Highlights
- Chronic kidney disease (CKD) in atrial fibrillation (AF) patients is associated with several clinical factors.
- There is a linear association between creatinine clearance and time in therapeutic range (TTR).
- CKD is a risk factor for stroke and death, while TTR > 70% is a predictor for stroke, major bleeding and death.
The concomitant presence of atrial fibrillation (AF) and chronic kidney disease (CKD) confers an increased risk of both ithromboembolism and bleeding. Oral anticoagulation with vitamin K antagonist (VKA) are an effective treatment in preventing thromboembolic events, but good anticoagulation control as reflected by a high time in therapeutic range (TTR) is crucial in reducing thromboembolic and bleeding adverse events. Our results showed a close relationship between CKD, TTR and adverse events. In the decision-making process of prescribing VKA, clinicians need to consider renal function as well as good anticoagulation control (e.g. TTR > 70%) to ensure best efficacy and safety.
Abstract
Background
Chronic kidney disease (CKD) is highly prevalent in atrial fibrillation (AF) patients and associated with an increased risk of adverse outcomes. Our objectives were to study clinical features associated with CKD in AF patients and the impact of CKD on anticoagulation control, as reflected by time in therapeutic range (TTR). We also determined the impact of CKD and TTR in predicting adverse outcomes.
Methods and Results
We analysed pooled datasets from SPORTIF III and V trials, including 3646 patients assigned to warfarin with data on renal function. CKD (creatinine clearance < 60 ml/min) was present in 952 (26%) patients. TTR was higher in patients with normal renal function compared to those with CKD (p < 0.001).
On logistic analysis, chronic AF and male sex were associated with TTR > 70%, whilst diabetes mellitus, aspirin use and CKD were inversely associated with TTR > 70%. On Cox regression analysis, CKD was an independent predictor for stroke (p = 0.006) and death (p < 0.001). TTR > 70% was independently associated with a lower risk of stroke (p = 0.024), death (p = 0.001) and major bleeding (p = 0.001).
Conclusions
CKD is highly prevalent amongst AF patients and a risk factor for stroke and death. Adjusting for CKD, good quality anticoagulation control (TTR > 70%) was an independent predictor for lower risks of stroke, death and major bleeding.
Keywords
Non-valvular atrial fibrillation ; Chronic kidney disease ; Stroke ; Major bleeding ; Clinical outcomes
1. Introduction
Chronic kidney disease (CKD) is highly prevalent in non-valvular AF patients, ranging from 26% to 32%, especially in the elderly (Apostolakis et al., 2013 and Banerjee et al., 2013 ). The presence of CKD in AF patients is associated with a greater risk of adverse clinical outcomes, including stroke, thromboembolism and bleeding. In the Danish nationwide cohort study, for example, the presence of CKD amongst patients with AF was significantly associated with an increased risk for stroke/systemic embolism (by 1.49-fold), all-cause mortality (by 2.37-fold), myocardial infarction (by 2-fold) and major bleeding (by 1.33-fold) (Olesen et al., 2012 ). Warfarin use amongst such patients has been associated with a substantial risk reduction in overall event rates, with a positive net clinical benefit (Bonde et al., 2014 ). Similarly, the Loire Valley Atrial Fibrillation Project confirmed the high risk associated with CKD in patients with AF (Banerjee et al., 2014 and Banerjee et al., 2013 ). In clinical trial populations, CKD also confers a much higher relative risk for both stroke and bleeding (Apostolakis et al., 2013 and Piccini et al., 2013 ).
When Vitamin K Antagonists (VKAs, e.g. warfarin) are used for thromboprophylaxis, good quality anticoagulation control, as reflected by time in therapeutic range (TTR) > 70% is recommended (De Caterina et al., 2013 ). Limited information been reported on the relationship between CKD and TTR in AF patients, but good TTR has been associated with lower risks of thromboembolism and bleeding (Connolly et al., 2008 , Friberg et al., 2014 and Pokorney et al., 2015 ).
In this study, our objectives were to study clinical features associated with CKD in AF patients; second, we studied the impact of CKD on anticoagulation control, as reflected by the time in therapeutic range (TTR). Third, we determined the impact of CKD as a risk factor for stroke, myocardial infarction, major bleeding and death amongst warfarin-treated AF patients, after adjustment for TTR as measure of anticoagulation control.
2. Methods
For the present analysis, we used the pooled study populations of the Stroke Prevention using an Oral Thrombin Inhibitor in patients with atrial Fibrillation (SPORTIF) III and V trials. The original protocol and principal results have been previously described (Albers et al., 2005 , Halperin, 2003 and Olsson, 2003 ). In brief, the SPORTIF trials were two multicentre phase III clinical trials comparing the efficacy and safety of the direct thrombin inhibitor, ximelagatran, against warfarin in patients with non-valvular AF. SPORTIF III was an open label trial, while SPORTIF V was a double blind study (Halperin, 2003 ). Signed, informed consent was required from each participant in accordance with protocol regulations approved by the local review boards governing research involving human subjects, and the Declaration of Helsinki. In both SPORTIF trials, ximelagatran was non-inferior in efficacy and safety as warfarin for stroke prevention in non-valvular AF patients (Lip, 2005 ) but due to hepatotoxicity, ximelagatran was withdrawn. For the purpose of this study, we studied only SPORTIF patients assigned to warfarin treatment where complete information on renal function was available.
2.1. Thromboembolic Risk and Anticoagulation Control
Thromboembolic risk was categorised according to both CHADS2 and CHA2 DS2 -VASc scores (Gage et al., 2004 and Lip et al., 2010 ). Based on CHADS2 , patients were categorised as “low risk” when with CHADS2 = 0, “moderate risk” with CHADS2 = 1 and “high risk” when CHADS2 ≥ 2 (Gage et al., 2004 ). Based on the CHA2 DS2 -VASc score, “low risk” was defined as a CHA2 DS2 -VASc 0 in males or 1 in females; “moderate risk” as male patients with CHA2 DS2 -VASc = 1; and “high risk” with CHA2 DS2 -VASc ≥ 2 (Camm et al., 2012 and Lip et al., 2015 ).
