Summary
Inferior vena cava (IVC) thrombus is a rare presentation of adrenal carcinoma. Hyperladosteronism is rarely associated with it. We report a case of an aldosterone-producing left adrenal carcinoma with IVC thrombus and invasion of multiple organs, treated with neoadjuvant chemotherapy and salvage surgery. The patient is alive and asymptomatic after 46 months. Surgical aspects and therapeutic options are discussed and compared with the current medical literature. This is believed to be the first report of multiple organ resection combined with IVC thrombus removal for a functioning adrenal carcinoma.
Keywords
adrenocortical cancer;hyperaldosteronism;inferior vena cava thrombus
1. Introduction
Adrenal carcinomas are rare, with an incidence of 0.7 to 2 cases per 1 million population per year.1 They are more common in women, with a female to male ratio of 1.5 to 1.2 Female patients seem to have a higher probability of presenting with functioning malignant tumors, which apparently hold a better prognosis than nonfunctioning adrenal neoplasms.2 Also, adrenal cancers have been associated with familial syndromes and gene mutations, and rarely with hyperaldosteronism.3 ; 4
There is limited experience in the treatment of advanced adrenal carcinoma. Current strategies are based on retrospective analysis and case reports. Recently, large multicenter trials have elucidated some aspects of multiagent chemotherapy usage for advanced disease.5 ; 6 However, salvage surgery is not routine, and is performed in a minority of patients.5 ; 6
Adrenal carcinomas in general do not invade adjacent organs.2 Left-sided adrenal carcinoma with inferior vena cava (IVC) thrombus is a rare presentation for this uncommon malignancy.7; 8; 9 ; 10 Salvage surgery for adrenal carcinoma with IVC thrombus and multiple organ invasion has never been previously reported.
We describe an unusual case of a 42-year-old female patient diagnosed with a left-sided adrenal carcinoma with invasion of multiple organs and an IVC thrombus subjected to neoadjuvant chemotherapy and salvage surgery.
2. Case report
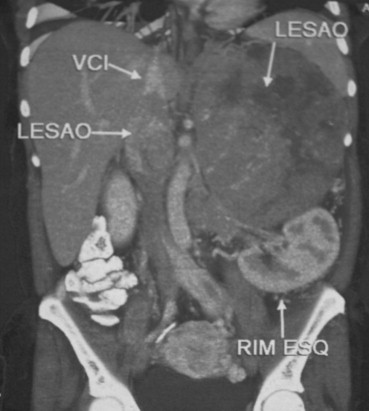
A 42-year-old woman was referred to our Surgical Oncology Department after an unsuccessful attempt to remove a voluminous left adrenal carcinoma with an IVC thrombus extending to the retrohepatic cava (Fig. 1). The patient also complained of bilateral inferior limb edema and refractory hypertension due to hyperaldosteronism (supine aldosterone blood levels = 36.4 ng/dL). Potassium blood levels were normal (3.5 mEq/L). Family history was negligible.
|
|
|
Figure 1. Computed tomography scan showing a voluminous mass, with inferior vena cava thrombus, extending to the suprahepatic vein level. VCI = inferior vena cava; LESAO = adrenal cancer and the vena cava thrombus; RIM ESQ = left kidney (Note that abbreviations were in Portuguese). |
Salvage chemotherapy with etoposide, doxorubicin, cisplatin, and mitotane was administered for 6 months. During chemotherapy, two hypertensive crisis episodes were diagnosed. After chemotherapy, a 30% mass reduction was observed and salvage surgery was indicated. No metastasis had been found before surgery.
At laparotomy, a large mass with mesocolon, left colic artery, spleen, pancreas, and left kidney invasion was seen. Left radical adrenalectomy with left nephrectomy, splenectomy, caudal pancreatectomy, and resection of the left mesocolon with ligation of the left colic artery were performed. The IVC was clamped at the retrohepatic diaphragmatic level. A right cavotomy with total removal of the thrombus was performed and the IVC was sutured. An associated left-sided retroperitoneal lymphadenectomy was executed. No postoperative complications were diagnosed, and resection was considered to leave no residual disease (R0). Blood pressure and aldosterone levels normalized soon after surgery and mitotane was maintained for 6 months.
After 18 months, bilateral pulmonary metastases were detected. Aldosterone blood levels were normal and hypertension was not present. Gemcitabine was administered for six months and continuous daily mitotane was initiated. Metastases stability was achieved.
Currently, the patient has survived for 46 months and is asymptomatic with stable disease. No hypertensive episodes have been documented since the radical surgical procedure.
3. Discussion
Left adrenal carcinoma with IVC thrombus and multiple organ invasion has rarely, if ever, been reported.
Our case demonstrates an initially inoperable female patient with a voluminous adrenal tumor and refractory hypertension. A combination chemotherapy regimen with etoposide, cisplatin, doxorubicin, and mitotane was started and seems to be the most active combination for advanced disease.5 ; 6 It was used as a salvage chemotherapy regimen and allowed surgery to be performed after a 6-month period.
Surgery remains the most important pillar in adrenal carcinoma treatment. In our opinion, salvage surgical procedures should always be attempted, because they probably represent the best option for the patient.6; 7 ; 11 Furthermore, they have been associated with extended overall survival.2; 4 ; 11 In this case, the patient remained asymptomatic for 24 months. It is important to note that recurrence occurred as nonfunctioning disease, demonstrating that its behavior cannot be predicted.
