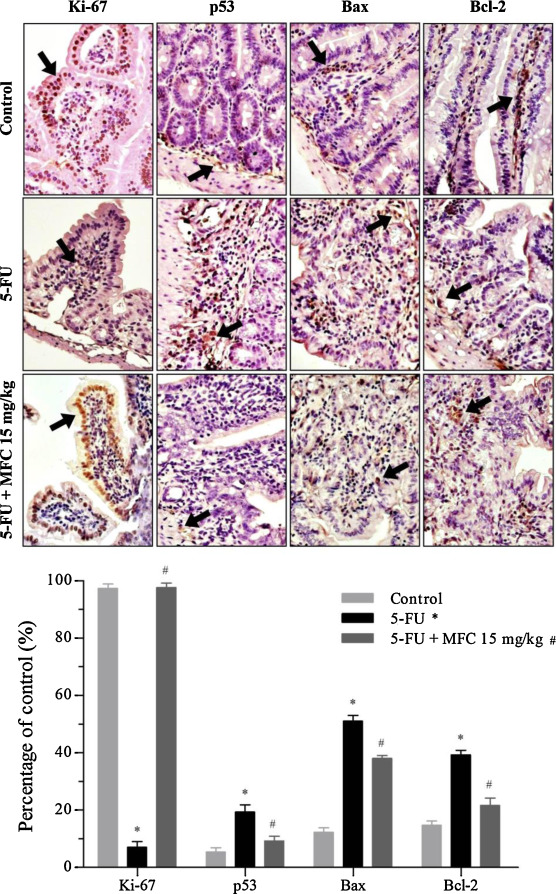
Chemotherapy-induced mucositis is able to lead intestinal basal stem cell to apoptosis, due its toxicity to the cellular turnover in bowel tissue. Proteins of the Bcl-2 family play a central role in the execution-phase of cell apoptosis together with p53 [3] and [33] . The pattern of positive immunoexpression shown in Fig. 5 demonstrates an increased expression of pro-apoptotic members p53, Bax and the anti-apoptotic member Bcl-2 in 5-FU photomicrographs (P < 0.05). Such high expression was not observed among treated animals (MFC 15 mg/kg), indicating less apoptosis (Fig. 6 ).
|
|
|
Fig. 5. Expression of Ki-67 (cell proliferation), p53 (pro-apoptotic), Bax (pro-apoptotic) and Bcl-2 (anti-apoptotic) in mice bearing intestinal mucositis and treated with MFC. See black arrows. Immunohistochemical staining, original magnification, 40X. Data expressed as mean ± SEM. *P < 0.05 comparing 5-FU vs Control, #P < 0.05 comparing 5-FU vs MFC 15 mg/kg. For statistical analysis, two-way ANOVA and Bonferroni post-tests were applied. |
|
|
|
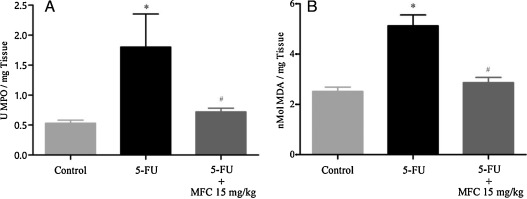
Fig. 6. Effect of MFC treatment on MPO activity and MDA determination in duodenal portions of mice bearing intestinal mucositis. Experiments performed in triplicate with 5 animals per group. Data expressed as mean ± SEM. *P < 0.05 comparing 5-FU vs Control, #P < 0.05 comparing 5-FU vs MFC 15 mg/kg. For statistical analysis, one-way ANOVA and Bonferroni post-tests were applied. |
Cell proliferation is usually described in the healing stage of chemotherapy-induced mucositis [1] , [2] , [4] and [6] . As expected, the Ki-67 level in 5-FU mice decreased by ∼90% compared to control (P < 0.05), and treatment with MFC at 15 mg/kg induced greater expression of Ki-67 protein (P < 0.05), shown by strong nuclear staining on the epithelium of villi and crypts, which indicates cellular proliferation (Fig. 5 ).
3.4. Effect of MFC in MPO activity and MDA formation
MPO is the most abundant enzyme present in azurophilic granules in neutrophils [20] . It is widely used as a neutrophil marker in inflamed tissue such as mucositis [10] , [13] and [22] . MPO activity in duodenal tissue of 5-FU-induced mucositis group was by 70% higher than in the control, and 60% higher relative to the treated group (Fig. 5 A, both P < 0.05). Additionally, there was no significant difference between treated (MFC 15 mg/kg) and control group (P < 0.05).