Anticoagulation control, as reflected by the TTR was calculated using the standardized Rosendaal interpolation method (Rosendaal et al., 1993 ), by assigning an INR value to each day between two successive observed INR values. The percentage of time that the interpolated INR remained between 2.0 and 3.0 was used to establish TTR value, and the cut-off value for defining optimal anticoagulation was a mean individual TTR of ≥ 70% (De Caterina et al., 2013 ).
2.2. Renal Function
Renal function was based on the estimated creatinine clearance (CrCl), using the 4-item Cockroft-Gault formula [(140 − age) ∗ (weight in kg) ∗ (0.85 if female) / (72 ∗ creatinine)]. Accordingly, an estimated CrCl level of < 60 ml/min was used to define CKD, based on established guidelines (“K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification.,” 2002 ).
2.3. Study Outcomes
Principal clinical outcomes were based on the original study protocol, and were centrally adjudicated (Halperin, 2003 ). Stroke was defined as the acute onset of a focal neurological deficit in any of the carotid, vertebral or cerebral artery distribution territories lasting > 24 h. As per the SPORTIF trials protocol, myocardial infarction (MI) diagnosis required 2 out of 3 of the following criteria: (i) typical chest pain lasting at least 20 min; (ii) electrocardiographic changes clearly indicating an acute MI; (iii) significant, i.e. > 2 × upper limit, elevation of cardiac biomarkers (creatine kinase-MB, troponin). Based on the International Society of Thrombosis and Haemostasis (ISTH) criteria (Schulman and Kearon, 2005 ), major bleeding was defined by one of the following: clinically overt bleeding with a concomitant fall in haemoglobin levels > 2 g/dl or requiring blood transfusion of at least 2 units of whole blood or erythrocytes; bleeding episode involving a critical site.
2.4. Statistical Analysis
All continuous variables were tested for normality with the Shapiro-Wilk test. Variables with normal distribution were expressed as means and standard deviations (SD), and tested for differences using a t -test. Non-parametric variables were expressed as median and interquartile range (IQR) and differences tested using the Mann-Whitney U test. Categorical variables, expressed as counts and percentages, were analysed by chi-squared test. A linear regression analysis between CrCl and TTR was performed, adjusted for type of AF and thromboembolic risk.
Regression analysis was performed in order to establish clinical factors significantly associated with the presence of CKD. All variables different between the two groups at the baseline with a level of significance of at least p < 0.10 underwent a univariate analysis, and those predictors with a level significance of p < 0.10 were inserted into a forward multivariate logistic model. To determine the factors associated with a TTR > 70%, both for general population and CKD-only population, all baseline clinical variables underwent univariate analyses and those predictors with a level significance of p < 0.10 were inserted into a forward multivariate logistic model. In order to explore the incremental value of TTR in determining events occurrence, a correlation analysis between TTR and hazard risk for study outcomes was performed using Pearson product-moment correlation test, both for the overall cohort and stratified according to CKD presence. Correlation coefficients were then compared using the z test, after being processed with Fisher transformation.
Survival analysis, assessed by an intention-to-treat approach, was performed according to the presence of CKD and differences in survival distributors between subgroups were analysed using the Log-Rank test. Evaluation of the clinical characteristics significantly associated with outcomes was performed using a Cox proportional hazards analysis, corrected according to thromboembolic risk, performed with CKD both as a categorical variable and CrCl as a continuous variable. All demographic variables, comorbidities and risk factors analysed at baseline underwent a univariate analysis. All variables with a p -value < 0.10 for the association with clinical outcome at the univariate analysis, were inserted into the forward stepwise multivariate model. A backward model was then performed to verify the results of the forward one. A two-sided p value < 0.05 was considered statistically significant. All analyses were performed using SPSS v. 22.0 (IBM, NY, USA) and R v. 3.2.1 (The R Foundation).
3. Results
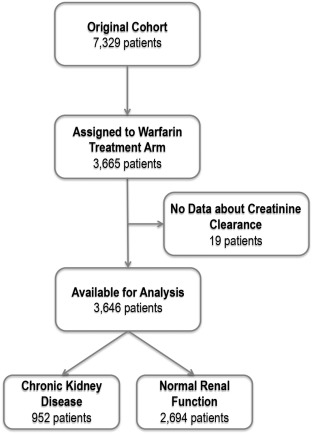
The pooled study population constituted 7329 patients, of which 3665 were assigned to the warfarin treatment arm. Complete data on renal function were available for 3646 (99.5%) [[[#f0005|Fig. 1]] ]. Of the selected cohort, 30.5% (n = 1113) were female, median age was 72 [IQR 66–77] years, and median CrCl was 78.60 [59.1–102.1] ml/min. Of the entire cohort, 10.8% (n = 393) had paroxysmal AF.
|
|
|
Fig. 1. Patients included in this analysis. |
3.1. Prevalence of CKD and Associated Clinical Characteristics
CKD (defined as CrCl < 60 ml/min) was present in 952 (26.1%) patients. Clinical characteristics of patients with and without CKD are shown in Table 1 . Patients with CKD were significantly older (p < 0.001), more commonly female (p < 0.001) or paroxysmal AF (p = 0.009) and a lower median weight (p < 0.001). CKD patients had more prevalent coronary heart disease (CHD) (p = 0.042) and previous stroke or transient ischemic attack (TIA) (p < 0.001). Thromboembolic risk was higher in patients with CKD compared to those with normal renal function, whether based on CHADS2 (median [IQR] 2 [2–2] vs. 2 [1–2] respectively, p < 0.001) and CHA2 DS2 -VASc (median [IQR] 4 [3–5] vs. 3 [2–3], p < 0.001).