Multiple organ invasion is rare in adrenal cancer. Nevertheless, it should not preclude surgical treatment, which can be safely performed by an experienced surgical team.7; 8 ; 12
Mitotane has been widely used in adrenal carcinoma. It seems capable of prolonging survival, especially in patients previously subjected to radical surgical resection.13
High aldosterone blood levels are found in 1.5–5% of hypertensive patients. However, aldosterone-producing adrenal cancer is rarely the cause.14; 15 ; 16 Removal of functioning adrenal tumors usually corrects hypertension. Nevertheless, some patients can remain hypertensive in spite of R0 resection.17 It is important to mention that primary functioning adrenal carcinomas seem not to correlate with the metastasis pattern. Our patient had nonfunctioning metastases, whereas the opposite has been described elsewhere.4 ; 17
Finally, it seems that aldosterone-producing tumors have a better prognosis than cortisol-producing neoplasms.18 ; 19 Our patient had aldosterone-producing adrenal cancer associated with refractory hypertension. Surgical resection gave the only possibility of palliative control. The prolonged survival seems to ratify the current belief that aldosterone-producing neoplasms, either benign or malignant, do have a better outcome than cortisol-producing tumors.
References
- 1 S.H. Golden, K.A. Robinson, I. Saldanha, B. Anton, P.W. Ladenson; Prevalence and incidence of endocrine and metabolic disorders in the United States: a comprehensive review; J Clin Endocrinol Metab, 94 (2009), pp. 1853–1878
- 2 M.S. Didolkar, R.A. Bescher, E.G. Elias, R.H. Moore; Natural history of adrenal cortical carcinoma. A clinicopathologic study of 42 patients; Cancer, 47 (1981), pp. 2153–2161
- 3 J.E. Griniatsos, N. Dimitriou, A. Zilos, et al.; Bilateral adrenocortical carcinoma in a patient with multiple endocrine neoplasia type 1 (MEN1) and a novel mutation in the MEN1 gene; World J Surg Oncol, 9 (2011), p. 6
- 4 E.M. Abma, P.M. Kluin, R.P. Dullaart; Malignant aldosterone-producing adrenal tumor: reoccurrence with glucocorticoid excess without hyperaldosteronism; Neth J Med, 66 (2008), pp. 252–255
- 5 Y. Shudo, G. Matsumia, T. Sakaguchi, et al.; Combination chemotherapy in advanced adrenocortical carcinoma; N Engl J Med, 366 (2012), pp. 2189–2197
- 6 A. Berruti, M. Terzolo, P. Sperone, et al.; Etoposide, doxorubicin and cisplatin plus mitotane in the treatment of advanced adrenocortical carcinoma: a large prospective phase II trial; Endoc Relat Cancer, 12 (2005), pp. 657–666
- 7 L. Chiche, B. Dousset, E. Kleffer, Y. Chapuis; Adrenocortical carcinoma extending into the inferior vena cava: presentation of a 15-patient series and review of the literature; Surgery, 139 (2006), pp. 15–27
- 8 J. Hardwigsen, P. Baqué, B. Crespy, V. Moutardier, J.R. Delpero, Y.P. Le Treut; Resection of the inferior vena cava for neoplasms with or without prosthetic replacement: a 14-patient series; Ann Surg, 233 (2001), pp. 242–249
- 9 T. Nakanoma, M. Ueno, S. Nonaka, T. Tsukamoto, N. Deguchi; Left adrenocortical cancer with inferior vena cava tumor thrombus – a case report; Nihon Hinyokika Gakkai Zasshi, 92 (2001), pp. 34–37
- 10 A.N. Hisham, A. Sarajah, H. Zanariah; Large adrenocortical carcinoma extending into the inferior vena cava and right atrium; Asian J Surg, 26 (2003), pp. 40–42
- 11 O.D. Winkel, J. Pfannchmidt, T. Muley, et al.; Metastatic adrenocortical carcinoma: results of 56 pulmonary metastasectomies in 24 patients; Ann Thorac Surg, 92 (2011), pp. 1965–1970
- 12 S. Gaujoux, M.F. Brennan; Recommendation for standardized surgical management of primary adrenocortical carcinoma; Surgery, 152 (2012), pp. 123–132
- 13 M. Terzolo, A. Angeli, M. Fassnacht, et al.; Adjuvant mitotane treatment for adrenocortical carcinoma; N Engl J Med, 356 (2007), pp. 2372–2380
- 14 K. Weingartner, E.W. Gerharz, A. Bittinger, J. Rosai, R. Leppek, H. Riedmiller; Isolated clinical syndrome of primary aldosteronism in a patient with adrenocortical carcinoma: case report and review of the literature; Urol Int, 55 (1995), pp. 232–235
- 15 L. Ammar, P.F. Plouin, O. Steichen; Aldosterone-producing adenoma and other surgically correctable forms of primary aldosteronism; Orphanet J Rare Dis, 19 (2010), pp. 5–9
- 16 C. Yang, W. Qiang, L. Lil, Y. Lin, J. Zhu, S. Han; Clinical analysis of 149 cases of aldosterone-producing adrenal cortical neoplasms; Zhonghua Wai Ke Za Zhi, 399 (2001), pp. 937–939
- 17 T.M. Seccia, A. Fassina, G.G. Nussdorfer, A.C. Pessina, G.P. Rossi; Aldosterone-producing adrenocortical carcinoma: an unusual cause of Conns syndrome with an ominous clinical course; Endoc Relat Cancer, 12 (2005), pp. 149–159
- 18 S. Balasubramaniam, T. Fojo; Practical considerations in the evaluation and management of adrenocortical cancer; Semin Oncol, 37 (2010), pp. 19–26
- 19 M.J. Bolland, I.M. Holdaway, J.E. Berkeley, et al.; Mortality and morbidity in Cushings syndrome in New Zealand; Clin Endocrinol, 75 (2011), pp. 436–442
Document information
Published on 26/05/17
Submitted on 26/05/17
Licence: Other
Share this document
claim authorship
Are you one of the authors of this document?