MDA is one of the final products widely adopted for a sensitive assay of lipid peroxidation and oxidative stress induced by ROS in intestinal tissue [13] , [23] and [24] . Fig. 5 B demonstrates there was an increase by ∼51% in MDA formation when mice which received 5-FU were compared to control group (P < 0.05). Treatment with MFC 15 mg/kg significantly reduced tissue oxidative stress and free radical production in about 44% (P < 0.05). Likewise, in MDA formation assay, there was no significant difference between treated and control group (P < 0.05).
4. Discussion
As no broadly curative drug treatment for mucositis is commercially available, palliative procedures have been employed to alleviate its signs and symptoms [25] , [26] , [27] and [28] . For its intestinal manifestation, therapeutic and/or preventive approaches are applied for infection and pain control, induction of cell proliferation and reepithelialization [4] , [6] , [7] , [8] and [9] . Considering the range of cancers and the occurrence of intestinal mucositis by antineoplastic agents, it is necessary to search for therapeutic alternatives to alleviate this serious side effect. In this study, MFC exerted protective action against 5-FU-induced morphological and histological intestinal changes, especially at the dose of 15 mg/kg. Such effectiveness is demonstrated by other anti-mucositis approaches [10] , [11] , [12] , [13] , [29] and [30] .
Body weight analysis showed that MFC was capable of attenuating weight loss induced by 5-FU. Intestinal mucositis generated by chemotherapy potentially causes a decrease in food intake, which linked to a loss in intestinal absorptive capacity lead to diarrheal episodes and body mass loss [1] and [3] . Diarrhea is reported as high as 50–80% in patients undergoing chemotherapy, and tends to become a limiting factor when severe [31] . Our data suggested improvements in food intake and nutrition in mice bearing mucositis treated with MFC. This result is consistent with other proposed experimental treatments for mucositis [10] , [11] , [12] , [13] , [14] and [32] .
The occurrence of intestinal mucositis induced by chemotherapy is directly associated with decreased cell proliferation and increased apoptosis activity in mucosal epithelial cells and, consequently, the modulation of regulatory proteins, such as the Bcl-2 family [33] . In this context, the tumor suppressor gene p53 interacts with members of the Bcl-2 family in the mitochondrial membrane promoting the release of cytochrome c, thereby increasing the transcription of the Fas death receptor gene and inhibiting the secretion of growth factors during chemotherapy sections [3] and [33] .
To elucidate the influence of MFC in cell proliferation/apoptosis activity in mucosal epithelial cells of mice bearing mice, an immunohistochemical identification of proteins Ki-67, p53, Bax and Bcl-2 was performed. Treatment with MFC 15 mg/kg induced a slightly greater increase in the expression of Bcl-2 (anti-apoptotic) than in Bax (pro-apoptotic) in the epithelium of villi and crypts. Likewise, treatment discretely reduced p53 expression in the animal mucosa. On the other hand, Ki-67 protein increased approximately 90% of expression in villi and crypts epithelium duodenal, when compared to 5-FU group, which indicates that the formulation acted on the rate of intestinal cell proliferation. Recently, Reinke et al. [10] , Wright et al. [11] , Ávila et al. [13] , and Al-Refai [34] , also found that antineoplastic agents administration result in increased apoptosis and decreased cellularity in the small-intestinal mucosa. Like our findings, the imbalance of the rate cell proliferation observed by these authors was modulated by the treatment.
With a view to measuring neutrophil infiltration in duodenal tissue, intestinal MPO activity was investigated. MPO levels were increased in duodenal tissue of 5-FU group, when compared to control. Otherwise, MPO amount in duodenal segments of mice treated with MFC was almost equivalent of normal levels, indicating less neutrophil infiltration and consequently less inflammation. Our data is consistent with the findings of Reinke et al. [10] and Ferreira et al. [32] .
MDA quantification has been used as a marker for the production of reactive oxygen species (ROS). ROS are involved in numerous mechanisms of injury, and one of the most significant is lipid peroxidation present in cell membranes. In this analysis, we measured lipid peroxidation associated to oxidative stress through the production of MDA in duodenal tissue samples. MDA values found in mice bearing mucositis and treated with MFC were about 50% lower than the values of 5-FU group, and equivalent to those of the control group. Our data correlates to Marques et al. [35] and Cunha et al. [36] . Thus, considering MDA levels found, it is suggested that 5-FU acted as a pro-oxidant on intestinal epithelium by keeping mucosa under constant oxidative damage. Furthermore, we propose that MFC was capable of protecting this epithelium, through the antioxidant properties of curcuminoids and mucoadhesive physical protection effect of poloxamer 407.