| N (%) | Normal renal function (≥ 60 ml/min)N = 2694 | Chronic kidney disease (< 60 ml/min)N = 952 | p |
|---|---|---|---|
| Age, years (median [IQR]) | 70 [64–75] | 78 [74–82] | < 0.001 |
| Weight, kg (median [IQR]) | 89 [78–100] | 70 [61–79] | < 0.001 |
| Female | 656 (24.4) | 457 (48.0) | < 0.001 |
| Creatinine clearance, ml/min | 88.86 [74.02–111.57] | 49.77 [43.01–55.25] | < 0.001 |
| Type of atrial fibrillation (n = 3644) | 0.009 | ||
| Paroxysmal | 269 (10.0) | 124 (13.0) | |
| Chronic | 2424 (90.0) | 827 (87.0) | |
| Heart rate (n = 3510), per min | 76 [67–87] | 73 [64–86] | < 0.001 |
| Hypertension | 2082 (77.3) | 717 (75.3) | 0.216 |
| Diabetes mellitus | 652 (24.2) | 205 (21.5) | 0.095 |
| Current smoker | 272 (10.1) | 58 (6.1) | < 0.001 |
| Coronary heart disease | 1162 (43.2) | 447 (46.9) | 0.042 |
| Chronic heart failure | 988 (36.7) | 378 (39.7) | 0.097 |
| Previous stroke/TIA | 485 (18.0) | 263 (27.6) | < 0.001 |
| Previous bleeding | 154 (5.7) | 54 (5.7) | 0.958 |
| Aspirin use | 518 (19.2) | 205 (21.6) | 0.119 |
| TTR (n = 3606), % (median [IQR]) | 69.6 [56.7–80.2] | 66.6 [51.9–76.8] | < 0.001 |
| TTR > 70% (n = 3606) | 1307 (48.9) | 390 (41.8) | < 0.001 |
| Median [IQR] CHADS2 | 2 [1–2] | 2 [2–2] | < 0.001 |
| CHADS2 score | |||
| 0 | 99 (3.7) | 13 (1.4) | |
| 1 | 1233 (45.8) | 196 (20.6) | < 0.001 |
| ≥ 2 | 1362 (50.6) | 743 (78.0) | |
| Median [IQR] CHA2 DS2 -VASc | 3 [2–3] | 4 [3–5] | < 0.001 |
| CHA2 DS2 -VASC risk class | < 0.001 | ||
| Low risk | 8 (0.3) | 1 (0.1) | |
| Intermediate risk | 457 (17.0) | 19 (2.0) | |
| High risk | 2229 (82.7) | 932 (97.9) |
Legend : BPM = beats per minute; IQR = interquartile range; TIA = transient ischemic attack; TTR = time in therapeutic range. Low risk = male patients with CHA2 DS2 -VASC 0 or female patients with CHA2 DS2 -VASC 1; intermediate risk = male patients with CHA2 DS2 -VASC 1; high risk = all patients with CHA2 DS2 -VASC ≥ 2.
On multivariable logistic regression analysis, clinical characteristics significantly associated with the presence of CKD were age (Odds Ratio (OR) 1.16 per year, 95% confidence interval (CI) 1.15–1.18, p < 0.001), weight (OR for any kg 0.92, 95% CI 0.91–0.92, p < 0.001), paroxysmal AF (OR 1.67, 95% CI 1.23–2.27, p = 0.001), diabetes mellitus (OR 1.47, 95% CI 1.16–1.86, p = 0.001), chronic heart failure (CHF) (OR 1.61, 95% CI 1.32–1.96, p < 0.001), and previous stroke/TIA (OR 1.41, 95% CI 1.13–1.75, p = 0.002).
3.2. Relationship between CKD and TTR
Median TTR in the whole cohort was 68.6% [IQR 56.5–80.6%], being significantly higher in patients with normal renal function when compared with CKD patients (p < 0.001). Also, the proportion with TTR > 70% was more prevalent in patients with normal renal function (p < 0.001).
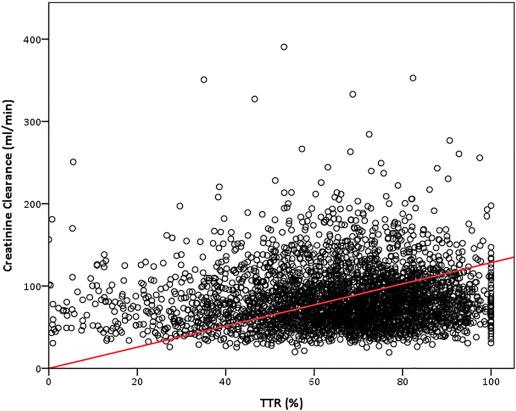
After adjustment for type of AF and thromboembolic risk, a linear relationship was found between CrCl and TTR (standardized beta: 0.054, p = 0.003) [[[#f0010|Fig. 2]] ]. On multivariable logistic analysis, the presence of chronic AF (OR 1.56, 95% CI 1.25–1.94, p < 0.001) and male sex (OR 1.18, 95% CI 1.01–1.35, p = 0.033) were positively associated with a TTR > 70%. Conversely, a previous diagnosis of diabetes mellitus (OR 0.82, 95% CI 0.70–0.96, p = 0.011), concomitant use of aspirin (OR 0.75, 95% CI 0.63–0.88, p < 0.001) and the presence of CKD (OR 0.75, 95% CI 0.67–0.92, p = 0.002) were negatively associated with achieving a TTR > 70%.
|
|
|
Fig. 2. Scatterplot and regression line between creatinine clearance and TTR Legend = TTR: time in therapeutic range. |
In the CKD subgroup (n = 952), the weight (OR per kg 1.01, 95% CI 1.00–1.02, p = 0.041), the presence of CHF (OR 1.38, 95% CI 1.05–1.81, p = 0.022) and chronic AF (OR 1.85, 95% CI 1.22–2.81, p = 0.004) were significantly associated with TTR > 70%.
3.3. Impact of CKD in Predicting Adverse Clinical Outcomes
After a median [IQR] follow-up time of 1.6 [1.3–1.8] years, 504 events (13.8% of patients) were recorded, as follows: 90 strokes (17.9% of total events), 52 myocardial infarctions (10.3%), 198 deaths (39.3%) and 164 major bleeding events (32.5%).
3.3.1. Stroke
Stroke was more common in CKD patients than in patients with normal renal function (2.6% patient-years vs. 1.3% patient-years, respectively), with the unadjusted hazard ratio (HR) for stroke being 1.99 (95% CI: 1.30–3.03, p = 0.001). With CrCl as a continuous variable, every decrease of 10 ml/min was associated with a higher risk of stroke (HR 1.07, 95% CI 1.00–1.14, p = 0.048).
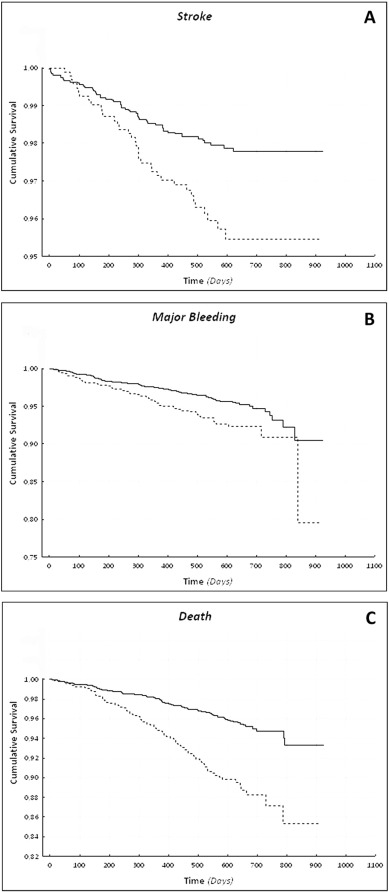
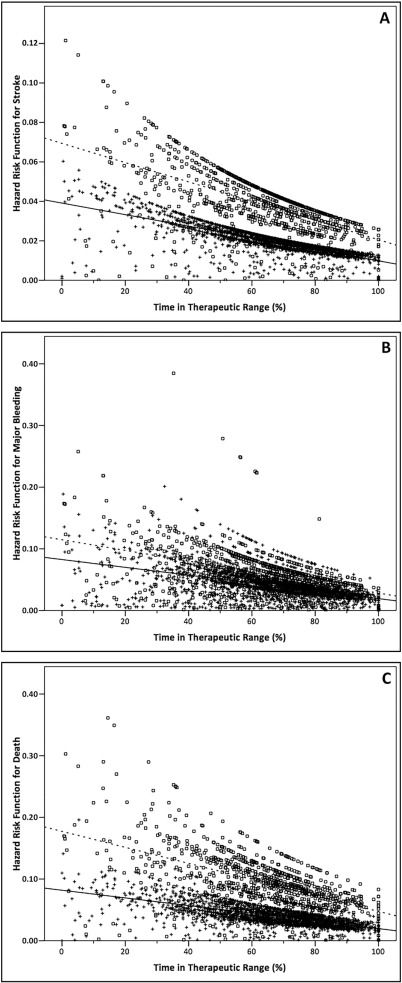
On Kaplan-Meier analysis, CKD patients had a higher risk of stroke compared to patients with normal renal function (Log-Rank test: 10.56; p = 0.001) [[[#f0015|Fig. 3]] , Panel A]. On multivariable Cox regression analysis, a clinical history of CHD (p = 0.045), a previous stroke/TIA (p < 0.001) and the presence of CKD (p = 0.006) were independent risk factors associated with the occurrence of stroke, while TTR > 70% (p = 0.004) was inversely associated with the risk of stroke (Table 2 ). Cox regression analysis performed using CrCl as a continuous variable ( Table 2 ) shows that its influence in determining stroke risk became non-significant, after adjusting for other clinical risk factors (p = 0.337). A backward selection model also confirmed these results. TTR and HR for occurrence of stroke were inversely correlated (r = − 0.772, p < 0.001). Similarly, there was a significant correlation between TTRs and stroke HRs according to renal function subgroups [[[#f0020|Fig. 4]] , Panel A] (r = − 0.569, p < 0.001). A significant difference when comparing the two correlation coefficients for the overall cohort and stratified according to CKD presence, respectively (z = 8.61, p < 0.001).
|
|
|
Fig. 3. Cumulative event-free survival in patients according to the presence of chronic kidney disease. Panel A) Stroke p = 0.001; Panel B) Major bleeding p = 0.002; Panel C) Death p < 0.001. Legend = Solid line: normal renal function; dashed line: CKD. |
| Multivariable analysis | |||
|---|---|---|---|
| Hazard ratio | 95% CI | p | |
| Categorical variable | |||
| Stroke | |||
| TTR > 70% | 0.63 | 0.41–0.98 | 0.040 |
| Coronary heart disease | 1.54 | 1.01–2.34 | 0.042 |
| Chronic kidney diseasea | 1.82 | 1.19–2.80 | 0.006 |
| Previous stroke/TIA | 2.23 | 1.44–3.44 | < 0.001 |
| Major bleeding | |||
| TTR > 70% | 0.58 | 0.42–0.80 | 0.001 |
| Age (per year) | 1.03 | 1.01–1.05 | 0.009 |
| Chronic heart failure | 1.40 | 1.02–1.92 | 0.036 |
| Death | |||
| TTR > 70% | 0.63 | 0.47–0.84 | 0.002 |
| Female | 0.66 | 0.47–0.92 | 0.013 |
| Chronic heart failure | 1.53 | 1.14–2.04 | 0.003 |
| Current smoker | 1.54 | 1.00–2.38 | 0.049 |
| Coronary heart disease | 1.77 | 1.32–2.37 | < 0.001 |
| Chronic kidney diseasea | 2.58 | 1.92–3.46 | < 0.001 |
| Continuous variable | |||
| Stroke | |||
| TTR > 70% | 0.60 | 0.39–0.93 | 0.024 |
| Coronary heart disease | 1.54 | 1.01–2.34 | 0.045 |
| Previous stroke/TIA | 2.18 | 1.40–3.38 | 0.001 |
| Major bleeding | |||
| TTR > 70% | 0.58 | 0.42–0.80 | 0.001 |
| Age (per year) | 1.03 | 1.01–1.05 | 0.009 |
| Chronic heart failure | 1.40 | 1.02–1.92 | 0.036 |
| Death | |||
| TTR > 70% | 0.61 | 0.46–0.82 | 0.001 |
| Female | 0.69 | 0.49–0.96 | 0.029 |
| Current smoker | 1.55 | 1.00–2.38 | 0.047 |
| Chronic heart failure | 1.58 | 1.19–2.12 | 0.002 |
| Coronary heart disease | 1.75 | 1.30–2.35 | < 0.001 |
| Creatinine clearance (per 10 ml/min decrease) | 1.13 | 1.07–1.19 | < 0.001 |
Legend : CI = confidence interval; TIA = transient ischemic attack; TTR = time in therapeutic range.
a. Creatinine clearance < 60 ml/min.
|
|
|
Fig. 4. Correlation between TTR and hazard risk according to presence of chronic kidney disease. Panel A) Stroke p < 0.001; Panel B) Major bleeding p < 0.001; Panel C) Death p < 0.001. Legend = Cross and solid line: normal renal function; square and dashed line: CKD. |
3.3.2. Myocardial Infarction
The crude incidence rate of MI was similar between those with CKD and normal renal function (1.1% and 0.9% patient-years, respectively). No significant association was found between CKD and MI (unadjusted HR 1.19, 95% CI 0.66–2.18, p = 0.563).