Mucoadhesive formulations allow intimate contact between the pharmaceutical form and the absorbing membrane by creating a film on the mucus layer lining of mucosa, thereby reducing its spread and protecting the drug from possible degradation in the intestinal lumen [37] . Therefore, the benefit rendered by mucoadhesive formulations would be a once daily regime, which would improve treatment compliance and reduce adverse effects [38] . Chen et al. [39] evaluated the potential of Poloxamer 407 as a thermogelling mucoadhesive polymer to improve curcuminoids bioavailability in primary culture cells. Their results revealed controlled curcuminoid release over a 24 h period. In our study, it can suggest that the components of mucoadhesive formulation acted as vehicle for curcuminoids and probably increased the contact time between extract components and intestinal mucosa.
In different parts of the world, the dry extract obtained from C. longa L. has been used for centuries associated with numerous biological activities [15] . However, as unformulated curcuminoids have relatively low bioavailability and poor aqueous solubility [16] and [40] . Yang et al. [41] observed that intestinal absorption of curcuminoids in murine models is only around 60%. Thus, our data further emphasizes the importance of the mucoadhesive formulation in reaching the expected clinical outcome against mucositis.
A study conducted by Henrotin et al. [42] demonstrated that there are many molecular evidences to justify the potential of C. longa L. components (monoterpenes, diterpenes, sesquiterpenes) for the treatment of various inflammatory diseases. However, in animal bearing mucositis and treated with MFC we still observed inflammatory infiltrate, which could be attributed to the presence of other inflammatory cells than neutrophils, such as lymphocyte infiltration.
Our data demonstrate that MFC exerted protective action against duodenal mucosa damage caused by 5-FU. It also stimulated cell proliferation, reduced weight loss, MPO levels and MDA formation. Supplementary studies are underway searching for the elucidation of mechanisms involved in the protective effects of MFC in order to make this formulation a clinical tool for the prevention and/or treatment of mucositis.
Conflicts of interest
The authors indicate no potential conflicts of interest.
Transparency document
Acknowledgements
The authors acknowledge the support of Fundação de Apoio a Pesquisa da Universidade Federal de Goiás (FUNAPE), Fundo de Amparo a Pesquisa do Estado de Goiás (FAPEG), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Financiadora de Estudos e Projetos (FINEP) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
References
- [1] S.T. Sonis, L.S. Elting, D. Keefe, D.E. Peterson, M. Schubert, M. Hauer-Jensen, B.N. Bekele, J. Raber-Durlacher, J.P. Donnelly, E.B. Rubenstein, Mucositis Study Section of the Multinational Association for Supportive Care in Cancer, International Society for Oral Oncology; Perspectives on cancer therapy-induced mucosal injury: pathogenesis, measurement, epidemiology, and consequences for patients; Cancer, 100 (2004), pp. 1995–2025 http://dx.doi.org/10.1002/cncr.20162
- [2] S.T. Sonis; Regimen-related gastrointestinal toxicities in cancer patients; Curr. Opin. Support Palliat Care, 4 (2010), pp. 26–30 http://dx.doi.org/10.1097/SPC.0b013e328335fb76
- [3] M.J. Van Vliet, H.J. Harmsen, E.S. de Bont, W.J. Tissing; The role of intestinal microbiota in the development and severity of chemotherapy-induced mucositis; PLoS Pathog., 6 (2010), p. e1000879 http://dx.doi.org/10.1371/journal.ppat.1000879