3.3.3. Major Bleeding
Major bleeding was more common in patients with CKD than in those with normal renal function (4.6% patient-years, vs. 2.8% patient-years, respectively). Similar to stroke, patients with CKD had a significantly higher risk of a major bleeding event, compared to those with normal renal function (Log-Rank: 10.03; p = 0.002) [[[#f0015|Fig. 3]] , Panel B].
CKD conferred a higher risk of major bleeding (unadjusted HR 1.67, 95% CI 1.21–2.31, p = 0.002). This risk was non-significant when CrCl was considered as a continuous variable (per decrease of 10 ml/min, HR 1.04, 95% CI 0.99–1.09, p = 0.093). On multivariable analysis (see Table 2 ), increasing age (p = 0.009) and clinical history of CHF (p = 0.036) were risk factors for major bleeding; conversely, TTR > 70% (p = 0.007) was independently associated with less major bleeding. Entering CrCl in the analysis as a continuous variable did not affect the multivariable model (Table 2 ). Similar data were obtained when performing a backward selection model.
When correlating TTR and HR for major bleeding, a significant inverse relationship was found in the overall cohort (r = − 0.527, p < 0.001). When performed with HR according to renal function subgroups [[[#f0020|Fig. 4]] , Panel B], there was a numerical reduction in correlation coefficient (r = − 0.490, p < 0.001), but this was non-significant (z = 1.57, p = 0.116).
3.3.4. All-cause Death
A higher rate of death was found in patients with CKD compared to those with normal renal function (crude incidence rate 6.4% patient-years vs. 2.5% patient-years; unadjusted HR 2.54 (95% CI 1.92–3.36, p < 0.001)). As for stroke, a decrease of 10 ml/min in CrCl was found associated with a higher risk of death (HR 1.14, 95% CI 1.08–1.20, p < 0.001).
Survival analysis confirmed a higher mortality of patients with CKD compared to patients with normal renal function (Log-Rank test: 45.62; p < 0.001) [[[#f0015|Fig. 3]] , Panel C]. Multivariable Cox regression analysis (Table 2 ) shows that female sex (p = 0.013) and TTR > 70% (p = 0.002) were independently associated with a lower risk of death; conversely, a clinical history of CHF (p = 0.004), CHD (p < 0.001), smoking habit (p = 0.049) and the presence of CKD (p < 0.001) were risk factors associated with an increased risk of death. When the Cox multivariable analysis was performed using CrCl as a continuous variable, a decrease of 10 ml/min was associated with a higher risk of death (Table 2 ). No difference was found when backward selection model was performed.
Similar to stroke, there was significant inverse correlation between unadjusted HR for death and TTR (r = − 0.676, p < 0.001), but the correlation coefficient was reduced (r = − 0.478, p < 0.001) when stratifying according to CKD presence [[[#f0020|Fig. 4]] , Panel C]. The difference between correlation coefficients was significant (z = 8.40, p < 0.001).
4. Discussion
In this study, we demonstrate that more than a quarter of AF patients have CKD, and that age, female sex, CHF, previous stroke/TIA and the occurrence of paroxysmal AF were significantly associated with CKD in AF patients. Second, we found a linear association between CrCl and TTR in AF, and those patients with CKD had poorer anticoagulation control, as shown by a lower TTR when compared to patients with normal renal function. Importantly, the presence of CKD was negatively associated with achieving a TTR > 70%. Third, CKD was an independent risk factor for the occurrence of stroke and death, independently from TTR; however, no significant association was found between CKD and both myocardial infarction or major bleeding after adjusting for TTR. Conversely, adequate anticoagulation control as expressed by TTR > 70% was inversely associated with major bleeding.
Clinical factors associated with CKD have been previously reported in a smaller real-world’ populations of AF patients and our data are broadly confirmatory of the role of age, diabetes mellitus and CHF in predicting the presence of CKD in AF patients (Roldán et al., 2013 ). Indeed, both increasing age and diabetes mellitus are major predictors of CKD in the general population (Fox, 2004 ). Conversely, hypertension (an important risk factor for CKD in general population (Fox, 2004 )) had no significant influence on CKD amongst AF patients in our trial cohort.
CKD is predictive of stroke in AF patients, whilst oral anticoagulation with VKA lowers stroke risk albeit at a higher risk of bleeding (Hart et al., 2011 ). The Loire Valley Atrial Fibrillation Project showed that CKD, defined as an estimated glomerular filtration rate (eGFR) below 60 ml/min/1.73 m2 , was associated with a higher incidence of ischemic stroke/thromboembolism events, but multivariable Cox regression analysis did not demonstrate an independent effect of renal impairment on predicting major adverse events, after adjusting for CHA2 DS2 -VASc score risk factors (Banerjee et al., 2014 and Banerjee et al., 2013 ).
Our results demonstrate that even in AF patients treated with warfarin, CKD was still associated with a higher incidence of stroke, independent of other concomitant risk factors. These data support previous findings from another prospective randomised trial, where the presence of a CrCl < 60 ml/min was associated with two-fold higher risk of stroke compared with those with CrCl ≥ 60 ml/min (Apostolakis et al., 2013 ). Importantly, we show that the predictive ability of CKD in identifying patients with a greater risk of stroke was independent of anticoagulation control, as reflected by the TTR, notwithstanding the latter being a strong predictor of adverse outcomes in patients with AF (White et al., 2007 ).