- [4] A. Villa, S.T. Sonis; Mucositis: pathobiology and management; Curr. Opin. Oncol., 27 (2015), pp. 159–164 http://dx.doi.org/10.1097/CCO.0000000000000180
- [5] C. Benz, M. Degregorio, S. Saks, N. Sambol, W. Holleran, R. Ignoffo, B. Lewis, E. Cadman; Sequential infusions of methotrexate and 5-fluorouracil in advanced cancer: pharmacology, toxicity, and response; Cancer Res., 45 (1985), pp. 3354–3358
- [6] Y.Z. Van Sebille, R. Stansborough, H.R. Wardill, E. Bateman, R.J. Gibson, D.M. Keefe; Management of mucositis during chemotherapy: from pathophysiology to pragmatic therapeutics; Curr. Oncol. Rep., 17 (2015) http://dx.doi.org/10.1007/s11912-015-0474-9
- [7] M.R. Posner, R.I. Haddad; Novel agents for the treatment of mucositis; J. Support Oncol., 5 (2007), pp. 33–39
- [8] E.B. Rubenstein, D.E. Peterson, M. Schubert, D. Keefe, D. McGuire, J. Epstein, L.S. Elting, P.C. Fox, C. Cooksley, S.T. Sonis; Mucositis study section of the multinational association for supportive care in cancer; International Society for Oral Oncology,clinical practice guidelines for the prevention and treatment of cancer therapy-induced oral and gastrointestinal mucositis; Cancer, 100 (2004), pp. 2026–2046 http://dx.doi.org/10.1002/cncr.20163
- [9] D.M. Keefe, S.T. Sonis, J.M. Bowen; Emerging drugs for chemotherapy-induced mucositis; Expert Opin. Emerg. Drugs, 13 (2008), pp. 511–522 http://dx.doi.org/10.1517/14728214.13.3.511
- [10] D. Reinke, S. Kritas, P. Polychronopoulos, A.L. Skaltsounis, N. Aligiannis, C.D. Tran; Herbal substance, acteoside, alleviates intestinal mucositis in mice; Gastroenterol Res. Pract., 2015 (2015) http://dx.doi.org/10.1155/2015/327872
- [11] T.H. Wright, R. Yazbeck, K.A. Lymn, E.J. Whitford, K.Y. Cheah, R.N. Butler, C. Feinle-Bisset, A.N. Pilichiewicz, S. Mashtoub, G.S. Howarth; The herbal extract, iberogast, improves jejunal integrity in rats with 5-Fluorouracil (5-FU)-induced mucositis; Cancer Biol. Ther., 8 (2009), pp. 923–929 http://dx.doi.org/10.4161/cbt.8.10.8146
- [12] Z. Liu, J. Xi, S. Schröder, W. Wang, T. Xie, Z. Wang, S. Bao; Chimonanthus nitens var. salicifolius aqueous extract protects against 5-fluorouracil induced gastrointestinal mucositis in a mouse model ; Evid. Based Compl. Alt. Med., 2013 (2013) http://dx.doi.org/10.1155/2013/789263
- [13] P.H.M. Ávila, R.I. Ávila, E.X.S. Filho, C.C.C. Bastos, A.C. Batista, E.F. Mendonça, R.C. Serpa, R.N. Marreto, A.F. Cruz, E.M. Lima, M.C. Valadares; Mucoadhesive formulation of Bidens pilosa L. (Asteraceae) reduces intestinal injury from 5-fluorouracil-induced mucositis in mice ; Toxicol. Rep., 2 (2015), pp. 563–573 http://dx.doi.org/10.1016/j.toxrep.2015.03.003
- [14] W.T. Loo, L.J. Jin, L.W. Chow, M.N. Cheung, M. Wang; Rhodiola algida improves chemotherapy-induced oral mucositis in breast cancer patients ; Expert Opin. Investig. Drugs, 19 (Suppl. 1) (2010), pp. 91–100 http://dx.doi.org/10.1517/13543781003727057
- [15] S.C. Gupta, B. Sung, J.H. Kim, S. Prasad, S. Li, B.B. Aggarwal; Multitargeting by turmeric, the golden spice: from kitchen to clinic; Mol. Nutr. Food Res., 57 (2013), pp. 1510–1528 http://dx.doi.org/10.1002/mnfr.201100741
- [16] O. Naksuriya, S. Okonogi, R.M. Schiffelers, W.E. Hennink; Curcumin nanoformulations: a review of pharmaceutical properties and preclinical studies and clinical data related to cancer treatment; Biomaterial, 35 (2014), pp. 3365–3383 http://dx.doi.org/10.1016/j.biomaterials.2013.12.090