The lack of significant association between CrCl when analysed as a continuous variable, and the risk of stroke could be attributed to more complex and multifactorial physiological alterations present in patients with established CKD that statistical adjustments would not be able to fully adjust for. Indeed, the risk of cardiovascular disease is non-linear in the transition from one functional CKD class to another, but increases exponentially with greater impairment of renal function (Schiffrin et al., 2007 ).
Our study also supports previous evidence showing that CKD is associated with a higher risk for major bleeding (Apostolakis et al., 2013 and Banerjee et al., 2014 ), even when stratified according to VKA treatment (Banerjee et al., 2014 ). Nonetheless, major bleeding is strongly related to the quality of anticoagulation control, and supra-therapeutic INR values amongst CKD patients are associated with a higher risk of developing a major bleed especially with progressive renal impairment (Limdi et al., 2015 ). In contrast to other previous studies (Olesen et al., 2012 ), we failed to show a significant association with MI risk, but given the low number of MIs recorded, this lack of association could partially be due to an inadequate sample size, and being unpowered. Finally, we found that CKD was an independent predictor for a higher risk of all cause death, consistent with prior studies (Apostolakis et al., 2013 , Banerjee et al., 2014 and Olesen et al., 2012 ). Importantly, the high risk for death in our study was independently related to CKD and inversely to effective anticoagulation (TTR > 70%).
Our data strengthen and reinforce the relationship between renal function and TTR (Kleinow et al., 2011 and Kooiman et al., 2014 ). One study derived from a cohort of anticoagulated AF patients found that patients with moderate and severe CKD had higher TTR than patients with normal renal function, although CKD patients spent more time above therapeutic range and INR-variability significantly increased in patients with moderate and severe CKD (Kooiman et al., 2014 ). Recently, an analysis from the Outcomes Registry for Better Informed Treatment of Atrial Fibrillation (ORBIT-AF) registry found that an estimated glomerular filtration rate below 60 ml/min/1.73 m2 was significantly associated with a reduced TTR (≤ 53%) (Pokorney et al., 2015 ). Our results strongly reinforce this relationship between renal function and anticoagulation control, and that CKD patients derived benefit from VKA treatment with good TTRs (Bonde et al., 2014 ).
Good quality anticoagulation control (TTR > 70%) was an independent predictor for a lower risk of stroke, death and major bleeding. There was a significant inverse correlation between TTR and HR risk function for stroke, death and major bleeding overall, but when this analysis was repeated in the CKD subgroup, the degree of correlation was reduced for stroke and death risk, perhaps reflecting how CKD modulates the risk of adverse events.
4.1. Limitations
Renal function data was only available based on the Cockroft-Gault formula and not by more recent and validated methods of eGFR evaluation (Levey et al., 2015 ). The original trial protocol excluded patients with a severe CKD, and this may have led to an underestimate in the prevalence of CKD in AF population. Also, the absence of subjects with severe CKD precluded a more detailed analysis of the wider spectrum of renal function and its influence on TTR and clinical outcomes. Moreover, other parameters used to evaluate anticoagulation control (e.g. time spent below or above the target, proportion of INRs in range, etc.) were not available for this analysis. Since the original trials were run between 2000 and 2002, current clinical practice in managing AF patients could have changed, with possible implications for the generalizability of our results. However, VKAs are still very widely used worldwide for thromboprophylaxis, in particular for treating AF patients with CKD (Potpara et al., 2015 ). Thus, the association between CKD and TTR remains relevant during the decision-making process to prescribe the right drug for the right patient (Shields and Lip, 2015 ). Finally, the association of CKD with paroxysmal AF could be spurious, since only a low proportion of these subjects were enrolled in the original trial cohort.
In conclusion , CKD is highly prevalent amongst AF patients and associated with several clinical comorbidities. Importantly, CKD in AF patients is an independent risk factor for stroke and death, but not major bleeding after adjustment for TTR. Even after adjusting for CKD, good quality anticoagulation control (TTR > 70%) was an independent predictor for a lower risk of stroke, death and major bleeding.
Author Contributions
MP and GYHL conceived the study, analysed data, interpreted results and drafted the manuscript. DAL provided critical revision of the manuscript.
Funding
None directly related to this manuscript.
Disclosures
GYHL : Chairman, Scientific Documents Committee, European Heart Rhythm Association (EHRA). Reviewer for various guidelines and position statements from ESC, EHRA, NICE etc. Steering Committees/trials: Includes steering committees for various Phase II and III studies, Health Economics & Outcomes Research, etc. Investigator in various clinical trials in cardiovascular disease, including those on antithrombotic therapies in atrial fibrillation, acute coronary syndrome, lipids, etc. Consultant for Bayer/Janssen, Astellas, Merck, Sanofi, BMS/Pfizer, Biotronik, Medtronic, Portola, Boehringer Ingelheim, Microlife and Daiichi-Sankyo.
Speaker for Bayer, BMS/Pfizer, Medtronic, Boehringer Ingelheim, Microlife, Roche and Daiichi-Sankyo.
DAL : Investigator-initiated educational grants from Bayer Healthcare and Boehringer Ingelheim. Speakers bureau for Boehringer Ingelheim, Bristol-Myers-Squibb and Bayer for lectures at educational meetings. Dr Lane is also on the Steering Committee of a Phase IV clinical trial sponsored by Bristol-Myers-Squibb.