- [17] S.Y. Li, W. Yuan, G.R. Deng, P. Wang, P. Yang, B.B. Aggarwal; Chemical composition and product quality control of turmeric (Curcuma longa L.) ; Pharm. Crops., 2 (2011), pp. 28–54
- [18] F.J. Varum, E.L. McConnell, J.J. Sousa, F. Veiga, A.W. Basit; Mucoadhesion and the gastrointestinal tract; Crit. Rev. Ther. Drug Carrier. Syst., 25 (2008), pp. 207–258 i3.10 http://dx.doi.org/10.1615/CritRevTherDrugCarrierSyst.v25. i3.10
- [19] R. Hubrecht, J. Kirkwood; The UFAW Handbook on the Care and Management of Laboratory and Other Research Animals; (8th ed.)Wiley-Blackwell, United Kingdom (2010)
- [20] P.P. Bradley, R.D. Christensen, G. Rothstein; Cellular and extracellular myeloperoxidase in pyogenic inflammation; Blood, 60 (1982), pp. 618–622
- [21] H. Ohkawa, N. Ohishi, K. Yagi; Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction; Anal Biochem., 95 (1979), pp. 351–358
- [22] R.C. Lima-Júnior, A.A. Figueiredo, H.C. Freitas, M.L. Melo, D.V. Wong, C.A. Leite, R.P. Medeiros, R.D. Marques-Neto, M.L. Vale, G.A. Brito, R.B. Oriá, M.H. Souza, F.Q. Cunha, R.A. Ribeiro; Involvement of nitric oxide on the pathogenesis of irinotecan-induced intestinal mucositis: role of cytokines on inducible nitric oxide synthase activation; Cancer. Chemother. Pharmacol., 4 (2012), pp. 931–942 http://dx.doi.org/10.1007/s00280-011-1780-z
- [23] H. Okur, M. Küçükaydin, K. Köse, O. Kontaş, P. Doğam, A. Kazez; Hypoxia-induced necrotizing enterocolitis in the immature rat: the role of lipid peroxidation and management by vitamin E; J. Pediatr. Surg., 10 (1995), pp. 1416–1419
- [24] O.M.S. Sá, N.N.F. Lopes, M.T.S. Alves, R.V. Lalla, M.L.V. Oliva, E.M.M. Caran; Glycine supplementation reduces the severity of chemotherapy-induced oral mucositis in hamsters; Nat. Sci., 5 (2013), pp. 972–978 http://dx.doi.org/10.4236/ns.2013.59118
- [25] D.B. McGuire, J.S. Fulton, J. Park, C.G. Brown, M.E.P. Correa, J. Eilers, S. Elad, F. Gibson, L.K. Oberle-Edwards, J. Bowen, R.V. Lalla, Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO); Systematic review of basic oral care for the management of oral mucositis in cancer patients; Support. Care Cancer, 21 (2013), pp. 3165–3177 http://dx.doi.org/10.1007/s00520-013-1942-0
- [26] O. Nicolatou-Galitis, T. Sarri, J. Bowen, M. Di Palma, V.E. Kouloulias, P. Niscola, D. Riesenbeck, M. Stokman, W. Tissing, E. Yeoh, S. Elad, R.V. Lalla, Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO); Systematic review of anti-inflammatory agents for the management of oral mucositis in cancer patients; Support. Care Cancer, 21 (2013), pp. 3179–3189 http://dx.doi.org/10.1007/s00520-013-1847-y
- [27] D.P. Saunders, J.B. Epstein, S. Elad, J. Allemano, P. Bossi, M.D. van de Wetering, N.G. Rao, C. Potting, K.K. Cheng, A. Freidank, M.T. Brennan, J. Bowen, K. Dennis, R.V. Lalla, Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO); Systematic review of antimicrobials, mucosal coating agents, anesthetics, and analgesics for the management of oral mucositis in cancer patients; Support. Care Cancer, 21 (2013), pp. 3191–3207 http://dx.doi.org/10.1007/s00520-013-1871-y
- [28] A.F. Oton-Leite, G.B. Silva, M.O. Morais, T.A. Silva, C.R. Leles, M.C. Valadares, J.C. Pinezi, A.C. Batista, E.F. Mendonça; Effect of low-level laser therapy on chemoradiotherapy-induced oral mucositis and salivary inflammatory mediators in head and neck cancer patients; Lasers Surg. Med., 47 (2015), pp. 296–305 http://dx.doi.org/10.1002/lsm.22349
- [29] J.C. Cool, J.L. Dyer, C.J. Xian, R.N. Butler, M.S. Geier, G.S. Howarth; Pre-treatment with insulin-like growth factor-I partially ameliorates 5-fluorouracil-induced intestinal mucositis in rats; Growth Horm. IGF Res., 15 (2005), pp. 72–82 http://dx.doi.org/10.1016/j.ghir.2004.12.002