Appendix A. Supplementary data
Supplementary Tables
References
- Albers et al., 2005 G.W. Albers, H.-C. Diener, L. Frison, M. Grind, M. Nevinson, S. Partridge, J.L. Halperin, J. Horrow, S.B. Olsson, P. Petersen, A. Vahanian; Ximelagatran vs warfarin for stroke prevention in patients with nonvalvular atrial fibrillation: a randomized trial; JAMA, 293 (2005), pp. 690–698 http://dx.doi.org/10.1001/jama.293.6.690
- Anon., 2002 Am. J. Kidney Dis., 39 (2002), pp. S1–266
- Apostolakis et al., 2013 S. Apostolakis, Y. Guo, D.A. Lane, H. Buller, G.Y.H. Lip; Renal function and outcomes in anticoagulated patients with non-valvular atrial fibrillation: the AMADEUS trial; Eur. Heart J., 34 (2013), pp. 3572–3579 http://dx.doi.org/10.1093/eurheartj/eht328
- Banerjee et al., 2013 A. Banerjee, L. Fauchier, P. Vourc’h, C.R. Andres, S. Taillandier, J.M. Halimi, G.Y.H. Lip; Renal impairment and ischemic stroke risk assessment in patients with atrial fibrillation: the Loire Valley atrial fibrillation project; J. Am. Coll. Cardiol., 61 (2013), pp. 2079–2087 http://dx.doi.org/10.1016/j.jacc.2013.02.035
- Banerjee et al., 2014 A. Banerjee, L. Fauchier, P. Vourc'h, C.R. Andres, S. Taillandier, J.M. Halimi, G.Y.H. Lip; A prospective study of estimated glomerular filtration rate and outcomes in patients with atrial fibrillation: the Loire Valley atrial fibrillation project; Chest, 145 (2014), pp. 1370–1382 http://dx.doi.org/10.1378/chest.13-2103
- Bonde et al., 2014 A.N. Bonde, G.Y.H. Lip, A.-L. Kamper, P.R. Hansen, M. Lamberts, K. Hommel, M.L. Hansen, G.H. Gislason, C. Torp-Pedersen, J.B. Olesen; Net clinical benefit of antithrombotic therapy in patients with atrial fibrillation and chronic kidney disease; J. Am. Coll. Cardiol., 64 (2014), pp. 2471–2482 http://dx.doi.org/10.1016/j.jacc.2014.09.051
- Camm et al., 2012 A. Camm, G. Lip, R. De Caterina, I. Savelieva, D. Atar, S. Hohnloser, G. Hindricks, P. Kirchhof; 2012 focused update of the ESC guidelines for the management of atrial fibrillation: an update of the 2010 ESC guidelines for the management of atrial fibrillation. Developed with the special contribution of the European Heart Rhythm Association; Eur. Heart J., 33 (2012), pp. 2719–2747 http://dx.doi.org/10.1093/eurheartj/ehs253
- Connolly et al., 2008 S.J. Connolly, J. Pogue, J. Eikelboom, G. Flaker, P. Commerford, M.G. Franzosi, J.S. Healey, S. Yusuf; Benefit of oral anticoagulant over antiplatelet therapy in atrial fibrillation depends on the quality of international normalized ratio control achieved by centers and countries as measured by time in therapeutic range; Circulation, 118 (2008), pp. 2029–2037 http://dx.doi.org/10.1161/CIRCULATIONAHA.107.750000
- De Caterina et al., 2013 R. De Caterina, S. Husted, L. Wallentin, F. Andreotti, H. Arnesen, F. Bachmann, C. Baigent, K. Huber, J. Jespersen, S.D. Kristensen, G.Y.H. Lip, J. Morais, L.H. Rasmussen, A. Siegbahn, F.W.A. Verheugt, J.I. Weitz; Vitamin K antagonists in heart disease: current status and perspectives (Section III); Thromb. Haemost., 110 (2013), pp. 1087–1107 http://dx.doi.org/10.1160/TH13-06-0443
- Fox, 2004 C.S. Fox; Predictors of new-onset kidney disease in a community-based population; JAMA, 291 (2004), p. 844 http://dx.doi.org/10.1001/jama.291.7.844
- Friberg et al., 2014 L. Friberg, L. Benson, G.Y.H. Lip; Balancing stroke and bleeding risks in patients with atrial fibrillation and renal failure: the Swedish Atrial Fibrillation Cohort study; Eur. Heart J. (2014), pp. 297–306 http://dx.doi.org/10.1093/eurheartj/ehu139
- Gage et al., 2004 B.F. Gage, C. van Walraven, L. Pearce, R.G. Hart, P.J. Koudstaal, B.S.P. Boode, P. Petersen; Selecting patients with atrial fibrillation for anticoagulation: stroke risk stratification in patients taking aspirin; Circulation, 110 (2004), pp. 2287–2292 http://dx.doi.org/10.1161/01.CIR.0000145172.55640.93
- Halperin, 2003 J.L. Halperin; Ximelagatran compared with warfarin for prevention of thromboembolism in patients with nonvalvular atrial fibrillation: rationale, objectives, and design of a pair of clinical studies and baseline patient characteristics (SPORTIF III and V); Am. Heart J., 146 (2003), pp. 431–438 http://dx.doi.org/10.1016/S0002-8703(03)00325-9
- Hart et al., 2011 R.G. Hart, L.A. Pearce, R.W. Asinger, C.A. Herzog; Warfarin in atrial fibrillation patients with moderate chronic kidney disease; Clin. J. Am. Soc. Nephrol., 6 (2011), pp. 2599–2604 http://dx.doi.org/10.2215/CJN.02400311
- Kleinow et al., 2011 M.E. Kleinow, C.L. Garwood, J.L. Clemente, P. Whittaker; Effect of chronic kidney disease on warfarin management in a pharmacist-managed anticoagulation clinic; J. Manag. Care Pharm., 17 (2011), pp. 523–530
- Kooiman et al., 2014 J. Kooiman, N. van Rein, B. Spaans, K.A.J. van Beers, J.R. Bank, W.R. van de Peppel, A.I. del Sol, S.C. Cannegieter, T.J. Rabelink, G.Y.H. Lip, F.A. Klok, M.V. Huisman; Efficacy and safety of vitamin K-antagonists (VKA) for atrial fibrillation in non-dialysis dependent chronic kidney disease; PLoS One, 9 (2014), Article e94420 http://dx.doi.org/10.1371/journal.pone.0094420