- [30] T.G. Son, E.J. Gong, M.J. Bae, S.D. Kim, K. Heo, C. Moon, K. Yang, J.S. Kim; Protective effect of genistein on radiation-induced intestinal injury in tumor bearing mice; BMC Complement Altern. Med., 13 (2013) http://dx.doi.org/10.1186/1472-6882-13-103
- [31] A. Stein, W. Voigt, K. Jordan; Chemotherapy-induced diarrhea: pathophysiology, frequency and guideline-based management; Ther. Adv. Med. Oncol., 2 (2010), pp. 51–63 http://dx.doi.org/10.1007/s00520-013-1871-y
- [32] T.M. Ferreira, A.J. Leonel, M.A. Melo, R.R. Santos, D.C. Cara, V.N. Cardoso, M.I. Correia, J.I. Alvarez-Leite; Oral supplementation of butyrate reduces mucositis and intestinal permeability associated with 5-fluorouracil administration; Lipids, 47 (2012), pp. 669–678 http://dx.doi.org/10.1007/s11745-012-3680-3
- [33] D.M. Pritchard, C.S. Potten, J.A. Hickman; The relationships between p53-dependent apoptosis, inhibition of proliferation, and 5-fluorouracil-induced histopathology in murine intestinal epithelia; Cancer Res., 58 (1998), pp. 5453–5465
- [34] A.S. Al-Refai; Immunohistochemical study of the effect of chamomile extract on 5-fluorouracil induced intestinal mucositis in Albino rats; J. Clin. Cell Immunol., 5 (2014) http://dx.doi.org/10.4172/2155-9899.1000232
- [35] L.H.S. Marques, C.M.G. Silva, T.M.M. Lameiro, M.G. Almeida, F.L. Cunha, J.A. Pereira, C.A.R. Martinez; Evaluation of lipid peroxidation levels on mucosa colonic cells after application of hydrogen peroxide in enemas. Experimental study in rats; Rev. Bras. Colo-Proctol., 30 (2010), pp. 272–280
- [36] F.L. Cunha, C.M.G. Silva, M.G. Almeida, T.M.M. Lameiro, L.H.S. Marques, N.F. Margarido, C.A. Martinez; Reduction in oxidative stress levels in the colonic mucosa without fecal stream after the application of enemas containing aqueous Ilex paraguariensis extract; Acta Cir. Bras., 26 (2011), pp. 289–296
- [37] V. Grabovac, D. Guggi, A.A. Bernkop-Schnürch; Comparison of the mucoadhesive properties of various polymers; Adv. Drug Deliv. Rev., 57 (2005), pp. 1713–1723 http://dx.doi.org/10.1016/j.addr.2005.07.006
- [38] P. Chinna Reddy, K.S. Chaitanya, Y. Madhusudan Rao; A Review on bioadhesive buccal drug delivery systems: current status of formulation and evaluation methods; Daru, 19 (2011), pp. 385–403
- [39] M.J. Chen, Y.M. Cheng, P.H. Lai, J.F. Wu, Y.C. Hsu; In vitro biocompatibility of thermally gelling liquid mucoadhesive loaded curcuminoids in colorectal cancer chemoprevention ; Int. J. Colorectal Dis., 27 (2012), pp. 869–878 http://dx.doi.org/10.1007/s00384-011-1393-3
- [40] S. Prasad, A.K. Tyagi, B.B. Aggarwal; Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: the golden pigment from golden spice; Cancer Res. Treat., 46 (2014), pp. 2–18 http://dx.doi.org/10.4143/crt.2014.46.1.2
- [41] K.Y. Yang, L.C. Lin, T.Y. Tseng, S.C. Wang, T.H. Tsai; Oral bioavailability of curcumin in rat and the herbal analysis from Curcuma longa by LC–MS/MS ; J. Chromatogr. B Analyt. Technol. Biomed. Life Sci., 853 (2007), pp. 183–189 http://dx.doi.org/10.1016/j.jchromb.2007.03.010
- [42] Y. Henrotin, F. Priem, A. Mobasheri; Curcumin: a new paradigm and therapeutic opportunity for the treatment of osteoarthritis curcumin for osteoarthritis management; Springerplus, 2 (2013) http://dx.doi.org/10.1186/2193-1801-2-56
Document information
Published on 12/05/17
Accepted on 12/05/17
Submitted on 12/05/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?