- Levey et al., 2015 A.S. Levey, C. Becker, L.A. Inker; Glomerular filtration rate and albuminuria for detection and staging of acute and chronic kidney disease in adults: a systematic review; JAMA, 313 (2015), pp. 837–846 http://dx.doi.org/10.1001/jama.2015.0602
- Limdi et al., 2015 N.A. Limdi, T.D. Nolin, S.L. Booth, A. Centi, M.B. Marques, M.R. Crowley, M. Allon, T.M. Beasley; Influence of kidney function on risk of supratherapeutic international normalized ratio-related hemorrhage in warfarin users: a prospective cohort study; Am. J. Kidney Dis., 65 (2015), pp. 701–709 http://dx.doi.org/10.1053/j.ajkd.2014.11.004
- Lip, 2005 G.Y.H. Lip; Preventing stroke in atrial fibrillation: the SPORTIF programme; Pathophysiol. Haemost. Thromb., 34 (Suppl. 1) (2005), pp. 25–30 http://dx.doi.org/10.1159/000083081
- Lip et al., 2010 G.Y.H. Lip, R. Nieuwlaat, R. Pisters, D.A. Lane, H.J.G.M. Crijns; Refining clinical risk stratification for predicting stroke and thromboembolism in atrial fibrillation using a novel risk factor-based approach: the euro heart survey on atrial fibrillation; Chest, 137 (2010), pp. 263–272 http://dx.doi.org/10.1378/chest.09-1584
- Lip et al., 2015 G.Y.H. Lip, F. Skjøth, L.H. Rasmussen, T.B. Larsen; Oral anticoagulation, aspirin, or no therapy in patients with nonvalvular AF with 0 or 1 stroke risk factor based on the CHA2DS2-VASc score; J. Am. Coll. Cardiol., 65 (2015), pp. 1385–1394 http://dx.doi.org/10.1016/j.jacc.2015.01.044
- Olesen et al., 2012 J.B. Olesen, G.Y.H. Lip, A.-L. Kamper, K. Hommel, L. Køber, D.A. Lane, J. Lindhardsen, G.H. Gislason, C. Torp-Pedersen; Stroke and bleeding in atrial fibrillation with chronic kidney disease; N. Engl. J. Med., 367 (2012), pp. 625–635 http://dx.doi.org/10.1056/NEJMoa1105594
- Olsson, 2003 S.B. Olsson; Stroke prevention with the oral direct thrombin inhibitor ximelagatran compared with warfarin in patients with non-valvular atrial fibrillation (SPORTIF III): randomised controlled trial; Lancet, 362 (2003), pp. 1691–1698
- Piccini et al., 2013 J.P. Piccini, S.R. Stevens, Y. Chang, D.E. Singer, Y. Lokhnygina, A.S. Go, M.R. Patel, K.W. Mahaffey, J.L. Halperin, G. Breithardt, G.J. Hankey, W. Hacke, R.C. Becker, C.C. Nessel, K.A.A. Fox, R.M. Califf; Renal dysfunction as a predictor of stroke and systemic embolism in patients with nonvalvular atrial fibrillation: validation of the R(2)CHADS(2) index in the ROCKET AF (rivaroxaban once-daily, oral, direct factor Xa inhibition compared with vitamin K ant; Circulation, 127 (2013), pp. 224–232 http://dx.doi.org/10.1161/CIRCULATIONAHA.112.107128
- Pokorney et al., 2015 S.D. Pokorney, D.N. Simon, L. Thomas, G.C. Fonarow, P.R. Kowey, P. Chang, D.E. Singer, J. Ansell, R.G. Blanco, B. Gersh, K.W. Mahaffey, E.M. Hylek, A.S. Go, J.P. Piccini, E.D. Peterson; Patients' time in therapeutic range on warfarin among US patients with atrial fibrillation: results from ORBIT-AF registry; Am. Heart J., 170 (2015), pp. 141–148, 148.e1 http://dx.doi.org/10.1016/j.ahj.2015.03.017
- Potpara et al., 2015 T.S. Potpara, R. Lenarczyk, T.B. Larsen, J.-C. Deharo, J. Chen, N. Dagres, Conducted by the Scientific Initiatives Committee, E.H.R.A., Conducted by the Scientific Initiatives Committee European Heart Rhythm Association; Management of atrial fibrillation in patients with chronic kidney disease in Europe results of the European Heart Rhythm Association survey; Europace, 17 (2015), pp. 1862–1867 http://dx.doi.org/10.1093/europace/euv416
- Roldán et al., 2013 V. Roldán, F. Marín, H. Fernández, S. Manzano-Fernández, P. Gallego, M. Valdés, V. Vicente, G.Y.H. Lip; Renal impairment in a “real-life” cohort of anticoagulated patients with atrial fibrillation (implications for thromboembolism and bleeding); Am. J. Cardiol., 111 (2013), pp. 1159–1164 http://dx.doi.org/10.1016/j.amjcard.2012.12.045
- Rosendaal et al., 1993 F.R. Rosendaal, S.C. Cannegieter, F.J. van der Meer, E. Briët; A method to determine the optimal intensity of oral anticoagulant therapy; Thromb. Haemost., 69 (1993), pp. 236–239
- Schiffrin et al., 2007 E.L. Schiffrin, M.L. Lipman, J.F.E. Mann; Chronic kidney disease: effects on the cardiovascular system; Circulation, 116 (2007), pp. 85–97 http://dx.doi.org/10.1161/CIRCULATIONAHA.106.678342
- Schulman and Kearon, 2005 S. Schulman, C. Kearon; Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients; J. Thromb. Haemost., 3 (2005), pp. 692–694 http://dx.doi.org/10.1111/j.1538-7836.2005.01204.x
- Shields and Lip, 2015 A.M. Shields, G.Y.H. Lip; Choosing the right drug to fit the patient when selecting oral anticoagulation for stroke prevention in atrial fibrillation; J. Intern. Med., 278 (2015), pp. 1–18 http://dx.doi.org/10.1111/joim.12360
- White et al., 2007 H.D. White, M. Gruber, J. Feyzi, S. Kaatz, H.-F. Tse, S. Husted, G.W. Albers; Comparison of outcomes among patients randomized to warfarin therapy according to anticoagulant control: results from SPORTIF III and V; Arch. Intern. Med., 167 (2007), pp. 239–245 http://dx.doi.org/10.1001/archinte.167.3.239
Document information
Published on 06/04/17
Licence: Other
Share this document
Keywords
claim authorship
Are you one of the authors of this document?